1 Shashikala TK, 2 Kavya Madhyastha
1Professor; 2Resident
Department of Anesthesiology, Mysore Medical College & Research Institute, Irwin Road, Mysuru, Karnataka 570001, (India)
Correspondence: Dr Shashikala TK, No 156, 1st cross, 4th main, Bank colony, Bogadhi, Mysore – 570026 (India); Phone: 9480057506; E-mail: drshi72@gmail.com
ABSTRACT
Background: Dexmedetomidine, highly selective α2 agonist is used as an effective adjuvant to various local anesthetics in regional anesthesia. We have conducted this study to know the efficacy of dexmedetomidine through two routes i.e intravenous and perineural routes with ropivacaine for supraclavicular brachial plexus block in elective forearm surgeries.
Methodology: After the institutional ethical committee clearance, 60 patients aged between 18 to 60 years, belonging to ASA class I and II were randomly divided into two groups of 30 each after a detailed informed consent. Group DPN received 0.5% ropivacaine 28 ml + 2 ml of dexmedetomidine 50 μg perineurally and 100 ml of 0.9% normal saline intravenously. Group DIV received 0.5% ropivacaine 28 ml + 2 ml 0.9% normal saline perineurally and 50 μg of Dexmedetomidine in 100 ml of 0.9% normal saline intravenously. Primary objectives was to know the time of sensory onset, motor onset, time for complete motor and sensory block, total duration of sensory and motor block and total duration of analgesia. Secondary objectives were hemodynamic parameters, sedation scores and adverse events. Statistical methods were carried out through the SPSS for Windows (version 16.0)
Results: The demographic profile of the patients like age, sex, height, weight, BMI were similar in both groups. The sensory and motor onset time as well as time for complete sensory and motor block was earlier in Group DPN than Group DIV. The sensory block duration was prolonged in Group DPN (686.33 ± 52.22 min) compared to Group DIV (371 ± 38.98 min) [p < 0.001]. The motor block duration was also significantly prolonged in Group DPN (593.50 ± 58.12 min) compared to Group DIV (319.50 ± 26.47 min) [p < 0.001]. The total duration of analgesia was also significantly prolonged in Group DPN (701.83 ± 44.92 min) compared to Group DIV (405.16 ± 30.86 min)[p < 0.001]. Two patients had bradycardia and one patient had hypotension in Group DIV. Ramsay sedation scores in both the groups were < 3.
Conclusion: Dexmedetomidine is an excellent adjuvant to ropivacaine for supraclavicular block. Perineural dexmedetomidine offers a better block quality and a prolonged duration of analgesia compared to intravenous dexmedetomidine with minimal side effects
Key words: Dexmedetomidine, Ropivacaine, brachial plexus block
Citation: Shashikala TK, Madhyastha K. A prospective randomized double blinded study to evaluate the efficacy of dexmedetomidine 50 μg intravenously and perineurally as an adjunct to 0.5% ropivacaine for supraclavicular brachial plexus block. Anaesth Pain & Intensive Care 2017;21(4):413-419
Received: 12 Dec 2017; Reviewed: 13, 14, 22 Dec 2017; Corrected & Accepted: 24 Dec 2017
INTRODUCTION
Regional anesthesia is the most common and popular technique of choice for upper limb surgeries in the recent days – sometimes as a sole mode of anesthesia when general anesthesia is not indicated and sometimes as an adjunct to general anesthesia.1,2 It has several advantages credited to itself being- safe, patient friendly, better surgical field, good relaxation, better hemodynamic profile, superior quality of analgesia- both intra and postoperatively, avoidance of poly pharmacy, early recovery, decreased incidence of postoperative nausea and vomiting, decreased postoperative pulmonary complications, early mobilization and shorter hospital stay.2,3 Thus regional anesthesia is preferred over general anesthesia, wherever feasible. Supraclavicular brachial plexus block, is often compared to spinal anesthesia of lower limb. The reason being, it approaches the brachial plexus where the relatively compact trunks / divisions track under the clavicle and over the first rib, residing posterior, lateral and cephalad to the subclavian artery.3 This approach provides anesthesia for upper arm, elbow and forearm surgeries. Shoulder surgeries can also be performed with supplementation of supraclavicular nerve block.
The intra and postoperative analgesia duration depends on the choice of local anesthetic made. Using a short acting local anesthetic can make regional anesthesia a shortcoming. Hence an intermediate or long acting local anesthetic is preferred over the conventional local anesthetic lignocaine.4 Bupivacaine became more commonly used local anesthetic for regional anesthesia. The major drawback for Bupivacaine was its cardiotoxic profile, which is a serious concern in supraclavicular block where brachial plexus lies in close proximity to major vessels.1,2 The search for a better local anesthetic continued and in 1996, ropivacaine was introduced which was an S enantiomer.3 Being less lipophilic than bupivacaine and due to its stereoselective property, ropivacaine has a cardiosafe profile.2,3,5 Another unique feature of ropivacaine is its preferential action on Aδ and C fibers (which transmit pain) than on Aβ fibers (which provides motor function). Thus it provides a prolonged sensory blockade and shorter motor blockade which makes it a better choice for regional anesthesia.1,2,3
Ropivacaine when used alone may provide analgesia for 4-6 hours. If we need a prolonged postoperative analgesia we may look into other options like – use of continuous infusion catheters or adding an additive to local anesthetic.3,6 Continuous infusion catheter placement requires resource availability, skilled person, cost, monitoring facility.3 Hence use of additives to local anesthetics makes a better and a safe option to prolong the analgesia. various additives have been tried and are being studied for its safety and efficacy like – epinephrine, opioids (fentanyl, buprenorphine, tramadol), ketamine, α2 agonist (clonidine, dexmedetomidine), steroids (dexamethasone) etc.1,4 α2 agonists have analgesic, anxiolytic, sedative and sympatholytic properties, which makes it a better choice for local anesthetic.3 Dexmedetomidine, introduced in 1999 in US, is a more selective α 2 agonist. It was initially approved for short term ICU sedation. Later on it was used for procedural sedation inside the operating room as well as outside the operating room.7 Off label studies have shown that it has increased the duration of analgesia of local anesthetic when used as an adjunct.1 Its safety profile, opioid sparing effect and its lack of respiratory depression makes it even more attractive choice of additive to a local anesthetic. Though we know about the analgesic effect of dexmedetomidine, we wanted to study the efficacy of this wonder drug in various routes. Hence we undertook this study to know the efficacy of dexmedetomidine as an adjunct to ropivacaine in intravenous route and perineural route for supraclavicular brachial plexus block
METHODOLOGY
This study was conducted on 60 patients who were posted for elective forearm surgeries at K. R. Hospital attached to Mysore Medical College and Research Institute, Mysore. The study was conducted from June 2016 to July 2017.
Patients aged between 18-60 years, belonging to ASA class I-II, without any serious comorbid diseases, admitted for elective forearm surgeries were included in the study.
Exclusion Criteria included patient refusal, patients with ASA class III, IV &V, patients with any serious comorbid conditions like diabetes mellitus, asthma, hypertension, cardiac diseases, hematological diseases, ischemic heart disease, epilepsy, coagulopathies etc, patients allergic to local anesthetics, patients posted for emergency surgeries, patients with BMI more than 30 kg/m2, pregnant patients, patients with significant psychiatric illness and preexisting neurological deficits or neuropathy affecting brachial plexus. After institutional ethical committee clearance, 60 patients in the age group 18-60 years of ASA class I-II, posted for elective upper limb surgeries without any serious comorbid diseases, were selected. The study group was randomly divided into two groups of thirty each, by closed sealed opaque envelope method.
Group DPN received – 28 ml 0.5% ropivacaine + 50 µg dexmedetomidine (2 ml) perineurally and 100 ml of 0.9% normal saline iv. Group DIV received – 28 ml 0.5% Ropivacaine + 2 ml normal saline perineurally and 50 μg dexmedetomidine in 100 ml 0.9% normal saline iv
Data was collected in a pretested proforma meeting the objectives of the study. Preoperative assessment was done for each patient and written informed consent was taken. Patients were premedicated with tablet Ranitidine 150 mg and tablet alprazolam 0.5 mg on the night before the surgery. Intravenous line were obtained with 18G cannula in the unaffected upper limb and was co loaded with ringer lactate 15 ml/kg bodyweight. Patients were connected to multi-channel monitor (Star Plus; Larsen Toubro Ltd, India) for monitoring pulse, SpO2, ECG, NIBP, MAP.
Patients were premedicated with midazolam 0.02 mg/kg. They received the intravenous preparation as per the group allocation over 10 min prior to the block and were positioned in supine position with head turned opposite to the side of procedure. All patients were given supraclavicular brachial plexus block using Winnie’s perivascular, subclavian approach. The interscalene groove was identified, then traced downwards and Subclavian artery is palpated in the groove. The artery is then pushed downwards with the non-dominant hand’s thumb. The point just above the thumb was the entry point for the needle. 1mA selected as the current in the peripheral nerve stimulator (Plexigon™) and 5 cm Stimuplex™ stimulating needle was used. The needle entered at 300 angle with the direction, caudal and posterior, till the muscle twitches were elicited in the hand and fingers. The current was reduced and when twitches were still present at 0.4 mA and disappearing below that, 30 ml of study drug as per the group allocation was injected at the incremental doses of 3 ml with negative aspiration for blood. Intercostobrachial nerve blockade was done separately using 5 ml of 1% lignocaine with adrenaline.
The following parameters were noted;
Hypotension was defined as fall in systolic blood pressure > 30% of the basal systolic blood pressure and was treated with incremental doses of injection mephenteramine 6 mg and fluid bolus IV.
Bradycardia was defined as decrease in heart rate by >20% of the basal heart rate or heart rate < 50 beats per min and was treated with injection atropine 0.6 mg iv.
Postoperatively all the patients were monitored in the post anesthesia care unit for sedation, hypotension, bradycardia, pain any adverse reactions.
Statistical analysis: All the statistical methods were carried out through the SPSS for Windows (version 16.0). Age, height, weight, body mass index (BMI), sensory and motor onset time, time for complete sensory and motor blockade, duration of sensory and motor blockade and total duration of analgesia were analyzed by using independent student’s t – test. Sex ratio and sedation scores were compared using repeated ANOVA test. Systolic, diastolic and mean arterial pressure, heart rate and peripheral oxygen saturation was compared using one way ANOVA test.
RESULTS
There was no statistically significant difference between the two groups with respect to demographic data like age, sex ratio, weight, height, BMI.
The sensory onset at C5, C6, C8, T1 dermatomes was earlier in Group DPN (4.73 ±1.14 min, 5.33 ± 1.03 min, 7.43 ± 0.93 min, 8.10 ± 0.71 min respectively) compared to Group DIV (5.00 ± 0.64 min, 5.76 ± 0.57 min, 7.50 ± 0.73 min, 8.30 ± 0.65 min), but this was not statistically significant with p > 0.05. Sensory onset at C7 was earlier in Group DPN (6.66 ± 1.06 min) compared to Group DIV (6.83 ± 0.70 min) and was statistically significant (p = 0.048). The motor onset time at shoulder level was earlier in Group DPN (7.33 ± 1.09 min) compared to Group DIV (6.46 ± 0.82 min) and it was statistically highly significant (p = 0.001). Though the motor onset at elbow and wrist level were earlier in Group DPN (8.70 ± 0.87 min and 11.13 ± 1.07 min) compared to Group DIV (9.26 ± 1.28 min and 11.26 ± 1.23 min), it was not statistically significant (p > 0.05). The time for complete sensory blockade and motor blockade was earlier in Group DPN (10.13 ± 1.28 min and 14.20 ± 1.44 min) compared to Group DIV (14.43 ± 0.90 min and 17.03 ± 1.03 min) and it was statistically highly significant (p < 0.001)
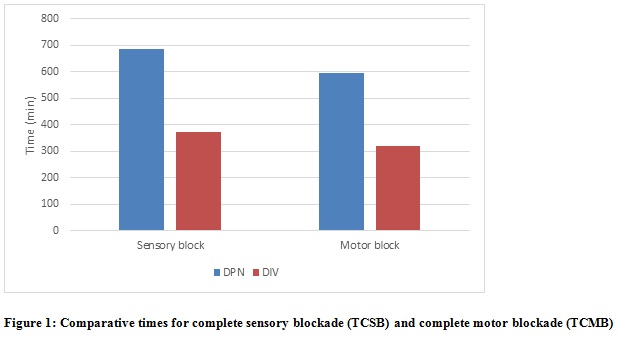
DPN=Group Perineural; DIV= Group Intravenous
The mean total sensory block duration was more in Group DPN (686.33 ± 52.22 min) compared to Group DIV (371± 38.98 min). The mean motor block duration was prolonged in Group DPN (593.50 ± 58.12 min) compared to Group DIV (319.50 ± 26.47 min). The total duration of analgesia was more in Group DPN (701.83 ± 44.92 min) compared to Group DIV (405.16 ± 30.86 min). The difference in means of all these parameters were statistically highly significant (p < 0.001)(Figure 2).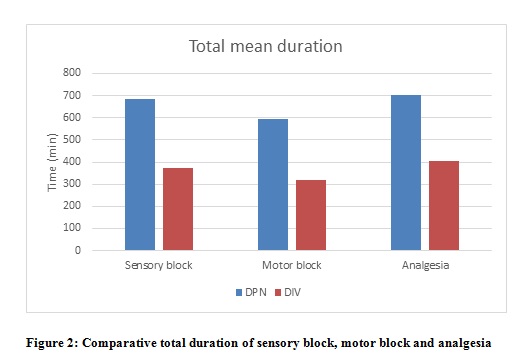
DPN=Group perineural; DIV= Group intravenous
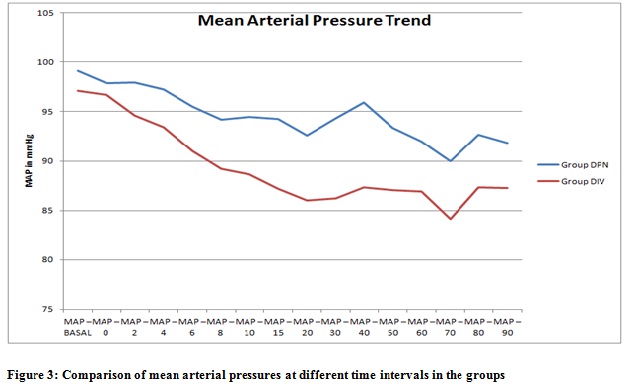
The mean systolic blood pressure at various intervals of time was lower in Group DIV compared to Group DPN. This difference was statistically significant from 6th min onwards after the administration of block. The mean diastolic blood pressure values were less in Group DIV compared to Group DPN, but it was statistically significant only in 20th and 30th min. The mean arterial pressure values were less in Group DIV compared to Group DPN. This difference was statistically significant in 15th, 20th, 30th and 40th min after block (Figure 3).
The mean heart rate values were lower in Group DIV compared to Group DPN and it was statistically significant after the 8th minute after the block (Figure 4). The differences in mean peripheral oxygen saturation values and sedation scores were in both groups were not significant.
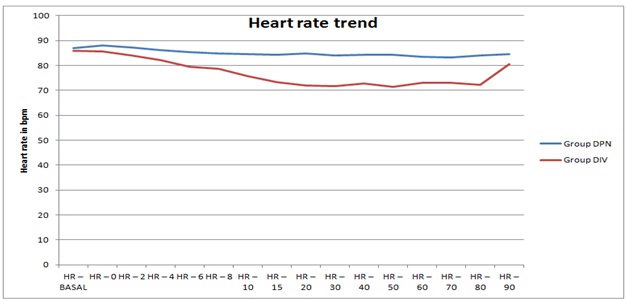
Figure 4: Comparison of mean heart rate at different time intervals in the groups
Two patients had bradycardia and one patient had hypotension in Group DIV which were treated accordingly with 0.6 mg atropine and inj mephenteramine 6 mg in graded doses. No adverse events were noted in DPN.
DISCUSSION
Brachial plexus block is the most commonly used regional anesthesia technique in upper limb surgeries. It is safe alternative to general anesthesia, which provides good intra operative and prolonged postoperative analgesia with minimal side effects like – respiratory depression, hypotension and, bradycardia.8 With the intervention of ultrasound and nerve stimulator technology, efficacy and safety of the brachial plexus block were greatly improved. It helps in reducing the hospital stay, early mobilization and less postoperative respiratory complications, less financial burden and also undesirable side effects of general anesthesia. Conventionally, lignocaine and bupivacaine have been used as the local anesthetic for regional anesthesia till recently. Ropivacaine is an amide group of local anesthetic with a prolonged duration of action with minimal cardiotoxicity when compared to Bupivacaine.8 However, these early advantages can be short-lived which results in block resolution before the period of worst postoperative pain.9 Many of the adjuvants have been used to prolong the postoperative analgesia. Dexmedetomidine is a newer α 2 adrenoceptor agonist is currently gained wide popularity for its sedative, anxiolytic and analgesic properties. Pre and intra operative intravenous administration of dexmedetomidine has shown to prolong the duration of sensory block with local anesthetics.10 Adding dexmedetomidine to local anesthetics during the peripheral nerve blockade and regional anesthesia may also prove more efficacious than systemic routine.8 Our current study was designed to test the hypothesis that dexmedetomidine when added as an adjuvant to ropivacaine in supraclavicular brachial plexus block (perineural), enhance the duration of sensory and motor block, duration of analgesia and quality of block which is superior to systemic use of dexmedetomidine.
In our study, we chose 0.5% ropivacaine for supraclavicular block. The rationale for choosing this concentration is supported by the study conducted by Klein et al in 1998.11 The study was to compare the efficacy of bupivacaine 0.5%, ropivacaine 0.5% and ropivacaine 0.75%, the volume being 30 ml each. They found that there was no significant difference in time of onset and recovery or any improvement in duration of analgesia. Ropivacaine causes a greater sensory and motor differential blockade than bupivacaine which is dose-dependant. Higher concentrations (1%) causing greater degree of motor blockade than lower concentrations 0.5% and 0.75%).11 Hickey and coworkers have shown that 0.25% ropivacaine when used for subclavian perivascular brachial plexus block for upper limb surgeries required frequent analgesia supplementation due to low concentration of local anesthetic.12
In 2013 F.W Abdallah and R Brull conducted a meta-analysis where they concluded that dexmedetomidine is potential local anesthetic adjuvant intrathecally as well as for peripheral nerve blockade, but its safety profile was not conclusively approved due to lack of clinical trials.9 In 2017, they conducted meta-analysis study to re-evaluate the basis of using perineural dexmedetomidine, they concluded that perineural dexmedetomidine was safe. In fact there is some evidence that perineural dexmedetomidine may be neuroprotective against the local anesthetic induced inflammatory response in few animal studies,13 thereby decreasing the potential for peripheral nerve injury.10 In human beings, the beneficial effect of adding dexmedetomidine to local anesthetics during regional anesthesia and some peripheral nerve blockade procedures have proved to be efficacious for surgical patients.10
The unique mechanism of action of dexmedetomidine sets it apart from other sedatives. The α2 adrenoceptors are found in abundant number in locus ceruleus in CNS. The presynaptic activation of α2 adrenoceptors inhibits the release of norepinephrine which results in sedative and hypnotic effects. Also, it is the site of origin for the descending medullospinal pathway, which is an important modulator of nociceptive neurotransmission. Thus stimulation of adrenoceptors leads to analgesia. Postsynaptic activation leads sympathetic outflow blockade leading to bradycardia and hypotension. Sedation offered by dexmedetomidine mimics NREM sleep. Thus the patient appears to be deeply sedated but easily arousable as in natural sleep.7,14
Zhang Y et al15 conducted a study to know the effect of dexmedetomidine in different doses as an adjuvant with ropivacaine in supraclavicular brachial plexus block i.e. 50 and 100 µg with 40 ml of 0.33% ropivacaine. It was concluded that dexmedetomidine may prolong the duration of block when added to ropivacaine, but may also have side effects such as bradycardia, hypertension and hypotension when used in high dose. Hence we chose 50 µg of dexmedetomidine as an adjuvant to 0.5% ropivacaine.
In our study there was no statistically significant difference between the two groups with respect to demographic data like age, sex ratio, weight, height, BMI.
The time for complete sensory blockade as well as complete motor blockade was early in Group DPN compared to Group DIV, which is statistically highly significant (p < 0.001). The results can be compared with the results of the study by Kathuria S et al.10
The total sensory and motor block duration were prolonged in Group DPN when compared to Group DIV (p < 0.001). The results are comparable with the study conducted by Kathuria et al,10 in which they got similar results. In present study the total duration of analgesia was prolonged in Group DPN when compared to Group DIV which is statistically highly significant (p < 0.001).
Abdallah F W et al16 conducted similar study, they observed prolonged analgesia in perineural approach when compared to intravenous route, which is statistically significant, but they induced general anesthesia after giving blocks. So our study cannot be compared with this study.
CONCLUSION
We conclude that in supraclavicular brachial plexus addition of dexmedetomidine to 0.5% ropivacaine shortens the sensory and motor block onset time, significantly prolongs the duration of sensory and motor block compared to systemic use of dexmedetomidine. The perineural approach significantly delays the need for rescue analgesia with minimal side effects compared to IV route of dexmedetomidine.
Conflict of interest: No conflict declared by the authors.
Author contribution: STK - Concept, conduct of study, editing of manuscript
KM - Conduct of study
REFERENCES
1Professor; 2Resident
Department of Anesthesiology, Mysore Medical College & Research Institute, Irwin Road, Mysuru, Karnataka 570001, (India)
Correspondence: Dr Shashikala TK, No 156, 1st cross, 4th main, Bank colony, Bogadhi, Mysore – 570026 (India); Phone: 9480057506; E-mail: drshi72@gmail.com
ABSTRACT
Background: Dexmedetomidine, highly selective α2 agonist is used as an effective adjuvant to various local anesthetics in regional anesthesia. We have conducted this study to know the efficacy of dexmedetomidine through two routes i.e intravenous and perineural routes with ropivacaine for supraclavicular brachial plexus block in elective forearm surgeries.
Methodology: After the institutional ethical committee clearance, 60 patients aged between 18 to 60 years, belonging to ASA class I and II were randomly divided into two groups of 30 each after a detailed informed consent. Group DPN received 0.5% ropivacaine 28 ml + 2 ml of dexmedetomidine 50 μg perineurally and 100 ml of 0.9% normal saline intravenously. Group DIV received 0.5% ropivacaine 28 ml + 2 ml 0.9% normal saline perineurally and 50 μg of Dexmedetomidine in 100 ml of 0.9% normal saline intravenously. Primary objectives was to know the time of sensory onset, motor onset, time for complete motor and sensory block, total duration of sensory and motor block and total duration of analgesia. Secondary objectives were hemodynamic parameters, sedation scores and adverse events. Statistical methods were carried out through the SPSS for Windows (version 16.0)
Results: The demographic profile of the patients like age, sex, height, weight, BMI were similar in both groups. The sensory and motor onset time as well as time for complete sensory and motor block was earlier in Group DPN than Group DIV. The sensory block duration was prolonged in Group DPN (686.33 ± 52.22 min) compared to Group DIV (371 ± 38.98 min) [p < 0.001]. The motor block duration was also significantly prolonged in Group DPN (593.50 ± 58.12 min) compared to Group DIV (319.50 ± 26.47 min) [p < 0.001]. The total duration of analgesia was also significantly prolonged in Group DPN (701.83 ± 44.92 min) compared to Group DIV (405.16 ± 30.86 min)[p < 0.001]. Two patients had bradycardia and one patient had hypotension in Group DIV. Ramsay sedation scores in both the groups were < 3.
Conclusion: Dexmedetomidine is an excellent adjuvant to ropivacaine for supraclavicular block. Perineural dexmedetomidine offers a better block quality and a prolonged duration of analgesia compared to intravenous dexmedetomidine with minimal side effects
Key words: Dexmedetomidine, Ropivacaine, brachial plexus block
Citation: Shashikala TK, Madhyastha K. A prospective randomized double blinded study to evaluate the efficacy of dexmedetomidine 50 μg intravenously and perineurally as an adjunct to 0.5% ropivacaine for supraclavicular brachial plexus block. Anaesth Pain & Intensive Care 2017;21(4):413-419
Received: 12 Dec 2017; Reviewed: 13, 14, 22 Dec 2017; Corrected & Accepted: 24 Dec 2017
INTRODUCTION
Regional anesthesia is the most common and popular technique of choice for upper limb surgeries in the recent days – sometimes as a sole mode of anesthesia when general anesthesia is not indicated and sometimes as an adjunct to general anesthesia.1,2 It has several advantages credited to itself being- safe, patient friendly, better surgical field, good relaxation, better hemodynamic profile, superior quality of analgesia- both intra and postoperatively, avoidance of poly pharmacy, early recovery, decreased incidence of postoperative nausea and vomiting, decreased postoperative pulmonary complications, early mobilization and shorter hospital stay.2,3 Thus regional anesthesia is preferred over general anesthesia, wherever feasible. Supraclavicular brachial plexus block, is often compared to spinal anesthesia of lower limb. The reason being, it approaches the brachial plexus where the relatively compact trunks / divisions track under the clavicle and over the first rib, residing posterior, lateral and cephalad to the subclavian artery.3 This approach provides anesthesia for upper arm, elbow and forearm surgeries. Shoulder surgeries can also be performed with supplementation of supraclavicular nerve block.
The intra and postoperative analgesia duration depends on the choice of local anesthetic made. Using a short acting local anesthetic can make regional anesthesia a shortcoming. Hence an intermediate or long acting local anesthetic is preferred over the conventional local anesthetic lignocaine.4 Bupivacaine became more commonly used local anesthetic for regional anesthesia. The major drawback for Bupivacaine was its cardiotoxic profile, which is a serious concern in supraclavicular block where brachial plexus lies in close proximity to major vessels.1,2 The search for a better local anesthetic continued and in 1996, ropivacaine was introduced which was an S enantiomer.3 Being less lipophilic than bupivacaine and due to its stereoselective property, ropivacaine has a cardiosafe profile.2,3,5 Another unique feature of ropivacaine is its preferential action on Aδ and C fibers (which transmit pain) than on Aβ fibers (which provides motor function). Thus it provides a prolonged sensory blockade and shorter motor blockade which makes it a better choice for regional anesthesia.1,2,3
Ropivacaine when used alone may provide analgesia for 4-6 hours. If we need a prolonged postoperative analgesia we may look into other options like – use of continuous infusion catheters or adding an additive to local anesthetic.3,6 Continuous infusion catheter placement requires resource availability, skilled person, cost, monitoring facility.3 Hence use of additives to local anesthetics makes a better and a safe option to prolong the analgesia. various additives have been tried and are being studied for its safety and efficacy like – epinephrine, opioids (fentanyl, buprenorphine, tramadol), ketamine, α2 agonist (clonidine, dexmedetomidine), steroids (dexamethasone) etc.1,4 α2 agonists have analgesic, anxiolytic, sedative and sympatholytic properties, which makes it a better choice for local anesthetic.3 Dexmedetomidine, introduced in 1999 in US, is a more selective α 2 agonist. It was initially approved for short term ICU sedation. Later on it was used for procedural sedation inside the operating room as well as outside the operating room.7 Off label studies have shown that it has increased the duration of analgesia of local anesthetic when used as an adjunct.1 Its safety profile, opioid sparing effect and its lack of respiratory depression makes it even more attractive choice of additive to a local anesthetic. Though we know about the analgesic effect of dexmedetomidine, we wanted to study the efficacy of this wonder drug in various routes. Hence we undertook this study to know the efficacy of dexmedetomidine as an adjunct to ropivacaine in intravenous route and perineural route for supraclavicular brachial plexus block
METHODOLOGY
This study was conducted on 60 patients who were posted for elective forearm surgeries at K. R. Hospital attached to Mysore Medical College and Research Institute, Mysore. The study was conducted from June 2016 to July 2017.
Patients aged between 18-60 years, belonging to ASA class I-II, without any serious comorbid diseases, admitted for elective forearm surgeries were included in the study.
Exclusion Criteria included patient refusal, patients with ASA class III, IV &V, patients with any serious comorbid conditions like diabetes mellitus, asthma, hypertension, cardiac diseases, hematological diseases, ischemic heart disease, epilepsy, coagulopathies etc, patients allergic to local anesthetics, patients posted for emergency surgeries, patients with BMI more than 30 kg/m2, pregnant patients, patients with significant psychiatric illness and preexisting neurological deficits or neuropathy affecting brachial plexus. After institutional ethical committee clearance, 60 patients in the age group 18-60 years of ASA class I-II, posted for elective upper limb surgeries without any serious comorbid diseases, were selected. The study group was randomly divided into two groups of thirty each, by closed sealed opaque envelope method.
Group DPN received – 28 ml 0.5% ropivacaine + 50 µg dexmedetomidine (2 ml) perineurally and 100 ml of 0.9% normal saline iv. Group DIV received – 28 ml 0.5% Ropivacaine + 2 ml normal saline perineurally and 50 μg dexmedetomidine in 100 ml 0.9% normal saline iv
Data was collected in a pretested proforma meeting the objectives of the study. Preoperative assessment was done for each patient and written informed consent was taken. Patients were premedicated with tablet Ranitidine 150 mg and tablet alprazolam 0.5 mg on the night before the surgery. Intravenous line were obtained with 18G cannula in the unaffected upper limb and was co loaded with ringer lactate 15 ml/kg bodyweight. Patients were connected to multi-channel monitor (Star Plus; Larsen Toubro Ltd, India) for monitoring pulse, SpO2, ECG, NIBP, MAP.
Patients were premedicated with midazolam 0.02 mg/kg. They received the intravenous preparation as per the group allocation over 10 min prior to the block and were positioned in supine position with head turned opposite to the side of procedure. All patients were given supraclavicular brachial plexus block using Winnie’s perivascular, subclavian approach. The interscalene groove was identified, then traced downwards and Subclavian artery is palpated in the groove. The artery is then pushed downwards with the non-dominant hand’s thumb. The point just above the thumb was the entry point for the needle. 1mA selected as the current in the peripheral nerve stimulator (Plexigon™) and 5 cm Stimuplex™ stimulating needle was used. The needle entered at 300 angle with the direction, caudal and posterior, till the muscle twitches were elicited in the hand and fingers. The current was reduced and when twitches were still present at 0.4 mA and disappearing below that, 30 ml of study drug as per the group allocation was injected at the incremental doses of 3 ml with negative aspiration for blood. Intercostobrachial nerve blockade was done separately using 5 ml of 1% lignocaine with adrenaline.
The following parameters were noted;
- Onset of sensory blockade
- Sensory blockade was tested using pin prick method with a 27G hypodermic needle every 2 min after giving block in each of the dermatomes C5, C6, C7, C8 and T1 separately.
- Onset of motor blockade at the level of shoulder, elbow and wrist.
- the time taken for complete sensory and motor blockade
- Total duration of analgesia
- All the patients were monitored during the period of block and peri operative period employing multi-channel monitor which displays heart rate, systolic and diastolic blood pressure, mean arterial pressure, SpO2
- Time of motor regression to score 0
- Time of sensory regression to score 0
- Sedation was assessed by a Modified Ramsay Sedation Score.
Hypotension was defined as fall in systolic blood pressure > 30% of the basal systolic blood pressure and was treated with incremental doses of injection mephenteramine 6 mg and fluid bolus IV.
Bradycardia was defined as decrease in heart rate by >20% of the basal heart rate or heart rate < 50 beats per min and was treated with injection atropine 0.6 mg iv.
Postoperatively all the patients were monitored in the post anesthesia care unit for sedation, hypotension, bradycardia, pain any adverse reactions.
Statistical analysis: All the statistical methods were carried out through the SPSS for Windows (version 16.0). Age, height, weight, body mass index (BMI), sensory and motor onset time, time for complete sensory and motor blockade, duration of sensory and motor blockade and total duration of analgesia were analyzed by using independent student’s t – test. Sex ratio and sedation scores were compared using repeated ANOVA test. Systolic, diastolic and mean arterial pressure, heart rate and peripheral oxygen saturation was compared using one way ANOVA test.
RESULTS
There was no statistically significant difference between the two groups with respect to demographic data like age, sex ratio, weight, height, BMI.
The sensory onset at C5, C6, C8, T1 dermatomes was earlier in Group DPN (4.73 ±1.14 min, 5.33 ± 1.03 min, 7.43 ± 0.93 min, 8.10 ± 0.71 min respectively) compared to Group DIV (5.00 ± 0.64 min, 5.76 ± 0.57 min, 7.50 ± 0.73 min, 8.30 ± 0.65 min), but this was not statistically significant with p > 0.05. Sensory onset at C7 was earlier in Group DPN (6.66 ± 1.06 min) compared to Group DIV (6.83 ± 0.70 min) and was statistically significant (p = 0.048). The motor onset time at shoulder level was earlier in Group DPN (7.33 ± 1.09 min) compared to Group DIV (6.46 ± 0.82 min) and it was statistically highly significant (p = 0.001). Though the motor onset at elbow and wrist level were earlier in Group DPN (8.70 ± 0.87 min and 11.13 ± 1.07 min) compared to Group DIV (9.26 ± 1.28 min and 11.26 ± 1.23 min), it was not statistically significant (p > 0.05). The time for complete sensory blockade and motor blockade was earlier in Group DPN (10.13 ± 1.28 min and 14.20 ± 1.44 min) compared to Group DIV (14.43 ± 0.90 min and 17.03 ± 1.03 min) and it was statistically highly significant (p < 0.001)

DPN=Group Perineural; DIV= Group Intravenous
The mean total sensory block duration was more in Group DPN (686.33 ± 52.22 min) compared to Group DIV (371± 38.98 min). The mean motor block duration was prolonged in Group DPN (593.50 ± 58.12 min) compared to Group DIV (319.50 ± 26.47 min). The total duration of analgesia was more in Group DPN (701.83 ± 44.92 min) compared to Group DIV (405.16 ± 30.86 min). The difference in means of all these parameters were statistically highly significant (p < 0.001)(Figure 2).

DPN=Group perineural; DIV= Group intravenous
The mean systolic blood pressure at various intervals of time was lower in Group DIV compared to Group DPN. This difference was statistically significant from 6th min onwards after the administration of block. The mean diastolic blood pressure values were less in Group DIV compared to Group DPN, but it was statistically significant only in 20th and 30th min. The mean arterial pressure values were less in Group DIV compared to Group DPN. This difference was statistically significant in 15th, 20th, 30th and 40th min after block (Figure 3).

The mean heart rate values were lower in Group DIV compared to Group DPN and it was statistically significant after the 8th minute after the block (Figure 4). The differences in mean peripheral oxygen saturation values and sedation scores were in both groups were not significant.

Figure 4: Comparison of mean heart rate at different time intervals in the groups

Two patients had bradycardia and one patient had hypotension in Group DIV which were treated accordingly with 0.6 mg atropine and inj mephenteramine 6 mg in graded doses. No adverse events were noted in DPN.
DISCUSSION
Brachial plexus block is the most commonly used regional anesthesia technique in upper limb surgeries. It is safe alternative to general anesthesia, which provides good intra operative and prolonged postoperative analgesia with minimal side effects like – respiratory depression, hypotension and, bradycardia.8 With the intervention of ultrasound and nerve stimulator technology, efficacy and safety of the brachial plexus block were greatly improved. It helps in reducing the hospital stay, early mobilization and less postoperative respiratory complications, less financial burden and also undesirable side effects of general anesthesia. Conventionally, lignocaine and bupivacaine have been used as the local anesthetic for regional anesthesia till recently. Ropivacaine is an amide group of local anesthetic with a prolonged duration of action with minimal cardiotoxicity when compared to Bupivacaine.8 However, these early advantages can be short-lived which results in block resolution before the period of worst postoperative pain.9 Many of the adjuvants have been used to prolong the postoperative analgesia. Dexmedetomidine is a newer α 2 adrenoceptor agonist is currently gained wide popularity for its sedative, anxiolytic and analgesic properties. Pre and intra operative intravenous administration of dexmedetomidine has shown to prolong the duration of sensory block with local anesthetics.10 Adding dexmedetomidine to local anesthetics during the peripheral nerve blockade and regional anesthesia may also prove more efficacious than systemic routine.8 Our current study was designed to test the hypothesis that dexmedetomidine when added as an adjuvant to ropivacaine in supraclavicular brachial plexus block (perineural), enhance the duration of sensory and motor block, duration of analgesia and quality of block which is superior to systemic use of dexmedetomidine.
In our study, we chose 0.5% ropivacaine for supraclavicular block. The rationale for choosing this concentration is supported by the study conducted by Klein et al in 1998.11 The study was to compare the efficacy of bupivacaine 0.5%, ropivacaine 0.5% and ropivacaine 0.75%, the volume being 30 ml each. They found that there was no significant difference in time of onset and recovery or any improvement in duration of analgesia. Ropivacaine causes a greater sensory and motor differential blockade than bupivacaine which is dose-dependant. Higher concentrations (1%) causing greater degree of motor blockade than lower concentrations 0.5% and 0.75%).11 Hickey and coworkers have shown that 0.25% ropivacaine when used for subclavian perivascular brachial plexus block for upper limb surgeries required frequent analgesia supplementation due to low concentration of local anesthetic.12
In 2013 F.W Abdallah and R Brull conducted a meta-analysis where they concluded that dexmedetomidine is potential local anesthetic adjuvant intrathecally as well as for peripheral nerve blockade, but its safety profile was not conclusively approved due to lack of clinical trials.9 In 2017, they conducted meta-analysis study to re-evaluate the basis of using perineural dexmedetomidine, they concluded that perineural dexmedetomidine was safe. In fact there is some evidence that perineural dexmedetomidine may be neuroprotective against the local anesthetic induced inflammatory response in few animal studies,13 thereby decreasing the potential for peripheral nerve injury.10 In human beings, the beneficial effect of adding dexmedetomidine to local anesthetics during regional anesthesia and some peripheral nerve blockade procedures have proved to be efficacious for surgical patients.10
The unique mechanism of action of dexmedetomidine sets it apart from other sedatives. The α2 adrenoceptors are found in abundant number in locus ceruleus in CNS. The presynaptic activation of α2 adrenoceptors inhibits the release of norepinephrine which results in sedative and hypnotic effects. Also, it is the site of origin for the descending medullospinal pathway, which is an important modulator of nociceptive neurotransmission. Thus stimulation of adrenoceptors leads to analgesia. Postsynaptic activation leads sympathetic outflow blockade leading to bradycardia and hypotension. Sedation offered by dexmedetomidine mimics NREM sleep. Thus the patient appears to be deeply sedated but easily arousable as in natural sleep.7,14
Zhang Y et al15 conducted a study to know the effect of dexmedetomidine in different doses as an adjuvant with ropivacaine in supraclavicular brachial plexus block i.e. 50 and 100 µg with 40 ml of 0.33% ropivacaine. It was concluded that dexmedetomidine may prolong the duration of block when added to ropivacaine, but may also have side effects such as bradycardia, hypertension and hypotension when used in high dose. Hence we chose 50 µg of dexmedetomidine as an adjuvant to 0.5% ropivacaine.
In our study there was no statistically significant difference between the two groups with respect to demographic data like age, sex ratio, weight, height, BMI.
The time for complete sensory blockade as well as complete motor blockade was early in Group DPN compared to Group DIV, which is statistically highly significant (p < 0.001). The results can be compared with the results of the study by Kathuria S et al.10
The total sensory and motor block duration were prolonged in Group DPN when compared to Group DIV (p < 0.001). The results are comparable with the study conducted by Kathuria et al,10 in which they got similar results. In present study the total duration of analgesia was prolonged in Group DPN when compared to Group DIV which is statistically highly significant (p < 0.001).
Abdallah F W et al16 conducted similar study, they observed prolonged analgesia in perineural approach when compared to intravenous route, which is statistically significant, but they induced general anesthesia after giving blocks. So our study cannot be compared with this study.
CONCLUSION
We conclude that in supraclavicular brachial plexus addition of dexmedetomidine to 0.5% ropivacaine shortens the sensory and motor block onset time, significantly prolongs the duration of sensory and motor block compared to systemic use of dexmedetomidine. The perineural approach significantly delays the need for rescue analgesia with minimal side effects compared to IV route of dexmedetomidine.
Conflict of interest: No conflict declared by the authors.
Author contribution: STK - Concept, conduct of study, editing of manuscript
KM - Conduct of study
REFERENCES
- Gujarala I, Thipparampalli AK, Durga P, Gopinath R. Effect of perineural dexmedetomidine on the quality of supraclavicular brachial plexus block with 0.5% Ropivacaine and its interaction with general anaesthesia. Indian J Anaesth. 2015 Feb;59(2):89–95. doi: 10.4103/0019-5049.151369. [PubMed] [Free full text]
- Rashmi HD, Komala HK . Effect of Dexmedetomidine as an Adjuvant to 0.75% Ropivacaine in Interscalene Brachial Plexus Block Using Nerve Stimulator: A Prospective, Randomized Double Blind Study. Anaesth Essays Res. 2017 Jan–Mar;11(1):134-9. doi: 10.4103/0259-1162.181431 [PubMed] [Free full text]
- Hajashareef HM, Murugan T. Comparitive study of ropivacaine 0.5% and ropivacaine 0.5% with dexmedetomidine 50 μg in ultrasound guided supraclavicular brachial plexus block for upper limb orthopaedic surgery. Int J Sci Stud. 2017;4(12):169-75. [Free full text]
- Wu HH, Wang HT, Jin JJ, Cui GB, Zhou KC, Chen Y, et al. Does dexmedetomidine as a neuraxial adjuvant facilitate better anesthesia and analgesia? A systematic review and meta analysis. PLoS ONE. 2014 Mar 26;9(3):e93114. doi:10.1371/journal.pone.0093114. [PubMed] [Free full text]
- Bangera A, Manasa M, Krishna P. Comparison of effects of ropivacaine with and without dexmedetomidine in axillary brachial plexus block: A randomized double blinded clinical trial. Saudi J Anaesth. 2016 Jan-Mar;10(1):38–44. doi: 10.4103/1658-354X.169473 [PubMed] [Free full text]
- Marhofer D, Kettner SC, Marhofer P, Pils S, Weber M, Zeitlinger M. Dexmedetomidine as an adjuvant to ropivacaine prolongs peripheral nerve block : a volunteer study. Br J Anaesth. 2012 Mar;110(3):438-42. doi: 10.1093/bja/aes400 [PubMed] [Free full text]
- Gurudatt C L. Dexmedetomidine – the wonder drug, for the present and the future. RGUHS J Med Sciences. 2013 Jun;3(3):170- 6.
- Das A, Majumdar S, Haider S, Chattopadhyay S, Pal S, Kundu R, et al. Effect of Dexmedetomidine as an adjuvant in Ropivacaine – induced supraclavicular brachial plexus block: A prospective, double blinded and randomized controlled study. Saudi J Anaesth. 2014 Nov;8(Suppl 1):S72-7. doi: 10.4103/1658-354X.144082. [PubMed] [Free full text]
- Abdallah F W, Brull R. Facilitatory effects of perineural dexmedetomidine on neuarxial and peripheral nerve block : a systematic review and meta- analysis. Br J Anaesth. 2013 Jun;110(6):915-25. [PubMed] [Free full text]
- Kathuria S, Gupta S, Dhawan I. Dexmedetomidine as an adjuvant to ropivacaine in supraclavicular brachial plexus block. Saudi J Anaesth. 2015 Apr-Jun;9(2):148–54. doi: 10.4103/1658-354X.152841.[PubMed] [Free full text]
- Klein SM, Greengrass RA, Steele SM, D’Ercole FJ, Speer KP, Gleason DH, et al. A comparison of 0.5% bupivacaine, 0.5% ropivacaine and 0.75% ropivacaine for interscalene brachial plexus block. Anesth Analg. 1998 Dec;87(6):1316-9. [PubMed] .
- Hickey R, Rowley CL, Candido KD, Hoffman J, Ramamurthy S, Winnie AP. A comparative study of 0.25% ropivacaine and 0.25% bupivacaine for brachial plexus block. Anesth Analg. 1992 Oct;75(4):602-6. [PubMed]
- Vorobeichik L, Brull R, Abdallah FW. Evidence basis for using perineural dexmedetomidine to enhance the quality of brachial plexus nerve blocks : a systematic review and meta – analysis of randomized controlled trials. Br J Anaesth. 2017 Feb;118(2):167-81. doi: 10.1093/bja/aew411. [PubMed] [Free full text]
- Sudheesh K, Harsoor S. Dexmedetomidine in anaesthesia practice: A wonder drug?. Indian J Anaesth. 2011 Jul;55(4):323-4. doi: 10.4103/0019-5049.84824. [PubMed] [Free full text]
- Zhang Y, Wang C, Shi J, Sun B, Liu S, Li P, et al. Perineural administration of dexmedetomidine in combination with ropivacaine prolongs axillary brachial plexus block. Int J Clin Exp Med. 2014 Mar 15;7(3):680–5. [PubMed] [Free full text]
- Abdallah FW, Dwyer T, Chan VW, Niazi AU, Harris DJO, Oldfield S, et al. IV and Perineural Dexmedetomidine Similarly Prolong the Duration of Analgesia after Interscalene Brachial Plexus Block – A Randomized, Three arm, Triple Masked, Placebo Controlled Trial. Anesthesiology. 2016 Mar;124(3):683-95. doi: 10.1097/ALN.0000000000000983 [PubMed] [Free full text]