Samah Medhat Eid *1 , Amr Essam Eldin 2 , Ahmed Mohamed Khamis 3 ,
Mohamed Ibrahim Hassan Mohamed 4
Author affiliations:
Background & objective: Early detection of staple-line leak (SLL) after laparoscopic sleeve gastrectomy (LSG) is important for prompt intervention and to reduce the morbidity and mortality of this group of patients. We evaluated neutrophil-to-lymphocyte ratio (NLR) and C-reactive protein/albumin ratio (CAR) for early detection of staple-line leak (SLL) after laparoscopic sleeve gastrectomy (LSG) in high-risk patients.
Methodology: This prospective diagnostic study was carried out at Ain Shams University Hospitals, from Aug 2022 to Jul 2023. Fifty adult patients with a BMI >50 kg/m², ASA III, who had undergone LSG, had CBC, CRP, and albumin measured preoperatively and on postoperative days (POD) 1–3; NLR and CAR were calculated. To confirm the presence of SLL required two positive abdominal ultrasounds. ROC analyses estimated AUC, optimal cut-offs, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) , and accuracy.
Results: SLL occurred in 5/50 (10%), diagnosed at 11.8 ± 3.5 days. Baseline markers were similar (all P > 0.17). On POD1–3, CRP, CAR, and NLR were higher with SLL (all P < 0.001); albumin was lower on POD3 (P < 0.001). NLR performed best: POD1 cutoff ≥2.6—AUC 0.78, sensitivity 60%, specificity 100%, accuracy 96%; POD2 ≥3.1—AUC 0.94, sensitivity 80%, specificity 100%, accuracy 98%; POD3 ≥3.5—AUC 1.00 with 100% sensitivity, specificity, and accuracy. On POD3, CRP ≥8.1 (AUC 0.91) and CAR ≥2.16 (AUC 0.95) were inferior.
Conclusion: In high-risk LSG, serial NLR—especially by POD3—provides near-perfect discrimination of SLL and outperforms CRP and CAR. Routine NLR monitoring may expedite diagnosis and intervention.
Abbreviations: AUC: Area under curve, CRP: C-reactive protein, CAR: C-reactive protein/albumin ratio, CBC: Complete blood count, NLR: neutrophil-to-lymphocyte ratio, POD: Postoperative day, SLL: staple-line leak,
Keywords: Staple Line Leak, Neutrophil-to-Lymphocyte Ratio, Laparoscopic Sleeve Gastrectomy, CRP/Albumin Ratio, Bariatric Surgery.
Citation: Eid SM, Eldin AE, Khamis AM, Mohamed MIH. Diagnostic accuracy of CRP/albumin and neutrophil/lymphocyte ratios for early detection of staple line leak after laparoscopic sleeve gastrectomy in high-risk patients: a prospective observational study. Anaesth. pain intensive care 2026;30(2):232-239. DOI: 10.35975/apic.v30i2.3131
Received: May 09, 2025; Revised: October 26, 2025; Accepted: January 01, 2026
Obesity has evolved into a global health epidemic, with its prevalence escalating substantially over the past two decades. It is intrinsically linked to a broad spectrum of comorbidities, including hypertension, type 2 diabetes mellitus, cardiovascular disease, and obstructive sleep apnea, thereby imposing profound clinical challenges and socioeconomic costs. 1 Bariatric surgery has been established as the most efficacious and durable therapeutic modality for morbid obesity, achieving sustained weight reduction and marked amelioration of obesity-associated comorbid conditions. Among the surgical interventions, laparoscopic sleeve gastrectomy (LSG) has attained international acceptance owing to its technical feasibility, therapeutic effectiveness, and favorable safety profile.2
Despite its recognized benefits, LSG is associated with inherent risks, the most critical being staple line leak (SLL), which has been reported in 1% to 7% of cases. SLL represents a potentially fatal complication that, if not promptly diagnosed, may progress to peritonitis, sepsis, and ultimately multi-organ failure. Clinical indicators such as fever, tachycardia, and abdominal pain, though critical, are often nonspecific and tend to manifest late in the clinical course, delaying diagnosis and worsening outcomes. Radiological modalities, while useful, are not always practical for repeated monitoring in the immediate postoperative period. Consequently, there is a pressing need for simple, reliable, and cost-effective biomarkers that can facilitate early detection of SLL, especially in high-risk bariatric cases.3
In recent years, systemic inflammatory markers have gained attention as prognostic tools in surgical cases. Among them, the neutrophil-to-lymphocyte ratio (NLR) has emerged as a readily available index of systemic inflammatory response, reflecting the balance between neutrophil-mediated inflammation and lymphocyte-mediated regulation. Similarly, the C-reactive protein (CRP)/albumin ratio (CAR) integrates both positive and negative acute-phase reactants, offering a more comprehensive assessment of inflammatory status than either marker alone. Both NLR and CAR have shown predictive value in oncological, cardiovascular, and critical care settings, yet their role in the early detection of staple line complications following LSG remains underexplored.4
Given the high morbidity associated with delayed diagnosis of SLL, particularly in severe obesity cases (BMI >50 kg/m²) and ASA class III status, evaluating novel, easily accessible biomarkers is of paramount clinical relevance. Therefore, this study aims to investigate the diagnostic accuracy of postoperative NLR and CAR in predicting SLL after LSG in high-risk cases.
This prospective observational diagnostic accuracy study was undertaken in the Department of Anaesthesiology, Intensive Care, and Pain Management at Ain Shams University Hospitals, Cairo, Egypt, during a 12-month interval extending from August 2022 to July 2023.
Eligible participants comprised adult cases of both sexes, aged 18–45 years, with a BMI exceeding 50 kg/m², classified as ASA physical status III, and scheduled to undergo elective LSG under general anesthesia. Exclusion criteria encompassed refusal to participate, use of therapeutic anticoagulants, acute coronary syndrome, pulmonary edema, severe renal or hepatic disease, immunosuppressive or hormonal therapy, malignancy, previous multi-visceral resections, and presence of active infection.
Sample size estimation was performed using NCSS PASS software version 11.0, based on data from Romano et al. which reported an AUC of 0.724 for inflammatory markers in predicting SLL.5 Assuming a null hypothesis AUC of 0.5, a sample of 50 cases achieved 82% power to detect a significant difference at α = 0.05.
Induction of anesthesia was standardized across cases, employing propofol (1–2 mg/kg), fentanyl (1 μg/kg), and atracurium (0.5 mg/kg), followed by endotracheal intubation. Maintenance consisted of 1–2% isoflurane in a 50% oxygen/air mixture with additional atracurium administered intermittently. Postoperative care included routine admission to the surgical intensive care unit for monitoring.
Baseline venous sampling was performed preoperatively, followed by collections on postoperative days 1, 2, and 3. Laboratory measurements included complete blood count with differential, CRP, and serum albumin levels. NLR and CAR were subsequently calculated. Abdominal ultrasonography was performed routinely and repeated whenever leakage was clinically suspected (fever, tachycardia, or abdominal tenderness). SLL was confirmed by two positive ultrasound examinations. Cases were stratified into two groups: leak and no-leak. The primary endpoint was the diagnostic accuracy of NLR and CAR for predicting SLL after LSG, expressed as AUC of the ROC curve. Secondary endpoints included optimal cutoff values, sensitivity, specificity, PPV, NPV, and diagnostic accuracy of CRP, albumin, and derived ratios.
2.1. Statistical methods
All statistical analyses were conducted using IBM SPSS Statistics version 28.0 (IBM Corp., Chicago, IL, USA). The Shapiro–Wilk test was used to assess normality of quantitative data, which were expressed as mean ± SD and compared using either independent-samples t-test or repeated measures ANOVA with Dunnett’s post hoc test. Categorical data were presented as frequencies and percentages and analyzed using Fisher’s exact test. ROC curve analysis was applied to examine the diagnostic performance of NLR, CRP, and CAR, with calculation of sensitivity, specificity, PPV, NPV, overall accuracy, and Youden’s index. Statistical significance was defined as a two-sided p-value less than 0.05.
Within this investigation, the incidence of SLL was 10% (5/50) at a mean time of 11.8 ± 3.5 days. Age (P = 0.43), gender distribution (P = 0.595), BMI (P = 0.256), and operation duration (P = 0.247) did not markedly differ between cases with and without SLL (Table 1).
Baseline values of CRP (P = 0.213), serum albumin (P = 0.705), CAR (P = 0.209), and NLR (P = 0.172) did not differ substantially between cases with and without SLL. However, postoperative values on days 1, 2, and 3 showed significant differences between groups for CRP (all P < 0.001), CAR (all P < 0.001), and NLR (all P < 0.001), as well as for serum albumin on day 3 (P < 0.001). Moreover, repeated-measures analysis demonstrated that the postoperative changes in CRP, serum albumin, CAR, and NLR were all significantly different from their respective baseline values (all §P < 0.001) (Table 2).
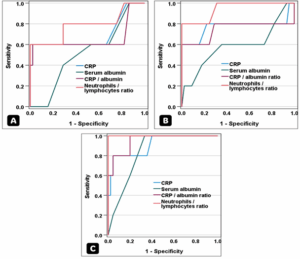
The NLR with a cutoff ≥ 2.6 demonstrated the highest significant diagnostic performance on day 1 for predicting SLL, followed by CRP and the CRP/albumin ratio. On day 2, NLR with a cutoff ≥3.1 again showed the best diagnostic performance, followed by CRP and the CRP/albumin ratio. By day 3, NLR with a cutoff ≥3.5 achieved perfect diagnostic accuracy, outperforming CRP and the CRP/albumin ratio (Table 3, Figure 1).
Following LSG, SLL is linked to higher morbidity, mortality, and extended hospitalization, with incidence rates in the literature ranging from 0% to 7% 6, 7. In our study, the incidence was 10% (5/50) among high-risk cases (BMI > 50 kg/m², ASA III).
SLL diagnosis after LSG is traditionally based on clinical manifestations such as abdominal pain, fever, and tachycardia, along with leukocytosis and radiological confirmation using contrast studies or CT scans. However, these conventional methods may delay diagnosis, as early clinical signs are often subtle and nonspecific 8, 9. Aurora et al. 10, in a systematic review of 4,888 cases, reported that 50% of leaks occurred after postoperative day 10. In our study, the mean time to diagnosis was 11.8 ± 3.5 days, underscoring the potential for late clinical presentation.
Hence, an effective biochemical marker is crucial to facilitate earlier detection of leakage before overt clinical deterioration, and in this context, in this prospective diagnostic accuracy study, we evaluated the utility of postoperative inflammatory markers—namely the NLR, CRP, and CAR—for early detection of SLL following LSG in high-risk bariatric cases. Our findings demonstrated that NLR, particularly on postoperative day 3, exhibited superior diagnostic accuracy compared with CRP and CAR, achieving an AUC of 1.0 with 100% sensitivity and specificity at a cutoff value of ≥3.5. CRP and CAR also showed predictive ability but with lower diagnostic accuracy and specificity.
Previous research has consistently highlighted CRP as an important marker of postoperative septic complications, particularly after colorectal and upper gastrointestinal surgery 11, 12. In the context of LSG, several authors have noted that a sustained postoperative rise in CRP often precedes clinical or radiological evidence of leakage, making it a potentially valuable early warning sign. For instance, a persistently elevated CRP beyond postoperative day 3 has been suggested as a threshold to prompt further imaging for suspected sepsis.
Albanopoulos et al. (2013) demonstrated that CRP achieved 100% sensitivity and specificity on day 3 and maintained high diagnostic value on day 5, while Kassir et al. (2015) reported markedly higher mean CRP levels in cases with complications relative to those without, although they argued that its predictive power diminished at the individual level. Ruiz-Tovar et al. ((2025).13-15 , in a larger cohort (208 cases), also found that CRP at 24 h postoperatively had good discriminatory capacity for SLL, with 85% sensitivity and 90% specificity. In contrast, our study confirmed CRP as a useful indicator but with lower diagnostic performance (AUC 0.91, sensitivity 80%, specificity 95.6% on day 3), and importantly, it was clearly outperformed by NLR, which achieved perfect accuracy in identifying leaks.
The CAR has recently emerged as a composite biomarker reflecting both systemic inflammation and nutritional status, and has been increasingly studied across various surgical and medical settings. Yang et al. 15 analyzed 20,733 cases from the NHANES database, including 1,744 with rheumatoid arthritis, and demonstrated that higher CAR was substantially correlated with disease incidence, with an AUC of 0.749 for their prediction model. In a meta-analysis of nine studies including 2,954 stroke cases, Yang et al. 16 further confirmed that elevated CAR was independently correlated with poor functional outcomes (OR 2.24) and mortality (OR 2.11). Onder et al. 17 evaluated 177 cases with rheumatoid arthritis and identified a CAR cutoff of 2.67 for early RA, achieving 80% sensitivity and 85% specificity, thus supporting its use as a marker of disease activity. In pediatric practice, Khedr et al. 18 studied 154 preterm neonates and showed that a CAR >1.5 predicted neonatal sepsis with 100% sensitivity and 95% specificity (AUC 0.99). In gastrointestinal surgery, Zhao et al. 19 reported on 534 cases undergoing surgery for Crohn’s disease and found that a postoperative CAR ≥3.25 was strongly predictive of complications (OR 13.2, AUC 0.77), outperforming CRP alone. Similarly, Lee et al. 20 examined 128 gastric cancer cases and showed that preoperative CAR independently predicted postoperative morbidity, with a cutoff of 0.265 significantly associated with complications (HR 2.83). Finally, in a recent meta-analysis of 2,020 gastrointestinal surgery cases, Shaukat et al. 21 demonstrated that both CRP and CAR had strong predictive value for infectious complications (pooled OR 2.43), reinforcing their clinical utility. In our high-risk LSG cohort, CAR was substantially higher in SLL cases relative to those without (2.78 vs. 1.55 on POD3 [post operative day], P < 0.001) and achieved good diagnostic accuracy (AUC 0.95, 80% sensitivity, 95.6% specificity). However, although CAR outperformed CRP alone, it was still inferior to NLR, which reached perfect accuracy in our series.
The NLR, a simple index derived from the differential WBC count, has gained attention as a reliable marker of systemic inflammation. Neutrophilia reflects the acute inflammatory response to tissue injury, while postoperative lymphocytopenia is often linked to cortisol release and neuroendocrine stress; therefore, NLR captures the balance between these opposing immune pathways, simultaneously indicating heightened inflammation and suppressed immune regulation 4, 22, 23. Previous studies have suggested that persistently elevated NLR values (>7–8) should raise suspicion of postoperative infectious complications, as values in uncomplicated cases typically remain lower 24. In gastrointestinal surgery, Mohamed et al. 25 showed that an NLR cutoff of 5.01 on POD1 predicted early complications after intestinal obstruction surgery with an AUC of 0.873, 84.6% sensitivity, and 78.4% specificity, underscoring its reliability as a general surgical biomarker.
In the bariatric setting, Romano et al. 5 prospectively demonstrated in 151 LSG cases that NLR achieved perfect diagnostic accuracy (AUC = 1.0) for predicting leak, clearly outperforming CRP, fibrinogen, and WBC counts. Da Silva et al. 26 retrospectively analyzed 737 bariatric cases and demonstrated that an elevated POD1 NLR was independently correlated with higher rates of 30-day complications, reoperation, and prolonged hospital stay. Consistent with these observations, our findings confirm that NLR is superior to CRP and CAR, reaching an AUC of 1.0 with 100% sensitivity and specificity by POD3 in high-risk LSG cases.
Despite these strengths, the study has limitations. First, the sample size was modest, and only five cases of SLL were observed, which may limit generalizability and increase the risk of overestimating diagnostic accuracy. Second, the diagnosis of SLL relied primarily on ultrasonographic findings, which, although non-invasive and repeatable, may lack the sensitivity of CT with oral contrast—the current gold standard. Third, the study focused exclusively on high-risk cases (BMI >50 kg/m², ASA III), and results may not be directly extrapolated to lower-risk populations. Finally, while NLR and CAR were predictive, we did not evaluate other promising biomarkers such as procalcitonin or presepsin, which may offer complementary diagnostic value.
NLR, particularly on day 3, demonstrated perfect diagnostic accuracy for SLL after LSG, outperforming CRP and CAR. Routine NLR monitoring may enable timely intervention and improved outcomes in high-risk cases.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Ethical considerations
The protocol received approval from the Research Ethics Committee (REC) of the Faculty of Medicine, Ain Shams University (Approval No. FMASU MD305/2022) and prospectively registered with the Pan African Clinical Trial Registry (PACTR202507562026819).
Owing to its observational nature, the requirement for informed consent was waived.
11. Authors’ contribution
SME: Concept, study design, data collection, manuscript drafting and editing.
AEE: Literature search, methodology development, critical review, and manuscript editing.
AMK: Statistical analysis, interpretation of data, and manuscript preparation.
MIHM: Data collection, clinical support, and contribution to discussion writing
Mohamed Ibrahim Hassan Mohamed 4
Author affiliations:
- Samah Medhat Eid, Assistant Lecturer of Anesthesia, Ain Shams University, Cairo, Egypt; Email: dr.omadam3216@gmail.com 2. Amr Essam Eldin, Professor of Anesthesia, Ain Shams University, Cairo, Egypt; Email: Email: Amresam87@gmail.com
- Ahmed Mohamed Khamis, MD, Email: Khamisba2020@gmail.com; ORCID ID: https://orcid.org/0000-0002-1665-0628
- Mohamed Ibrahim Hassan Mohamed, Ain Shams University Hospitals, Cairo, Egypt; Email: T.majestic@hotmail.com
ABSTRACT
Background & objective: Early detection of staple-line leak (SLL) after laparoscopic sleeve gastrectomy (LSG) is important for prompt intervention and to reduce the morbidity and mortality of this group of patients. We evaluated neutrophil-to-lymphocyte ratio (NLR) and C-reactive protein/albumin ratio (CAR) for early detection of staple-line leak (SLL) after laparoscopic sleeve gastrectomy (LSG) in high-risk patients.
Methodology: This prospective diagnostic study was carried out at Ain Shams University Hospitals, from Aug 2022 to Jul 2023. Fifty adult patients with a BMI >50 kg/m², ASA III, who had undergone LSG, had CBC, CRP, and albumin measured preoperatively and on postoperative days (POD) 1–3; NLR and CAR were calculated. To confirm the presence of SLL required two positive abdominal ultrasounds. ROC analyses estimated AUC, optimal cut-offs, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) , and accuracy.
Results: SLL occurred in 5/50 (10%), diagnosed at 11.8 ± 3.5 days. Baseline markers were similar (all P > 0.17). On POD1–3, CRP, CAR, and NLR were higher with SLL (all P < 0.001); albumin was lower on POD3 (P < 0.001). NLR performed best: POD1 cutoff ≥2.6—AUC 0.78, sensitivity 60%, specificity 100%, accuracy 96%; POD2 ≥3.1—AUC 0.94, sensitivity 80%, specificity 100%, accuracy 98%; POD3 ≥3.5—AUC 1.00 with 100% sensitivity, specificity, and accuracy. On POD3, CRP ≥8.1 (AUC 0.91) and CAR ≥2.16 (AUC 0.95) were inferior.
Conclusion: In high-risk LSG, serial NLR—especially by POD3—provides near-perfect discrimination of SLL and outperforms CRP and CAR. Routine NLR monitoring may expedite diagnosis and intervention.
Abbreviations: AUC: Area under curve, CRP: C-reactive protein, CAR: C-reactive protein/albumin ratio, CBC: Complete blood count, NLR: neutrophil-to-lymphocyte ratio, POD: Postoperative day, SLL: staple-line leak,
Keywords: Staple Line Leak, Neutrophil-to-Lymphocyte Ratio, Laparoscopic Sleeve Gastrectomy, CRP/Albumin Ratio, Bariatric Surgery.
Citation: Eid SM, Eldin AE, Khamis AM, Mohamed MIH. Diagnostic accuracy of CRP/albumin and neutrophil/lymphocyte ratios for early detection of staple line leak after laparoscopic sleeve gastrectomy in high-risk patients: a prospective observational study. Anaesth. pain intensive care 2026;30(2):232-239. DOI: 10.35975/apic.v30i2.3131
Received: May 09, 2025; Revised: October 26, 2025; Accepted: January 01, 2026
1. INTRODUCTION
Obesity has evolved into a global health epidemic, with its prevalence escalating substantially over the past two decades. It is intrinsically linked to a broad spectrum of comorbidities, including hypertension, type 2 diabetes mellitus, cardiovascular disease, and obstructive sleep apnea, thereby imposing profound clinical challenges and socioeconomic costs. 1 Bariatric surgery has been established as the most efficacious and durable therapeutic modality for morbid obesity, achieving sustained weight reduction and marked amelioration of obesity-associated comorbid conditions. Among the surgical interventions, laparoscopic sleeve gastrectomy (LSG) has attained international acceptance owing to its technical feasibility, therapeutic effectiveness, and favorable safety profile.2
Despite its recognized benefits, LSG is associated with inherent risks, the most critical being staple line leak (SLL), which has been reported in 1% to 7% of cases. SLL represents a potentially fatal complication that, if not promptly diagnosed, may progress to peritonitis, sepsis, and ultimately multi-organ failure. Clinical indicators such as fever, tachycardia, and abdominal pain, though critical, are often nonspecific and tend to manifest late in the clinical course, delaying diagnosis and worsening outcomes. Radiological modalities, while useful, are not always practical for repeated monitoring in the immediate postoperative period. Consequently, there is a pressing need for simple, reliable, and cost-effective biomarkers that can facilitate early detection of SLL, especially in high-risk bariatric cases.3
In recent years, systemic inflammatory markers have gained attention as prognostic tools in surgical cases. Among them, the neutrophil-to-lymphocyte ratio (NLR) has emerged as a readily available index of systemic inflammatory response, reflecting the balance between neutrophil-mediated inflammation and lymphocyte-mediated regulation. Similarly, the C-reactive protein (CRP)/albumin ratio (CAR) integrates both positive and negative acute-phase reactants, offering a more comprehensive assessment of inflammatory status than either marker alone. Both NLR and CAR have shown predictive value in oncological, cardiovascular, and critical care settings, yet their role in the early detection of staple line complications following LSG remains underexplored.4
Given the high morbidity associated with delayed diagnosis of SLL, particularly in severe obesity cases (BMI >50 kg/m²) and ASA class III status, evaluating novel, easily accessible biomarkers is of paramount clinical relevance. Therefore, this study aims to investigate the diagnostic accuracy of postoperative NLR and CAR in predicting SLL after LSG in high-risk cases.
2. METHODOLOGY
This prospective observational diagnostic accuracy study was undertaken in the Department of Anaesthesiology, Intensive Care, and Pain Management at Ain Shams University Hospitals, Cairo, Egypt, during a 12-month interval extending from August 2022 to July 2023.
Eligible participants comprised adult cases of both sexes, aged 18–45 years, with a BMI exceeding 50 kg/m², classified as ASA physical status III, and scheduled to undergo elective LSG under general anesthesia. Exclusion criteria encompassed refusal to participate, use of therapeutic anticoagulants, acute coronary syndrome, pulmonary edema, severe renal or hepatic disease, immunosuppressive or hormonal therapy, malignancy, previous multi-visceral resections, and presence of active infection.
Sample size estimation was performed using NCSS PASS software version 11.0, based on data from Romano et al. which reported an AUC of 0.724 for inflammatory markers in predicting SLL.5 Assuming a null hypothesis AUC of 0.5, a sample of 50 cases achieved 82% power to detect a significant difference at α = 0.05.
Induction of anesthesia was standardized across cases, employing propofol (1–2 mg/kg), fentanyl (1 μg/kg), and atracurium (0.5 mg/kg), followed by endotracheal intubation. Maintenance consisted of 1–2% isoflurane in a 50% oxygen/air mixture with additional atracurium administered intermittently. Postoperative care included routine admission to the surgical intensive care unit for monitoring.
Baseline venous sampling was performed preoperatively, followed by collections on postoperative days 1, 2, and 3. Laboratory measurements included complete blood count with differential, CRP, and serum albumin levels. NLR and CAR were subsequently calculated. Abdominal ultrasonography was performed routinely and repeated whenever leakage was clinically suspected (fever, tachycardia, or abdominal tenderness). SLL was confirmed by two positive ultrasound examinations. Cases were stratified into two groups: leak and no-leak. The primary endpoint was the diagnostic accuracy of NLR and CAR for predicting SLL after LSG, expressed as AUC of the ROC curve. Secondary endpoints included optimal cutoff values, sensitivity, specificity, PPV, NPV, and diagnostic accuracy of CRP, albumin, and derived ratios.
2.1. Statistical methods
All statistical analyses were conducted using IBM SPSS Statistics version 28.0 (IBM Corp., Chicago, IL, USA). The Shapiro–Wilk test was used to assess normality of quantitative data, which were expressed as mean ± SD and compared using either independent-samples t-test or repeated measures ANOVA with Dunnett’s post hoc test. Categorical data were presented as frequencies and percentages and analyzed using Fisher’s exact test. ROC curve analysis was applied to examine the diagnostic performance of NLR, CRP, and CAR, with calculation of sensitivity, specificity, PPV, NPV, overall accuracy, and Youden’s index. Statistical significance was defined as a two-sided p-value less than 0.05.
3. RESULTS
Within this investigation, the incidence of SLL was 10% (5/50) at a mean time of 11.8 ± 3.5 days. Age (P = 0.43), gender distribution (P = 0.595), BMI (P = 0.256), and operation duration (P = 0.247) did not markedly differ between cases with and without SLL (Table 1).
| Table 1: Demographic characteristics among studied cases
according to SLL |
||||
| Parameters | Total
(n = 50) |
Staple line leak | P-value | |
| Positive
(n = 5) |
Negative
(n = 45) |
|||
| Age (years) | 30.3 ± 5.5 | 28.4 ± 2.6 | 30.5 ± 5.8 | 0.43 |
| Gender | ||||
| · Male | 13 (26.0) | 2 (40.0) | 11 (24.4) | 0.595 |
| · Female | 37 (74.0) | 3 (60.0) | 34 (75.6) | |
| BMI (kg/m²) | 48.6 ± 4.4 | 46.5 ± 4.7 | 48.8 ± 4.3 | 0.256 |
| Operation duration
(minutes) |
54.2 ± 6.9 | 57.6 ± 7.7 | 53.8 ± 6.8 | 0.247 |
| Data presented as mean ± SD or n (%); P < 0.05 considered as significant | ||||
Baseline values of CRP (P = 0.213), serum albumin (P = 0.705), CAR (P = 0.209), and NLR (P = 0.172) did not differ substantially between cases with and without SLL. However, postoperative values on days 1, 2, and 3 showed significant differences between groups for CRP (all P < 0.001), CAR (all P < 0.001), and NLR (all P < 0.001), as well as for serum albumin on day 3 (P < 0.001). Moreover, repeated-measures analysis demonstrated that the postoperative changes in CRP, serum albumin, CAR, and NLR were all significantly different from their respective baseline values (all §P < 0.001) (Table 2).
| Table 2: Laboratory findings among studied cases according to SLL | ||||
| Laboratory Tests | Total (n = 50) | Staple line leak | ^P-value | |
| Positive (n = 5) | Negative (n = 45) | |||
| CRP (mg/L) | ||||
| Baseline | 0.9 ± 0.2 | 1.0 ± 0.1 | 0.9 ± 0.2 | 0.213 |
| Day-1 | 5.3 ± 0.8◊ | 6.8 ± 1.9◊ | 5.2 ± 0.3◊ | < 0.001* |
| Day-2 | 7.0 ± 0.9◊ | 8.3 ± 2.2◊ | 6.9 ± 0.5◊ | < 0.001* |
| Day-3 | 6.6 ± 1.9◊ | 10.3 ± 3.4◊ | 6.2 ± 1.1◊ | < 0.001* |
| §P-value | < 0.001* | < 0.001* | < 0.001* | |
| Serum albumin (g/dL) | ||||
| Baseline | 4.4 ± 0.2 | 4.4 ± 0.2 | 4.4 ± 0.2 | 0.705 |
| Day-1 | 4.2 ± 0.2◊ | 4.1 ± 0.2◊ | 4.2 ± 0.2◊ | 0.882 |
| Day-2 | 4.0 ± 0.2◊ | 3.9 ± 0.3◊ | 4.0 ± 0.2◊ | 0.333 |
| Day-3 | 4.0 ± 0.2◊ | 3.7 ± 0.1◊ | 4.0 ± 0.2◊ | < 0.001* |
| §P-value | < 0.001* | < 0.001* | < 0.001* | |
| CAR | ||||
| Baseline | 0.21 ± 0.04 | 0.23 ± 0.03 | 0.21 ± 0.04 | 0.209 |
| Day-1 | 1.29 ± 0.21◊ | 1.65 ± 0.50◊ | 1.25 ± 0.10◊ | < 0.001* |
| Day-2 | 1.75 ± 0.26◊ | 2.13 ± 0.64◊ | 1.71 ± 0.14◊ | < 0.001* |
| Day-3 | 1.67 ± 0.51◊ | 2.78 ± 0.90◊ | 1.55 ± 0.26◊ | < 0.001* |
| §P-value | < 0.001* | < 0.001* | < 0.001* | |
| NLR | ||||
| Baseline | 1.86 ± 0.09 | 1.80 ± 0.07 | 1.86 ± 0.09 | 0.172 |
| Day-1 | 2.14 ± 0.25◊ | 2.65 ± 0.53◊ | 2.09 ± 0.11◊ | < 0.001* |
| Day-2 | 2.18 ± 0.53◊ | 3.55 ± 0.82◊ | 2.02 ± 0.11◊ | < 0.001* |
| Day-3 | 2.25 ± 0.97◊ | 5.07 ± 0.46◊ | 1.93 ± 0.11◊ | < 0.001* |
| §P-value | < 0.001* | < 0.001* | < 0.001* | |
| CRP: C-reactive protein, NLR: Neutrophil-to-lymphocyte ratio, CAR: CRP/albumin ratio, ^: Independent t-test, §: Repeated-measures ANOVA (RMANOVA), RMANOVA: Repeated-measures analysis of variance, ◊: Time point significantly different from baseline, *Significant P-value. | ||||
The NLR with a cutoff ≥ 2.6 demonstrated the highest significant diagnostic performance on day 1 for predicting SLL, followed by CRP and the CRP/albumin ratio. On day 2, NLR with a cutoff ≥3.1 again showed the best diagnostic performance, followed by CRP and the CRP/albumin ratio. By day 3, NLR with a cutoff ≥3.5 achieved perfect diagnostic accuracy, outperforming CRP and the CRP/albumin ratio (Table 3, Figure 1).
| Table 3: Diagnostic performance and postoperative laboratory findings to predict SLE | ||||
| Characteristics | CRP | Serum albumin | CAR | NLR |
| Day 1 | ||||
| AUC (95% CI) | 0.693 (0.366 - 1) | 0.518 (0.271 - 0.764) | 0.658 (0.295 - 1) | 0.784 (0.510 - 1) |
| Cut point | ≥6.1 | ≤4.3 | ≥1.53 | ≥2.6 |
| Sensitivity | 60.00% | 60.00% | 60.00% | 60.00% |
| Specificity | 97.80% | 46.70% | 97.80% | 100.00% |
| PPV | 75.00% | 11.10% | 75.00% | 100.00% |
| NPV | 95.70% | 91.30% | 95.70% | 95.70% |
| Diagnostic accuracy | 94.00% | 48.00% | 94.00% | 96.00% |
| Youden’s Index | 57.80% | 6.70% | 57.80% | 60.00% |
| P-value | 0.16 | 0.897 | 0.251 | 0.038* |
| Day 2 | ||||
| AUC (95% CI) | 0.773 (0.450 - 1) | 0.587 (0.271 - 0.903) | 0.747 (0.405 - 1) | 0.944 (0.841 - 1) |
| Cut point | ≥7.6 | ≤4.2 | ≥1.98 | ≥3.1 |
| Sensitivity | 60.00% | 60.00% | 60.00% | 80.00% |
| Specificity | 100.00% | 64.40% | 100.00% | 100.00% |
| PPV | 100.00% | 15.80% | 100.00% | 100.00% |
| NPV | 95.70% | 93.50% | 95.70% | 97.80% |
| Diagnostic accuracy | 96.00% | 64.00% | 96.00% | 98.00% |
| Youden’s Index | 60.00% | 24.40% | 60.00% | 80.00% |
| P-value | 0.047* | 0.528 | 0.073 | 0.001* |
| Day 3 | ||||
| AUC (95% CI) | 0.911 (0.775 - 1) | 0.840 (0.717 - 0.963) | 0.951 (0.873 - 1) | 1 (1 - 1) |
| Cut point | ≥8.1 | ≤3.8 | ≥2.16 | ≥3.5 |
| Sensitivity | 80.00% | 100.00% | 80.00% | 100.00% |
| Specificity | 95.60% | 66.70% | 95.60% | 100.00% |
| PPV | 66.70% | 25.00% | 66.70% | 100.00% |
| NPV | 97.70% | 100.00% | 97.70% | 100.00% |
| Diagnostic accuracy | 94.00% | 70.00% | 94.00% | 100.00% |
| Youden’s Index | 75.60% | 66.70% | 75.60% | 100.00% |
| P-value | 0.003* | 0.013* | 0.001* | < 0.001* |
| CRP: C-reactive protein, AUC: Area under the curve, CAR: CRP/albumin ratio, CI: Confidence interval, PPV: Positive predictive value, NLR: Neutrophil-to-lymphocyte ratio, NPV: Negative predictive value, SLE: Staple line leak, *: Significant P-value. | ||||

Figure 1: ROC curves showing the diagnostic performance of CRP, serum albumin, CAR, and NLR in predicting SLE on A) postoperative day 1, B) postoperative day 2, and C) postoperative day 3
4. DISCUSSION
Following LSG, SLL is linked to higher morbidity, mortality, and extended hospitalization, with incidence rates in the literature ranging from 0% to 7% 6, 7. In our study, the incidence was 10% (5/50) among high-risk cases (BMI > 50 kg/m², ASA III).
SLL diagnosis after LSG is traditionally based on clinical manifestations such as abdominal pain, fever, and tachycardia, along with leukocytosis and radiological confirmation using contrast studies or CT scans. However, these conventional methods may delay diagnosis, as early clinical signs are often subtle and nonspecific 8, 9. Aurora et al. 10, in a systematic review of 4,888 cases, reported that 50% of leaks occurred after postoperative day 10. In our study, the mean time to diagnosis was 11.8 ± 3.5 days, underscoring the potential for late clinical presentation.
Hence, an effective biochemical marker is crucial to facilitate earlier detection of leakage before overt clinical deterioration, and in this context, in this prospective diagnostic accuracy study, we evaluated the utility of postoperative inflammatory markers—namely the NLR, CRP, and CAR—for early detection of SLL following LSG in high-risk bariatric cases. Our findings demonstrated that NLR, particularly on postoperative day 3, exhibited superior diagnostic accuracy compared with CRP and CAR, achieving an AUC of 1.0 with 100% sensitivity and specificity at a cutoff value of ≥3.5. CRP and CAR also showed predictive ability but with lower diagnostic accuracy and specificity.
Previous research has consistently highlighted CRP as an important marker of postoperative septic complications, particularly after colorectal and upper gastrointestinal surgery 11, 12. In the context of LSG, several authors have noted that a sustained postoperative rise in CRP often precedes clinical or radiological evidence of leakage, making it a potentially valuable early warning sign. For instance, a persistently elevated CRP beyond postoperative day 3 has been suggested as a threshold to prompt further imaging for suspected sepsis.
Albanopoulos et al. (2013) demonstrated that CRP achieved 100% sensitivity and specificity on day 3 and maintained high diagnostic value on day 5, while Kassir et al. (2015) reported markedly higher mean CRP levels in cases with complications relative to those without, although they argued that its predictive power diminished at the individual level. Ruiz-Tovar et al. ((2025).13-15 , in a larger cohort (208 cases), also found that CRP at 24 h postoperatively had good discriminatory capacity for SLL, with 85% sensitivity and 90% specificity. In contrast, our study confirmed CRP as a useful indicator but with lower diagnostic performance (AUC 0.91, sensitivity 80%, specificity 95.6% on day 3), and importantly, it was clearly outperformed by NLR, which achieved perfect accuracy in identifying leaks.
The CAR has recently emerged as a composite biomarker reflecting both systemic inflammation and nutritional status, and has been increasingly studied across various surgical and medical settings. Yang et al. 15 analyzed 20,733 cases from the NHANES database, including 1,744 with rheumatoid arthritis, and demonstrated that higher CAR was substantially correlated with disease incidence, with an AUC of 0.749 for their prediction model. In a meta-analysis of nine studies including 2,954 stroke cases, Yang et al. 16 further confirmed that elevated CAR was independently correlated with poor functional outcomes (OR 2.24) and mortality (OR 2.11). Onder et al. 17 evaluated 177 cases with rheumatoid arthritis and identified a CAR cutoff of 2.67 for early RA, achieving 80% sensitivity and 85% specificity, thus supporting its use as a marker of disease activity. In pediatric practice, Khedr et al. 18 studied 154 preterm neonates and showed that a CAR >1.5 predicted neonatal sepsis with 100% sensitivity and 95% specificity (AUC 0.99). In gastrointestinal surgery, Zhao et al. 19 reported on 534 cases undergoing surgery for Crohn’s disease and found that a postoperative CAR ≥3.25 was strongly predictive of complications (OR 13.2, AUC 0.77), outperforming CRP alone. Similarly, Lee et al. 20 examined 128 gastric cancer cases and showed that preoperative CAR independently predicted postoperative morbidity, with a cutoff of 0.265 significantly associated with complications (HR 2.83). Finally, in a recent meta-analysis of 2,020 gastrointestinal surgery cases, Shaukat et al. 21 demonstrated that both CRP and CAR had strong predictive value for infectious complications (pooled OR 2.43), reinforcing their clinical utility. In our high-risk LSG cohort, CAR was substantially higher in SLL cases relative to those without (2.78 vs. 1.55 on POD3 [post operative day], P < 0.001) and achieved good diagnostic accuracy (AUC 0.95, 80% sensitivity, 95.6% specificity). However, although CAR outperformed CRP alone, it was still inferior to NLR, which reached perfect accuracy in our series.
The NLR, a simple index derived from the differential WBC count, has gained attention as a reliable marker of systemic inflammation. Neutrophilia reflects the acute inflammatory response to tissue injury, while postoperative lymphocytopenia is often linked to cortisol release and neuroendocrine stress; therefore, NLR captures the balance between these opposing immune pathways, simultaneously indicating heightened inflammation and suppressed immune regulation 4, 22, 23. Previous studies have suggested that persistently elevated NLR values (>7–8) should raise suspicion of postoperative infectious complications, as values in uncomplicated cases typically remain lower 24. In gastrointestinal surgery, Mohamed et al. 25 showed that an NLR cutoff of 5.01 on POD1 predicted early complications after intestinal obstruction surgery with an AUC of 0.873, 84.6% sensitivity, and 78.4% specificity, underscoring its reliability as a general surgical biomarker.
In the bariatric setting, Romano et al. 5 prospectively demonstrated in 151 LSG cases that NLR achieved perfect diagnostic accuracy (AUC = 1.0) for predicting leak, clearly outperforming CRP, fibrinogen, and WBC counts. Da Silva et al. 26 retrospectively analyzed 737 bariatric cases and demonstrated that an elevated POD1 NLR was independently correlated with higher rates of 30-day complications, reoperation, and prolonged hospital stay. Consistent with these observations, our findings confirm that NLR is superior to CRP and CAR, reaching an AUC of 1.0 with 100% sensitivity and specificity by POD3 in high-risk LSG cases.
5. LIMITATIONS
Despite these strengths, the study has limitations. First, the sample size was modest, and only five cases of SLL were observed, which may limit generalizability and increase the risk of overestimating diagnostic accuracy. Second, the diagnosis of SLL relied primarily on ultrasonographic findings, which, although non-invasive and repeatable, may lack the sensitivity of CT with oral contrast—the current gold standard. Third, the study focused exclusively on high-risk cases (BMI >50 kg/m², ASA III), and results may not be directly extrapolated to lower-risk populations. Finally, while NLR and CAR were predictive, we did not evaluate other promising biomarkers such as procalcitonin or presepsin, which may offer complementary diagnostic value.
6. CONCLUSIONS
NLR, particularly on day 3, demonstrated perfect diagnostic accuracy for SLL after LSG, outperforming CRP and CAR. Routine NLR monitoring may enable timely intervention and improved outcomes in high-risk cases.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Ethical considerations
The protocol received approval from the Research Ethics Committee (REC) of the Faculty of Medicine, Ain Shams University (Approval No. FMASU MD305/2022) and prospectively registered with the Pan African Clinical Trial Registry (PACTR202507562026819).
Owing to its observational nature, the requirement for informed consent was waived.
11. Authors’ contribution
SME: Concept, study design, data collection, manuscript drafting and editing.
AEE: Literature search, methodology development, critical review, and manuscript editing.
AMK: Statistical analysis, interpretation of data, and manuscript preparation.
MIHM: Data collection, clinical support, and contribution to discussion writing
12. REFERENCES
- Shariq OA, McKenzie TJ. Obesity-related hypertension: a review of pathophysiology, management, and the role of metabolic surgery. Gland Surg. 2020;9(1):80-93.
- 2Aderinto N, Olatunji G, Kokori E, Olaniyi P, Isarinade T, Yusuf IA. Recent advances in bariatric surgery: a narrative review of weight loss procedures. Ann Med Surg (Lond). 2023;85(12):6091-104.
- Mukhtar R, Abduljabbar M. Surgical management of staple line leak after laparoscopic sleeve gastrectomy. Journal of the Faculty of Medicine-Baghdad. 2017;59:1-4.
- Chmielewski PP, Strzelec B, Mozdziak P, Kempisty B. Neutrophil-to-Lymphocyte Ratio as a Prognostic Biomarker for Long-Term Survival in Older Adults at a Mental Health Care Center: A Historical Cohort Analysis. J Clin Med. 2025;14(7).
- Romano L, Mattei A, Colozzi S, Giuliani A, Cianca G, Lazzarin G, et al. Laparoscopic sleeve gastrectomy: A role of inflammatory markers in the early detection of gastric leak. J Minim Access Surg. 2021;17(3):342-50.
- Bellanger DE, Greenway FL. Laparoscopic sleeve gastrectomy, 529 cases without a leak: short-term results and technical considerations. Obes Surg. 2011;21(2):146-50.
- Stroh C, Birk D, Flade-Kuthe R, Frenken M, Herbig B, Höhne S, et al. A nationwide survey on bariatric surgery in Germany--results 2005-2007. Obes Surg. 2009;19(1):105-12.
- Di Furia M, Romano L, Salvatorelli A, Brandolin D, Lomanto D, Cianca G, et al. Indocyanine Green Fluorescent Angiography During Laparoscopic Sleeve Gastrectomy: Preliminary Results. Obes Surg. 2019;29(12):3786-90.
- Schietroma M, Pessia B, Mattei A, Romano L, Giuliani A, Carlei F. Temperature-Neutrophils-Multiple Organ Failure Grading for Complicated Intra-Abdominal Infections. Surg Infect (Larchmt). 2020;21(1):69-74.
- Aurora AR, Khaitan L, Saber AA. Sleeve gastrectomy and the risk of leak: a systematic analysis of 4,888 patients. Surg Endosc. 2012;26(6):1509-15.
- Alsaif SH, Rogers AC, Pua P, Casey PT, Aherne GG, Brannigan AE, et al. Preoperative C-reactive protein and other inflammatory markers as predictors of postoperative complications in patients with colorectal neoplasia. World J Surg Oncol. 2021;19(1):74.
- Plat VD, Voeten DM, Daams F, van der Peet DL, Straatman J. C-reactive protein after major abdominal surgery in daily practice. Surgery. 2021;170(4):1131-9.
- Albanopoulos K, Alevizos L, Natoudi M, Dardamanis D, Menenakos E, Stamou K, et al. C-reactive protein, white blood cells, and neutrophils as early predictors of postoperative complications in patients undergoing laparoscopic sleeve gastrectomy. Surg Endosc. 2013;27(3):864-71.
- Kassir R, Blanc P, Bruna Tibalbo LM, Breton C, Lointier P. C-Reactive protein and procalcitonin for the early detection of postoperative complications after sleeve gastrectomy: preliminary study in 97 patients. Surg Endosc. 2015;29(6):1439-44.
- Yang F, Sang W, Liu Y, Wang J. The C-reactive protein-to-albumin ratio as a diagnostic biomarker for rheumatoid arthritis: a cross-sectional NHANES analysis. Front Med (Lausanne). 2025;12:1624527.
- Yang J, Chen Y, Wan J, Li F, Yang X, Shen B, et al. Prognostic value of the C-reactive protein to albumin ratio in patients with stroke: a meta-analysis. Sci Rep. 2025;15(1):21150.
- 1Onder M, Orucoglu N, Omar F, Canataroglu A. C-Reactive Protein-to-Albumin Ratio: A Novel Inflammatory Marker and Disease Activity Sign in Early Rheumatoid Arthritis. Aktuelle Rheumatologie. 2021;47.
- Khedr D, Nabil S, Abdulrazek AA, Abdelnaby A, Lotfy S. The C-Reactive Protein/Albumin Ratio As An Early Diagnostic Marker Of Neonatal Sepsis In Preterm Neonates: A Case-Control Study. Pediatri Sci J. 2024;4(2):103-10.
- Zhao H, Liu H, Qi W, Liu W, Ye L, Cao Q, et al. Postoperative Ratio of C-Reactive Protein to Albumin as a Predictive Marker in Patients with Crohn's Disease Undergoing Bowel Resection. Gastroenterol Res Pract. 2021;2021:6629608.
- Lee JW, Sharma AR, Lee SS, Chun WJ, Kim HS. The C-reactive protein to albumin ratio predicts postoperative complication in patients who undergo gastrectomy for gastric cancer. Heliyon. 2020;6(6):e04220.
- Shaukat W, Baig AM, Ali Z, Kumari K, Tariq T, Soomro AK, et al. Prognostic Value of C-reactive Protein and Neutrophil-to-Lymphocyte Ratio in Predicting Postoperative Infections After Gastrointestinal Surgery: A Meta-Analysis. Cureus. 2025;17(8).
- Buonacera A, Stancanelli B, Colaci M, Malatino L. Neutrophil to Lymphocyte Ratio: An Emerging Marker of the Relationships between the Immune System and Diseases. Int J Mol Sci. 2022;23(7).
- Islam MM, Satici MO, Eroglu SE. Unraveling the clinical significance and prognostic value of the neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, systemic immune-inflammation index, systemic inflammation response index, and delta neutrophil index: An extensive literature review. Turk J Emerg Med. 2024;24(1):8-19.
- Medina Fernández FJ, Muñoz-Casares FC, Arjona-Sánchez A, Casado-Adam A, Gómez-Luque I, Garcilazo Arismendi DJ, et al. Postoperative time course and utility of inflammatory markers in patients with ovarian peritoneal carcinomatosis treated with neoadjuvant chemotherapy, cytoreductive surgery, and HIPEC. Ann Surg Oncol. 2015;22(4):1332-40.
- Mohamed AAA, Elghandour MMA, Nashed GA, Abdulaziz A. Role of neutrophil-to-lymphocyte ratio in the prediction of early postoperative complications after surgery for intestinal obstruction. Egypt J Surg. 2023;41(4):-.
- Da Silva M, Cleghorn MC, Elnahas A, Jackson TD, Okrainec A, Quereshy FA. Postoperative day one neutrophil-to-lymphocyte ratio as a predictor of 30-day outcomes in bariatric surgery patients. Surg Endosc. 2017;31(6):2645-50.