Manal Atallah Khwein 1 , Ferdous A. Jabir 2
Authors affiliations:
Background & objective: Type 2 Diabetes Mellitus (T2DM) is a multifactorial disorder, in which genetic susceptibility and dysregulation of iron metabolism and coagulation play significant roles. The NRAMP1 gene (SLC11A1), which is involved in innate immunity and iron homeostasis, is a candidate for influencing T2DM risk, although population-specific data are limited. We study aimed to investigate the association between the NRAMP1 rs3731864 polymorphism and hepcidin, D-dimer, and human thrombin levels in Iraqi patients with T2DM.
Methodology: A case-control study was conducted on 120 subjects (60 T2DM patients and 60 healthy controls). Genotyping of the rs3731864 polymorphism was performed by PCR. Serum hepcidin and thrombin levels were measured using Enzyme-Linked Fluorescent Assay (ELFA), and plasma D-dimer was quantified with a FineCare™ FIA meter.
Results: The minor A allele of rs3731864 was associated with enhanced risk of T2DM (OR = 0.28, 95%CI: 0.144-0.58, P = 0.0005). T2DM patients had significantly higher circulating D-dimer (P < 0.0001) and thrombin levels (P = 0.0035), but lower hepcidin levels (P = 0.0025) than controls. But the levels of hepcidin were statistically higher in poor-controlled glycemic (HbA1c ≥7%) patients. Good diagnostic accuracy of hepcidin (0.73) and D-dimer was revealed by analysis of ROC curve (AUC=0.69).
Conclusion: NRAMP1 rs3731864 A allele is a significant risk factor for T2DM in the Iraqi population. This genetic predisposition is accompanied by a prothrombotic state and altered iron metabolism, highlighting the interplay between innate immunity, coagulation, and iron regulation in T2DM.
Keywords: Diabetes Mellitus, Type 2; NRAMP1 Protein; Hepcidins; Fibrinogen; Thrombin; Iraq.
Citation: Khwein MA, Jabir FA. Association of NRAMP1 (rs3731864) gene polymorphism with hepcidin, D-dimer, and thrombin levels in Iraqi patients with type 2 diabetes mellitus. Anaesth. pain intensive care 2025;30(2):207-214. DOI: 10.35975/apic.v30i2.3128
Received: December 07, 2025; Revised: January 03, 2026; Accepted: January 03, 2026
Type 2 Diabetes Mellitus (T2DM) is a major global public health challenge characterized by chronic hyperglycemia that results from a progressive loss of beta-cell insulin secretion, often in the context of insulin resistance.1,2 According to the International Diabetes Federation (IDF), the number of diabetes mellitus cases is growing exponentially, reaching several hundreds of millions affected among global population. It is expected that this number will rise dramatically in the next few decades, especially in low- and middle-income countries.3,4 T2DM is a complex, heterogeneous disease which leads to long-term morbidity and morality through both microvascular and macrovascular complications as consequences of chronic inflammation and sustained pro-thrombotic state.5
T2DM aetiology is characterized by the important interaction of genetic susceptibility and environmental factors. Growing evidence has emphasized the role of innate immunity and iron homeostasis in shaping T2DM susceptibility and development.6 Natural Resistance-Associated Macrophage Protein 1 (NRAMP1), which is encoded by the SLC11A1 gene, can be regarded as one of the most important proteins in this sense. NRAMP1 is dually specific for divalent metals and operates primarily in the phagolysosomal membrane of macrophages to control the intracellular levels of essential metals, including iron and manganese by regulating their concentration.7,8 This function associates SLC11A1 with macrophage activation, inflammatory pathways and iron homeostasis which are emerging as contributors to metabolic disturbance and T2DM.9 For example, Polymorphisms of SLC11A1 gene (i.e., rs3731864) have been studied for its potential susceptibility to T2DM and metabolic traits, but the results are controversial between ethnic subgroups.10
In addition to genetic predisposition, the hypercoagulable state in T2DM patients is a significant factor in the development of vascular complications. Chronic hyperglycemia and inflammation lead to endothelial dysfunction and activation of the coagulation cascade.11 Hepcidin, the master regulator of systemic iron metabolism, is an acute-phase reactant whose levels are elevated in inflammatory conditions and has been strongly associated with an increased long-term risk of mortality and nonfatal cardiovascular events in T2DM patients.12 Furthermore, D-dimer and Human Thrombin are essential biomarkers of coagulation and fibrinolysis. Elevated plasma D-dimer levels, a product of fibrin degradation, and increased thrombin generation are direct indicators of a persistent pro-thrombotic milieu in T2DM, which significantly accelerates atherosclerosis and increases the risk of stroke and myocardial infarction.13,14
Despite the established links between SLC11A1 polymorphisms, iron dysregulation, and hypercoagulability in T2DM, a significant gap remains in the understanding of the integrated relationship between the NRAMP1 (rs3731864) gene polymorphism and the specific profile of these key biochemical parameters (Hepcidin, D-dimer, and Human Thrombin) in the context of T2DM susceptibility and complication risk within the Iraqi population. Genetic associations are known to exhibit population-specific variations owing to distinct genetic backgrounds and environmental exposure. Therefore, a localized, comprehensive study is necessary to accurately assess how the NRAMP1 rs3731864 variant influences the levels of these critical iron and coagulation biomarkers in patients with Iraqi T2DM, a relationship that has not been adequately explored in this specific demographic. The primary aim of this study was to investigate the association between the NRAMP1 (rs3731864) gene polymorphism and the levels of selected biochemical parameters, namely Hepcidin, D-dimer, and Human Thrombin, in Iraqi patients with type 2 Diabetes Mellitus.
A case-control study was conducted from November 2024 to May 2025. The study enrolled a total of 120 Iraqi participants, comprising 60 patients diagnosed with T2DM and 60 age- and sex-matched healthy controls. T2DM patients were recruited from the outpatient diabetes clinic of Al-Hamza General Hospital in Diwaniyah Governorate. The diagnosis of T2DM was confirmed based on the ADA criteria: fasting blood glucose ≥126 mg/dL and/or HbA1c ≥6.5%. Healthy controls were individuals with no personal history of diabetes, normal fasting blood glucose (<100 mg/dL), and HbA1c <5.7%. Exclusion criteria for both groups included a history of type 1 diabetes, acute infections, liver or renal dysfunction, hematological disorders, recent thromboembolic events (within the last 6 months), pregnancy, and the use of anticoagulant therapy.
For each individual, 7 ml of venous blood was collected in three vacutainer tubes. One milliliter was separated in an EDTA tube for genetic polymorphism analysis. Three ml of the blood was transferred into a sodium citrate tube for plasmas separation whereas corresponding to another 3ml was drawn into a gel tube for serums preparation. EDTA-tubes whole blood was subjected to DNA extraction directly. Serum was separated by centrifugation (3000 × g for 10 min) from blood collected in gel tubes. The prepared sera and plasma aliquots were placed in Eppendorf tubes and stored in a deep freezer at -20°C until biochemical analysis was done.
2.3. Biochemical Assays
Biochemical parameters were done in the laboratories of Al-Hamza General Hospital, Al-Ameen Center for Research and Advanced Biotechnologies, Najaf & Clinical Biochemistry Unit Al-Manar Laboratory. Fasting Blood Glucose (FBG) and HbA1c were determined on routine automated clinical chemistry analyzers. Serum Hepcidin and Human Thrombin were detected by ELISA kit (Elabscience, USA) in accordance with the manufacturer's protocol. Plasma concentration of D-dimer was determined with use of a FineCare™ FIA meter.
2.4. Genotyping of NRAMP1 Polymorphisms
Genomic DNA was extracted from whole blood samples collected in EDTA tubes. NRAMP1 gene polymorphisms (rs3731864) were analyzed using polymerase chain reaction (PCR). Specific outer and inner primer sets were designed for each of the SNPs. The primer sequences for rs3731864 were: Forward Outer 5'-CAGGACTTTGGGACTGCAGCCCCAAACT-3,’ forward inner 5'-ACCCCTGGACCAGGCTGGGCTGACACA-3,’ reverse outer 5'-CTCCCAGGCTGTTCGATGTCAGAGCCCT-3,’ and reverse inner 5'-CAGAGTGGGATTCTGAAACCAGAGTGGACC-3.
For PCR, a 25 μL reaction mixture was used, which includes 4.5 μL of DNA template, and 2μl each for forward and reverse primers with addition to 12.5 μl green master mix. The thermo-cycling conditions for amplification of the rs3731865 variant were 95°C for 5 min, then 33 cycles at 95°C for 25 s, primer annealing at 60°C for 30 s, and extension at 72 °C for 35 s, followed by a final extension step at 72 °C for another10 min. For SNP rs3731864, the program consisted of an initial 5 min at 95°C and then 30 cycles of: 5s at 95°C, 30 s at 54.5°C, and finally extending for a further forty seconds at72°C followed by an additional elongation phase for five minutes. The PCR products were analyzed by 1.5% agarose gel electrophoresis and diluted.
2.5. Statistical analysis
Data were collected, and the system was placed on a data file in Microsoft Excel.Afterward the data were analysed with GraphPad Prism version 9.0. Variables are presented as n (%) for categorical data and mean ± SD for continuous variables. Unpaired independent sample t-test was used to compare continuous variables between groups, for data with normal distribution. Categorical variables were compared using the chi-squared (χ²) test. Receiver Operating Characteristic (ROC) curve analysis was used to evaluate the diagnostic accuracy of the biomarkers, and area under the curve (AUC) with optimal cut-off value, sensitivity and specificity were obtained. The genotype and allele frequencies between the cases and controls of both BD samples were compared using chi squared test to estimate Hardy-Weinberg equilibrium (HWE) within the control sample.
Table 1 provides a summary of demographics and clinical factors comparing patients with T2DM to healthy individuals. The T2DM group was substantially older and heavier (mean BMI), but this difference did not reach statistical significance (P = 0.08 and P = 0.09, respectively). Unsurprisingly, fasting blood sugar and HbA1c values were significantly higher in T2DM group (P < 0.0001) signifying diabetic condition. Most importantly the main biomarkers were also significantly different in both studies: hepcidin was lowest in T2DM (13.84 vs 17.6 ng/ml, P = 0.0025) and D-dimer as well as human thrombin were significantly highest (P < 0.0001 and P = 0.0035, respectively).
This table demonstrates the diagnosis value of serum iron, hepcidin, D-dimer and human thrombin in T2DM by analyzing ROC curve. Serum iron concentration was not diagnostically useful (AUC=0.54, P = 0.45). On the other hand, hepcidin, D-dimer and human thrombin showed good discriminatory power. D-dimer was the most reliable (AUC: 0.69), and exhibited high specificity (91.7%) and acceptable sensitivity (74.6%) at a cutoff value of more than (>230 ng/mL). Hepcidin also had good diagnostic accuracy (AUC=0.73), while human thrombin was highly specific (96.7%) but less sensitive (52.6%).
The distribution of genotypes and alleles of the NRAMP1 rs3731864 polymorphism in T2DM patients and controls, evaluating the risk of developing disease. There was a significant difference in genotype distribution (P = 0.0051). The wild-type GG genotype was higher in controls (70%), whereas the GA and AA genotypes, were more prevalent in T2DM patients (45% and 20%, respectively). Therefore, the minor A allele was significantly associated with risk for T2DM (OR=0.28, 95%CI:0.144- 0.58, P = 0.0005) which suggested that A allele could be a hypothetical risk alleleor developing T2DM in this Iraqi population. The genotype distribution was consistent with the Hardy-Weinberg Equilibrium for the control group, which represented a random sample.
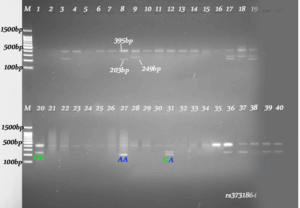
Figure1: Gel electrophoresis of NRAMP1 rs3731864 genotyping: Products of PCR were run on 1.5% agarose gel (72 V, 80 min). Lanes, 1-40: Participant samples (Lane M is a 100 bp DNA ladder as molecular weight marker). The bands in the gels refer to the predicted product sizes 395 bp, 249 bp and 203 bp for each genotype of SNP rs3731864.
4. DISCUSSION
The present study presents a complete correlation between the NRAMP1 gene polymorphism, iron metabolism and hypercoagulability in Iraqi patients with T2DM. The main results showed significant association of rs3731864 polymorphism with susceptibility to T2DM as well as severe disturbances in major biomarkers of coagulation and iron homeostasis. Shot text These findings not only confirm the multifactorial aetiology of T2DM, but also emphasize the role of population-specific SNPs as well as indicate the clinical significance of prothrombotic and inflammatory process involved in T2DM.
The present study identified significantly elevated levels of D-dimer and human thrombin in T2DM patients compared to those in healthy controls, confirming the presence of a hypercoagulable state. This is consistent with the large body of evidence from the last decade demonstrating that T2DM is a prothrombotic condition.11,15 The elevated D-dimer level, a specific product of cross-linked fibrin degradation, indicates ongoing activation of both coagulation and fibrinolysis. This suggests a dynamic but ultimately pathological state in which the body continuously forms and dissolves microclots, placing patients at a heightened risk for vascular events. Our findings align with a 2022 study by Cheng et al., which established high D-dimer concentration as an independent predictor of adverse cardiovascular outcomes in a T2DM population, even after adjusting for conventional risk factors.16 Furthermore, a recent study by Khatun et al. (2025) similarly reported significantly higher D-dimer and fibrinogen levels in diabetic patients, reinforcing the concept of a persistent thrombotic alert in this group.13
The concurrent increase in human thrombin provides a mechanistic basis for this observation. Thrombin is the central enzyme in the coagulation cascade and its increased generation, as observed in our cohort, directly reflects a more active coagulation process. This is likely driven by hyperglycemia and insulin resistance, which upregulate coagulation factors and promote endothelial dysfunction.17 Recent research by Han et al. (2024) further solidified this link, showing that elevated thrombin generation potential is associated with incident T2DM, suggesting that it is not just a consequence but also a potential contributor to the progression of the disease.18 This prothrombotic milieu is a critical driver of accelerated atherosclerosis and an increased risk of myocardial infarction and stroke that characterizes T2DM.
Our study of iron metabolism produced a tangle of confusing, but nevertheless illuminating, findings. Although T2DM group had lower hepcidin levels, a subgroup analysis showed that patients with inadequate glycemic control (HbA1c ≥ 7%) presented higher hepcidin level. This observation illustrates the complex regulation of hepcidin by body iron levels, inflammation, and metabolic cues. The lower hepcidin levels in the total group may represent an adaptive mechanism to underlying iron deficiency or other measures, but the increase in the uncontrolled group is remarkable. This finding indicates the possibility that chronic inflammation and strong metabolic stress, which are characteristics of uncontrolled diabetes, may supersede the other signals stimulating hepcidin production.
This finding is of high clinical relevance, as a landmark prospective study by Mantovani et al. (2024) recently demonstrated that elevated plasma hepcidin levels are strongly associated with an increased long-term risk of mortality and non-fatal cardiovascular events in T2DM patients.19 The proposed mechanisms involve hepcidin-mediated iron deposition in macrophages within atherosclerotic plaques, which exacerbates lipid peroxidation, foam cell formation, and ferroptosis, an iron-dependent form of cell death implicated in cardiovascular pathology.19,20 Therefore, the elevated hepcidin levels observed in our poorly controlled patients could be a key link between metabolic dysregulation and increased cardiovascular risk. This aligns with broader research indicating that iron dysregulation, characterized by either overload or deficiency, is a significant risk factor for metabolic and cardiovascular disease.21 The causal role of iron in T2DM has been further strengthened by large-scale Mendelian randomization studies, such as that of Wang et al. (2021), which provided genetic evidence supporting a causal link between increased systemic iron status and higher T2DM risk.22
A central finding of our study is the significant association of SLC11A1 rs3731864 polymorphism with T2DM in the Iraqi population, where the minor 'A' allele conferred an increased risk of the disease. SLC11A1, encoding a divalent metal transporter in macrophages, is a critical component of innate immunity and inflammatory pathways.23 Its role in regulating macrophage activation and iron homeostasis makes it a strong biological candidate for influencing T2DM susceptibility, a disease fundamentally linked to chronic low-grade inflammation.
The wider function of SLC11A1 as a key immune response regulator is well-recognised. A larger systematic review and meta-analysis of Archer et al. (20152) showed that genetic variants in the whole SLC11A1 region are undoubtedly associated with susceptibility to infectious (e.g tuberculosis) as well as autoimmune diseases.24 This places the gene at a critical juncture in the balance between host defense and immune tolerance. Various polymorphisms have been associated with various autoimmune diseases such as rheumatoid arthritis and inflammatory bowel disease exhibiting contradictory effects. For example, one study found that the 469+14G/C polymorphism (rs3731865) is related to increased susceptibility of autoimmune diseases especially rheumatoid arthritis according to a meta-analysis.24 This particular variant has also been linked to susceptibility to T1D in European populations.25
Interestingly, our results contrast with a 2023 study by Kavian et al. in an Iranian population, which found that the same rs3731864 'A' allele was associated with a protective effect against T2DM.26 This discrepancy underscores the well-established principle that the impact of genetic polymorphisms on complex diseases can vary significantly across different ethnic populations owing to distinct genetic backgrounds and environmental exposures. While our study points to the 'A' allele as a risk factor in Iraqis, the study by Kavian et al. suggests a different role in a neighboring population. Such population-specific effects are crucial for the future of personalized medicine and highlight the necessity of conducting localized genetic association studies. The functional consequence of the rs3731864 variant, which is located in an intronic region, may be related to altered splicing of the SLC11A1 transcript, as suggested by in silico analysis from the study by Kavian et al., potentially leading to a dysregulated inflammatory response in macrophages.26
The case control design does not allow us to draw conclusions about causality and although the sample size is adequate for initial association, replication in a larger multicenter Iraqi cohort would be an advantage. We also did not directly measure the inflammatory cytokines to match with our genetic and biochemical results. Further research should include prospective studies to validate the predictive role of these biomarkers in Iraqi population. Functional studies are needed to confirm the specific molecular mechanism by which rs3731864 polymorphism affects macrophage function and T2DM predisposition in this ethnic group. Examination of an interaction between this genetic variant, hepcidin expression and the prothrombotic state is necessary to better understand the pathogenesis of T2DM and design treatment approaches.
6. CONCLUSION
The present study provides strong evidence that SLC11A1 rs3731864 polymorphism is a major T2DM risk factor in the Iraqi population. This genetic predisposition is paralleled by a clear prothrombotic phenotype, consisting of increased levels D-dimer and thrombin, and by an altered iron inflammatory axis with changes in hepcidin levels particularly among patients with poor glycemic control. Our results indicate that innate immunity, coagulation, and iron metabolisms closely intersect with the T2DM process. Biomarkers that were studied, hepcidin, D-dimer and thrombin, hold the potential to be used for risk stratification and potentially identifying those at risk for CVD in whom aggressive modification of cardiovascular risk such as lipid reduction may be beneficial. This work highlights the need for population-specific genetic research and opens doors for further study of individualized therapy with a focus on these interconnected pathways.
7. Acknowledgment
The authors wish to extend their sincere gratitude to all the patients and healthy volunteers who participated in this study. We also acknowledge the valuable support of the staff at the outpatient diabetes clinic of Al-Hamza General Hospital for their assistance in recruiting patients. We are thankful for the technical assistance provided by the laboratory teams at the Al-Ameen Center for Research and Advanced Biotechnologies in Najaf and the Clinical Biochemistry Unit at Al-Manar Laboratory for conducting the biochemical assays
8. Ethics considerations
Ethical approval for the study was obtained from the Institutional Review Board of the College of Medicine and Al‐Diwaniyah Health Directorate reviewed and approved the study protocol before it started (No.:4679/30 on 26//2024). Written informed consent was secured from all participants prior to their inclusion in the study.
9. Availability of Data
The datasets generated and/or analyzed during the current study are not publicly available due to participant privacy and confidentiality concerns but are available from the corresponding author on reasonable request.
11. Competing interests
The authors declare that they have no competing interests, financial or otherwise, that could have influenced the work reported in this paper.
12. Funding
Non.
13. Authors’ Contributions
MAK: Conceptualization, methodology, investigation, data curation, formal analysis, writing the original draft, and project administration. FAJ: Supervision, validation, visualization, and critical review and editing of the manuscript. All authors have read and approved the final version of the manuscript.
Authors affiliations:
- Manal Atallah Khwein. Department of Medical Chemistry, College of Medicine, University of AL -Qadisiyah, Al-Diywaniyah, Iraq; Email: manalatalah614@gmail.com
- Ferdous A Jabir. Department of Medical Chemistry, College of Medicine, University of AL -Qadisiyah, Al-Diywaniyah, Iraq. Email: Ferdousabbas@gmail.com
ABSTRACT
Background & objective: Type 2 Diabetes Mellitus (T2DM) is a multifactorial disorder, in which genetic susceptibility and dysregulation of iron metabolism and coagulation play significant roles. The NRAMP1 gene (SLC11A1), which is involved in innate immunity and iron homeostasis, is a candidate for influencing T2DM risk, although population-specific data are limited. We study aimed to investigate the association between the NRAMP1 rs3731864 polymorphism and hepcidin, D-dimer, and human thrombin levels in Iraqi patients with T2DM.
Methodology: A case-control study was conducted on 120 subjects (60 T2DM patients and 60 healthy controls). Genotyping of the rs3731864 polymorphism was performed by PCR. Serum hepcidin and thrombin levels were measured using Enzyme-Linked Fluorescent Assay (ELFA), and plasma D-dimer was quantified with a FineCare™ FIA meter.
Results: The minor A allele of rs3731864 was associated with enhanced risk of T2DM (OR = 0.28, 95%CI: 0.144-0.58, P = 0.0005). T2DM patients had significantly higher circulating D-dimer (P < 0.0001) and thrombin levels (P = 0.0035), but lower hepcidin levels (P = 0.0025) than controls. But the levels of hepcidin were statistically higher in poor-controlled glycemic (HbA1c ≥7%) patients. Good diagnostic accuracy of hepcidin (0.73) and D-dimer was revealed by analysis of ROC curve (AUC=0.69).
Conclusion: NRAMP1 rs3731864 A allele is a significant risk factor for T2DM in the Iraqi population. This genetic predisposition is accompanied by a prothrombotic state and altered iron metabolism, highlighting the interplay between innate immunity, coagulation, and iron regulation in T2DM.
Keywords: Diabetes Mellitus, Type 2; NRAMP1 Protein; Hepcidins; Fibrinogen; Thrombin; Iraq.
Citation: Khwein MA, Jabir FA. Association of NRAMP1 (rs3731864) gene polymorphism with hepcidin, D-dimer, and thrombin levels in Iraqi patients with type 2 diabetes mellitus. Anaesth. pain intensive care 2025;30(2):207-214. DOI: 10.35975/apic.v30i2.3128
Received: December 07, 2025; Revised: January 03, 2026; Accepted: January 03, 2026
1. INTRODUCTION
Type 2 Diabetes Mellitus (T2DM) is a major global public health challenge characterized by chronic hyperglycemia that results from a progressive loss of beta-cell insulin secretion, often in the context of insulin resistance.1,2 According to the International Diabetes Federation (IDF), the number of diabetes mellitus cases is growing exponentially, reaching several hundreds of millions affected among global population. It is expected that this number will rise dramatically in the next few decades, especially in low- and middle-income countries.3,4 T2DM is a complex, heterogeneous disease which leads to long-term morbidity and morality through both microvascular and macrovascular complications as consequences of chronic inflammation and sustained pro-thrombotic state.5
T2DM aetiology is characterized by the important interaction of genetic susceptibility and environmental factors. Growing evidence has emphasized the role of innate immunity and iron homeostasis in shaping T2DM susceptibility and development.6 Natural Resistance-Associated Macrophage Protein 1 (NRAMP1), which is encoded by the SLC11A1 gene, can be regarded as one of the most important proteins in this sense. NRAMP1 is dually specific for divalent metals and operates primarily in the phagolysosomal membrane of macrophages to control the intracellular levels of essential metals, including iron and manganese by regulating their concentration.7,8 This function associates SLC11A1 with macrophage activation, inflammatory pathways and iron homeostasis which are emerging as contributors to metabolic disturbance and T2DM.9 For example, Polymorphisms of SLC11A1 gene (i.e., rs3731864) have been studied for its potential susceptibility to T2DM and metabolic traits, but the results are controversial between ethnic subgroups.10
In addition to genetic predisposition, the hypercoagulable state in T2DM patients is a significant factor in the development of vascular complications. Chronic hyperglycemia and inflammation lead to endothelial dysfunction and activation of the coagulation cascade.11 Hepcidin, the master regulator of systemic iron metabolism, is an acute-phase reactant whose levels are elevated in inflammatory conditions and has been strongly associated with an increased long-term risk of mortality and nonfatal cardiovascular events in T2DM patients.12 Furthermore, D-dimer and Human Thrombin are essential biomarkers of coagulation and fibrinolysis. Elevated plasma D-dimer levels, a product of fibrin degradation, and increased thrombin generation are direct indicators of a persistent pro-thrombotic milieu in T2DM, which significantly accelerates atherosclerosis and increases the risk of stroke and myocardial infarction.13,14
Despite the established links between SLC11A1 polymorphisms, iron dysregulation, and hypercoagulability in T2DM, a significant gap remains in the understanding of the integrated relationship between the NRAMP1 (rs3731864) gene polymorphism and the specific profile of these key biochemical parameters (Hepcidin, D-dimer, and Human Thrombin) in the context of T2DM susceptibility and complication risk within the Iraqi population. Genetic associations are known to exhibit population-specific variations owing to distinct genetic backgrounds and environmental exposure. Therefore, a localized, comprehensive study is necessary to accurately assess how the NRAMP1 rs3731864 variant influences the levels of these critical iron and coagulation biomarkers in patients with Iraqi T2DM, a relationship that has not been adequately explored in this specific demographic. The primary aim of this study was to investigate the association between the NRAMP1 (rs3731864) gene polymorphism and the levels of selected biochemical parameters, namely Hepcidin, D-dimer, and Human Thrombin, in Iraqi patients with type 2 Diabetes Mellitus.
2. METHODOLOGY
A case-control study was conducted from November 2024 to May 2025. The study enrolled a total of 120 Iraqi participants, comprising 60 patients diagnosed with T2DM and 60 age- and sex-matched healthy controls. T2DM patients were recruited from the outpatient diabetes clinic of Al-Hamza General Hospital in Diwaniyah Governorate. The diagnosis of T2DM was confirmed based on the ADA criteria: fasting blood glucose ≥126 mg/dL and/or HbA1c ≥6.5%. Healthy controls were individuals with no personal history of diabetes, normal fasting blood glucose (<100 mg/dL), and HbA1c <5.7%. Exclusion criteria for both groups included a history of type 1 diabetes, acute infections, liver or renal dysfunction, hematological disorders, recent thromboembolic events (within the last 6 months), pregnancy, and the use of anticoagulant therapy.
For each individual, 7 ml of venous blood was collected in three vacutainer tubes. One milliliter was separated in an EDTA tube for genetic polymorphism analysis. Three ml of the blood was transferred into a sodium citrate tube for plasmas separation whereas corresponding to another 3ml was drawn into a gel tube for serums preparation. EDTA-tubes whole blood was subjected to DNA extraction directly. Serum was separated by centrifugation (3000 × g for 10 min) from blood collected in gel tubes. The prepared sera and plasma aliquots were placed in Eppendorf tubes and stored in a deep freezer at -20°C until biochemical analysis was done.
2.3. Biochemical Assays
Biochemical parameters were done in the laboratories of Al-Hamza General Hospital, Al-Ameen Center for Research and Advanced Biotechnologies, Najaf & Clinical Biochemistry Unit Al-Manar Laboratory. Fasting Blood Glucose (FBG) and HbA1c were determined on routine automated clinical chemistry analyzers. Serum Hepcidin and Human Thrombin were detected by ELISA kit (Elabscience, USA) in accordance with the manufacturer's protocol. Plasma concentration of D-dimer was determined with use of a FineCare™ FIA meter.
2.4. Genotyping of NRAMP1 Polymorphisms
Genomic DNA was extracted from whole blood samples collected in EDTA tubes. NRAMP1 gene polymorphisms (rs3731864) were analyzed using polymerase chain reaction (PCR). Specific outer and inner primer sets were designed for each of the SNPs. The primer sequences for rs3731864 were: Forward Outer 5'-CAGGACTTTGGGACTGCAGCCCCAAACT-3,’ forward inner 5'-ACCCCTGGACCAGGCTGGGCTGACACA-3,’ reverse outer 5'-CTCCCAGGCTGTTCGATGTCAGAGCCCT-3,’ and reverse inner 5'-CAGAGTGGGATTCTGAAACCAGAGTGGACC-3.
For PCR, a 25 μL reaction mixture was used, which includes 4.5 μL of DNA template, and 2μl each for forward and reverse primers with addition to 12.5 μl green master mix. The thermo-cycling conditions for amplification of the rs3731865 variant were 95°C for 5 min, then 33 cycles at 95°C for 25 s, primer annealing at 60°C for 30 s, and extension at 72 °C for 35 s, followed by a final extension step at 72 °C for another10 min. For SNP rs3731864, the program consisted of an initial 5 min at 95°C and then 30 cycles of: 5s at 95°C, 30 s at 54.5°C, and finally extending for a further forty seconds at72°C followed by an additional elongation phase for five minutes. The PCR products were analyzed by 1.5% agarose gel electrophoresis and diluted.
2.5. Statistical analysis
Data were collected, and the system was placed on a data file in Microsoft Excel.Afterward the data were analysed with GraphPad Prism version 9.0. Variables are presented as n (%) for categorical data and mean ± SD for continuous variables. Unpaired independent sample t-test was used to compare continuous variables between groups, for data with normal distribution. Categorical variables were compared using the chi-squared (χ²) test. Receiver Operating Characteristic (ROC) curve analysis was used to evaluate the diagnostic accuracy of the biomarkers, and area under the curve (AUC) with optimal cut-off value, sensitivity and specificity were obtained. The genotype and allele frequencies between the cases and controls of both BD samples were compared using chi squared test to estimate Hardy-Weinberg equilibrium (HWE) within the control sample.
3. RESULTS
Table 1 provides a summary of demographics and clinical factors comparing patients with T2DM to healthy individuals. The T2DM group was substantially older and heavier (mean BMI), but this difference did not reach statistical significance (P = 0.08 and P = 0.09, respectively). Unsurprisingly, fasting blood sugar and HbA1c values were significantly higher in T2DM group (P < 0.0001) signifying diabetic condition. Most importantly the main biomarkers were also significantly different in both studies: hepcidin was lowest in T2DM (13.84 vs 17.6 ng/ml, P = 0.0025) and D-dimer as well as human thrombin were significantly highest (P < 0.0001 and P = 0.0035, respectively).
| Table 1: Baseline characteristics and biochemical parameters in study participants | ||||
| Characteristic | T2DM Group
(n = 60) |
Control Group
(n = 60) |
P-value | |
| Age (years) Mean ± SD | 53.1 ± 10.2 | 41.8 ± 9.9 | 0.08 | |
| Gender | Male, n (%) | 35 (58%) | 30 (50%) | 0.4 |
| Female, n (%) | 25 (42%) | 30 (50%) | ||
| BMI (kg/m²) Mean ±SD | 26.7 ± 3.4 | 25.3 ± 2.1 | 0.09 | |
| Fasting Blood Sugar (mg/dL) | 188.4 ± 77.6 | 90.6 ± 7.7 | < 0.0001 | |
| HbA1c (%) | 8.8 ± 3.9 | 4.7 ± 0.4 | < 0.0001 | |
| Serum Iron (μg/dL) | 103.2 ± 58.4 | 99.1 ± 19.6 | 0.61 | |
| Hepcidin (ng/ml) | 13.84 ± 7.7 | 17.6 ± 5.45 | 0.0025** | |
| D-dimer (ng/mL) | 288.7 ± 170 | 171.6 ± 58.2 | < 0.0001 | |
| Human Thrombin (pg/ml) | 7.8 ± 6.2 | 5.3 ± 2.1 | 0.0035** | |
| Data presented as mean ± SD or n (%); independent sample t-test. Chi-square. NS: Not Significant. * P < 0.01: statistically significant | ||||
| Table 2: Subgroup analysis of biochemical parameters by glycemic control status in the T2DM Group | |||
| Parameter | T2DM Controlled
(n = 16) |
T2DM Uncontrolled
(n = 44) |
P-value |
| HbA1c (%) | 6.1 ± 0.8 | 9.7 ± 4.2 | 0.0014 |
| Fasting Glucose (mg/dL) | 143.8 ± 61.2 | 204 ± 77.2 | 0.0062 |
| Hepcidin (ng/mL) | 11.2 ± 4.8 | 14.8 ± 8.3 | 0.03 |
| D-dimer (ng/mL) | 208.5 ± 79.5 | 316.1 ± 184 | 0.033 |
| Human Thrombin (pg/mL) | 5.2 ± 2.9 | 8.1 ± 6.8 | 0.11 |
| * P < 0.01: statistical significance. Independent sample t-test. Data presented as Mean ± SD | |||
The distribution of genotypes and alleles of the NRAMP1 rs3731864 polymorphism in T2DM patients and controls, evaluating the risk of developing disease. There was a significant difference in genotype distribution (P = 0.0051). The wild-type GG genotype was higher in controls (70%), whereas the GA and AA genotypes, were more prevalent in T2DM patients (45% and 20%, respectively). Therefore, the minor A allele was significantly associated with risk for T2DM (OR=0.28, 95%CI:0.144- 0.58, P = 0.0005) which suggested that A allele could be a hypothetical risk alleleor developing T2DM in this Iraqi population. The genotype distribution was consistent with the Hardy-Weinberg Equilibrium for the control group, which represented a random sample.
| Table 3: Diagnostic performance of biomarkers for identifying type 2 diabetes mellitus. | |||||
| Biomarker | AUC (95% CI) | P-value | Optimal Cut-off | Sensitivity (%) | Specificity (%) |
| Serum Iron (μg/dL) | 0.54 (0.44 to 0.63) | 0.45 | ≤ 76 | 25 | 95 |
| Hepcidin (ng/ml) | 0.73 (0.66 to 0.80) | < 0.001 | ≤ 13.6 | 72.9 | 68.3 |
| D-dimer (ng/mL) | 0.69 (0.61 to 0.77) | < 0.001 | >230 | 74.6 | 91.7 |
| Human Thrombin(pg/ml) | 0.65 (0.56 to 0.74) | 0.004 | >7.0 | 52.6 | 96.7 |
| Abbreviation: AUC, Area Under the Curve; CI, Confidence Interval. | |||||
| Table 4: Association of NRAMP1 rs3731864 genotype and allele frequencies with T2DM susceptibility. | |||||
| Genotype | Control
(n = 40) |
T2DM Group
(n = 40) |
OR (95% CI)* | P-value | |
| NRAMP1 rs3731864 | GG | 28 (70) | 14 (35) | 1 [reference] | --- |
| GA | 10 (25) | 18 (45) | 0.45 (0.084 to 1.75) | 0.0051** | |
| AA | 2 (5) | 8 (20) | |||
|
HWE |
Expected GG | 27 (70) | 13 (35) | 1 [reference] | --- |
| Expected GG | 12 (25) | 20 (45) | 0.23 (0.019 to 1.8) | 0.003** | |
| Expected GG | 1 (5) | 7 (20) | |||
| Allele | |||||
| NRAMP1 rs3731864 | G | 66 (82) | 46 (58) | 1 [reference] | --- |
| A | 14 (18) | 34 (42) | 0.28 (0.144 to 0.58) | 0.0005 | |
| Total | 80 | 80 | |||
| Data presented as n (%); HWE: Hardy-Weinberg Equilibrium, Chi-square, Significant at P < 0.001. | |||||

Figure1: Gel electrophoresis of NRAMP1 rs3731864 genotyping: Products of PCR were run on 1.5% agarose gel (72 V, 80 min). Lanes, 1-40: Participant samples (Lane M is a 100 bp DNA ladder as molecular weight marker). The bands in the gels refer to the predicted product sizes 395 bp, 249 bp and 203 bp for each genotype of SNP rs3731864.
4. DISCUSSION
The present study presents a complete correlation between the NRAMP1 gene polymorphism, iron metabolism and hypercoagulability in Iraqi patients with T2DM. The main results showed significant association of rs3731864 polymorphism with susceptibility to T2DM as well as severe disturbances in major biomarkers of coagulation and iron homeostasis. Shot text These findings not only confirm the multifactorial aetiology of T2DM, but also emphasize the role of population-specific SNPs as well as indicate the clinical significance of prothrombotic and inflammatory process involved in T2DM.
The present study identified significantly elevated levels of D-dimer and human thrombin in T2DM patients compared to those in healthy controls, confirming the presence of a hypercoagulable state. This is consistent with the large body of evidence from the last decade demonstrating that T2DM is a prothrombotic condition.11,15 The elevated D-dimer level, a specific product of cross-linked fibrin degradation, indicates ongoing activation of both coagulation and fibrinolysis. This suggests a dynamic but ultimately pathological state in which the body continuously forms and dissolves microclots, placing patients at a heightened risk for vascular events. Our findings align with a 2022 study by Cheng et al., which established high D-dimer concentration as an independent predictor of adverse cardiovascular outcomes in a T2DM population, even after adjusting for conventional risk factors.16 Furthermore, a recent study by Khatun et al. (2025) similarly reported significantly higher D-dimer and fibrinogen levels in diabetic patients, reinforcing the concept of a persistent thrombotic alert in this group.13
The concurrent increase in human thrombin provides a mechanistic basis for this observation. Thrombin is the central enzyme in the coagulation cascade and its increased generation, as observed in our cohort, directly reflects a more active coagulation process. This is likely driven by hyperglycemia and insulin resistance, which upregulate coagulation factors and promote endothelial dysfunction.17 Recent research by Han et al. (2024) further solidified this link, showing that elevated thrombin generation potential is associated with incident T2DM, suggesting that it is not just a consequence but also a potential contributor to the progression of the disease.18 This prothrombotic milieu is a critical driver of accelerated atherosclerosis and an increased risk of myocardial infarction and stroke that characterizes T2DM.
Our study of iron metabolism produced a tangle of confusing, but nevertheless illuminating, findings. Although T2DM group had lower hepcidin levels, a subgroup analysis showed that patients with inadequate glycemic control (HbA1c ≥ 7%) presented higher hepcidin level. This observation illustrates the complex regulation of hepcidin by body iron levels, inflammation, and metabolic cues. The lower hepcidin levels in the total group may represent an adaptive mechanism to underlying iron deficiency or other measures, but the increase in the uncontrolled group is remarkable. This finding indicates the possibility that chronic inflammation and strong metabolic stress, which are characteristics of uncontrolled diabetes, may supersede the other signals stimulating hepcidin production.
This finding is of high clinical relevance, as a landmark prospective study by Mantovani et al. (2024) recently demonstrated that elevated plasma hepcidin levels are strongly associated with an increased long-term risk of mortality and non-fatal cardiovascular events in T2DM patients.19 The proposed mechanisms involve hepcidin-mediated iron deposition in macrophages within atherosclerotic plaques, which exacerbates lipid peroxidation, foam cell formation, and ferroptosis, an iron-dependent form of cell death implicated in cardiovascular pathology.19,20 Therefore, the elevated hepcidin levels observed in our poorly controlled patients could be a key link between metabolic dysregulation and increased cardiovascular risk. This aligns with broader research indicating that iron dysregulation, characterized by either overload or deficiency, is a significant risk factor for metabolic and cardiovascular disease.21 The causal role of iron in T2DM has been further strengthened by large-scale Mendelian randomization studies, such as that of Wang et al. (2021), which provided genetic evidence supporting a causal link between increased systemic iron status and higher T2DM risk.22
A central finding of our study is the significant association of SLC11A1 rs3731864 polymorphism with T2DM in the Iraqi population, where the minor 'A' allele conferred an increased risk of the disease. SLC11A1, encoding a divalent metal transporter in macrophages, is a critical component of innate immunity and inflammatory pathways.23 Its role in regulating macrophage activation and iron homeostasis makes it a strong biological candidate for influencing T2DM susceptibility, a disease fundamentally linked to chronic low-grade inflammation.
The wider function of SLC11A1 as a key immune response regulator is well-recognised. A larger systematic review and meta-analysis of Archer et al. (20152) showed that genetic variants in the whole SLC11A1 region are undoubtedly associated with susceptibility to infectious (e.g tuberculosis) as well as autoimmune diseases.24 This places the gene at a critical juncture in the balance between host defense and immune tolerance. Various polymorphisms have been associated with various autoimmune diseases such as rheumatoid arthritis and inflammatory bowel disease exhibiting contradictory effects. For example, one study found that the 469+14G/C polymorphism (rs3731865) is related to increased susceptibility of autoimmune diseases especially rheumatoid arthritis according to a meta-analysis.24 This particular variant has also been linked to susceptibility to T1D in European populations.25
Interestingly, our results contrast with a 2023 study by Kavian et al. in an Iranian population, which found that the same rs3731864 'A' allele was associated with a protective effect against T2DM.26 This discrepancy underscores the well-established principle that the impact of genetic polymorphisms on complex diseases can vary significantly across different ethnic populations owing to distinct genetic backgrounds and environmental exposures. While our study points to the 'A' allele as a risk factor in Iraqis, the study by Kavian et al. suggests a different role in a neighboring population. Such population-specific effects are crucial for the future of personalized medicine and highlight the necessity of conducting localized genetic association studies. The functional consequence of the rs3731864 variant, which is located in an intronic region, may be related to altered splicing of the SLC11A1 transcript, as suggested by in silico analysis from the study by Kavian et al., potentially leading to a dysregulated inflammatory response in macrophages.26
5. LIMITATIONS
The case control design does not allow us to draw conclusions about causality and although the sample size is adequate for initial association, replication in a larger multicenter Iraqi cohort would be an advantage. We also did not directly measure the inflammatory cytokines to match with our genetic and biochemical results. Further research should include prospective studies to validate the predictive role of these biomarkers in Iraqi population. Functional studies are needed to confirm the specific molecular mechanism by which rs3731864 polymorphism affects macrophage function and T2DM predisposition in this ethnic group. Examination of an interaction between this genetic variant, hepcidin expression and the prothrombotic state is necessary to better understand the pathogenesis of T2DM and design treatment approaches.
6. CONCLUSION
The present study provides strong evidence that SLC11A1 rs3731864 polymorphism is a major T2DM risk factor in the Iraqi population. This genetic predisposition is paralleled by a clear prothrombotic phenotype, consisting of increased levels D-dimer and thrombin, and by an altered iron inflammatory axis with changes in hepcidin levels particularly among patients with poor glycemic control. Our results indicate that innate immunity, coagulation, and iron metabolisms closely intersect with the T2DM process. Biomarkers that were studied, hepcidin, D-dimer and thrombin, hold the potential to be used for risk stratification and potentially identifying those at risk for CVD in whom aggressive modification of cardiovascular risk such as lipid reduction may be beneficial. This work highlights the need for population-specific genetic research and opens doors for further study of individualized therapy with a focus on these interconnected pathways.
7. Acknowledgment
The authors wish to extend their sincere gratitude to all the patients and healthy volunteers who participated in this study. We also acknowledge the valuable support of the staff at the outpatient diabetes clinic of Al-Hamza General Hospital for their assistance in recruiting patients. We are thankful for the technical assistance provided by the laboratory teams at the Al-Ameen Center for Research and Advanced Biotechnologies in Najaf and the Clinical Biochemistry Unit at Al-Manar Laboratory for conducting the biochemical assays
8. Ethics considerations
Ethical approval for the study was obtained from the Institutional Review Board of the College of Medicine and Al‐Diwaniyah Health Directorate reviewed and approved the study protocol before it started (No.:4679/30 on 26//2024). Written informed consent was secured from all participants prior to their inclusion in the study.
9. Availability of Data
The datasets generated and/or analyzed during the current study are not publicly available due to participant privacy and confidentiality concerns but are available from the corresponding author on reasonable request.
11. Competing interests
The authors declare that they have no competing interests, financial or otherwise, that could have influenced the work reported in this paper.
12. Funding
Non.
13. Authors’ Contributions
MAK: Conceptualization, methodology, investigation, data curation, formal analysis, writing the original draft, and project administration. FAJ: Supervision, validation, visualization, and critical review and editing of the manuscript. All authors have read and approved the final version of the manuscript.
14. REFERENCES
- Khalid M, Petroianu G, Adem A. Advanced Glycation End Products and Diabetes Mellitus: Mechanisms and Perspectives. Biomolecules. 2022;12(4). [PubMed] DOI: 3390/biom12040542
- Maity M, Kumari A. Understanding Diabetes Mellitus: A Descriptive Study of Its Types, Causes and Health Impacts. Bharati Int J Multidiscip Res Dev. 2025;3:34–9.
- Ye J, Wu Y, Yang S, Zhu D, Chen F, Chen J, et al. The global, regional and national burden of type 2 diabetes mellitus in the past, present and future: a systematic analysis of the Global Burden of Disease Study 2019. Front Endocrinol (Lausanne). 2023;14:1192629. [PubMed] DOI: 3389/fendo.2023.1192629
- GBD 2021 Diabetes Collaborators. Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: a systematic analysis for the Global Burden of Disease Study 2021. Lancet. 2023;402(10397):203-234. [PubMed] DOI: 1016/S0140-6736(23)01301-6
- Guan H, Tian J, Wang Y, Niu P, Zhang Y, Zhang Y, et al. Advances in secondary prevention mechanisms of macrovascular complications in type 2 diabetes mellitus patients: a comprehensive review. Eur J Med Res. 2024;29(1):152. [PubMed] DOI: 1186/s40001-024-01739-1
- Pechlaner R, Weiss G, Bansal S, Mayr M, Santer P, Pallhuber B, et al. Inadequate hepcidin serum concentrations predict incident type 2 diabetes mellitus. Diabetes Metab Res Rev. 2016;32(2):187–92. [PubMed] DOI: 1002/dmrr.2711
- Cellier MFM. Cell-Type Specific Determinants of NRAMP1 Expression in Professional Phagocytes. Biology (Basel). 2013;2:233–83. [PubMed] DOI: 3390/biology2010233
- Wessling-Resnick M. Nramp1 and other transporters involved in metal withholding during infection. J Biol Chem. 2015;290(31):18984–90. [PubMed] DOI: 1074/jbc.R115.643973
- Liu K, Liu Y, Li T, Li Q, Wang J, An Y, et al. Association Analysis of SLC11A1 Polymorphisms with Somatic Cell Score in Chinese Holstein Cows. Animals (Basel). 2025;15(10). [PubMed] DOI: 3390/ani15101370
- Pauline R, Devaraj DV, Sivasubramanian J, Velmurugan S, Stephen SB, Yasam SK, et al. Systemic assessment of solute carrier family 11-member A1 (rs17235409) gene polymorphism and Mycobacterium Tuberculosis Risk in Asian and caucasian population: A comprehensive updated meta-analysis. Int J Mycobacteriol. 2023;12(4):467–77. [PubMed] DOI: 4103/ijmy.ijmy_180_23
- Safdar NZ, Kietsiriroje N, Ajjan RA. The Cellular and Protein Arms of Coagulation in Diabetes: Established and Potential Targets for the Reduction of Thrombotic Risk. Int J Mol Sci. 2023;24(20). [PubMed] DOI: 3390/ijms242015328
- Reichert CO, da Cunha J, Levy D, Maselli LMF, Bydlowski SP, Spada C. Hepcidin: Homeostasis and Diseases Related to Iron Metabolism. Acta Haematol. 2017;137(4):220–36. [PubMed] DOI: 1159/000471838
- Zannat-e-Khuda, Khatun M, Chakma T, Jim TR. Coagulation Profile and D Dimer in Diabetes Patients. Barind Med Coll J. 2025;11(1):108–12. [PubMed] DOI: 70818/bmcj.v011i01.0182
- Tripodi A, Branchi A, Chantarangkul V, Clerici M, Merati G, Artoni A, et al. Hypercoagulability in patients with type 2 diabetes mellitus detected by a thrombin generation assay. J Thromb Thrombolysis. 2011;31(2):165–72. [PubMed] DOI: 1007/s11239-010-0506-0
- Ebrahim H, Asrie F, Getaneh Z. Basic Coagulation Profiles and Platelet Parameters Among Adult Type 1 and Type 2 Diabetes Patients at Dessie Referral Hospital, Northeast Ethiopia: Comparative Cross-Sectional Study. J Blood Med. 2021;12:33–42. [PubMed] DOI: 2147/JBM.S287136
- Cheng L, Fu Q, Zhou L, Fan Y, Liu F, Fan Y, et al. D-dimer as a predictor of cardiovascular outcomes in patients with diabetes mellitus. BMC Cardiovasc Disord. 2022;22(1):82. [PubMed] DOI: 1186/s12872-022-02531-x
- Olefsky JM, Glass CK. Macrophages, inflammation, and insulin resistance. Annu Rev Physiol. 2010;72:219–46. [PubMed] DOI: 1146/annurev-physiol-021909-135846
- Han J, van Hylckama Vlieg A, de Mutsert R, Rosendaal FR, van der Velde JHPM, Boone SC, et al. Associations of coagulation parameters and thrombin generation potential with the incidence of type 2 diabetes: mediating role of glycoprotein acetylation. Eur J Epidemiol. 2024;39(10):1171–81. [PubMed] DOI: 1007/s10654-024-01162-0
- Mantovani A, Busti F, Borella N, Scoccia E, Pecoraro B, Sani E, et al. Elevated plasma hepcidin concentrations are associated with an increased risk of mortality and nonfatal cardiovascular events in patients with type 2 diabetes: a prospective study. Cardiovasc Diabetol. 2024;23(1):305. [PubMed] DOI: 1186/s12933-024-02377-x
- Wang YB, Li ZP, Wang P, Wang RB, Ruan YH, Shi Z, et al. Iron dysregulation, ferroptosis, and oxidative stress in diabetic osteoporosis: Mechanisms, bone metabolism disruption, and therapeutic strategies. World J Diabetes. 2025;16(6):106720. [PubMed] DOI: 4239/wjd.v16.i6.106720
- Qiu F, Wu L, Yang G, Zhang C, Liu X, Sun X, et al. The role of iron metabolism in chronic diseases related to obesity. Mol Med. 2022;28(1):130. [PubMed] DOI: 1186/s10020-022-00558-6
- Wang X, Fang X, Zheng W, Zhou J, Song Z, Xu M, et al. Genetic Support of A Causal Relationship Between Iron Status and Type 2 Diabetes: A Mendelian Randomization Study. J Clin Endocrinol Metab. 2021;106(11):e4641–51. [PubMed] DOI: 1210/clinem/dgab454
- Cellier MFM. Developmental Control of NRAMP1 (SLC11A1) Expression in Professional Phagocytes. Biology (Basel). 2017;6(2):28. [PubMed] DOI: 3390/biology6020028
- Archer NS, Nassif NT, O’Brien BA. Genetic variants of SLC11A1 are associated with both autoimmune and infectious diseases: systematic review and meta-analysis. Genes Immun. 2015;16(4):275–83. [PubMed] DOI: 1038/gene.2015.8
- Yang JHM, Downes K, Howson JMM, Nutland S, Stevens HE, Walker NM, et al. Evidence of association with type 1 diabetes in the SLC11A1 gene region. BMC Med Genet. 2011;12:59. [PubMed] DOI: 1186/1471-2350-12-59
- Kavian Z, Sargazi S, Majidpour M, Sarhadi M, Saravani R, Shahraki M, et al. Association of SLC11A1 polymorphisms with anthropometric and biochemical parameters describing Type 2 Diabetes Mellitus. Sci Rep. 2023;13(1):6195. [PubMed] DOI: 1038/s41598-023-33239-3