Mona Raafat Elghamry 1 , Sameh Mohamed Fathy 2 , Nashwa Ebrahim Hamoda 3 , Marwa Ahmed Eloraby Elfert 4
Background & objective: Nalbuphine (agonist-antagonist) is very commonly used in the clinical practice, because of its property to alleviates pain without the side effects associated with other narcotics. This research assessed the analgesic efficacy of intrathecal morphine compared to nalbuphine in spine surgery.
Methodology: Eighty participants were randomly separated into two groups; Group M received 0.2 mg morphine, while Group N patients were administered 0.8 mg nalbuphine. Both drugs were mixed with saline to make a volume of 3 mL for intrathecal injection. The primary outcome was the duration of effective analgesia. Secondary outcomes included visual analog scale (VAS) scores, fentanyl consumption, and any observed adverse effects.
Results: Group M experienced a significantly prolonged duration of effective analgesia (P < 0.001) with significantly less postoperative fentanyl consumption compared to Group N (P < 0.001). The VAS scores were significantly lower in Group M than Group N at 1, 2, 4, and 6 hours after the procedure (all P < 0.001). Conversely, VAS scores were significantly higher in Group M than in Group N at 18 and 24 hours postoperative (P = 0.002 and 0.022, respectively). A significant increase in the incidence of postoperative nausea and vomiting (P = 0.011) and pruritus (P < 0.001) was observed in Group M compared to Group N.
Conclusion: Intrathecal nalbuphine provided analgesia in the early postoperative period and is more effective than morphine in reducing the risk of side effects.
Abbreviations: ERAS: enhanced recovery after surgery, ITM: intrathecal morphine, ITN: intrathecal nalbuphine, Keywords: Lumber spine surgery; Morphine; Nalbuphine; Postoperative pain; intrathecal
Citation: Elghamry MR, Fathy SM, Hamoda NE, Elfert MAE. Efficacy and safety of preemptive intrathecal morphine vs nalbuphine in lumbar spine. Anaesth. pain intensive care 2025;30(1):7-15. DOI: 10.35975/apic.v30i1.3096
Received: July 03, 2025; Revised: October 03, 2025; Accepted: October 03, 2025
Patients undergoing spine surgery often experience severe acute pain, particularly in the first three days postoperative, due to several factors, such as pre-existingchronic pain, opioid dependence therapy, and extensive tissue dissection during the surgical procedure.
Unfortunately, many of these patients are vulnerable to developing chronic pain that may persist for six months postoperatively.1,2
The stated objective of the enhanced recovery after surgery (ERAS) protocol is to mitigate the body's negative response to surgery. One component of this protocol is preemptive analgesia, which aims to reduce opioid consumption through effective management with regional blocks and spinal and epidural analgesia.3
Narcotic analgesics are the most popular and effective medications for managing severe pain; however, their use is limited by undesirable side effects, including nausea, respiratory depression, and vomiting. Although intrathecal (IT) opioids can reduce overall opioid consumption, they may cause delayed respiratory depression, particularly with morphine. When used in conjunction with IT local anesthetics (LA), they provide a synergistic effect that decreases opioid consumption and, consequently, their adverse effects.4
Nalbuphine, a synthetic opioid, functions as a µ-receptor antagonist and a k-receptor agonist. When used with IT LA, it offers analgesia without the undesirable side effects associated with µ-agonists, such as respiratory depression, making it a valuable adjuvant for postoperative pain management.5
We hypothesized that utilizing intrathecal nalbuphine (ITN) instead of morphine could provide effective analgesia with fewer side effects. This investigation assessed the safety and efficacy of preemptive intrathecal morphine (ITM) analgesia in comparison to nalbuphine for patients undergoing lumbar spine surgery under general anesthesia (GA).
A prospective, double-blind, randomized clinical trial was conducted at Tanta University Hospitals from May 2023 to December 2023. The trial was registered with the Pan African Clinical Trials Registry (PACTR202304865602085) following approval from the institutional ethics committee (No: 36264PR115/2/23), granted on April 4, 2023. All participants provided written informed consent before enrollment in the study.
The study population comprised 80 patients of both genders, aged between 40 and 70, with an American Society of Anesthesiologists (ASA) physical status classification of I or II. These individuals were scheduled to undergo elective posterior lumbar interbody fusion surgery at one or two levels, with or without concomitant laminectomy, under general anesthesia (GA).
Exclusion criteria included individuals with contraindications to intrathecal injection, known allergies or contraindications to the investigational drugs, a body mass index (BMI) over 35 kg/m2, chronic alcohol abuse, substance use disorders, and a documented history of major psychiatric or cognitive impairments that could impede their ability to comprehend and respond to the visual analog scale (VAS). Additionally, patients with previous lumbar surgeries or revision procedures or pre-existing neurological deficits, such as symptoms of lumbar injury or intervertebral nerve damage, as detected by a neurosurgical physician based on imaging and physical investigations, were excluded.
Randomization was facilitated by a computer-generated random number sequence, with assignments concealed in sealed, opaque envelopes to maintain allocation concealment.
Participants were allocated to two equal groups at random (40 each) with a 1:1 allocation ratio. Group M (control group) consisted of patients who received an intrathecal (IT) injection of 0.2 mg morphine sulphate diluted in 0.9% saline solution, resulting in a total volume of 3 mL. Group N (experimental group) comprised participants who were administered an IT injection of 0.8 mg nalbuphine hydrochloride (Nalfin®, Memphis, Egypt) diluted in 0.9% saline solution, yielding a total volume of 3 mL.
The nurse was unaware of the specifics of the research and could not see the assigned individuals in their respective enrollment groups; she prepared the anesthetic mixtures in two identical syringes labeled syringes 1 and 2. The IT injection and administration of GA were performed by an anesthetist who had no additional role in the study, while another anesthetist, unaware of group allocation, recorded the outcomes. Furthermore, both patients and nurses in the post-anesthesia care unit (PACU) were unaware of the groups to which they were assigned.
At pre-anesthetic counseling, a physical examination, laboratory investigations, and documentation of any sensory or motor deficits were conducted for all patients. Furthermore, the patients received instructions on how to use the 10-cm VAS for pain (0 = no pain, 10 = worst imaginable pain) and the patient-controlled analgesia (PCA) equipment.
Before the IT injection, a peripheral intravenous (IV) catheter was inserted in the operating room and monitors for non-invasive blood pressure, pulse oximetry, and electrocardiography were connected. Initial oxygen saturation measurements, mean arterial blood pressure (MAP), and heart rate (HR) were documented, followed by ongoing observation for any abnormal changes, which would be recorded as potential side effects.
The IT injection was administered while the patient was seated and under strict aseptic conditions, following the group assignment. The procedure was performed at least 20 minutes before the administration of GA, utilizing a 25G Whitacre Pencil Point spinal needle to inject into the L3-4 or L4-5 interspinous region.
Every patient received the same GA protocol, consisting of IV induction with 2-2.5 mg/kg of propofol, 1.5 mg/kg of lidocaine, 2 µg/kg of fentanyl, and 0.2 mg/kg of cisatracurium. Following endotracheal intubation, the concentration of carbon dioxide at the end of each breath (end-tidal CO2 or EtCO2) was measured. Electrodes were used to monitor the bispectral index (BIS), and a Foley catheter was inserted. Additionally, 8 mg of dexamethasone and 10 mg per kilogram of body weight of tranexamic acid were administered IV unless contraindicated. Anesthesia was maintained using sevoflurane in a balanced combination of oxygen and air, with the sevoflurane concentration adjusted to keep the BIS value within the range of 40 to 60, thus providing an appropriate level of anesthesia. Positive pressure ventilation was calibrated with a tidal volume of 6-8 mL/kg, and the respiratory rate was adjusted to maintain EtCO2 levels between 30 and 40 mmHg. To maintain a train-of-four count of 1, a dose of 0.03 mg/kg of cisatracurium was delivered as needed. In all groups, an additional dose of fentanyl (1 µg/kg) was administered IV if there was a 20% increase in HR or MAP compared to the initial measurement. The total amount of fentanyl used during surgery was recorded.
Thirty minutes before the end of the surgery, patients were administered 4 mg of ondansetron IV, 1 g of paracetamol IV every six hours, and 30 mg of IV ketorolac every eight hours. Subsequently, the neuromuscular blockade was reversed, and the endotracheal tube was removed. All patients were then transferred to the PACU and received IV fentanyl through PCA. Additionally, the Foley catheter was removed. The PCA pump was configured with the following parameters: a concentration of 10 µg/mL, an on-demand dose of 20 µg, a lockout interval of 15 minutes, no background infusion, and a maximum dose of 300 µg within four hours.
The pump was initiated for patients in both groups upon their first request for analgesia or when VAS > 3, whichever occurred first. The bolus dose and lockout interval were adjusted to optimize pain control.
The primary outcome of this research was the length of effective analgesia, defined as the interval from the end of the IT injection to the first dose of rescue analgesia. The VAS was utilized to evaluate pain after surgery upon arrival to the PACU and then at 1, 2, 4, 6, 12, 18, and 24 hours postoperatively. Total fentanyl consumption (intraoperative and up to 24 hours postoperative) was calculated, excluding the induction dose. Adverse effects observed up to 24 hours postoperative were secondary measurements.
The adverse consequences assessed included the prevalence of postoperative nausea and vomiting (PONV), which were evaluated using a categorical scoring system. This standardized classification facilitated a systematic assessment and documentation of PONV episodes following the operation, enabling appropriate clinical management and data collection for subsequent analysis. The scoring designations were as follows: a score of 0 indicated an absence of symptoms; a score of 1 denoted the presence of symptoms (nausea and/or vomiting) without the need for pharmacological intervention; and a score of 2 was assigned when symptoms were present and required treatment, which was administered via IV with 4 mg of ondansetron.
Pruritus was assessed as follows: 0 = none, 1 = present. If pruritus was severe and intolerable, naloxone was administered.
Respiratory depression was characterized by the concurrent manifestation of bradypnea, defined as a respiratory rate below 10 breaths per minute, and hypoxia, a state of reduced oxygen levels in the blood or tissues (oxygen saturation < 95%). Initially, patients were treated with supportive therapy, including oxygen administration via face mask, and if symptoms persisted, mechanical ventilation and naloxone administration were employed.
The sedation level was assessed in the PACU one hour after the surgical procedure using the Ramsay sedation scale. This validated instrument employs a numerical scoring system ranging from 1 to 6, each representing a distinct level of consciousness and responsiveness. Specifically, a score of 1 denotes an anxious and agitated state; 2 indicates a cooperative demeanor; 3 signifies a state of responsiveness to verbal commands only; 4 corresponds to sleep with arousability to light tactile stimuli or simple verbal cues; 5 represents a deeper level of sleep, requiring strong physical stimulation for arousal; and a score of 6 is assigned when the individual is in a state of unrousable sleep.6
Urine retention was managed with the insertion of a Foley catheter.
2.1. Statistical analysis
The sample size was determined using G-power software , with a statistical power of 80% and a confidence level of 95%. Based on a previous study.7, the effect size was calculated to be 0.592, determined by comparing the average duration of effective pain relief, which was 275 ± 228 minutes in the group receiving ITM and 176 ± 62 minutes in the group receiving nalbuphine. To identify a substantial disparity between the two groups, a minimum of 36 individuals per group was required. Assuming a dropout rate of 10%, the calculated minimum total sample size was 80 patients, with 40 patients assigned to each group.
The data were entered into the computer and analyzed using the IBM SPSS software package version 20.0 (Armonk, NY: IBM Corp). Categorical data were quantified using numerical values and percentages, and the chi-square test was employed to compare the two groups. Fisher's exact test was used instead if the expected count of fewer than 5 was observed in more than 20% of the cells. The Shapiro-Wilk test was utilized to assess the normality of the continuous data. Quantitative data were represented by the range (minimum and maximum values), mean, standard deviation, median, and interquartile range (IQR). The Student's t-test was employed to compare two groups based on quantitative data that followed a normal distribution, whereas the Mann-Whitney test was utilized for quantitative variables that did not follow a normal distribution. The Friedman test was used to compare more than two periods or stages for quantitative variables that were irregularly distributed. Subsequently, Dunn's post hoc test was conducted for pairwise comparisons. The statistical significance of the results was assessed at a significance level of 5%.
Initially, 93 individuals were evaluated for eligibility during the eight-month research period. However, only 80 patients were eligible to participate in the experiment after applying all the inclusion and exclusion criteria. The remaining applicants were excluded because they did not want to participate (5 patients) or did not fulfill the requirements (8 patients). Eighty patients were eligible for inclusion, with forty allocated to each research group at random. Two patients were not followed up during the trial, and two had a more complex and lengthier surgical procedure than planned. Hence, the final analysis was performed on 38 participants in group M and 40 in group N (Figure 1).
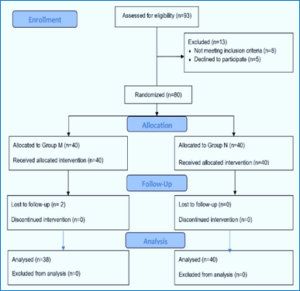
Figure 1: Study flow diagram
The patient demographics and operations were consistent across all groups (Table 1).
The effective analgesia period was significantly prolonged in group M (P < 0.001). Fentanyl consumption after surgery was significantly decreased in group M (P < 0.001), although no participant in either group required additional intraoperative fentanyl rescue doses (Table 2).
Group M had significantly lower VAS scores than Group N at 1, 2, 4, and 6 hours postoperatively (P<0.001 for all). The results differed at 18 and 24 hours, with group M showing higher scores in respective P values of 0.002 and 0.022. The VAS scores for the groups were similar upon arrival at the PACU and twelve hours after surgery, with respective P values of 0.481 and 0.318 (Figure 2).
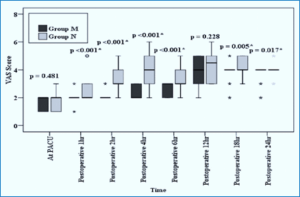
Figure 2: Comparative VAS scores in two groups at different time intervals
Group M had a significantly higher occurrence of PONV and pruritus than group N, with respective P values of 0.011 and <0.001. No significant differences were detected between the groups regarding sedation and urine retention. No other adverse consequences were noted (Table 2).
In spinal procedures, our work contributes modestly to the ongoing scholarly discussion on intrathecal adjuvant drugs. Up to our best knowledge, it is the first research to discuss administration of ITN alone not as an adjuvant drug and this study one of few studies that delt with intrathecal analgesia for spine surgeries. In this particular clinical setting, the results indicate that ITN has the potential to serve as a viable substitute for pure opioid agonists in pain management without significant adverse effects. Notably, ITN has not been extensively studied regarding its analgesic effects during the postoperative period. The results of our study enhance the discussion on the effectiveness of ITN for achieving pain relief after surgery. The data indicated that, regardless of a longer duration of pain alleviation and a lesser amount of fentanyl used postoperatively in patients receiving ITM, the inclusion of morphine was significantly associated with a higher occurrence of negative outcomes, including PONV and pruritus.
The mechanism of action of intrathecal opioids is complex. After their administration intrathecally, they distributed into multiple compartments and their duration of action depends mainly on their lipid solubility. The more lipid soluble drugs have short duration of action.11
Intrathecal opioids pass towards the brain via the CSF. Moreover, opioids diffuse spinally to bind nonspecific receptors in the white matter and to specific receptors in the gray matter. Also, they pass into the epidural space with binding to the epidural fat then pass to the blood by vascular reuptake of epidural vessels.12
The literature addresses the optimal dosing of ITN, which remains a point of contention. In our study, our cases were administered ITN at a dose of 0.8 mg, based on previous researches.7,8,9 Nalbuphine has a ceiling effect, meaning that the analgesic effect levels off after a specific dose, and increasing the dosage does not provide additional pain relief. Optimal analgesia with minimal adverse consequences was observed with ITN doses ranging from 0.8 to 1.6 mg, according to a meta-analysis conducted by Yu et al. (2022),10 which examined doses between 0.4 and 2.4 mg. They suggested that ITN provides comparable pain relief to opioids but with a lower incidence of adverse effects such as hypotension, shivering, and itching.
Contrary to our results, Mukherjee et al. (2011) conducted a study examining three varying doses (0.2, 0.4, and 0.8 mg) of ITN.11 They aimed to identify the most effective dose for long-lasting pain relief while minimizing side effects. Their findings demonstrated that both 0.4 and 0.8 mg doses resulted in extended periods of pain relief; however, the 0.8 mg dose was associated with a greater occurrence of adverse effects, including hypotension, bradycardia, nausea, vomiting, and pruritus.
In alignment with our results, Culebras et al.(2000) compared various dosages of ITN and morphine in cesarean deliveries.7 Their findings indicated that administering ITN at a dosage of 0.8 mg resulted in effective pain relief during and after surgery, with no notable adverse effects. In contrast, morphine demonstrated a longer-lasting analgesic effect but was linked to a higher incidence of unwanted symptoms, such as itching and PONV.
Supporting our findings, Jyothi et al.(2014) determined that 0.8 mg of ITN (as opposed to 1.6 or 2.4 mg) had a longer half-life, provided high-quality analgesia, and did not lead to any major adverse effects.8 Borah et al. (2018) reached a similar conclusion, finding that 0.4 and 0.8 mg of ITN offered prolonged analgesia with comparable efficacy and a more favorable side effect profile than the 1.6 mg dose.9
In contrast, Singhal et al. (2028) established that 0.8 mg of ITN extended analgesia but significantly increased side effects, particularly PONV, compared to 0.4 mg.13 Conversely, Ahluwalia et al. (2015) found that 0.8 mg of ITN effectively reduced intraoperative and postoperative pain while producing minimal adverse effects compared to a placebo group.14
Regarding ITM, De Bie et al. (2020) discovered that patients undergoing lumbar fusion surgery who received 100 µg of ITM had reduced morphine consumption at 24 and 48 hours postoperatively, although there was no change in the incidence of morphine-related complications.15 In a study by Al Khawaja. (2021), subarachnoid administration of 0.2 mg morphine was conducted directly during the intraoperative period.16 The author concluded that this approach facilitated early hospital discharge by providing prolonged postoperative analgesia, promoting early mobilization without complications, and reducing the need for parenteral opioids or PCA.
Evidence from multiple studies.6, 17 supports using 0.2 mg of ITM as effective and safe for analgesia following lumbar surgery. Postoperative analgesia remained effective even at higher doses (0.35% ITM).18 compared to placebo, with no discernible increase in adverse effects. Consequently, we used a 0.2 mg ITM dosage in our trial.
Our findings are consistent with those of Yoon et al. (2002), who compared ITM, ITN, and a combination of both for elective cesarean procedures, albeit using different dosages.19
Researchers discovered that while the ITN group experienced a decreased incidence of pruritus, the duration of analgesia was significantly prolonged when ITN was administered alone. Remarkably, all three groups exhibited similar rates of PONV. Similarly, a clinical trial conducted by Hassan et al. (2002) confirmed that nalbuphine effectively reduced the occurrence of pruritus, although morphine was found to be a superior analgesic.20
In the context of orthopedic surgery, Fournier et al. (2002) found that ITN produced analgesia more rapidly but had a shorter duration.21 In contrast, the ITM group required less analgesic medication postoperatively. Nevertheless, contrary to our expectations, both groups reported similar rates of adverse effects.
Morphine as a hydrophilic drug crosses then blood brain barrier slowly with less affinity to bind to the epidural fat. While morphine has a high affinity to bind to the specific grey matter receptors along with slow vascular reuptake. This distribution results in longer-lasting and higher concentrations of morphine in the CSF which in turn leads to slow onset and prolonged duration of action with the possibility of delayed onset of respiratory depression.22
When comparing opioids other than morphine, Jain et al. (2022) found that ITN provided a longer duration of analgesia and reduced the need for postoperative rescue analgesia compared to fentanyl.23 In contrast, our research showed no significant differences in hemodynamic parameters or adverse effects between the groups. Similarly, Bindra et al. (2018) suggested that intravenous naloxone could replace intrathecal fentanyl (IVF) during cesarean sections.24
Amin et al. (2020) discovered that adding 1 mg of ITN to 12.5 mg of bupivacaine improved analgesia, accelerated the onset of the block, and slowed the regression of sensory block compared to 2.5 mg of midazolam.25 Our findings are consistent with those of Shakooh et al. (2014), who investigated the combination of nalbuphine with bupivacaine for analgesic efficacy.26 They found that adding ITN improved the quality of analgesia both intraoperatively and postoperatively, with negligible adverse effects.
Being a lipophilic drug, nalbuphine has rapid onset and short duration of action because of rapid vascular reuptake and more affinity to bind epidural fat which in turn lead to fast decrease in its CSF concentration. It has agonist activity on kappa receptor and antagonist activity on mu receptor so regarding to respiratory depression it has a ceiling effect.27
According to Tiwari et al. (2013), incorporating ITN with bupivacaine prolonged postoperative analgesia without adverse effects.28 Their study employed lower doses of nalbuphine (0.2 mg and 0.4 mg), which could explain the discrepancy in findings.
So, from the results of the study, we introduce an idea to apply perioperative analgesia for spine surgeries through the intrathecal administration of nalbuphine. This technique is well known in anesthesia practice and no need for special training nor special equipment like that of nerve block or fascial plane block. Moreover, nalbuphine is an available, cheap drug with no significant adverse effects and unlike other opioids no need to have license to obtain it.
This study has a few limitations. First, pain scores were not evaluated during movement. Second, the study duration was short, and we did not follow up with the patients for chronic pain outcomes. Third, a single-shot injection was used to determine nalbuphine's exact duration of action at the lowest effective dose and also, we could not add it with local anesthetics for the same cause. Fourth, it was single center study for one type of surgery and small sample size so further multi-center studies are recommended in different types of surgeries to explore the addition of other adjuvants, such as dexamethasone, to ITN to extend the duration of the analgesic effect. Additionally, catheter insertion could be investigated.
In conclusion, patients undergoing lumbar spine surgery could benefit from early analgesia after surgery with 0.8 mg of ITN, which may reduce the likelihood of adverse effects, making it a viable alternative to ITM.
6. Data availability
The data supporting this study's findings are available upon reasonable request sent to the e-mail of the corresponding author (drmonagh19802000@gmail.com). The data are not publicly available due to privacy or ethical restrictions.
7. Conflict of interest
All authors declare that there was no potential conflict of interest is relevant to this article.
8. Funding
No external funding to declare.
9. Authors contribution
M R E: concept, study design, conduction of the work, data collection, read and approved the final manuscript
S M F: concept, study design, data collection, revising it critically for important intellectual content, read and approved the final manuscript
N E H: concept, study design, data analysis, first draft of the manuscript, read and approved the final manuscript
M AE E: concept, study design, conduction of the work, data collection, first draft of the manuscript, read and approved the final manuscript
- Mona Raafat Elghamry, Assistant professor, Department of Anesthesia, Surgical ICU, and Pain Medicine, Faculty of Medicine, Tanta University, Elgeish Street, Tanta, Egypt (Zip code: 31527) Tel: +201060101867 Email: drmonagh19802000@gmail.com
- Sameh Mohamed Fathy, Assistant professor, Department of Anesthesia, Surgical ICU, and Pain Medicine, Faculty of Medicine, Tanta University, Elgeish Street, Tanta, Egypt (Zip code: 31527) Email:Samehsadek923@gmail.com
- Nashwa Ebrahim Hamoda Lecturer, Department of Anesthesia, Surgical ICU, and Pain Medicine, Faculty of Medicine, TantaUniversity, Elgeish Street, Tanta, Egypt (Zip code: 31527) Email: nashwa741987@gmail.com
- Marwa Ahmed Eloraby Elfert Lecturer, Department of Anesthesia, Surgical ICU, and Pain Medicine, Faculty of Medicine, Tanta University, Elgeish Street, Tanta, Egypt (Zip code: 31527) E-mail: marwafert@gmail.com
ABSTRACT
Background & objective: Nalbuphine (agonist-antagonist) is very commonly used in the clinical practice, because of its property to alleviates pain without the side effects associated with other narcotics. This research assessed the analgesic efficacy of intrathecal morphine compared to nalbuphine in spine surgery.
Methodology: Eighty participants were randomly separated into two groups; Group M received 0.2 mg morphine, while Group N patients were administered 0.8 mg nalbuphine. Both drugs were mixed with saline to make a volume of 3 mL for intrathecal injection. The primary outcome was the duration of effective analgesia. Secondary outcomes included visual analog scale (VAS) scores, fentanyl consumption, and any observed adverse effects.
Results: Group M experienced a significantly prolonged duration of effective analgesia (P < 0.001) with significantly less postoperative fentanyl consumption compared to Group N (P < 0.001). The VAS scores were significantly lower in Group M than Group N at 1, 2, 4, and 6 hours after the procedure (all P < 0.001). Conversely, VAS scores were significantly higher in Group M than in Group N at 18 and 24 hours postoperative (P = 0.002 and 0.022, respectively). A significant increase in the incidence of postoperative nausea and vomiting (P = 0.011) and pruritus (P < 0.001) was observed in Group M compared to Group N.
Conclusion: Intrathecal nalbuphine provided analgesia in the early postoperative period and is more effective than morphine in reducing the risk of side effects.
Abbreviations: ERAS: enhanced recovery after surgery, ITM: intrathecal morphine, ITN: intrathecal nalbuphine, Keywords: Lumber spine surgery; Morphine; Nalbuphine; Postoperative pain; intrathecal
Citation: Elghamry MR, Fathy SM, Hamoda NE, Elfert MAE. Efficacy and safety of preemptive intrathecal morphine vs nalbuphine in lumbar spine. Anaesth. pain intensive care 2025;30(1):7-15. DOI: 10.35975/apic.v30i1.3096
Received: July 03, 2025; Revised: October 03, 2025; Accepted: October 03, 2025
1. INTRODUCTION
Patients undergoing spine surgery often experience severe acute pain, particularly in the first three days postoperative, due to several factors, such as pre-existingchronic pain, opioid dependence therapy, and extensive tissue dissection during the surgical procedure.
Unfortunately, many of these patients are vulnerable to developing chronic pain that may persist for six months postoperatively.1,2
The stated objective of the enhanced recovery after surgery (ERAS) protocol is to mitigate the body's negative response to surgery. One component of this protocol is preemptive analgesia, which aims to reduce opioid consumption through effective management with regional blocks and spinal and epidural analgesia.3
Narcotic analgesics are the most popular and effective medications for managing severe pain; however, their use is limited by undesirable side effects, including nausea, respiratory depression, and vomiting. Although intrathecal (IT) opioids can reduce overall opioid consumption, they may cause delayed respiratory depression, particularly with morphine. When used in conjunction with IT local anesthetics (LA), they provide a synergistic effect that decreases opioid consumption and, consequently, their adverse effects.4
Nalbuphine, a synthetic opioid, functions as a µ-receptor antagonist and a k-receptor agonist. When used with IT LA, it offers analgesia without the undesirable side effects associated with µ-agonists, such as respiratory depression, making it a valuable adjuvant for postoperative pain management.5
We hypothesized that utilizing intrathecal nalbuphine (ITN) instead of morphine could provide effective analgesia with fewer side effects. This investigation assessed the safety and efficacy of preemptive intrathecal morphine (ITM) analgesia in comparison to nalbuphine for patients undergoing lumbar spine surgery under general anesthesia (GA).
2. METHODOLOGY
A prospective, double-blind, randomized clinical trial was conducted at Tanta University Hospitals from May 2023 to December 2023. The trial was registered with the Pan African Clinical Trials Registry (PACTR202304865602085) following approval from the institutional ethics committee (No: 36264PR115/2/23), granted on April 4, 2023. All participants provided written informed consent before enrollment in the study.
The study population comprised 80 patients of both genders, aged between 40 and 70, with an American Society of Anesthesiologists (ASA) physical status classification of I or II. These individuals were scheduled to undergo elective posterior lumbar interbody fusion surgery at one or two levels, with or without concomitant laminectomy, under general anesthesia (GA).
Exclusion criteria included individuals with contraindications to intrathecal injection, known allergies or contraindications to the investigational drugs, a body mass index (BMI) over 35 kg/m2, chronic alcohol abuse, substance use disorders, and a documented history of major psychiatric or cognitive impairments that could impede their ability to comprehend and respond to the visual analog scale (VAS). Additionally, patients with previous lumbar surgeries or revision procedures or pre-existing neurological deficits, such as symptoms of lumbar injury or intervertebral nerve damage, as detected by a neurosurgical physician based on imaging and physical investigations, were excluded.
Randomization was facilitated by a computer-generated random number sequence, with assignments concealed in sealed, opaque envelopes to maintain allocation concealment.
Participants were allocated to two equal groups at random (40 each) with a 1:1 allocation ratio. Group M (control group) consisted of patients who received an intrathecal (IT) injection of 0.2 mg morphine sulphate diluted in 0.9% saline solution, resulting in a total volume of 3 mL. Group N (experimental group) comprised participants who were administered an IT injection of 0.8 mg nalbuphine hydrochloride (Nalfin®, Memphis, Egypt) diluted in 0.9% saline solution, yielding a total volume of 3 mL.
The nurse was unaware of the specifics of the research and could not see the assigned individuals in their respective enrollment groups; she prepared the anesthetic mixtures in two identical syringes labeled syringes 1 and 2. The IT injection and administration of GA were performed by an anesthetist who had no additional role in the study, while another anesthetist, unaware of group allocation, recorded the outcomes. Furthermore, both patients and nurses in the post-anesthesia care unit (PACU) were unaware of the groups to which they were assigned.
At pre-anesthetic counseling, a physical examination, laboratory investigations, and documentation of any sensory or motor deficits were conducted for all patients. Furthermore, the patients received instructions on how to use the 10-cm VAS for pain (0 = no pain, 10 = worst imaginable pain) and the patient-controlled analgesia (PCA) equipment.
Before the IT injection, a peripheral intravenous (IV) catheter was inserted in the operating room and monitors for non-invasive blood pressure, pulse oximetry, and electrocardiography were connected. Initial oxygen saturation measurements, mean arterial blood pressure (MAP), and heart rate (HR) were documented, followed by ongoing observation for any abnormal changes, which would be recorded as potential side effects.
The IT injection was administered while the patient was seated and under strict aseptic conditions, following the group assignment. The procedure was performed at least 20 minutes before the administration of GA, utilizing a 25G Whitacre Pencil Point spinal needle to inject into the L3-4 or L4-5 interspinous region.
Every patient received the same GA protocol, consisting of IV induction with 2-2.5 mg/kg of propofol, 1.5 mg/kg of lidocaine, 2 µg/kg of fentanyl, and 0.2 mg/kg of cisatracurium. Following endotracheal intubation, the concentration of carbon dioxide at the end of each breath (end-tidal CO2 or EtCO2) was measured. Electrodes were used to monitor the bispectral index (BIS), and a Foley catheter was inserted. Additionally, 8 mg of dexamethasone and 10 mg per kilogram of body weight of tranexamic acid were administered IV unless contraindicated. Anesthesia was maintained using sevoflurane in a balanced combination of oxygen and air, with the sevoflurane concentration adjusted to keep the BIS value within the range of 40 to 60, thus providing an appropriate level of anesthesia. Positive pressure ventilation was calibrated with a tidal volume of 6-8 mL/kg, and the respiratory rate was adjusted to maintain EtCO2 levels between 30 and 40 mmHg. To maintain a train-of-four count of 1, a dose of 0.03 mg/kg of cisatracurium was delivered as needed. In all groups, an additional dose of fentanyl (1 µg/kg) was administered IV if there was a 20% increase in HR or MAP compared to the initial measurement. The total amount of fentanyl used during surgery was recorded.
Thirty minutes before the end of the surgery, patients were administered 4 mg of ondansetron IV, 1 g of paracetamol IV every six hours, and 30 mg of IV ketorolac every eight hours. Subsequently, the neuromuscular blockade was reversed, and the endotracheal tube was removed. All patients were then transferred to the PACU and received IV fentanyl through PCA. Additionally, the Foley catheter was removed. The PCA pump was configured with the following parameters: a concentration of 10 µg/mL, an on-demand dose of 20 µg, a lockout interval of 15 minutes, no background infusion, and a maximum dose of 300 µg within four hours.
The pump was initiated for patients in both groups upon their first request for analgesia or when VAS > 3, whichever occurred first. The bolus dose and lockout interval were adjusted to optimize pain control.
The primary outcome of this research was the length of effective analgesia, defined as the interval from the end of the IT injection to the first dose of rescue analgesia. The VAS was utilized to evaluate pain after surgery upon arrival to the PACU and then at 1, 2, 4, 6, 12, 18, and 24 hours postoperatively. Total fentanyl consumption (intraoperative and up to 24 hours postoperative) was calculated, excluding the induction dose. Adverse effects observed up to 24 hours postoperative were secondary measurements.
The adverse consequences assessed included the prevalence of postoperative nausea and vomiting (PONV), which were evaluated using a categorical scoring system. This standardized classification facilitated a systematic assessment and documentation of PONV episodes following the operation, enabling appropriate clinical management and data collection for subsequent analysis. The scoring designations were as follows: a score of 0 indicated an absence of symptoms; a score of 1 denoted the presence of symptoms (nausea and/or vomiting) without the need for pharmacological intervention; and a score of 2 was assigned when symptoms were present and required treatment, which was administered via IV with 4 mg of ondansetron.
Pruritus was assessed as follows: 0 = none, 1 = present. If pruritus was severe and intolerable, naloxone was administered.
Respiratory depression was characterized by the concurrent manifestation of bradypnea, defined as a respiratory rate below 10 breaths per minute, and hypoxia, a state of reduced oxygen levels in the blood or tissues (oxygen saturation < 95%). Initially, patients were treated with supportive therapy, including oxygen administration via face mask, and if symptoms persisted, mechanical ventilation and naloxone administration were employed.
The sedation level was assessed in the PACU one hour after the surgical procedure using the Ramsay sedation scale. This validated instrument employs a numerical scoring system ranging from 1 to 6, each representing a distinct level of consciousness and responsiveness. Specifically, a score of 1 denotes an anxious and agitated state; 2 indicates a cooperative demeanor; 3 signifies a state of responsiveness to verbal commands only; 4 corresponds to sleep with arousability to light tactile stimuli or simple verbal cues; 5 represents a deeper level of sleep, requiring strong physical stimulation for arousal; and a score of 6 is assigned when the individual is in a state of unrousable sleep.6
Urine retention was managed with the insertion of a Foley catheter.
2.1. Statistical analysis
The sample size was determined using G-power software , with a statistical power of 80% and a confidence level of 95%. Based on a previous study.7, the effect size was calculated to be 0.592, determined by comparing the average duration of effective pain relief, which was 275 ± 228 minutes in the group receiving ITM and 176 ± 62 minutes in the group receiving nalbuphine. To identify a substantial disparity between the two groups, a minimum of 36 individuals per group was required. Assuming a dropout rate of 10%, the calculated minimum total sample size was 80 patients, with 40 patients assigned to each group.
The data were entered into the computer and analyzed using the IBM SPSS software package version 20.0 (Armonk, NY: IBM Corp). Categorical data were quantified using numerical values and percentages, and the chi-square test was employed to compare the two groups. Fisher's exact test was used instead if the expected count of fewer than 5 was observed in more than 20% of the cells. The Shapiro-Wilk test was utilized to assess the normality of the continuous data. Quantitative data were represented by the range (minimum and maximum values), mean, standard deviation, median, and interquartile range (IQR). The Student's t-test was employed to compare two groups based on quantitative data that followed a normal distribution, whereas the Mann-Whitney test was utilized for quantitative variables that did not follow a normal distribution. The Friedman test was used to compare more than two periods or stages for quantitative variables that were irregularly distributed. Subsequently, Dunn's post hoc test was conducted for pairwise comparisons. The statistical significance of the results was assessed at a significance level of 5%.
3. RESULTS
Initially, 93 individuals were evaluated for eligibility during the eight-month research period. However, only 80 patients were eligible to participate in the experiment after applying all the inclusion and exclusion criteria. The remaining applicants were excluded because they did not want to participate (5 patients) or did not fulfill the requirements (8 patients). Eighty patients were eligible for inclusion, with forty allocated to each research group at random. Two patients were not followed up during the trial, and two had a more complex and lengthier surgical procedure than planned. Hence, the final analysis was performed on 38 participants in group M and 40 in group N (Figure 1).

Figure 1: Study flow diagram
The patient demographics and operations were consistent across all groups (Table 1).
| Table 1: Comparative demographic data and characteristics of the surgery | |||
| Variables | Group M (n = 38) |
Group N (n = 40) |
P-value |
| -Age (years) | 55.32 ± 6.54 | 55.80 ± 7.03 | 0.754 |
| Gender | |||
| · Male | 20 (52.6) | 22 (55) | 0.834 |
| · Female | 18 (47.4) | 18 (45) | |
| ASA classification | |||
| · I | 24 (63.2) | 28 (70) | 0.522 |
| · II | 14 (36.8) | 12 (30) | |
| BMI (kg/m2) | 28.02 ± 3.63 | 28.39 ± 3.5 | 0.649 |
| Level of surgery | |||
| · L4–5 | 14 (36.8) | 14 (35) | 0.967 |
| · L5–S1 | 16 (42.1) | 18 (45) | |
| · L4–S1 | 8 (21.1) | 8 (20) | |
| Laminectomy | |||
| · No | 17 (44.7) | 19 (47.5) | 0.807 |
| · Yes | 21 (55.3) | 21 (52.5) | |
| Duration of operation (min) | 132.24 ± 31.67 | 128.95 ± 29.87 | 0.638 |
| Data presented as mean ± SD or n (%); P < 0,05 | |||
The effective analgesia period was significantly prolonged in group M (P < 0.001). Fentanyl consumption after surgery was significantly decreased in group M (P < 0.001), although no participant in either group required additional intraoperative fentanyl rescue doses (Table 2).
Group M had significantly lower VAS scores than Group N at 1, 2, 4, and 6 hours postoperatively (P<0.001 for all). The results differed at 18 and 24 hours, with group M showing higher scores in respective P values of 0.002 and 0.022. The VAS scores for the groups were similar upon arrival at the PACU and twelve hours after surgery, with respective P values of 0.481 and 0.318 (Figure 2).

Figure 2: Comparative VAS scores in two groups at different time intervals
Group M had a significantly higher occurrence of PONV and pruritus than group N, with respective P values of 0.011 and <0.001. No significant differences were detected between the groups regarding sedation and urine retention. No other adverse consequences were noted (Table 2).
| Table 2: Comparative duration of effective analgesia, fentanyl consumption, and side effects | |||
| Variables | Group M (n = 38) |
Group N (n = 40) |
P-value |
| PONV | |||
| · Grade 0 | 26 (68.4) | 37 (92.5) | MC P = 0.011* |
| · Grade 1 | 7 (18.4) | 3 (7.5) | |
| · Grade 2 | 5 (13.2) | 0 (0) | |
| Pruritus | 14 (36.8) | 0 (0) | < 0.001* |
| Sedation score | |||
| · Cooperative | 22 (57.9) | 25 (62.5) | MC P = .652 |
| · Respond to verbal commands only | 13 (34.2) | 14 (35.0) | |
| · Asleep but response to a light tactile stimulus or a sample verbal command | 3 (7.9) | 1 (2.5) | |
| Urine retention | 5 (13.2) | 1 (2.5) | MC P = 0.104 |
| Postoperative fentanyl consumption (µg) | 65.26 ± 16.56 | 107.5 ± 22.5 | < 0.001* |
| Duration of effective analgesia (min) | 947.21 ± 70.93 | 311.23 ± 44.86 | < 0.001* |
| MC: Monte Carlo; Data presented as mean ± SD or n (%); *P < 0.05 is Statistically significant | |||
4. DISCUSSION
In spinal procedures, our work contributes modestly to the ongoing scholarly discussion on intrathecal adjuvant drugs. Up to our best knowledge, it is the first research to discuss administration of ITN alone not as an adjuvant drug and this study one of few studies that delt with intrathecal analgesia for spine surgeries. In this particular clinical setting, the results indicate that ITN has the potential to serve as a viable substitute for pure opioid agonists in pain management without significant adverse effects. Notably, ITN has not been extensively studied regarding its analgesic effects during the postoperative period. The results of our study enhance the discussion on the effectiveness of ITN for achieving pain relief after surgery. The data indicated that, regardless of a longer duration of pain alleviation and a lesser amount of fentanyl used postoperatively in patients receiving ITM, the inclusion of morphine was significantly associated with a higher occurrence of negative outcomes, including PONV and pruritus.
The mechanism of action of intrathecal opioids is complex. After their administration intrathecally, they distributed into multiple compartments and their duration of action depends mainly on their lipid solubility. The more lipid soluble drugs have short duration of action.11
Intrathecal opioids pass towards the brain via the CSF. Moreover, opioids diffuse spinally to bind nonspecific receptors in the white matter and to specific receptors in the gray matter. Also, they pass into the epidural space with binding to the epidural fat then pass to the blood by vascular reuptake of epidural vessels.12
The literature addresses the optimal dosing of ITN, which remains a point of contention. In our study, our cases were administered ITN at a dose of 0.8 mg, based on previous researches.7,8,9 Nalbuphine has a ceiling effect, meaning that the analgesic effect levels off after a specific dose, and increasing the dosage does not provide additional pain relief. Optimal analgesia with minimal adverse consequences was observed with ITN doses ranging from 0.8 to 1.6 mg, according to a meta-analysis conducted by Yu et al. (2022),10 which examined doses between 0.4 and 2.4 mg. They suggested that ITN provides comparable pain relief to opioids but with a lower incidence of adverse effects such as hypotension, shivering, and itching.
Contrary to our results, Mukherjee et al. (2011) conducted a study examining three varying doses (0.2, 0.4, and 0.8 mg) of ITN.11 They aimed to identify the most effective dose for long-lasting pain relief while minimizing side effects. Their findings demonstrated that both 0.4 and 0.8 mg doses resulted in extended periods of pain relief; however, the 0.8 mg dose was associated with a greater occurrence of adverse effects, including hypotension, bradycardia, nausea, vomiting, and pruritus.
In alignment with our results, Culebras et al.(2000) compared various dosages of ITN and morphine in cesarean deliveries.7 Their findings indicated that administering ITN at a dosage of 0.8 mg resulted in effective pain relief during and after surgery, with no notable adverse effects. In contrast, morphine demonstrated a longer-lasting analgesic effect but was linked to a higher incidence of unwanted symptoms, such as itching and PONV.
Supporting our findings, Jyothi et al.(2014) determined that 0.8 mg of ITN (as opposed to 1.6 or 2.4 mg) had a longer half-life, provided high-quality analgesia, and did not lead to any major adverse effects.8 Borah et al. (2018) reached a similar conclusion, finding that 0.4 and 0.8 mg of ITN offered prolonged analgesia with comparable efficacy and a more favorable side effect profile than the 1.6 mg dose.9
In contrast, Singhal et al. (2028) established that 0.8 mg of ITN extended analgesia but significantly increased side effects, particularly PONV, compared to 0.4 mg.13 Conversely, Ahluwalia et al. (2015) found that 0.8 mg of ITN effectively reduced intraoperative and postoperative pain while producing minimal adverse effects compared to a placebo group.14
Regarding ITM, De Bie et al. (2020) discovered that patients undergoing lumbar fusion surgery who received 100 µg of ITM had reduced morphine consumption at 24 and 48 hours postoperatively, although there was no change in the incidence of morphine-related complications.15 In a study by Al Khawaja. (2021), subarachnoid administration of 0.2 mg morphine was conducted directly during the intraoperative period.16 The author concluded that this approach facilitated early hospital discharge by providing prolonged postoperative analgesia, promoting early mobilization without complications, and reducing the need for parenteral opioids or PCA.
Evidence from multiple studies.6, 17 supports using 0.2 mg of ITM as effective and safe for analgesia following lumbar surgery. Postoperative analgesia remained effective even at higher doses (0.35% ITM).18 compared to placebo, with no discernible increase in adverse effects. Consequently, we used a 0.2 mg ITM dosage in our trial.
Our findings are consistent with those of Yoon et al. (2002), who compared ITM, ITN, and a combination of both for elective cesarean procedures, albeit using different dosages.19
Researchers discovered that while the ITN group experienced a decreased incidence of pruritus, the duration of analgesia was significantly prolonged when ITN was administered alone. Remarkably, all three groups exhibited similar rates of PONV. Similarly, a clinical trial conducted by Hassan et al. (2002) confirmed that nalbuphine effectively reduced the occurrence of pruritus, although morphine was found to be a superior analgesic.20
In the context of orthopedic surgery, Fournier et al. (2002) found that ITN produced analgesia more rapidly but had a shorter duration.21 In contrast, the ITM group required less analgesic medication postoperatively. Nevertheless, contrary to our expectations, both groups reported similar rates of adverse effects.
Morphine as a hydrophilic drug crosses then blood brain barrier slowly with less affinity to bind to the epidural fat. While morphine has a high affinity to bind to the specific grey matter receptors along with slow vascular reuptake. This distribution results in longer-lasting and higher concentrations of morphine in the CSF which in turn leads to slow onset and prolonged duration of action with the possibility of delayed onset of respiratory depression.22
When comparing opioids other than morphine, Jain et al. (2022) found that ITN provided a longer duration of analgesia and reduced the need for postoperative rescue analgesia compared to fentanyl.23 In contrast, our research showed no significant differences in hemodynamic parameters or adverse effects between the groups. Similarly, Bindra et al. (2018) suggested that intravenous naloxone could replace intrathecal fentanyl (IVF) during cesarean sections.24
Amin et al. (2020) discovered that adding 1 mg of ITN to 12.5 mg of bupivacaine improved analgesia, accelerated the onset of the block, and slowed the regression of sensory block compared to 2.5 mg of midazolam.25 Our findings are consistent with those of Shakooh et al. (2014), who investigated the combination of nalbuphine with bupivacaine for analgesic efficacy.26 They found that adding ITN improved the quality of analgesia both intraoperatively and postoperatively, with negligible adverse effects.
Being a lipophilic drug, nalbuphine has rapid onset and short duration of action because of rapid vascular reuptake and more affinity to bind epidural fat which in turn lead to fast decrease in its CSF concentration. It has agonist activity on kappa receptor and antagonist activity on mu receptor so regarding to respiratory depression it has a ceiling effect.27
According to Tiwari et al. (2013), incorporating ITN with bupivacaine prolonged postoperative analgesia without adverse effects.28 Their study employed lower doses of nalbuphine (0.2 mg and 0.4 mg), which could explain the discrepancy in findings.
So, from the results of the study, we introduce an idea to apply perioperative analgesia for spine surgeries through the intrathecal administration of nalbuphine. This technique is well known in anesthesia practice and no need for special training nor special equipment like that of nerve block or fascial plane block. Moreover, nalbuphine is an available, cheap drug with no significant adverse effects and unlike other opioids no need to have license to obtain it.
This study has a few limitations. First, pain scores were not evaluated during movement. Second, the study duration was short, and we did not follow up with the patients for chronic pain outcomes. Third, a single-shot injection was used to determine nalbuphine's exact duration of action at the lowest effective dose and also, we could not add it with local anesthetics for the same cause. Fourth, it was single center study for one type of surgery and small sample size so further multi-center studies are recommended in different types of surgeries to explore the addition of other adjuvants, such as dexamethasone, to ITN to extend the duration of the analgesic effect. Additionally, catheter insertion could be investigated.
5. CONCLUSION
In conclusion, patients undergoing lumbar spine surgery could benefit from early analgesia after surgery with 0.8 mg of ITN, which may reduce the likelihood of adverse effects, making it a viable alternative to ITM.
6. Data availability
The data supporting this study's findings are available upon reasonable request sent to the e-mail of the corresponding author (drmonagh19802000@gmail.com). The data are not publicly available due to privacy or ethical restrictions.
7. Conflict of interest
All authors declare that there was no potential conflict of interest is relevant to this article.
8. Funding
No external funding to declare.
9. Authors contribution
M R E: concept, study design, conduction of the work, data collection, read and approved the final manuscript
S M F: concept, study design, data collection, revising it critically for important intellectual content, read and approved the final manuscript
N E H: concept, study design, data analysis, first draft of the manuscript, read and approved the final manuscript
M AE E: concept, study design, conduction of the work, data collection, first draft of the manuscript, read and approved the final manuscript
10. REFERENCES
- NK Prabhakar, AL Chadwick, C Nwaneshiudu, A Aggarwal, V Salmasi, TR Lii, et al. Management of Postoperative Pain in Patients Following Spine Surgery: A Narrative Review. Int J Gen Med 2022;15:4535–49. [PubMed] DOI: DOI: 2147/IJGM.S292698
- Jacquelyn A Corley, Lefko T Charalambous , Vikram A Mehta , Timothy Y Wang , Jihad Abdelgadir , Khoi D Than , et al. Perioperative Pain Management for Elective Spine Surgery: Opioid Use and Multimodal Strategies. World Neurosurg. 2022;162:118-25.[PubMed]DOI: 1016/j.wneu.2022.03.084
- T Bansal, AD Sharan, B Garg. Enhanced recovery after surgery (ERAS) protocol in spine surgery. J Clin Orthop Trauma 2022; 31: 101944.[PubMed] DOI: 1016/j.jcot.2022.101944
- Nitin K Prabhakar, Andrea L Chadwick , Chinwe Nwaneshiudu , Anuj Aggarwal , Vafi Salmasi , Theresa R Lii ,et al. Management of Postoperative Pain in Patients Following Spine Surgery: A Narrative Review. Int J Gen Med. 2022;15:4535–49. [PubMed]DOI: 2147/IJGM.S292698
- Marzouk MM, Elsayed El Hennawy AM, Mohammad Kamal M, Abdnagho Abdelnour TN. Analgesic Effect of Intrathecal Nalbuphine versus Intrathecal Fentanyl as Adjuvant to 0.5% Bupivacaine for Cesarian Section under Spinal Anaesthesia. Int J Med.2024;117: hcae070-066.[FullText]
- Yujie Wang, Xiangyang Guo, Zhaoqing Guo, Mao Xu. Preemptive analgesia with a single low dose of intrathecal morphine in multilevel posterior lumbar interbody fusion surgery: a double-blind, randomized, controlled trial. Spine Journal 2020;20:989−97.[PubMed] DOI: 1016/j.spinee.2020.03.001
- X Culebras, G Gaggero, J Zatloukal, C Kern, RA Marti. Advantages of intrathecal nalbuphine, compared with intrathecal morphine, after cesarean delivery: an evaluation of postoperative analgesia and adverse effects. Anesth Analg 2000;91:601-5. [PubMed] DOI: 1097/00000539-200009000-00019
- Jyothi B, Gowda S, Shaikh SI. A comparison of analgesic effect of different doses of intrathecal nalbuphine hydrochloride with bupivacaine and bupivacaine alone for lower abdominal and orthopedic surgeries. Indian J Pain 2014;28:18-23 [FullText]
- Borah TJ, Dey S, Yunus MD, Dev P, Karim HR, Bhattacharyya P. Effect of different doses of intrathecal nalbuphine as adjuvant to ropivacaine in elective lower limb surgeries: a dose finding study. Indian J Anaesth. 2018, 62:865-70.[PubMed] DOI: 4103/ija.IJA_278_18
- P Yu, J Zhang, J Wang: Nalbuphine for spinal anesthesia: A systematic review and meta-analysis. Pain Pract 2022;22:91–106 [PubMed] DOI: 1111/papr.13021
- Mukherjee A, Pal A, Agrawal J, Mehrotra A, Dawar N. Intrathecal nalbuphine as an adjuvant to subarachnoid block: what is the most effective dose? Anesth Essays Res. 2011;5:171-5. [PubMed] DOI: 4103/0259-1162.94759
- Ummerhofer WC, Arends RH, Shen DD, et al. Comparative spinal distribution and clearance kinetics of intrathecally administered morphine, fentanyl, alfentanil, and sufentanyl. Anesthesiology.2000; 92(3):739-53.[PubMed] DOI: 1097/00000542-200003000-00018
- D Singhal, A Chowdhary , N Mehta. Two Different Doses of Nalbuphine as an Adjuvant to Bupivacaine Intrathecally in Lower Abdominal and Lower Limb Surgeries-A Comparative Study. J Med Dent Sci 2018; 17: 81-6. [Fulltext]
- P Ahluwalia, A Ahluwalia, R Varshney, S Thakur, S Bhandari. A Prospective Randomized Double-Blind Study to Evaluate the Effects of Intrathecal Nalbuphine in Patients of Lower Abdominal Surgeries under Spinal Anaesthesia. Int j sci study.2015;3:19-2. [FullText]
- A De Bie, R Siboni, MF Smati, X Ohl, S Bredin. Intrathecal morphine injections in lumbar fusion surgery: Case-control study. Orthop Traumatol Sur Res 2020;106:1187–90.[PubMed] DOI: 1016/j.otsr.2020.02.024
- D Al Khawaja. Intrathecal morphine injection in anterior lumbar and lateral lumbar spine surgery: technical note. J Spine Surg 2021;7:204-7.[PubMed] DOI: 21037/jss-20-610
- Dhaliwal P, Yavin D, Whittaker T, Hawboldt GS, Jewett GA, Casha S, et al. Intrathecal Morphine Following Lumbar Fusion: A Randomized, Placebo-Controlled Trial. Neurosurgery 2019;85:189-98.[PubMed] DOI: 1093/neuros/nyy384
- Yen D, Turner K, Mark D. Is a single low dose of intrathecal morphine a useful adjunct to patient-controlled analgesia for postoperative pain control following lumbar spine surgery? A preliminary report. Pain Res Manag 2015;20:129-32. [PubMed] DOI: 1155/2015/761390
- Yoon HJ, Jee YS, Hong JY. A Comparison of Analgesic Effects and Side Effects of Intrathecal Morphine, Nalbuphine and a Morphine-Nalbuphine Mixture for Pain Relief during a Cesarean Section. Korean J Anesthesiol 2002;42:627-33. [FullText]
- Hassan KM, Mahmood HA, Moustafa IK, Rezk GA. Comparative Study between Intrathecal Morphine and Nalbuphine Added to Hyperbaric Bupivacaine 0.5% in Elective Cesarean Delivery. Egy J Hosp Med 2021;82: 80-8. [PubMed] DOI: 23736/S0375-9393.18.12751-9
- R Fournier, E Van gessel, M Macksay, Z Gamulin. Onset and offset of intrathecal morphine versus nalbuphine for postoperative pain relief after total hip replacement. Acta Anaesthesiol Scand 2000; 44: 940–5. [PubMed] DOI: 1034/j.1399-6576.2000.440808.x
- Bujedo BM, Santos SG, Azpiazu AU. A review of epidural and intrathecal opioids used in the management of postoperative pain. J Opioid Manag. 2012;8(3):177-92. [PubMed] DOI: 5055/jom.2012.0114
- Jain N, Sethi SK, Saini AL, Patodi V, Jain K, Thada B A Comparison between Intrathecal Nalbuphine versus Fentanyl as an Adjuvant with 0.5% Hyperbaric Bupivacaine for Postoperative Analgesia in Parturients Undergoing Lower Segment Cesarean Section J obstet anaesth crit care 2022;12:127-32. [PubMed] PMID: 38638177
- Bindra TK, Kumar P, Jindal G. Postoperative Analgesia with Intrathecal Nalbuphine versus Intrathecal Fentanyl in Cesarean Section: A Double-Blind Randomized Comparative Study. Anesth Essays Res 2018; 12:561–5. [PubMed] DOI: 4103/aer.AER_41_18
- Amin OAI, Ibrahem MA, Salem DAE. Nalbuphine versus Midazolam as an Adjuvant to Intrathecal Bupivacaine for Postoperative Analgesia in Patients Undergoing Cesarean Section. J Pain Res2020:13 1369–76. [PubMed] DOI: 2147/JPR.S242545
- Shakooh S, Bhosle P. Intrathecal nalbuphine: an effective adjuvant for postoperative analgesia. Innov J Med Health Sci. 2014;4:79–82. [FullText]
- Bisht S, Rashmi D. Comparison of intrathecal fentanyl and nalbuphine: A prospective randomized controlled study in patients undergoing total abdominal hysterectomy. Anaesthesia, Pain & Intensive Care. 2019 Jan 19:194-8. [FullText]
- Tiwari AK, Tomar GS, Agrawal J. Intrathecal bupivacaine in comparison with a combination of nalbuphine and bupivacaine for subarachnoid block: a randomized prospective double – blind clinical study. Am J Ther. 2013;20:592–5. [PubMed] DOI: 1097/MJT.0b013e31822048db