Zulfikar Loka Wicaksana 1 , Bambang Pujo Semedi 2 , Made Walmiky Budi 3 , Dina Angelika 4
Authors affiliations:
Congenital diaphragmatic hernia (CDH) causes pulmonary hypoplasia and maladaptive pulmonary vascular remodeling, predisposing to persistent pulmonary hypertension of the newborn (PPHN) and, in severe cases, acute cor pulmonale (ACP). The resultant cardiopulmonary failure often necessitates prolonged ventilation, vasoactive support, and central venous access, which—together with systemic inflammation—increase vulnerability to nosocomial sepsis. In septic neonates, endothelial injury and a prothrombotic state can convert catheter-related thrombophlebitis into septic emboli that occlude peripheral arteries and threaten limb viability. Although sepsis is recognized in CDH, the specific sequence from CDH/PPHN to intensive support and catheterization, progressing to catheter-associated septic embolism with irreversible lower-limb ischemia, is rarely documented, and evidence to guide anticoagulation in this setting remains scarce. We describe a neonate with CDH complicated by PPHN, ACP, and septic embolism culminating in limb loss, illustrating this pathophysiologic linkage and the therapeutic dilemma of balancing anticoagulation against bleeding risk.
Keywords: Congenital diaphragmatic hernia; Persistent pulmonary hypertension of the newborn; Acute cor pulmonale; Neonatal limb ischemia; Neonatal critical care
Citation: Wicaksana ZL, Semedi BP, Budi MW, Angelika D. Multisystem challenges in neonatal critical care: a CDH survivor with PPHN and septic embolism. Anaesth. pain intensive care 2025;29(9):1333-38. DOI: 10.35975/apic.v29i9.3076
Received: October 02, 2025; Revised: October 18, 2024; Accepted: October 27, 2025
Congenital diaphragmatic hernia (CDH), a rare defect (1.7–5.7 per 10,000 live births), causes organ herniation into the thorax, leading to pulmonary hypoplasia and persistent pulmonary hypertension of the newborn (PPHN) in 16–50% of cases.1,2 PPHN results in severe hypoxemia and risks acute cor pulmonale. Critically ill neonates with long-term central venous access are prone to nosocomial sepsis and rare septic embolism, causing limb ischemia.3,4 We report a unique case of CDH, PPHN, intraoperative ACP, and irreversible limb ischemia from septic embolism, emphasising vigilant monitoring for early vascular complications and adaptive, individualised perioperative management.
2. CASE REPORT
2.1. Diagnosis and Preoperative Management
A full-term female neonate (39 weeks of gestation, 2800 g) was referred from a peripheral hospital with respiratory distress and cyanosis at birth. Initial Apgar scores were 1-1-2, with poor response to resuscitation. Endotracheal intubation and mechanical ventilation were initiated at the referring facility to address hypoxemia. A babygram revealed left-sided congenital diaphragmatic hernia (CDH) with intestinal herniation into the left hemithorax, mediastinal shift to the right, and an orogastric tube positioned in the left thorax (Figure 1)
The patient was reintubated and placed on high-frequency oscillatory ventilation (HFOV) with a FiO₂ of 70% upon arrival at Dr. Soetomo General Hospital. With the following data, the initial blood gas analysis (BGA) showed moderate-to-severe hypoxemia: pH 7.28, PCO₂ 51 mmHg, PO₂ 103 mmHg, HCO3 24 mEq/L, BE -2.7, SaO₂ 97%, and P/F ratio 147. In accordance with inadequate oxygenation and potential for alveolar overdistension in hypoplastic lungs, ventilation was transitioned to pressure-controlled ventilation (PCV, FiO₂ 70%), resulting in significant enhancement of gas exchange (pH 7.39, PCO₂ 35.7 mmHg, PO₂ 192 mmHg, HCO₃ 21.7 mEq/L, BE -3, SaO₂ 100%, P/F ratio 274). A pre/post-ductal oxygen saturation gradient more than 10% (100% vs. 88% at FiO₂ 100%) showed right-to-left shunting, which is indicative of PPHN. Echocardiography demonstrated PPHN, revealing right atrial/ventricular dilatation, a small atrial septal defect (ASD, 0.3 cm), a small patent ductus arteriosus (PDA, 0.2 cm) with predominant right-to-left shunting, mild tricuspid regurgitation, and preserved left ventricular systolic function (LVEF 62%).
Oral sildenafil and intravenous milrinone improved hemodynamic stability and oxygenation. PCV is the optimal breathing method due to adequate oxygen levels (PaO₂ >60 mmHg, PCO₂ normal). BGA showed extended metabolic acidosis (pH 7.23, PCO₂ 61 mmHg, PO₂ 193 mmHg, HCO₃ 25.5 mEq/L, BE -2.1, SaO₂ 100%; venous: pH 7.37, PCO₂ 33 mmHg, PO₂ 31 mmHg, HCO₃ 19.1 mEq/L, BE -6.2, SO₂ 57%), indicating increased oxygen demand, cellular hypoxia, and possible sepsis-induced circulatory failure (septic shock). Endotracheal sputum culture showed that Acinetobacter baumannii was susceptible to amikacin, ampicillin-sulbactam, cefoperazone-sulbactam, and meropenem, requiring empirical antibiotic adjustments.
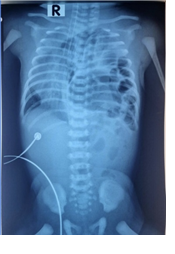
Figure 1: A babygram revealed left-sided CDH
2.2. Surgical Intervention, Post-operative Complications, and Recurrent Respiratory Deterioration
After one week of critical care with ventilatory support and broad-spectrum antibiotics (ampicillin, gentamicin), hemodynamic instability (BP 71/40 mmHg, HR 150–200 bpm) needed dobutamine (5 mcg/kg/min). A multidisciplinary team (Pediatric Surgery, Neonatology, Anesthesiology-Intensive Care) confirmed operative readiness on day 8 with stable oxygenation (SaO₂ 99% at FiO₂ 70%), BGA (pH 7.34, PCO₂ 38 mmHg, PO₂ 141 mmHg, HCO₃ 20.5 mEq/L, BE -5.3), and hemodynamics (BP 75/50 mmHg, MAP 50 mmHg, HR 150 bpm. An exploratory laparotomy showed a 10 cm left posterolateral diaphragmatic defect with intra-abdominal organ herniation, treated by reduction and synthetic patch closure. Although intraoperative lung expansion was adequate, hemodynamic instability developed (HR >200 bpm, MAP <45 mmHg, urine output <0.5 mL/kg/h). Fluid resuscitation with 5% plasbumin (10 mL/kg) raised CVP to 12–13 mmHg without improving MAP, indicating RV dysfunction and inadequate cardiac output. High-dose adrenaline (150–250 ng/kg/min) plus furosemide reduced preload and supported diuresis.
Dual vasopressors with adrenaline (150–250 ng/kg/min) and norepinephrine (150–250 ng/kg/min) were necessary to address persistent hemodynamic instability (BP 68/45 mmHg, HR 170–200 bpm) following surgery. SaO₂ was maintained at 90–96% by PCV (FiO₂ 50%). Adequate renal perfusion was demonstrated by the urine output (280 mL/16 h) in the presence of furosemide. The thoracic structure was improved in the postoperative babygram, but it indicated the presence of a pneumonia (Figure 2). Echocardiography demonstrated severe PPHN, including right atrial/ventricular dilatation, severe tricuspid regurgitation, mild pulmonary regurgitation, a small ASD, and a small PDA with left-to-right shunting. The ejection fraction was unmeasurable as a result of elevated pulmonary pressures. Persistent metabolic acidosis (pH 7.21, PCO₂ 38 mmHg, PO₂ 79 mmHg, HCO₃ 15.2 mEq/L, BE -12.7, SaO₂ 92%) suggested that suboptimal perfusion and refractory PPHN and severe sepsis. emergent multi-organ dysfunction were the result.
On postoperative day 2, severe retractions, asymmetric breath sounds, and hemodynamic deterioration (BP 80/55 mmHg) required increased vasopressors (adrenaline 300 ng/kg/min, norepinephrine 300 ng/kg/min), milrinone (0.3 mcg/kg/min), and sildenafil (1.5 mg q6h). After endotracheal sputum culture showed Enterobacter cloacae resistance to first antibiotics, cefoperazone-sulbactam was given.
The patient's condition fluctuated the next week. Furosemide administration likely caused severe metabolic acidosis to resolve and metabolic alkalosis to shift in a subsequent BGA. After being extubated on ICU day 9, her respiratory state suddenly deteriorated, requiring emergency re-intubation. After chest physiotherapy, passive mobilisation, suctioning, and nebulised bronchodilators, clinical improvement occurred. Nasal bubble CPAP and low-dose milrinone allowed re-extubation on ICU day 12. PDA and ASD resolved, and TR was minimal on ICU day 15 echocardiography. The patient would be transferred to the high-care unit the next day after tapering sildenafil. The arterial and central venous catheters were withdrawn, antibiotics were stopped, and CPAP was tapered to a nasal cannula one Lpm.
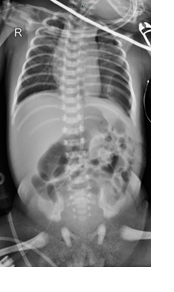
Figure 2: Postoperative babygram
2.3. Septic Thromboembolism and Comprehensive Management
A persistent fever (38.9°C) and a positive blood culture from the central venous catheter (CVC) on ICU day 18 confirmed the presence of Staphylococcus kloosii, indicating a systemic infection. Targeted meropenem and aggressive thermoregulation (paracetamol) were implemented, with a new CVC deployed at an alternative location. The patient sustained acute desaturation (SpO₂ 77%) and respiratory distress that necessitated emergency reintubation late that night.
On day 19, cutis marmorata, hematoma, and bullae on both lower limbs (Figure 3), strongly indicating septic thromboembolism and acute limb ischemia. Vascular examination showed no right posterior tibial/dorsalis pedis pulses, cold right lower limb, and unmeasurable peripheral SpO₂, with weak left limb pulses (SpO₂ 73%). Doppler ultrasound demonstrated irreversible right lower limb ischemia with a right posterior tibial/dorsalis pedis blockage (no flow) and adequate left limb flow (biphasic/triphasic). The Vascular Surgery Team advised amputation, but the family declined. Conservative treatment comprised heparin (5 units/kg/h), limb warming, and saline-soaked gauze to reduce ischemia and infection. Two days later, prolonged coagulation, thrombocytopenia, melena, and hematin via a nasogastric tube showed heparin-related bleeding, prompting heparin suspension, platelet/red cell transfusions, and proton pump inhibitors. After hemostatic stabilization, heparin was gently reinitiated with After hemostatic stabilization, heparin was gently reinitiated with close monitoring and continuous PPI support.
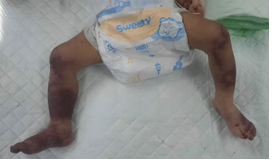
Figure 3: Clinical manifestations of septic thromboembolism and acute limb ischemia
2.4. Recovery, Transition, and Long-Term Complications
On ICU day 20, the patient exhibited respiratory distress, hemodynamic instability, severe anemia (Hb 5.2 g/dL), and thrombocytopenia (6000/µL), which required re-intubation, transfusions, and continued administration of meropenem, bronchodilator nebulization (salbutamol, ipratropium, saline), chest physiotherapy, and wound management.
Total parenteral nutrition (215–250 mL/24 h) was transitioned to enteral feeding (Neocate, 12–30 mL every 2 hours, 259 kcal/day) after confirming no gastrointestinal bleeding. Nutrition was critical for metabolic recovery and growth. By day 35, clinical stabilization allowed vasopressor/ventilator weaning, extubation, and transfer to low care with low-dose milrinone.
The low-care echocardiography (day 41) showed mild mitral regurgitation (gradient 9.9 mmHg), trivial tricuspid regurgitation, closed PDA, and a 0.2 cm ASD with left-to-right shunting. Sildenafil was discontinued, and a chest X-ray showed the resolution of pneumonia and a symmetrical diaphragmatic position (Figure 4). The patient was discharged with outpatient follow-up. Significant clinical improvement and weight gain were observed two weeks after discharge; however, autoamputation of all digits on the right foot was observed, underscoring the long-term consequences of bacterial limb ischemia.
The complexity of managing CDH with refractory PPHN and septic thromboembolism is demonstrated by this case, which emphasizes the importance of vigilant monitoring for infectious vascular complications, comprehensive PPHN management, and adaptive resuscitation strategies.
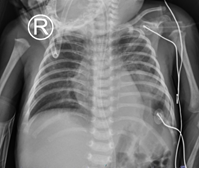
Figure 4: Chest X-ray showed the resolution of pneumonia and a symmetrical diaphragmatic position (day 40)
This case delineates an uncommon cascade in congenital diaphragmatic hernia (CDH): persistent pulmonary hypertension of the newborn (PPHN) progressing to intra-operative acute cor pulmonale (ACP), followed by catheter-associated septic embolism with irreversible lower-limb ischemia. Reports that link CDH/PPHN, central venous catheterization, and peripheral arterial limb loss are scarce, and guidance on anticoagulation in septic, postoperative neonates remains limited. 5,6 These features constitute the principal novelty and clinical relevance of the case.
The pathophysiology offers a coherent bridge from diaphragmatic pathology to vascular injury. Pulmonary hypoplasia and maladaptive vascular remodeling in CDH sustain high pulmonary vascular resistance, driving PPHN and right-ventricular pressure overload. 7,8 Surgical manipulation and positive-pressure ventilation may acutely increase right-ventricular afterload and precipitate ACP with septal shift and reduced left-sided filling, thereby lowering cardiac output. Low output, vasopressor dependence, and regional vasoconstriction diminish distal perfusion reserves and make peripheral tissues vulnerable to ischemia. 9
In parallel, central venous catheters, endothelial injury, and systemic inflammation promote thrombo-inflammation and catheter-related thrombophlebitis. Fragmentation of infected thrombus can seed the arterial tree as septic emboli, where limited perfusion reserve favors irreversible tissue loss. In neonatal series, septic emboli most often involve cerebral or pulmonary beds; documented peripheral arterial occlusion is unusual. Bonny et al. described several neonatal septic embolism cases without the CDH/PPHN context, and none progressed to documented limb loss. 5,10 Our case extends that literature by situating embolic limb ischemia within a cardiopulmonary milieu characterized by high pulmonary vascular resistance, risk of right-ventricular failure, and prolonged invasive support.
Diagnosis depends on vigilance rather than exhaustive parameter tracking. Echocardiography is central for recognizing ACP and guiding right-ventricular–protective strategies, while bedside Doppler ultrasonography enables early confirmation of limb malperfusion. A low threshold for repeat vascular imaging is warranted when clinical suspicion persists despite equivocal initial studies.
Therapeutically, clinicians face a recurrent dilemma. Anticoagulation may limit thrombus propagation and recurrent embolization, yet bleeding risk in septic neonates—especially with thrombocytopenia or gastrointestinal bleeding—often precludes standard dosing. 5,6 A pragmatic approach prioritizes rapid source control and catheter removal when feasible, targeted antibiotics, close coagulation monitoring, and individualized, time-staged anticoagulation once hemorrhagic risk abates. Serial vascular imaging can document trajectory and support adjustments.
The most actionable implications lie in prevention and systems-based care. Strict catheter stewardship—minimizing line number and dwell time, ensuring meticulous insertion and maintenance, and removing unnecessary catheters promptly—remains the cornerstone of CLABSI prevention. Peri-operative echocardiography can detect ACP early and inform right-ventricular–protective hemodynamics and selective pulmonary vasodilator use.6,10 Clinicians should maintain a low threshold for limb vascular assessment when color, temperature, or pulse symmetry changes are observed. Multidisciplinary coordination across neonatology, cardiology, infectious diseases, hematology, and surgery is essential to align anticoagulation timing, antimicrobial therapy, and surgical or rehabilitative planning.
This single case cannot establish causality, and several management decisions reflected clinical instability and local resource constraints. Nevertheless, the biological sequence is plausible and consistent with available evidence. In summary, the report underscores a rare but consequential intersection of CDH/PPHN, ACP, and catheter-associated septic embolism leading to limb ischemia as illustrated in Figure 5. It supports rigorous CLABSI prevention, vigilant vascular monitoring, early recognition of right-ventricular afterload crises, and individualized anticoagulation strategies when bleeding risk is high—practical signals for risk recognition and team-based management in neonatal critical care.
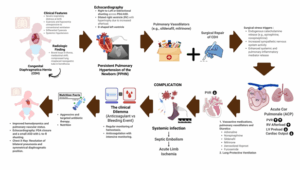
Figure 5: comprehensive case illustration
The neonatal presentation of this rare and complex case is congenital diaphragmatic hernia (CDH) with persistent pulmonary hypertension of the newborn (PPHN) which is further exacerbated by intraoperative acute cor pulmonale and septic embolism-induced limb ischemia. The therapeutic dilemma presented by the coexistence of infectious vascular complications and refractory PPHN in a hemostatically unstable neonate was the balance between anticoagulation and bleeding risks.
Multidisciplinary care, encompassing right ventricular optimization, aggressive infection control, and meticulous coagulation monitoring, yielded substantial clinical enhancement. However, long-term consequences, such as autoamputation of digits, highlight the necessity for careful cardiac and vascular surveillance, adaptive treatment approaches, and comprehensive long-term monitoring.
Authors affiliations:
- Zulfikar Loka Wicaksana, Department of Anesthesiology and Reanimation, Faculty of Medicine, Universitas Airlangga, Surabaya, Indonesia; Email: zulfikar.loka.wicaksana-2022@fk.unair.ac.id; {ORCID:0000-0002-7638-0754}
- Bambang Pujo Semedi, Department of Anesthesiology and Reanimation, Faculty of Medicine, Universitas Airlangga, Surabaya, Indonesia; Email: bambang-p-s@fk.unair.ac.id; {ORCID: 0000-0003-4499-3481}.
- Made Walmiky Budi, Division of Pediatric Surgery, Faculty of Medicine, Universitas Airlangga, Surabaya, Indonesia; Email: wallmicky@yahoo.com {ORCID:0000-0002-6555-7403}
- Dina Angelika, Departement of Pediatrics, Faculty of Medicine, Universitas Airlangga, Surabaya, Indonesia; Email: dina.angelika@yahoo.co.id; {ORCID:000000326033532}
ABSTRACT
Congenital diaphragmatic hernia (CDH) causes pulmonary hypoplasia and maladaptive pulmonary vascular remodeling, predisposing to persistent pulmonary hypertension of the newborn (PPHN) and, in severe cases, acute cor pulmonale (ACP). The resultant cardiopulmonary failure often necessitates prolonged ventilation, vasoactive support, and central venous access, which—together with systemic inflammation—increase vulnerability to nosocomial sepsis. In septic neonates, endothelial injury and a prothrombotic state can convert catheter-related thrombophlebitis into septic emboli that occlude peripheral arteries and threaten limb viability. Although sepsis is recognized in CDH, the specific sequence from CDH/PPHN to intensive support and catheterization, progressing to catheter-associated septic embolism with irreversible lower-limb ischemia, is rarely documented, and evidence to guide anticoagulation in this setting remains scarce. We describe a neonate with CDH complicated by PPHN, ACP, and septic embolism culminating in limb loss, illustrating this pathophysiologic linkage and the therapeutic dilemma of balancing anticoagulation against bleeding risk.
Keywords: Congenital diaphragmatic hernia; Persistent pulmonary hypertension of the newborn; Acute cor pulmonale; Neonatal limb ischemia; Neonatal critical care
Citation: Wicaksana ZL, Semedi BP, Budi MW, Angelika D. Multisystem challenges in neonatal critical care: a CDH survivor with PPHN and septic embolism. Anaesth. pain intensive care 2025;29(9):1333-38. DOI: 10.35975/apic.v29i9.3076
Received: October 02, 2025; Revised: October 18, 2024; Accepted: October 27, 2025
1. INTRODUCTION
Congenital diaphragmatic hernia (CDH), a rare defect (1.7–5.7 per 10,000 live births), causes organ herniation into the thorax, leading to pulmonary hypoplasia and persistent pulmonary hypertension of the newborn (PPHN) in 16–50% of cases.1,2 PPHN results in severe hypoxemia and risks acute cor pulmonale. Critically ill neonates with long-term central venous access are prone to nosocomial sepsis and rare septic embolism, causing limb ischemia.3,4 We report a unique case of CDH, PPHN, intraoperative ACP, and irreversible limb ischemia from septic embolism, emphasising vigilant monitoring for early vascular complications and adaptive, individualised perioperative management.
2. CASE REPORT
2.1. Diagnosis and Preoperative Management
A full-term female neonate (39 weeks of gestation, 2800 g) was referred from a peripheral hospital with respiratory distress and cyanosis at birth. Initial Apgar scores were 1-1-2, with poor response to resuscitation. Endotracheal intubation and mechanical ventilation were initiated at the referring facility to address hypoxemia. A babygram revealed left-sided congenital diaphragmatic hernia (CDH) with intestinal herniation into the left hemithorax, mediastinal shift to the right, and an orogastric tube positioned in the left thorax (Figure 1)
The patient was reintubated and placed on high-frequency oscillatory ventilation (HFOV) with a FiO₂ of 70% upon arrival at Dr. Soetomo General Hospital. With the following data, the initial blood gas analysis (BGA) showed moderate-to-severe hypoxemia: pH 7.28, PCO₂ 51 mmHg, PO₂ 103 mmHg, HCO3 24 mEq/L, BE -2.7, SaO₂ 97%, and P/F ratio 147. In accordance with inadequate oxygenation and potential for alveolar overdistension in hypoplastic lungs, ventilation was transitioned to pressure-controlled ventilation (PCV, FiO₂ 70%), resulting in significant enhancement of gas exchange (pH 7.39, PCO₂ 35.7 mmHg, PO₂ 192 mmHg, HCO₃ 21.7 mEq/L, BE -3, SaO₂ 100%, P/F ratio 274). A pre/post-ductal oxygen saturation gradient more than 10% (100% vs. 88% at FiO₂ 100%) showed right-to-left shunting, which is indicative of PPHN. Echocardiography demonstrated PPHN, revealing right atrial/ventricular dilatation, a small atrial septal defect (ASD, 0.3 cm), a small patent ductus arteriosus (PDA, 0.2 cm) with predominant right-to-left shunting, mild tricuspid regurgitation, and preserved left ventricular systolic function (LVEF 62%).
Oral sildenafil and intravenous milrinone improved hemodynamic stability and oxygenation. PCV is the optimal breathing method due to adequate oxygen levels (PaO₂ >60 mmHg, PCO₂ normal). BGA showed extended metabolic acidosis (pH 7.23, PCO₂ 61 mmHg, PO₂ 193 mmHg, HCO₃ 25.5 mEq/L, BE -2.1, SaO₂ 100%; venous: pH 7.37, PCO₂ 33 mmHg, PO₂ 31 mmHg, HCO₃ 19.1 mEq/L, BE -6.2, SO₂ 57%), indicating increased oxygen demand, cellular hypoxia, and possible sepsis-induced circulatory failure (septic shock). Endotracheal sputum culture showed that Acinetobacter baumannii was susceptible to amikacin, ampicillin-sulbactam, cefoperazone-sulbactam, and meropenem, requiring empirical antibiotic adjustments.

Figure 1: A babygram revealed left-sided CDH
2.2. Surgical Intervention, Post-operative Complications, and Recurrent Respiratory Deterioration
After one week of critical care with ventilatory support and broad-spectrum antibiotics (ampicillin, gentamicin), hemodynamic instability (BP 71/40 mmHg, HR 150–200 bpm) needed dobutamine (5 mcg/kg/min). A multidisciplinary team (Pediatric Surgery, Neonatology, Anesthesiology-Intensive Care) confirmed operative readiness on day 8 with stable oxygenation (SaO₂ 99% at FiO₂ 70%), BGA (pH 7.34, PCO₂ 38 mmHg, PO₂ 141 mmHg, HCO₃ 20.5 mEq/L, BE -5.3), and hemodynamics (BP 75/50 mmHg, MAP 50 mmHg, HR 150 bpm. An exploratory laparotomy showed a 10 cm left posterolateral diaphragmatic defect with intra-abdominal organ herniation, treated by reduction and synthetic patch closure. Although intraoperative lung expansion was adequate, hemodynamic instability developed (HR >200 bpm, MAP <45 mmHg, urine output <0.5 mL/kg/h). Fluid resuscitation with 5% plasbumin (10 mL/kg) raised CVP to 12–13 mmHg without improving MAP, indicating RV dysfunction and inadequate cardiac output. High-dose adrenaline (150–250 ng/kg/min) plus furosemide reduced preload and supported diuresis.
Dual vasopressors with adrenaline (150–250 ng/kg/min) and norepinephrine (150–250 ng/kg/min) were necessary to address persistent hemodynamic instability (BP 68/45 mmHg, HR 170–200 bpm) following surgery. SaO₂ was maintained at 90–96% by PCV (FiO₂ 50%). Adequate renal perfusion was demonstrated by the urine output (280 mL/16 h) in the presence of furosemide. The thoracic structure was improved in the postoperative babygram, but it indicated the presence of a pneumonia (Figure 2). Echocardiography demonstrated severe PPHN, including right atrial/ventricular dilatation, severe tricuspid regurgitation, mild pulmonary regurgitation, a small ASD, and a small PDA with left-to-right shunting. The ejection fraction was unmeasurable as a result of elevated pulmonary pressures. Persistent metabolic acidosis (pH 7.21, PCO₂ 38 mmHg, PO₂ 79 mmHg, HCO₃ 15.2 mEq/L, BE -12.7, SaO₂ 92%) suggested that suboptimal perfusion and refractory PPHN and severe sepsis. emergent multi-organ dysfunction were the result.
On postoperative day 2, severe retractions, asymmetric breath sounds, and hemodynamic deterioration (BP 80/55 mmHg) required increased vasopressors (adrenaline 300 ng/kg/min, norepinephrine 300 ng/kg/min), milrinone (0.3 mcg/kg/min), and sildenafil (1.5 mg q6h). After endotracheal sputum culture showed Enterobacter cloacae resistance to first antibiotics, cefoperazone-sulbactam was given.
The patient's condition fluctuated the next week. Furosemide administration likely caused severe metabolic acidosis to resolve and metabolic alkalosis to shift in a subsequent BGA. After being extubated on ICU day 9, her respiratory state suddenly deteriorated, requiring emergency re-intubation. After chest physiotherapy, passive mobilisation, suctioning, and nebulised bronchodilators, clinical improvement occurred. Nasal bubble CPAP and low-dose milrinone allowed re-extubation on ICU day 12. PDA and ASD resolved, and TR was minimal on ICU day 15 echocardiography. The patient would be transferred to the high-care unit the next day after tapering sildenafil. The arterial and central venous catheters were withdrawn, antibiotics were stopped, and CPAP was tapered to a nasal cannula one Lpm.

Figure 2: Postoperative babygram
2.3. Septic Thromboembolism and Comprehensive Management
A persistent fever (38.9°C) and a positive blood culture from the central venous catheter (CVC) on ICU day 18 confirmed the presence of Staphylococcus kloosii, indicating a systemic infection. Targeted meropenem and aggressive thermoregulation (paracetamol) were implemented, with a new CVC deployed at an alternative location. The patient sustained acute desaturation (SpO₂ 77%) and respiratory distress that necessitated emergency reintubation late that night.
On day 19, cutis marmorata, hematoma, and bullae on both lower limbs (Figure 3), strongly indicating septic thromboembolism and acute limb ischemia. Vascular examination showed no right posterior tibial/dorsalis pedis pulses, cold right lower limb, and unmeasurable peripheral SpO₂, with weak left limb pulses (SpO₂ 73%). Doppler ultrasound demonstrated irreversible right lower limb ischemia with a right posterior tibial/dorsalis pedis blockage (no flow) and adequate left limb flow (biphasic/triphasic). The Vascular Surgery Team advised amputation, but the family declined. Conservative treatment comprised heparin (5 units/kg/h), limb warming, and saline-soaked gauze to reduce ischemia and infection. Two days later, prolonged coagulation, thrombocytopenia, melena, and hematin via a nasogastric tube showed heparin-related bleeding, prompting heparin suspension, platelet/red cell transfusions, and proton pump inhibitors. After hemostatic stabilization, heparin was gently reinitiated with After hemostatic stabilization, heparin was gently reinitiated with close monitoring and continuous PPI support.

Figure 3: Clinical manifestations of septic thromboembolism and acute limb ischemia
2.4. Recovery, Transition, and Long-Term Complications
On ICU day 20, the patient exhibited respiratory distress, hemodynamic instability, severe anemia (Hb 5.2 g/dL), and thrombocytopenia (6000/µL), which required re-intubation, transfusions, and continued administration of meropenem, bronchodilator nebulization (salbutamol, ipratropium, saline), chest physiotherapy, and wound management.
Total parenteral nutrition (215–250 mL/24 h) was transitioned to enteral feeding (Neocate, 12–30 mL every 2 hours, 259 kcal/day) after confirming no gastrointestinal bleeding. Nutrition was critical for metabolic recovery and growth. By day 35, clinical stabilization allowed vasopressor/ventilator weaning, extubation, and transfer to low care with low-dose milrinone.
The low-care echocardiography (day 41) showed mild mitral regurgitation (gradient 9.9 mmHg), trivial tricuspid regurgitation, closed PDA, and a 0.2 cm ASD with left-to-right shunting. Sildenafil was discontinued, and a chest X-ray showed the resolution of pneumonia and a symmetrical diaphragmatic position (Figure 4). The patient was discharged with outpatient follow-up. Significant clinical improvement and weight gain were observed two weeks after discharge; however, autoamputation of all digits on the right foot was observed, underscoring the long-term consequences of bacterial limb ischemia.
The complexity of managing CDH with refractory PPHN and septic thromboembolism is demonstrated by this case, which emphasizes the importance of vigilant monitoring for infectious vascular complications, comprehensive PPHN management, and adaptive resuscitation strategies.

Figure 4: Chest X-ray showed the resolution of pneumonia and a symmetrical diaphragmatic position (day 40)
3. DISCUSSION
This case delineates an uncommon cascade in congenital diaphragmatic hernia (CDH): persistent pulmonary hypertension of the newborn (PPHN) progressing to intra-operative acute cor pulmonale (ACP), followed by catheter-associated septic embolism with irreversible lower-limb ischemia. Reports that link CDH/PPHN, central venous catheterization, and peripheral arterial limb loss are scarce, and guidance on anticoagulation in septic, postoperative neonates remains limited. 5,6 These features constitute the principal novelty and clinical relevance of the case.
The pathophysiology offers a coherent bridge from diaphragmatic pathology to vascular injury. Pulmonary hypoplasia and maladaptive vascular remodeling in CDH sustain high pulmonary vascular resistance, driving PPHN and right-ventricular pressure overload. 7,8 Surgical manipulation and positive-pressure ventilation may acutely increase right-ventricular afterload and precipitate ACP with septal shift and reduced left-sided filling, thereby lowering cardiac output. Low output, vasopressor dependence, and regional vasoconstriction diminish distal perfusion reserves and make peripheral tissues vulnerable to ischemia. 9
In parallel, central venous catheters, endothelial injury, and systemic inflammation promote thrombo-inflammation and catheter-related thrombophlebitis. Fragmentation of infected thrombus can seed the arterial tree as septic emboli, where limited perfusion reserve favors irreversible tissue loss. In neonatal series, septic emboli most often involve cerebral or pulmonary beds; documented peripheral arterial occlusion is unusual. Bonny et al. described several neonatal septic embolism cases without the CDH/PPHN context, and none progressed to documented limb loss. 5,10 Our case extends that literature by situating embolic limb ischemia within a cardiopulmonary milieu characterized by high pulmonary vascular resistance, risk of right-ventricular failure, and prolonged invasive support.
Diagnosis depends on vigilance rather than exhaustive parameter tracking. Echocardiography is central for recognizing ACP and guiding right-ventricular–protective strategies, while bedside Doppler ultrasonography enables early confirmation of limb malperfusion. A low threshold for repeat vascular imaging is warranted when clinical suspicion persists despite equivocal initial studies.
Therapeutically, clinicians face a recurrent dilemma. Anticoagulation may limit thrombus propagation and recurrent embolization, yet bleeding risk in septic neonates—especially with thrombocytopenia or gastrointestinal bleeding—often precludes standard dosing. 5,6 A pragmatic approach prioritizes rapid source control and catheter removal when feasible, targeted antibiotics, close coagulation monitoring, and individualized, time-staged anticoagulation once hemorrhagic risk abates. Serial vascular imaging can document trajectory and support adjustments.
The most actionable implications lie in prevention and systems-based care. Strict catheter stewardship—minimizing line number and dwell time, ensuring meticulous insertion and maintenance, and removing unnecessary catheters promptly—remains the cornerstone of CLABSI prevention. Peri-operative echocardiography can detect ACP early and inform right-ventricular–protective hemodynamics and selective pulmonary vasodilator use.6,10 Clinicians should maintain a low threshold for limb vascular assessment when color, temperature, or pulse symmetry changes are observed. Multidisciplinary coordination across neonatology, cardiology, infectious diseases, hematology, and surgery is essential to align anticoagulation timing, antimicrobial therapy, and surgical or rehabilitative planning.
This single case cannot establish causality, and several management decisions reflected clinical instability and local resource constraints. Nevertheless, the biological sequence is plausible and consistent with available evidence. In summary, the report underscores a rare but consequential intersection of CDH/PPHN, ACP, and catheter-associated septic embolism leading to limb ischemia as illustrated in Figure 5. It supports rigorous CLABSI prevention, vigilant vascular monitoring, early recognition of right-ventricular afterload crises, and individualized anticoagulation strategies when bleeding risk is high—practical signals for risk recognition and team-based management in neonatal critical care.

Figure 5: comprehensive case illustration
4. CONCLUSION
The neonatal presentation of this rare and complex case is congenital diaphragmatic hernia (CDH) with persistent pulmonary hypertension of the newborn (PPHN) which is further exacerbated by intraoperative acute cor pulmonale and septic embolism-induced limb ischemia. The therapeutic dilemma presented by the coexistence of infectious vascular complications and refractory PPHN in a hemostatically unstable neonate was the balance between anticoagulation and bleeding risks.
Multidisciplinary care, encompassing right ventricular optimization, aggressive infection control, and meticulous coagulation monitoring, yielded substantial clinical enhancement. However, long-term consequences, such as autoamputation of digits, highlight the necessity for careful cardiac and vascular surveillance, adaptive treatment approaches, and comprehensive long-term monitoring.
- Acknowledgements
- Conflicts of interest
- Authors contribution
8. REFERENCES
- Scavacini Marinonio AS, Harumi Miyoshi M, Testoni Costa-Nobre D, Sanudo A, Nema Areco KC, Daripa Kawakami M, et al. Congenital diaphragmatic hernia in a middle-income country: Persistent high lethality during a 12-year period. PLoS One. 2023 Feb 10;18(2):e0281723. PMCID: PMC9916629 DOI: 10.1371/journal.pone.0281723
- Kotecha S, Barbato A, Bush A, Claus F, Davenport M, Delacourt C, et al. Congenital diaphragmatic hernia. Eur Respir J. 2012 Apr;39(4):820–9. DOI: 10.1183/09031936.00066511
- van Vonderen JJ, Keus JMH, van Schaik J, Smiers FJ, Ten Harkel DJ, Lopriore E. A neonate with spontaneous arterial limb ischemia and an aneurysm of the oval foramen: a case report. J Med Case Rep. 2021 Oct 25;15(1):536. PMCID: PMC8543854 DOI: 10.1186/s13256-021-03078-9
- Muzaffar MM, Samani S, Van Der Velde P. Group A Streptococcus Bacteraemia Presenting as Acute Limb Ischaemia in a Neonate: A Case Report. Cureus. 2025 Mar;17(3):e80170. PMCID: PMC11972120 DOI: 10.7759/cureus.80170
- Bonny-Obro R, Aké YL, Midekor-Gonebo K, Kouassi-Dria AK, Ouattara SJ, Sounkere-Soro M, et al. Neonatal acute limb ischemia. J Pediatr Surg Case Rep. 2021 Mar;66(101784):101784.
- Shaniv D, Simpson-Lavy Y, Hershkovich Shporen C. Management of iatrogenic acute limb ischaemia in the neonate. BMJ Case Rep [Internet]. 2024 Sep 3;17(9). PMCID: PMC11590439 DOI: 10.1136/bcr-2024-261775
- Singh Y, Lakshminrusimha S. Pathophysiology and Management of Persistent Pulmonary Hypertension of the Newborn. Clin Perinatol. 2021 Aug;48(3):595–618. PMCID: PMC8351908 DOI: 10.1016/j.clp.2021.05.009
- Gupta VS, Harting MT. Congenital diaphragmatic hernia-associated pulmonary hypertension. Semin Perinatol. 2020 Feb;44(1):151167. DOI: 10.1053/j.semperi.2019.07.006
- Arrigo M, Price S, Harjola VP, Huber LC, Schaubroeck HAI, Vieillard-Baron A, et al. Diagnosis and treatment of right ventricular failure secondary to acutely increased right ventricular afterload (acute cor pulmonale): a clinical consensus statement of the Association for Acute CardioVascular Care of the European Society of Cardiology. Eur Heart J Acute Cardiovasc Care. 2024 Mar 11;13(3):304–12. PMCID: PMC10927027 DOI: 10.1093/ehjacc/zuad157
- Bakoush FBA, Azab AE, Yahya RAM. Neonatal sepsis: Insight into incidence, classification, risk factors, causative organisms, pathophysiology, prognosis, clinical manifestations, complications, systemic examination, and treatment. South Asian Res J App Med Sci. 2023 Nov 25;5(06):136–57. DOI: 10.36346/sarjams.2023.v05i06.004