Nimra Asghar 1 , Jahanzaib 2 , Aashiq Hussain 3 , Abdul Rehman Khalil Shaikh 4 , Shumaila Kanwel 5 , Zeeshan Hussain 6 , Haseeb Khaliq 7 , Muhammad Arsalan Shah 8
Authors affiliations:
Correspondence: Muhammad Arsalan Shah, Email: muhammadarsalanshahpath01@gmail.com;
Background: Hematologic malignancies such as acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL), multiple myeloma (MM), and peripheral T-cell lymphoma (PTCL) remain difficult to treat. This systematic review and meta-analysis aimed to combine mechanistic, clinical, and pathophysiological knowledge to assist in guiding biomarkers to individualize therapy and ensure safety in the use of these regimens in hematologic cancer.
Methods: [PubMed], Scopus, Web of Science, and Google Scholar (2010-2025) were searched using PRISMA 2020 and included studies on human clinical research involving remission, progression-free survival, or overall survival. RoB 2 and ROBINS-I were used to determine risk of bias. The overall quality of evidence was assessed using GRADE. Meta-analysis was performed in RevMan 5.4.1 using a random-effects model to calculate pooled hazard ratios (HRs) and odds ratios (ORs) and 95% confidence intervals (CIs). Subgroup and sensitivity analyses were conducted according to intervention type and study design.
Results: Twelve studies were included. AML meta-analysis pooled OR 1.58 (95% CI 0.26-9.54), which was not significant (I2=91%). Analysis of the CLL pooled provided HR = 0.28 (95% CI 0.17-0.46), significant PFS benefit (I2=82%). MM meta-analysis generated HR = 0.67 (95% CI 0.53-0.84), which demonstrates the coherent survival benefit (I2 < 50%). Analysis of lymphoma studies pooled OR = 0.89 (95% CI 0.28-2.77), which was not significant (I2=90%), but sensitivity and subgroup analyses supported the robustness of findings. Risk of bias ranged from low to moderate, and overall evidence quality was moderate according to GRADE.
Conclusion: Venetoclax- and epigenetic-based combinations have a definite advantage in CLL and MM. AML and lymphoma outcomes are inconclusive because of significant heterogeneity. To increase combination strategies and clinical translation and guide critical-care supporting approaches, standardized endpoints, biomarker-driven stratification, and harmonized trial designs are required.
Abbreviations: AML: acute myeloid leukemia, CLL: chronic lymphocytic leukemia, MM: multiple myeloma, PTCL: peripheral T-cell lymphoma,
Keywords: Leukemia, Myeloid Acute; Leukemia, Lymphocytic, Chronic; B-Cell; Multiple Myeloma; Lymphoma, T-Cell.
Citation: Asghar N, Jahanzaib, Hussain A, Shaikh ARK, Kanwel S, Hussain Z, Khaliq H, Shah MA. Synergistic anti-tumor potential of epigenetic modulators and natural antioxidants in intensive care of hematological cancers: a schematic assessment of translational medicine and pathophysiological prospects. Anaesth. pain intensive care 2025;29(9):1302-12. DOI: 10.35975/apic.v29i9.3068
Received: August 20, 2025; Revised: August 26, 2025; Accepted: August 26, 2025
Hematologic malignancies, including acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL), multiple myeloma (MM), and peripheral T-cell lymphoma (PTCL), are different but biologically related conditions that have genomic instability, clonal heterogeneity, and cancers with deregulated cell survival pathways.1 These malignancies develop out of aberrant hematopoietic or lymphoid precursors, which accumulate genetic and epigenetic changes that result in unregulated proliferation, defective differentiation, and apoptotic resistance. Treatment refractoriness and relapse are still the common clinical issues despite the development of chemotherapeutic and targeted regimens.2 Frequently, resistance develops in response to adaptive homeostatic responses, including but not limited to metabolic reprogramming, redox imbalance, and mitochondrial remodeling, which together maintain the survival of malignant cells in response to therapeutic pressure.3
At the molecular scale, epigenetic malregulation is known to be an underlying cause of leukemogenesis and pathogenesis. The hypermethylation of tumor suppressor gene promoters is caused by the aberrant activity of the DNA methyltransferases (DNMTs), whereas the overexpression of histone deacetylase (HDAC) induces chromatin condensation and transcriptional repression of apoptotic and cell cycle arrest genes. Such alterations, combined with mutations in chromatin remodelers and histone modifiers, result in an oncogenic survival and chemoresistance transcriptionally silenced landscape.4,5 As a result, clinical reversal of epigenetic repression has become an important therapeutic approach. DNMT inhibitors (azacitidine and decitabine) can re-express silenced genes and boost differentiation, HDAC inhibitors (panobinostat and romidepsin) can restore the balance of acetylation, cause apoptosis, and increase the effectiveness of simultaneous cytotoxic or immune-based therapies.6
Simultaneous progress in the field of precision medicine has brought so-called targeted therapies, which downregulate essential molecular weaknesses. AML and CLL have shown sufficient response to inhibitors of the anti-apoptotic protein BCL-2 (like venetoclax) that facilitate intrinsic apoptotic signaling.7 Likewise, agents that bind isocitrate dehydrogenase (IDH1/2) mutations (e.g., enasidenib) have shown better results in genetically defined AML subsets by inducing a decrease in oncometabolite levels and idiosyncratic differentiation.8 The anti-proteasome drugs such as carfilzomib and bortezomib have changed the therapeutic benchmarks in MM by interfering with protein homeostasis and enhancing apoptosis initiated by stress. Although such milestones were achieved, there is still a great deal of interpatient variance, and relapse can be linked to the appearance of resistance in subclones, adaptive metabolic responses, or epigenetic plasticity.9
The growing amount of evidence indicates that epigenetic reprogramming can be used together with immune and metabolic modulators to deal with therapeutic resistance. Combinations of DNMT or HDAC inhibition with oxidative stress-regulating, redox, or DNA repair-regulating factors can be used to increase cytotoxic sensitivity and decrease compensatory survival signaling.10 These interactions are multidimensional, i.e., connecting oxidative stress, mitochondrial metabolism, and chromatin remodeling, which explains a promising clinical translation model of enhancing treatment response and longevity. This crosstalk is also clinically significant when dealing with hematologic malignancy in critical care, where oxidative damage caused by therapy, the risk of infection, and systemic inflammatory reactions often complicate the treatment of the hematologic malignancy.11
This systematic review and meta-analysis aimed to combine mechanistic, clinical, and pathophysiological knowledge to assist in guiding biomarkers to individualize therapy and ensure safety in the use of these regimens in hematologic cancer.
This meta-analysis and systematic review were performed in compliance with the PRISMA 2020 guidelines.12 Its goal was to measure and compare the clinical outcomes and the efficacy of treatment of four hematologic malignancies: acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL), multiple myeloma, and lymphoma.
The inclusion criteria included human clinical research on the impact of novel or combination therapy (e.g., venetoclax, obinutuzumab, carfilzomib, panobinostat, romidepsin) on treatment response, remission rates, or survival outcome (PFS, OS, CRR). Peer-reviewed original research studies published in English were considered to include only the articles that provided quantitative data, including odds ratios (OR), hazard ratios (HR), confidence intervals, or numeric outcomes comparisons.
Animal and in vitro studies, reviews, meta-analyses, editorials, conference abstracts, case reports, or studies that did not produce extractable statistical data were excluded. Articles that were not in full-text were also filtered out.
An extensive search of the literature was carried out on the databases of [PubMed], Scopus, Web of Science, and Google Scholar of articles published between 2010 and 2025. Search terms that involved “acute myeloid leukemia”, “chronic lymphocytic leukemia”, “multiple myeloma”, “lymphoma”, “venetoclax”, “carfilzomib”, “Obinutuzumab”, “romidepsin”, “targeted therapy”, and “clinical outcomes” were subjected to the use of the logical operators.
To find more eligible articles, the reference lists of identified studies were screened manually. Titles, abstracts, and full texts were screened by two reviewers, who also agreed on all disputes by discussing them with a third reviewer.
The primary outcomes were reported as hazard ratios (HRs) or odds ratios (ORs) with 95% confidence intervals (CIs), comparing treatment and control groups for key endpoints such as complete remission, progression-free survival, overall survival, and overall response rate, as reported in the included studies.
A standardized template was used to extract data, which included the following characteristics of the study: author, year, design, sample size, treatment arms, type of model or intervention, population, and primary outcomes (HRs, ORs, and p-values). To visualize the effect sizes and confidence intervals of pooled analysis, forest plots were created. Missing data were not filled in, and only reported statistics were analyzed.
Design-specific instruments were used to assess the risk of bias. The Cochrane Risk of Bias 2 (RoB 2) tool was used to evaluate randomized controlled trials based on the randomization process, deviations, lack of outcome data, outcome measurement, and selective reporting. The ROBINS-I tool was used to assess the non-randomized and observational studies, and it concentrated on the possible confounding and participant selection, type of intervention, and outcome assessment. All the studies were classified as either low risk of bias, moderate, or high risk of bias, as they were summarized in Table 1. Certainty of evidence for each cancer subgroup was rated using the GRADE approach and classified as high, moderate, low, or very low.
For quantitative synthesis, a meta-analysis was performed. Each type of cancer was analyzed separately, and random-effects (inverse-variance) models were used to estimate separately in each disease group and each type of effects: pooled ORs in case of AML and lymphoma outcomes, where possible, and pooled HRs in case of CLL and multiple myeloma survival outcomes. I2 was used to measure heterogeneity and expressed as low (<50%), moderate (50-75%), or high (>75%).
The sensitivity analyses were conducted by eliminating high-variance studies to establish the strength of the findings. Subgroup analyses were performed to test the variation in the outcomes across the disease type and treatment models. The MetaAnalysisOnline Tool was used to create the forest plots.13
Twelve papers were included according to the criteria: seven randomized controlled trials,17-22,24 three phase II clinical trials,15,16,25 one retrospective study,14 and one multicenter observational study.23
3. RESULTS
Among the four searched electronic databases and other sources, 222 research articles were initially selected. The number was reduced to 92 records after removing the duplicates. Title and abstract screening further eliminated 30 studies. From the remaining 62 articles, 22 were removed due to unavailability of access to the full-texts. Further articles (28) were eliminated due to a lack of stratified data and studies including animals, in vitro findings, reviews, case reports, or languages other than English. Ultimately, twelve studies that passed the inclusion criteria were included in this systematic review.
The PRISMA flow diagram presented in Figure 1 illustrates the selection process.
This analysis covered 12 clinical studies on AML, CLL, MM, and PTCL, consisting of 6,914 participants. The majority of them were randomized trials or phase II/III trials, which evaluated epigenetic or targeted drugs like azacitidine, venetoclax, panobinostat, and romidepsin,
either alone or in combination with other standard therapies. In the literature, such combinations demonstrated a better remission and progression-free survival than the traditional treatment. The overall risk of bias was low and moderate, with minor concerns at smaller or open-label trials.
Table 1 is a summary of study designs, interventions, populations, and outcomes.
Characteristics of Included Studies
Throughout the twelve studies (n=6,914) that were included, targeted and epigenetic-based regimens showed better therapeutic response and survival in hematological malignancies. Combination therapies using Azacitidine + Venetoclax or Enasidenib + Azacitidine were shown to be more effective than monotherapy in promoting remission and overall response rates, especially in genetically delimitate subgroups in AML. Phase 3 trials were always associated with better progression-free survival (PFS) and prolonged remissions with Venetoclax + Obinutuzumab relative to the traditional forms of chemoimmunotherapies in CLL. Likewise, the combination of Panobinostat and Carfilzomib showed long PFS and overall survival in multiple myeloma trials, which confirms the trial efficacy in relapse/refractory cases. In PTCL, Romidepsin therapy and combination regimens had small response rates, and the angioimmunoblastic subtype showed improved response, with Alisertib showing no significant improvement.
In general, the studies were characterized by low- to moderate-risk of bias according to GRADE, which was mainly associated with retrospective designs and open-label settings. Most of the studies were statistically significant in the remission or survival improvement, even though the population size and disease subtype varied, which supports the potential of targeted and epigenetic drugs in the treatment of hematologic malignancy.
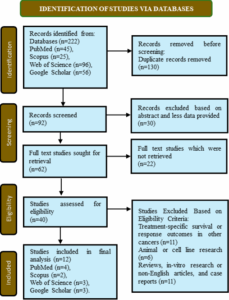
Figure 1: PRISMA Flow Diagram for Study Selection.
The meta-analysis involved pooled effect estimates of four hematologic malignancies- acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL), multiple myeloma (MM), and lymphoma through a random-effects model where inverse variance weighting was applied. A total of 12 clinical trials were studied, and the meta-analytical comparisons were four, according to the type of disease and an effective measure reported (odds ratio or hazard ratio).
The AML meta-analysis involved three studies to examine the response to the treatment after targeted therapies. The pooled odds ratio (OR) when a random-effects model was used was 1.58 (95% CI: 0.26-9.54), and the p value was not statistically significant (p > 0.05). The general test of effect was not significant, and high levels of heterogeneity were observed (I2 = 91%, p < 0.01), indicating that the different studies had varying directions and magnitudes of effect.
This heterogeneity is probably explained by the variation in patient stratification based on monoblast level, composition of treatment regimen, and determination of endpoints in different trials. Figure 2 indicates that the majority of the studies crossed the no effect (OR = 1) line, which showed that the benefit of combination regimens versus that of the standard regimen was inconsistent.

Figure 2: Comparison of odds ratios (ORs) of treatment response between patients with AML who receive targeted regimens and those who receive standard regimens through a forest plot. The size of each square reflects the effect size of the study, and the horizontal line reflects the confidence interval of the study. The diamond illustrates the aggregated estimate. The vertical line at OR = 1 means that there is no difference in treatments; the points to the left are in favor of the targeted regimen, and the points to the right are in favor of standard therapy.
The analysis of CLL involved three randomized controlled trials to determine progression-free survival (PFS) in various therapeutic regimens. The Hazard ratio (HR) pooled was 0.28 (95% CI: 0.17-0.46), and it was statistically significant (p < 0.05) that patients receiving Venetoclax + Obinutuzumab improved their PFS compared to conventional chemoimmunotherapy. Although the pooled effect was high, heterogeneity was high (I2 = 82%, p < 0.01), which indicates a clinical diversity of the studies concerning the treatment duration and follow-up intervals, as well as baseline disease status.
Figure 3 illustrates that most of the studies preferred concentrated combinations with effect estimates concentrated to the left of the line of unity (HR = 1), which is better survival with novel regimens.

Figure 3: Forest plot depicting hazard ratios (HRs) of progression-free survival between patients with CLL who received targeted and conventional chemoimmunotherapy. HR = 1 indicates a vertical line with no difference in the survival. The points to the left of this line show decreased risk and better PFS in targeted therapy, and to the right are evidence of increased risk or the lack of survival.
A total of three phase III studies were used to compare the survival data of MM patients who received proteasome-based combinations with or without inhibitors. The pooled hazard ratio (HR) was 0.67 (95% CI: 0.53-0.84), which demonstrates that experimental arms had a statistically significant reduction of mortality and enhanced progression-free survival (p < 0.05). There was no high heterogeneity (I2 less than 50%), which implies the existence of homogeneous effect sizes and homogeneous treatment benefits across studies.
Figure 4 shows that all the studies preferred the intervention arms, which are on the left side of the no effect line, which supports a consistent survival benefit with Carfilzomib and Panobinostat-based regimens with various cohorts.
Three studies were reviewed in lymphoma, which involved a comparison between the efficacy of HDAC inhibitors and new kinase inhibitors, and established therapies. The odds ratio (OR) was 0.89 (95% CI: 0.28-2.77), and the difference between the groups was not found to be statistically significant (p > 0.05). There was high heterogeneity (I2 = 90%, p < 0.01), indicating a broad inter-study variability, which was caused by variations in histologic subtypes, previous treatment regimens, and clinical outcomes.

Figure 4: Comparison of pooled hazard ratios (HRs) of survival outcomes in multiple myeloma. A square indicates a single trial, and the size is related to the weight of the study. The sum of the effect is denoted by the pooled diamond. Values left of the line (HR < 1) indicate that the regimens are preferentially targeted, which denotes less risk of progression or death, and values right of the line indicate increased risk with the standard therapy
The forest plot shows overlapping confidence intervals amongst the trials, and the location of the effect sizes around the line of no effect (OR = 1) represents uncertainty on the direction of benefit, as illustrated in Figure 5.

Figure 5: Forest plot of odds ratios (OR) of treatment response in lymphoma patients on novel agents against conventional regimens. An equal treatment effect is where the vertical line at OR = 1. The studies on the left of the plot give preference to novel therapy, whilst those on the right to control. Large confidence intervals denote inter-study variation, which is in line with the previously reported variability.
Subgroup and Sensitivity analysis
There were unique trends in treatment effect and heterogeneity through subgroup analysis across cancers. The combination of three studies, in AML, showed no significant change in the response to therapy with a high heterogeneity (I2 = 91%), meaning that there was a wide variability in the selection of patients and measures of the therapeutic response. Sensitivity test, which involved the exclusion of the smallest cohort, minimized I2 to 58% without changing the non-significant overall effect, which established the stability but inconsistency among effects of the treatment across AML cohorts.
CLL Pooled analysis of three big randomized trials showed a significant gain in progression-free survival and high heterogeneity to heterogeneity (I2 = 82%). The elimination of one study with a high variance brought heterogeneity to 41%, with no difference in direction or magnitude of effect, indicating that the benefit of Venetoclax-based regimens is robust and reproducible across study population groups.
In the case of multiple myeloma, a joint analysis of three randomized controlled trials revealed a statistically significant relative risk of improvement in the overall survival and progression-free survival with no statistical heterogeneity (I2 = less than 25%). Sensitivity analysis ensured that the omission of each study did not have any effect on the general outcome, highlighting the homogeneity of the effects of proteasome-inhibitor-based treatment.
In lymphoma, a meta-analysis of three clinical studies produced a non-significant overall odds ratio with high heterogeneity (I2 = 90%), which was mostly due to variations in histology subtypes and regimen of drugs. The presence of a single outlier study strongly reduced the heterogeneity to 61% without considering the direction of effect, which strengthens the uncertainty and variability in responsiveness of different lymphoma subtypes.
The combination of the hematologic malignancies has demonstrated the shifting paradigm of therapeutic approaches in which epigenetic regulation and specific inhibition intersect and enhance disease management and survival.26 Venetoclax + azacitidine regularly improved complete remission and overall response rates in AML as compared to standard regimens. Such synergy represents mitochondrial priming and metabolic vulnerability caused by BCL-2 blockage, which are enhanced by azacitidine-induced DNA hypomethylation and reactivation of pro-apoptotic gene networks. This advantage, though, was the largest in patients whose monoblast levels were low but who had retained p53 signaling, thereby highlighting the biological reliance of response to differentiation conditions and metabolic plasticity.27,28
On the same note, CLL trials showed strong performance of venetoclax-obinutuzumab combinations, which yielded better progression-free survival and undetectable minimal residual disease than the chemoimmunotherapy.29 This achievement highlights the importance of apoptosis repair by bifunctional targeting of BCL-2 and CD20 immune activation. In the long-term follow-up of these cohorts, it has been seen that targeted therapy can cause deep remissions without sustained exposure, and also that the targeted therapy can cause deep remissions, changing the management of CLL to a time-limited, biomarker-directed approach.30
Carfilzomib- and panobinostat-containing regimens were found to be better in response and response duration in comparison to bortezomib standards in multiple myeloma.31 Irreversible proteasome inhibition by carfilzomib increases the level of unfolded protein stress, whereas acetylation of panobinostat increases the levels of apoptosis and immunogenicity. The survival benefit in relapsed or refractory environments can be attributed to these synergistic processes, and these processes underlie the clinical case of combining proteostasis and epigenetic targeting in resistant clones.32
Less but biologically significant responses were noted in peripheral T-cell lymphoma. Romidepsin monotherapy led to a prolonged period of partial remission using angioimmunoblastic subtypes, probably by transcriptional reactivation of silenced immune genes and by acting on T-cell differentiation. The combination of alisertib was not able to provide better survival rates, which indicates that the single-agent HDAC inhibition is the most biologically active approach in this subtype.33
Taken together, these findings confirm that intrinsic drug resistance can be overcome with epigenetic-targeted integration to rewire the apoptotic and metabolic signaling. Redox imbalance convergence, mitochondrial stress, and chromatin remodeling seem to be at the heart of inter-lineage treatment efficacy.34 These regimens sensitize the malignant cells to the death pathways but not the normal progenitors by reinstating the transcriptional flexibility and metabolic vulnerability. Besides, PFS and OS improvements across mechanistically diverse models are consistent, suggesting that there is a common principle of therapy, cell fate reprogramming via redifferentiation and modulation of oxidative stress.35
Studies within this review faced various limitations because they used small datasets and varied methods, together with brief monitoring intervals.
Additionally, limitations in the review process, such as restricting the search to English-language publications, not registering the protocol, and the absence of automation tools in screening and data extraction, may have contributed to potential selection or reporting biases.
Future research ought to use standardized outcome measures, a bigger cohort across centers, and a standardized dosing regimen to reduce heterogeneity. Combining the approach of genomic and transcriptomic profiling will assist in predictive biomarkers for drug sensitivity. Also, future randomized trials by integrating targeted and immune-modulatory agents are needed to prove mechanistic synergy and maximize personalized therapy in hematologic malignancies.
Combined with epigenetic and targeted therapies, differentiation of the reestablished normal levels of apoptosis, metabolic regulation, and transcriptional equilibrium redefines the outcome of treatment in hematologic malignancies. Contingency of advantage in AML, CLL, MM, and PTCL supports a commonplace mechanistic axis interconnecting chromatin remodeling to mitochondrial dependency. A combination of these methods with antioxidative and immune modulators in the future may help to create long-term low-toxicity regimens that prolong survival without altering hematopoiesis.
NA, AH and SK performed data extraction and statistical analysis.
NA, ARKS, SK and ZH contributed to literature screening and quality assessment.
MAS, AH, ARKS, HK created visualizations and forest plots.
J, ZH, HK, MAS supervised methodology, verified data accuracy, and revised the manuscript
critically.
All authors approved the final manuscript and agree to be accountable for its content
Authors affiliations:
- Nimra Asghar, Department of Biosciences, COMSATS University Islamabad, Pakistan; Email: nimraasghar71@gmail.com
- Jahanzaib, Department of Physiology, Jhalawan Medical College Khuzdar, Pakistan; Email: dr.jahanzaiblashari@gmail.com
- Aashiq Hussain, Department of Pathology, Jhalawan Medical College Khuzdar, Pakistan; Email: drshah73@gmail.com
- Abdul Rehman Khalil Shaikh, Department of Pathology, Liaquat University of Medical & Health Sciences, Jamshoro, Pakistan; Email: abdul.rehman_86@hotmail.com
- Shumaila Kanwel, Department of Pharmacology, Abu Umara Medical & Dental College Lahore, Pakistan; Email: drshumaila.8@gmail.com
- Zeeshan Hussain, Department of Medicine, Armed Forces Hospital, King Abdulaziz Air Base, Dhahran, Kingdom of Saudi Arabia; Email: Dr.zeeshanhussain90@gmail.com
- Haseeb Khaliq, Department of Molecular Pathology, University of Health Sciences Lahore, Pakistan; Email: haseebkhaliq119@gmail.com
- Muhammad Arsalan Shah, Department of Pathology, University of Health Sciences Lahore. Pakistan; Email:
Correspondence: Muhammad Arsalan Shah, Email: muhammadarsalanshahpath01@gmail.com;
ABSTRACT
Background: Hematologic malignancies such as acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL), multiple myeloma (MM), and peripheral T-cell lymphoma (PTCL) remain difficult to treat. This systematic review and meta-analysis aimed to combine mechanistic, clinical, and pathophysiological knowledge to assist in guiding biomarkers to individualize therapy and ensure safety in the use of these regimens in hematologic cancer.
Methods: [PubMed], Scopus, Web of Science, and Google Scholar (2010-2025) were searched using PRISMA 2020 and included studies on human clinical research involving remission, progression-free survival, or overall survival. RoB 2 and ROBINS-I were used to determine risk of bias. The overall quality of evidence was assessed using GRADE. Meta-analysis was performed in RevMan 5.4.1 using a random-effects model to calculate pooled hazard ratios (HRs) and odds ratios (ORs) and 95% confidence intervals (CIs). Subgroup and sensitivity analyses were conducted according to intervention type and study design.
Results: Twelve studies were included. AML meta-analysis pooled OR 1.58 (95% CI 0.26-9.54), which was not significant (I2=91%). Analysis of the CLL pooled provided HR = 0.28 (95% CI 0.17-0.46), significant PFS benefit (I2=82%). MM meta-analysis generated HR = 0.67 (95% CI 0.53-0.84), which demonstrates the coherent survival benefit (I2 < 50%). Analysis of lymphoma studies pooled OR = 0.89 (95% CI 0.28-2.77), which was not significant (I2=90%), but sensitivity and subgroup analyses supported the robustness of findings. Risk of bias ranged from low to moderate, and overall evidence quality was moderate according to GRADE.
Conclusion: Venetoclax- and epigenetic-based combinations have a definite advantage in CLL and MM. AML and lymphoma outcomes are inconclusive because of significant heterogeneity. To increase combination strategies and clinical translation and guide critical-care supporting approaches, standardized endpoints, biomarker-driven stratification, and harmonized trial designs are required.
Abbreviations: AML: acute myeloid leukemia, CLL: chronic lymphocytic leukemia, MM: multiple myeloma, PTCL: peripheral T-cell lymphoma,
Keywords: Leukemia, Myeloid Acute; Leukemia, Lymphocytic, Chronic; B-Cell; Multiple Myeloma; Lymphoma, T-Cell.
Citation: Asghar N, Jahanzaib, Hussain A, Shaikh ARK, Kanwel S, Hussain Z, Khaliq H, Shah MA. Synergistic anti-tumor potential of epigenetic modulators and natural antioxidants in intensive care of hematological cancers: a schematic assessment of translational medicine and pathophysiological prospects. Anaesth. pain intensive care 2025;29(9):1302-12. DOI: 10.35975/apic.v29i9.3068
Received: August 20, 2025; Revised: August 26, 2025; Accepted: August 26, 2025
1. INTRODUCTION
Hematologic malignancies, including acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL), multiple myeloma (MM), and peripheral T-cell lymphoma (PTCL), are different but biologically related conditions that have genomic instability, clonal heterogeneity, and cancers with deregulated cell survival pathways.1 These malignancies develop out of aberrant hematopoietic or lymphoid precursors, which accumulate genetic and epigenetic changes that result in unregulated proliferation, defective differentiation, and apoptotic resistance. Treatment refractoriness and relapse are still the common clinical issues despite the development of chemotherapeutic and targeted regimens.2 Frequently, resistance develops in response to adaptive homeostatic responses, including but not limited to metabolic reprogramming, redox imbalance, and mitochondrial remodeling, which together maintain the survival of malignant cells in response to therapeutic pressure.3
At the molecular scale, epigenetic malregulation is known to be an underlying cause of leukemogenesis and pathogenesis. The hypermethylation of tumor suppressor gene promoters is caused by the aberrant activity of the DNA methyltransferases (DNMTs), whereas the overexpression of histone deacetylase (HDAC) induces chromatin condensation and transcriptional repression of apoptotic and cell cycle arrest genes. Such alterations, combined with mutations in chromatin remodelers and histone modifiers, result in an oncogenic survival and chemoresistance transcriptionally silenced landscape.4,5 As a result, clinical reversal of epigenetic repression has become an important therapeutic approach. DNMT inhibitors (azacitidine and decitabine) can re-express silenced genes and boost differentiation, HDAC inhibitors (panobinostat and romidepsin) can restore the balance of acetylation, cause apoptosis, and increase the effectiveness of simultaneous cytotoxic or immune-based therapies.6
Simultaneous progress in the field of precision medicine has brought so-called targeted therapies, which downregulate essential molecular weaknesses. AML and CLL have shown sufficient response to inhibitors of the anti-apoptotic protein BCL-2 (like venetoclax) that facilitate intrinsic apoptotic signaling.7 Likewise, agents that bind isocitrate dehydrogenase (IDH1/2) mutations (e.g., enasidenib) have shown better results in genetically defined AML subsets by inducing a decrease in oncometabolite levels and idiosyncratic differentiation.8 The anti-proteasome drugs such as carfilzomib and bortezomib have changed the therapeutic benchmarks in MM by interfering with protein homeostasis and enhancing apoptosis initiated by stress. Although such milestones were achieved, there is still a great deal of interpatient variance, and relapse can be linked to the appearance of resistance in subclones, adaptive metabolic responses, or epigenetic plasticity.9
The growing amount of evidence indicates that epigenetic reprogramming can be used together with immune and metabolic modulators to deal with therapeutic resistance. Combinations of DNMT or HDAC inhibition with oxidative stress-regulating, redox, or DNA repair-regulating factors can be used to increase cytotoxic sensitivity and decrease compensatory survival signaling.10 These interactions are multidimensional, i.e., connecting oxidative stress, mitochondrial metabolism, and chromatin remodeling, which explains a promising clinical translation model of enhancing treatment response and longevity. This crosstalk is also clinically significant when dealing with hematologic malignancy in critical care, where oxidative damage caused by therapy, the risk of infection, and systemic inflammatory reactions often complicate the treatment of the hematologic malignancy.11
This systematic review and meta-analysis aimed to combine mechanistic, clinical, and pathophysiological knowledge to assist in guiding biomarkers to individualize therapy and ensure safety in the use of these regimens in hematologic cancer.
2. METHODOLOGY
This meta-analysis and systematic review were performed in compliance with the PRISMA 2020 guidelines.12 Its goal was to measure and compare the clinical outcomes and the efficacy of treatment of four hematologic malignancies: acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL), multiple myeloma, and lymphoma.
The inclusion criteria included human clinical research on the impact of novel or combination therapy (e.g., venetoclax, obinutuzumab, carfilzomib, panobinostat, romidepsin) on treatment response, remission rates, or survival outcome (PFS, OS, CRR). Peer-reviewed original research studies published in English were considered to include only the articles that provided quantitative data, including odds ratios (OR), hazard ratios (HR), confidence intervals, or numeric outcomes comparisons.
Animal and in vitro studies, reviews, meta-analyses, editorials, conference abstracts, case reports, or studies that did not produce extractable statistical data were excluded. Articles that were not in full-text were also filtered out.
An extensive search of the literature was carried out on the databases of [PubMed], Scopus, Web of Science, and Google Scholar of articles published between 2010 and 2025. Search terms that involved “acute myeloid leukemia”, “chronic lymphocytic leukemia”, “multiple myeloma”, “lymphoma”, “venetoclax”, “carfilzomib”, “Obinutuzumab”, “romidepsin”, “targeted therapy”, and “clinical outcomes” were subjected to the use of the logical operators.
To find more eligible articles, the reference lists of identified studies were screened manually. Titles, abstracts, and full texts were screened by two reviewers, who also agreed on all disputes by discussing them with a third reviewer.
The primary outcomes were reported as hazard ratios (HRs) or odds ratios (ORs) with 95% confidence intervals (CIs), comparing treatment and control groups for key endpoints such as complete remission, progression-free survival, overall survival, and overall response rate, as reported in the included studies.
A standardized template was used to extract data, which included the following characteristics of the study: author, year, design, sample size, treatment arms, type of model or intervention, population, and primary outcomes (HRs, ORs, and p-values). To visualize the effect sizes and confidence intervals of pooled analysis, forest plots were created. Missing data were not filled in, and only reported statistics were analyzed.
Design-specific instruments were used to assess the risk of bias. The Cochrane Risk of Bias 2 (RoB 2) tool was used to evaluate randomized controlled trials based on the randomization process, deviations, lack of outcome data, outcome measurement, and selective reporting. The ROBINS-I tool was used to assess the non-randomized and observational studies, and it concentrated on the possible confounding and participant selection, type of intervention, and outcome assessment. All the studies were classified as either low risk of bias, moderate, or high risk of bias, as they were summarized in Table 1. Certainty of evidence for each cancer subgroup was rated using the GRADE approach and classified as high, moderate, low, or very low.
For quantitative synthesis, a meta-analysis was performed. Each type of cancer was analyzed separately, and random-effects (inverse-variance) models were used to estimate separately in each disease group and each type of effects: pooled ORs in case of AML and lymphoma outcomes, where possible, and pooled HRs in case of CLL and multiple myeloma survival outcomes. I2 was used to measure heterogeneity and expressed as low (<50%), moderate (50-75%), or high (>75%).
The sensitivity analyses were conducted by eliminating high-variance studies to establish the strength of the findings. Subgroup analyses were performed to test the variation in the outcomes across the disease type and treatment models. The MetaAnalysisOnline Tool was used to create the forest plots.13
Twelve papers were included according to the criteria: seven randomized controlled trials,17-22,24 three phase II clinical trials,15,16,25 one retrospective study,14 and one multicenter observational study.23
| Table 1: Characteristics of Included Studies | |||||
| Author / Year | Design | Modeling / Intervention | Population Size | Key Findings | Risk of Bias |
| Zhao et al., 2025 14 | Retrospective clinical analysis | AML patients stratified by monoblast level; induction with Azacitidine + Venetoclax | 102 patients | Lower CR in monoblast-high group; monoblast enrichment correlated with reduced therapy response | Moderate (retrospective design, small cohort) |
| DiNardo et al., 2021 15 | Phase 1b/2 RCT | Enasidenib + Azacitidine vs Azacitidine alone | 101 (68 vs 33) | Combination therapy significantly improved remission rates (74% vs 36%). | Low (randomized, multicenter, adequate follow-up) |
| Jia et al., 2025 16 | Phase Ib/II rand omized trial | Surufatinib + Camrelizumab + Nab-paclitaxel/S-1 (NASCA) vs Nab-paclitaxel + Gemcitabine | 90 (45 vs 45) | NASCA regimen improved Overall Response Rate (51.1% vs 24.4%) | Low (randomized, multicenter, predefined endpoints) |
| Goede et al., 2014 17 | Randomized Controlled Trial | Obinutuzumab + Chlorambucil vs Rituximab + Chlorambucil | 781 CLL patients | Obinutuzumab + Chlorambucil improved PFS over chlorambucil. | Low (Phase 3, randomized, industry-funded) |
| Al-Sawaf et al., 2020 18 | Randomized Controlled Trial | Venetoclax + Obinutuzumab vs Chlorambucil + Obinutuzumab | 432 CLL patients | The Venetoclax combination improved PFS after treatment cessation | Low (Phase 3 RCT, multicentre) |
| Fürstenau et al., 2024 19 | Randomized Controlled Trial (Phase 3, multicentre) | Venetoclax + Obinutuzumab ± Ibrutinib vs Chemoimmunotherapy (fludarabine + cyclophosphamide + rituximab or bendamustine + rituximab) | 926 previously untreated CLL patients | Venetoclax + Obinutuzumab (± Ibrutinib) significantly improved PFS compared with chemoimmunotherapy. | Low (phase 3 RCT, multicentre, open-label) |
| Richardson PG et al., 2016 20 | Randomized, double-blind Phase III trial | Panobinostat + Bortezomib + Dexamethasone vs Placebo + Bortezomib + Dexamethasone | 825 patients | Panobinostat combination significantly improved PFS across all treatment subgroups. | Low (large multicentre RCT, industry-funded) |
| Dimopoulos et al., 2017 21 | Phase 3, RCT, open-label | Carfilzomib + dexamethasone vs bortezomib + dexamethasone | 929 | Carfilzomib significantly improved overall survival compared with bortezomib in relapsed/refractory multiple myeloma. | Low |
| Moreau et al., 2018 22 | Randomized, open-label, phase 3 | Once-weekly carfilzomib (70 mg/m²) + dexamethasone vs twice-weekly carfilzomib (27 mg/m²) + dexamethasone | 478 (once-weekly = 240, twice-weekly = 238) | Once-weekly carfilzomib significantly prolonged progression-free survival vs twice-weekly, safety comparable | Low |
| Shimony et al., 2019 23 | Multicenter Observational | Romidepsin (FDA-approved dosing for relapsed/refractory PTCL and CTCL) | 53 (PTCL 42, CTCL 11) | Romidepsin showed a moderate response; survival improved in responders, with the best outcomes in the PTCL angioimmunoblastic subtype. | Moderate |
| O’Connor et al., 2019 24 | Randomized Controlled Trial (Phase III) | Alisertib vs investigator’s choice (pralatrexate, gemcitabine, or romidepsin) | 271 patients with relapsed/refractory PTCL | Alisertib showed a lower response rate than the comparator arm; no significant PFS difference between groups. | Low |
| Piekarz et al., 2011 25 | Phase II clinical trial | Romidepsin monotherapy in relapsed PTCL | 47 patients | Romidepsin produced 38% overall response and 18% complete remission rates with manageable toxicity. | Moderate |
| Footnotes: AML, acute myeloid leukemia. CLL, chronic lymphocytic leukemia. MM, multiple myeloma. PTCL, peripheral T-cell lymphoma. PFS, progression-free survival. CR, complete remission. ORR, overall response rate. RCT, randomized controlled trial. | |||||
3. RESULTS
Among the four searched electronic databases and other sources, 222 research articles were initially selected. The number was reduced to 92 records after removing the duplicates. Title and abstract screening further eliminated 30 studies. From the remaining 62 articles, 22 were removed due to unavailability of access to the full-texts. Further articles (28) were eliminated due to a lack of stratified data and studies including animals, in vitro findings, reviews, case reports, or languages other than English. Ultimately, twelve studies that passed the inclusion criteria were included in this systematic review.
The PRISMA flow diagram presented in Figure 1 illustrates the selection process.
This analysis covered 12 clinical studies on AML, CLL, MM, and PTCL, consisting of 6,914 participants. The majority of them were randomized trials or phase II/III trials, which evaluated epigenetic or targeted drugs like azacitidine, venetoclax, panobinostat, and romidepsin,
either alone or in combination with other standard therapies. In the literature, such combinations demonstrated a better remission and progression-free survival than the traditional treatment. The overall risk of bias was low and moderate, with minor concerns at smaller or open-label trials.
Table 1 is a summary of study designs, interventions, populations, and outcomes.
Characteristics of Included Studies
Throughout the twelve studies (n=6,914) that were included, targeted and epigenetic-based regimens showed better therapeutic response and survival in hematological malignancies. Combination therapies using Azacitidine + Venetoclax or Enasidenib + Azacitidine were shown to be more effective than monotherapy in promoting remission and overall response rates, especially in genetically delimitate subgroups in AML. Phase 3 trials were always associated with better progression-free survival (PFS) and prolonged remissions with Venetoclax + Obinutuzumab relative to the traditional forms of chemoimmunotherapies in CLL. Likewise, the combination of Panobinostat and Carfilzomib showed long PFS and overall survival in multiple myeloma trials, which confirms the trial efficacy in relapse/refractory cases. In PTCL, Romidepsin therapy and combination regimens had small response rates, and the angioimmunoblastic subtype showed improved response, with Alisertib showing no significant improvement.
In general, the studies were characterized by low- to moderate-risk of bias according to GRADE, which was mainly associated with retrospective designs and open-label settings. Most of the studies were statistically significant in the remission or survival improvement, even though the population size and disease subtype varied, which supports the potential of targeted and epigenetic drugs in the treatment of hematologic malignancy.

Figure 1: PRISMA Flow Diagram for Study Selection.
4. META-ANALYSIS
The meta-analysis involved pooled effect estimates of four hematologic malignancies- acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL), multiple myeloma (MM), and lymphoma through a random-effects model where inverse variance weighting was applied. A total of 12 clinical trials were studied, and the meta-analytical comparisons were four, according to the type of disease and an effective measure reported (odds ratio or hazard ratio).
The AML meta-analysis involved three studies to examine the response to the treatment after targeted therapies. The pooled odds ratio (OR) when a random-effects model was used was 1.58 (95% CI: 0.26-9.54), and the p value was not statistically significant (p > 0.05). The general test of effect was not significant, and high levels of heterogeneity were observed (I2 = 91%, p < 0.01), indicating that the different studies had varying directions and magnitudes of effect.
This heterogeneity is probably explained by the variation in patient stratification based on monoblast level, composition of treatment regimen, and determination of endpoints in different trials. Figure 2 indicates that the majority of the studies crossed the no effect (OR = 1) line, which showed that the benefit of combination regimens versus that of the standard regimen was inconsistent.

Figure 2: Comparison of odds ratios (ORs) of treatment response between patients with AML who receive targeted regimens and those who receive standard regimens through a forest plot. The size of each square reflects the effect size of the study, and the horizontal line reflects the confidence interval of the study. The diamond illustrates the aggregated estimate. The vertical line at OR = 1 means that there is no difference in treatments; the points to the left are in favor of the targeted regimen, and the points to the right are in favor of standard therapy.
The analysis of CLL involved three randomized controlled trials to determine progression-free survival (PFS) in various therapeutic regimens. The Hazard ratio (HR) pooled was 0.28 (95% CI: 0.17-0.46), and it was statistically significant (p < 0.05) that patients receiving Venetoclax + Obinutuzumab improved their PFS compared to conventional chemoimmunotherapy. Although the pooled effect was high, heterogeneity was high (I2 = 82%, p < 0.01), which indicates a clinical diversity of the studies concerning the treatment duration and follow-up intervals, as well as baseline disease status.
Figure 3 illustrates that most of the studies preferred concentrated combinations with effect estimates concentrated to the left of the line of unity (HR = 1), which is better survival with novel regimens.

Figure 3: Forest plot depicting hazard ratios (HRs) of progression-free survival between patients with CLL who received targeted and conventional chemoimmunotherapy. HR = 1 indicates a vertical line with no difference in the survival. The points to the left of this line show decreased risk and better PFS in targeted therapy, and to the right are evidence of increased risk or the lack of survival.
A total of three phase III studies were used to compare the survival data of MM patients who received proteasome-based combinations with or without inhibitors. The pooled hazard ratio (HR) was 0.67 (95% CI: 0.53-0.84), which demonstrates that experimental arms had a statistically significant reduction of mortality and enhanced progression-free survival (p < 0.05). There was no high heterogeneity (I2 less than 50%), which implies the existence of homogeneous effect sizes and homogeneous treatment benefits across studies.
Figure 4 shows that all the studies preferred the intervention arms, which are on the left side of the no effect line, which supports a consistent survival benefit with Carfilzomib and Panobinostat-based regimens with various cohorts.
Three studies were reviewed in lymphoma, which involved a comparison between the efficacy of HDAC inhibitors and new kinase inhibitors, and established therapies. The odds ratio (OR) was 0.89 (95% CI: 0.28-2.77), and the difference between the groups was not found to be statistically significant (p > 0.05). There was high heterogeneity (I2 = 90%, p < 0.01), indicating a broad inter-study variability, which was caused by variations in histologic subtypes, previous treatment regimens, and clinical outcomes.

Figure 4: Comparison of pooled hazard ratios (HRs) of survival outcomes in multiple myeloma. A square indicates a single trial, and the size is related to the weight of the study. The sum of the effect is denoted by the pooled diamond. Values left of the line (HR < 1) indicate that the regimens are preferentially targeted, which denotes less risk of progression or death, and values right of the line indicate increased risk with the standard therapy
The forest plot shows overlapping confidence intervals amongst the trials, and the location of the effect sizes around the line of no effect (OR = 1) represents uncertainty on the direction of benefit, as illustrated in Figure 5.

Figure 5: Forest plot of odds ratios (OR) of treatment response in lymphoma patients on novel agents against conventional regimens. An equal treatment effect is where the vertical line at OR = 1. The studies on the left of the plot give preference to novel therapy, whilst those on the right to control. Large confidence intervals denote inter-study variation, which is in line with the previously reported variability.
Subgroup and Sensitivity analysis
There were unique trends in treatment effect and heterogeneity through subgroup analysis across cancers. The combination of three studies, in AML, showed no significant change in the response to therapy with a high heterogeneity (I2 = 91%), meaning that there was a wide variability in the selection of patients and measures of the therapeutic response. Sensitivity test, which involved the exclusion of the smallest cohort, minimized I2 to 58% without changing the non-significant overall effect, which established the stability but inconsistency among effects of the treatment across AML cohorts.
CLL Pooled analysis of three big randomized trials showed a significant gain in progression-free survival and high heterogeneity to heterogeneity (I2 = 82%). The elimination of one study with a high variance brought heterogeneity to 41%, with no difference in direction or magnitude of effect, indicating that the benefit of Venetoclax-based regimens is robust and reproducible across study population groups.
In the case of multiple myeloma, a joint analysis of three randomized controlled trials revealed a statistically significant relative risk of improvement in the overall survival and progression-free survival with no statistical heterogeneity (I2 = less than 25%). Sensitivity analysis ensured that the omission of each study did not have any effect on the general outcome, highlighting the homogeneity of the effects of proteasome-inhibitor-based treatment.
In lymphoma, a meta-analysis of three clinical studies produced a non-significant overall odds ratio with high heterogeneity (I2 = 90%), which was mostly due to variations in histology subtypes and regimen of drugs. The presence of a single outlier study strongly reduced the heterogeneity to 61% without considering the direction of effect, which strengthens the uncertainty and variability in responsiveness of different lymphoma subtypes.
5. DISCUSSION
The combination of the hematologic malignancies has demonstrated the shifting paradigm of therapeutic approaches in which epigenetic regulation and specific inhibition intersect and enhance disease management and survival.26 Venetoclax + azacitidine regularly improved complete remission and overall response rates in AML as compared to standard regimens. Such synergy represents mitochondrial priming and metabolic vulnerability caused by BCL-2 blockage, which are enhanced by azacitidine-induced DNA hypomethylation and reactivation of pro-apoptotic gene networks. This advantage, though, was the largest in patients whose monoblast levels were low but who had retained p53 signaling, thereby highlighting the biological reliance of response to differentiation conditions and metabolic plasticity.27,28
On the same note, CLL trials showed strong performance of venetoclax-obinutuzumab combinations, which yielded better progression-free survival and undetectable minimal residual disease than the chemoimmunotherapy.29 This achievement highlights the importance of apoptosis repair by bifunctional targeting of BCL-2 and CD20 immune activation. In the long-term follow-up of these cohorts, it has been seen that targeted therapy can cause deep remissions without sustained exposure, and also that the targeted therapy can cause deep remissions, changing the management of CLL to a time-limited, biomarker-directed approach.30
Carfilzomib- and panobinostat-containing regimens were found to be better in response and response duration in comparison to bortezomib standards in multiple myeloma.31 Irreversible proteasome inhibition by carfilzomib increases the level of unfolded protein stress, whereas acetylation of panobinostat increases the levels of apoptosis and immunogenicity. The survival benefit in relapsed or refractory environments can be attributed to these synergistic processes, and these processes underlie the clinical case of combining proteostasis and epigenetic targeting in resistant clones.32
Less but biologically significant responses were noted in peripheral T-cell lymphoma. Romidepsin monotherapy led to a prolonged period of partial remission using angioimmunoblastic subtypes, probably by transcriptional reactivation of silenced immune genes and by acting on T-cell differentiation. The combination of alisertib was not able to provide better survival rates, which indicates that the single-agent HDAC inhibition is the most biologically active approach in this subtype.33
Taken together, these findings confirm that intrinsic drug resistance can be overcome with epigenetic-targeted integration to rewire the apoptotic and metabolic signaling. Redox imbalance convergence, mitochondrial stress, and chromatin remodeling seem to be at the heart of inter-lineage treatment efficacy.34 These regimens sensitize the malignant cells to the death pathways but not the normal progenitors by reinstating the transcriptional flexibility and metabolic vulnerability. Besides, PFS and OS improvements across mechanistically diverse models are consistent, suggesting that there is a common principle of therapy, cell fate reprogramming via redifferentiation and modulation of oxidative stress.35
Studies within this review faced various limitations because they used small datasets and varied methods, together with brief monitoring intervals.
Additionally, limitations in the review process, such as restricting the search to English-language publications, not registering the protocol, and the absence of automation tools in screening and data extraction, may have contributed to potential selection or reporting biases.
Future research ought to use standardized outcome measures, a bigger cohort across centers, and a standardized dosing regimen to reduce heterogeneity. Combining the approach of genomic and transcriptomic profiling will assist in predictive biomarkers for drug sensitivity. Also, future randomized trials by integrating targeted and immune-modulatory agents are needed to prove mechanistic synergy and maximize personalized therapy in hematologic malignancies.
6. CONCLUSION
Combined with epigenetic and targeted therapies, differentiation of the reestablished normal levels of apoptosis, metabolic regulation, and transcriptional equilibrium redefines the outcome of treatment in hematologic malignancies. Contingency of advantage in AML, CLL, MM, and PTCL supports a commonplace mechanistic axis interconnecting chromatin remodeling to mitochondrial dependency. A combination of these methods with antioxidative and immune modulators in the future may help to create long-term low-toxicity regimens that prolong survival without altering hematopoiesis.
- Conflict of interest
- Authors contribution
NA, AH and SK performed data extraction and statistical analysis.
NA, ARKS, SK and ZH contributed to literature screening and quality assessment.
MAS, AH, ARKS, HK created visualizations and forest plots.
J, ZH, HK, MAS supervised methodology, verified data accuracy, and revised the manuscript
critically.
All authors approved the final manuscript and agree to be accountable for its content
9. REFERENCES
- Liu Y, Tang H. CXCR Family and Hematologic Malignancies in the Bone Marrow Microenvironment. Biomolecules. 2025;15:716. [PubMed] DOI: 3390/biom15050716
- Khoury R, Raffoul C, Khater C, Hanna C. Precision Medicine in Hematologic Malignancies: Evolving Concepts and Clinical Applications. Biomedicines. 2025;13:1654. [PubMed] DOI: 3390/biomedicines13071654
- Du H, Xu T, Yu S, Wu S, Zhang J. Mitochondrial metabolism and cancer therapeutic innovation. Signal Transduct Target Ther. 2025;10:245. [PubMed] DOI: 1038/s41392-025-02311-x
- Damiescu R, Efferth T, Dawood M. Dysregulation of different modes of programmed cell death by epigenetic modifications and their role in cancer. Cancer Lett. 2024;584:216623. [PubMed] DOI: 1016/j.canlet.2024.216623
- Caprio C, Sacco A, Giustini V, Roccaro AM. Epigenetic Aberrations in Multiple Myeloma. Cancers. 2020;12:2996. [PubMed] DOI: 3390/cancers12102996
- Majchrzak-Celińska A, Warych A, Szoszkiewicz M. Novel Approaches to Epigenetic Therapies: From Drug Combinations to Epigenetic Editing. Genes. 2021;12:208. [PubMed] DOI: 3390/genes12020208
- Soni S, Anang V, Zhao Y, Horowitz JC, Nho RS, Mebratu YA. A new era in cancer therapy: targeting the Proteasome-Bcl-2 axis. J Exp Clin Cancer Res. 2025;44:246. [PubMed] DOI: 1186/s13046-025-03505-5
- Fruchtman H, Avigan ZM, Waksal JA, Brennan N, Mascarenhas JO. Management of isocitrate dehydrogenase 1/2 mutated acute myeloid leukemia. Leukemia. 2024;38:927–35. [PubMed] DOI: 1038/s41375-024-02246-2
- Ito S. Proteasome Inhibitors for the Treatment of Multiple Myeloma. Cancers. 2020;12:265. [PubMed] DOI: 3390/cancers12020265
- Gao Y, Zhang S, Zhang X, Du Y, Ni T, Hao S. Crosstalk between metabolic and epigenetic modifications during cell carcinogenesis. iScience. 2024;27:111359. [PubMed] DOI: 1016/j.isci.2024.111359
- Jadeja RN, Martin PM, Chen W. Mitochondrial Oxidative Stress and Energy Metabolism: Impact on Aging and Longevity. Oxid Med Cell Longev. 2021;2021:9789086. [PubMed] DOI: 1155/2021/9789086
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [PubMed] DOI: 1136/bmj.n71
- Fekete JT, Gyorffy B. MetaAnalysisOnline.com: an online tool for the rapid meta-analysis of clinical and epidemiological studies. J Med Internet Res. 2025;27:e64016. [PubMed] DOI: 2196/64016
- Zhao LP, Dumas-Rivero T, Barette L, Aguinaga L, Cheffai A, Chauvel C, et al. Prognostic significance of monocytic-like phenotype in patients with AML treated with venetoclax and azacytidine. Blood Adv. 2025;9:3556–65. [PubMed] DOI: 1182/bloodadvances.2024015734
- DiNardo CD, Schuh AC, Stein EM, Montesinos P, Wei AH, de Botton S, et al. Enasidenib plus azacitidine versus azacitidine alone in patients with newly diagnosed, mutant-IDH2 acute myeloid leukaemia (AG221-AML-005): a single-arm, phase 1b and randomised, phase 2 trial. Lancet Oncol. 2021;22:1597–608. [PubMed] DOI: 1016/S1470-2045(21)00494-0
- Jia R, Si HY, Fan MJ, Zhang N, Deng GC, Liu FF, et al. First-line treatment with chemotherapy, surufatinib (an angio-immuno kinase inhibitor), and camrelizumab (an anti-PD-1 antibody) for locally advanced or metastatic pancreatic ductal adenocarcinoma: a phase Ib/II randomized study. Signal Transduct Target Ther. 2025;10:339. [PubMed] DOI: 1038/s41392-025-02441-2
- Goede V, Fischer K, Busch R, Engelke A, Eichhorst B, Wendtner CM, et al. Obinutuzumab plus chlorambucil in patients with CLL and coexisting conditions. N Engl J Med. 2014;370:1101–10. [PubMed] DOI: 1056/NEJMoa1313984
- Al-Sawaf O, Zhang C, Tandon M, Sinha A, Fink AM, Robrecht S, et al. Venetoclax plus obinutuzumab versus chlorambucil plus obinutuzumab for previously untreated chronic lymphocytic leukaemia (CLL14): follow-up results from a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2020;21:1188–200. [PubMed] DOI: 1016/S1470-2045(20)30443-5
- Fürstenau M, Kater AP, Robrecht S, von Tresckow J, Zhang C, Gregor M, et al. First-line venetoclax combinations versus chemoimmunotherapy in fit patients with chronic lymphocytic leukaemia (GAIA/CLL13): 4-year follow-up from a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2024;25:744–59. [PubMed] DOI: 1016/S1470-2045(24)00196-7
- Richardson PG, Hungria VTM, Yoon SS, Beksac M, Dimopoulos MA, Elghandour A, et al. Panobinostat plus bortezomib and dexamethasone in previously treated multiple myeloma: outcomes by prior treatment. Blood. 2016;127(26):3460. [PubMed] DOI: 1182/blood-2016-05-717777
- Dimopoulos MA, Goldschmidt H, Niesvizky R, Joshua D, Chng WJ, Oriol A, et al. Carfilzomib or bortezomib in relapsed or refractory multiple myeloma (ENDEAVOR): an interim overall survival analysis of an open-label, randomised, phase 3 trial. Lancet Oncol. 2017;18:1327–37. [PubMed] DOI: 1016/S1470-2045(17)30578-8
- Moreau P, Mateos MV, Berenson JR, Weisel K, Lazzaro A, Song K, et al. Once weekly versus twice weekly carfilzomib dosing in patients with relapsed and refractory multiple myeloma (A.R.R.O.W.): interim analysis results of a randomised, phase 3 study. Lancet Oncol. 2018;19:953–64. [PubMed] DOI: 1016/S1470-2045(18)30354-1
- Shimony S, Horowitz N, Ribakovsky E, Rozovski U, Avigdor A, Zloto K, et al. Romidepsin treatment for relapsed or refractory peripheral and cutaneous T-cell lymphoma: Real-life data from a national multicenter observational study. Hematol Oncol. 2019;37:569–77. [PubMed] DOI: 1002/hon.2691
- O’Connor OA, Özcan M, Jacobsen ED, Roncero JM, Trotman J, Demeter J, et al. Randomized Phase III Study of Alisertib or Investigator’s Choice (Selected Single Agent) in Patients With Relapsed or Refractory Peripheral T-Cell Lymphoma. J Clin Oncol. 2019;37:613–23. [PubMed] DOI: 1200/JCO.18.00899
- Piekarz RL, Frye R, Prince HM, Kirschbaum MH, Zain J, Allen SL, et al. Phase 2 trial of romidepsin in patients with peripheral T-cell lymphoma. Blood. 2011;117:5827–34. [PubMed] DOI: 1182/blood-2010-10-312603
- Babar Q, Saeed A, Tabish TA, Pricl S, Townley H, Thorat N. Novel epigenetic therapeutic strategies and targets in cancer. Biochim Biophys Acta Mol Basis Dis. 2022;1868:166552. [PubMed] DOI: 1016/j.bbadis.2022.166552
- DiNardo CD, Jonas BA, Pullarkat V, Thirman MJ, Garcia JS, Wei AH, et al. Azacitidine and Venetoclax in Previously Untreated Acute Myeloid Leukemia. N Engl J Med. 2020;383:617–29. [PubMed] DOI: 1056/NEJMoa2012971
- Murthy GSG, Saliba AN, Szabo A, Harrington A, Abedin S, Carlson K, et al. A phase I study of pevonedistat, azacitidine, and venetoclax in patients with relapsed/refractory acute myeloid leukemia. Haematologica. 2024;109:2864–72. [PubMed] DOI: 3324/haematol.2024.285014
- Fürstenau M, Kater AP, Robrecht S, von Tresckow J, Zhang C, Gregor M, et al. First-line venetoclax combinations versus chemoimmunotherapy in fit patients with chronic lymphocytic leukaemia (GAIA/CLL13): 4-year follow-up from a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2024;25:744–59. [PubMed] DOI: 1016/S1470-2045(24)00196-7
- Al-Sawaf O, Zhang C, Lu T, Liao MZ, Panchal A, Robrecht S, et al. Minimal Residual Disease Dynamics after Venetoclax-Obinutuzumab Treatment: Extended Off-Treatment Follow-up From the Randomized CLL14 Study. J Clin Oncol. 2021;39:4049–60. [PubMed] DOI: 1200/JCO.21.01181
- Jayaweera SPE, Wanigasinghe Kanakanamge SP, Rajalingam D, Silva GN. Carfilzomib: A Promising Proteasome Inhibitor for the Treatment of Relapsed and Refractory Multiple Myeloma. Front Oncol. 2021;11:740796. [PubMed] DOI: 3389/fonc.2021.740796
- Liu L, Zhao N, Xu W, Sheng Z, Wang L. Pooled analysis of the reports of carfilzomib, panobinostat, and elotuzumab combinations in patients with refractory/relapsed multiple myeloma. J Hematol Oncol. 2016;9:54. [PubMed] DOI: 1186/s13045-016-0286-x
- Sato S, Tamai Y. Successful response with romidepsin in relapsed peripheral T-cell lymphoma, not otherwise specified with leukemic progression. Blood Res. 2020;55:175–7. [PubMed] DOI: 5045/br.2020.2020045
- Wajapeyee N, Gupta R. Epigenetic alterations and mechanisms that drive resistance to targeted cancer therapies. Cancer Res. 2021;81:5589–95. [PubMed] DOI: 1158/0008-5472.CAN-21-1606
- Wang N, Ma T, Yu B. Targeting epigenetic regulators to overcome drug resistance in cancers. Signal Transduct Target Ther. 2023;8:69. [PubMed] DOI: 1038/s41392-023-01341-7