Vernando Yanry Lameky 1 , Isak Roberth Akollo 2 , Jayanti Djarami 3 , Rosdiana Tisye Tiwery 4 , Ruthmeice Dualemban Ngarbingan 5
Authors affiliations:
Background & Objective: Low back pain (LBP) is a common musculoskeletal problem that reduces quality of life and work productivity. This study aimed to design, develop, and evaluate the effectiveness of the CLEMARA Patch, a clove leaf essential oil-based transdermal patch, in reducing LBP intensity.
Methodology: A quasi-experimental design with a pretest-posttest control group was used, involving 30 respondents divided equally into intervention and control groups. The intervention group received the CLEMARA Patch, which contains 4% pure eugenol, applied to the lumbar area for 6–8 hours, while the control group received standard therapy. Pain levels were assessed using the Numeric Rating Scale (NRS) at baseline, during treatment, and post-test.
Results: The intervention group showed a significant reduction in pain from 7.27 ± 2.120 to 1.13 ± 2.416 (P < 0.05), while the control group experienced only a slight, nonsignificant decrease from 6.80 ± 1.821 to 5.27 ± 2.434 (P > 0.05).
Conclusion: The CLEMARA Patch demonstrated superior effectiveness over standard therapy in reducing LBP, highlighting its potential as a safe, natural, and innovative herbal therapy.
Abbreviations: NRS: Numeric Rating Scale, NSAIDs: Non-Steroidal Anti-Inflammatory Drugs, PVP: PEG: Polyethylene Glycol, Polyvinylpyrrolidone, HPMC: Hydroxypropyl Methylcellulose, PG: Propylene Glycol, VCO: Virgin Coconut Oil, MgCl: Magnesium Chloride, SHM: Standardized Herbal Medicine, GRAS: Generally Recognized As Safe
Keywords: CLEMARA Patch; Clove Leaf Essential Oil; Eugenol; Transdermal Patch; Low Back Pain; Non-pharmacological Therapy
Citation: Lameky VY, Akollo IR, Djarami J, Tiwery RT, Ngarbingan RD. Effectiveness of a skin patch, based on clove leaf essential oil, for low back pain. Anaesth. pain intensive care 2025;29(9):1263-70. DOI: 10.35975/apic.v29i9.3063
Received: August 17, 2025; Revised: September 05, 2025; Accepted: September 06, 2025
Low Back Pain (LBP) is a global health problem affecting men and women across various age groups, significantly impacting quality of life and productivity.1 According to the World Health Organization (WHO) in 2022, approximately 1.71 billion people worldwide have musculoskeletal disorders, with LBP ranking third after osteoarthritis (528 million) and rheumatism (335 million), affecting 17.3 million people.2 By 2050, LBP cases are projected to increase to 843 million.3 In Indonesia, Riskesdas (2021) reported 12,914 LBP cases (3.71% of the population), making it the second most common condition after influenza.4 Data from PERDOSSIS (neurology specialist association) across 14 teaching hospitals further identified 4,456 LBP cases from 819 patient visits.5 In Maluku Province, prevalence rose from 3.2% in 2013 to 7.3% in 2018.6
A preliminary study by Lameky et al.7 involving port workers and drivers found that clove leaf oil in roll-on form provided only temporary warmth, lasting 10-15 minutes. Respondents also reported that heavy physical activity and sweating reduced the oil’s absorption, while frequent reliance on over-the-counter painkillers raised concerns. These findings highlight the urgency for effective, practical, and safe alternatives.
LBP management includes pharmacological and non-pharmacological approaches. Non-pharmacological therapies are preferred for their holistic benefits and fewer side effects, including the use of clove leaves.⁸˒⁹ Though often regarded as waste, clove leaves can be distilled into essential oils containing eugenol, β-caryophyllene, and caryophyllene oxide.¹⁰˒¹¹ Prior studies in India, Indonesia, and the United States have shown that clove oil relieves pain more effectively than standard methods.12,13,14 This study therefore sought to design the “CLEMARA Patch,” a clove leaf essential oil-based transdermal patch, leveraging eugenol’s analgesic and anti-inflammatory properties.14
Liquid clove oil poses limitations, such as high volatility, instability, and short duration of action. A transdermal patch offers advantages by ensuring controlled release, longer therapeutic effects, and better stability.15 Patches are also hygienic, practical, and support self-care without dependence on pharmacological drugs.16 From a pharmaceutical perspective, bioadhesive polymers enhance adhesion and transdermal delivery, with potential development as an OTC or phytopharmaceutical product.17
This aligns with WHO recommendations for non-pharmacological LBP therapy,18 Indonesia’s RPJM 2025–2029 (Asta Cita 4 and 5), and the National Research Master Plan (RIRN), supporting self-sufficiency in herbal medicines for musculoskeletal pain.7
Eugenol demonstrates strong analgesic and anti-inflammatory activity. Animal studies show eugenol inhibits acetic-acid-induced pain by up to 92.7% at 100 mg/kg, comparable to indomethacin.18 Its analgesic effects are more evident in the inflammatory phase, linked to inhibiting inflammatory mediators, calcium channels, and TRPV1 receptors. In formulation studies, eugenol has been identified as a natural permeation enhancer with safe and sustained release profiles. Gel preparations with HPMC further confirm its release dynamics, with 3% HPMC yielding optimal release and skin safety. 19
In Indonesia, clove leaf distillation yields 70–85% eugenol, confirming its potential as a local resource.19 WHO recognizes eugenol as Generally Recognized As Safe (GRAS). Eugenol exhibits analgesic, anti-inflammatory, antioxidant, antimicrobial, and neuroprotective properties. Despite these advantages, clinical evidence regarding formulations' long-term effectiveness and stability remains limited. No study has specifically developed a clove leaf essential oil transdermal patch for LBP.
This study, therefore, aimed to design, develop, and evaluate the effectiveness of the CLEMARA Patch in reducing LBP intensity. The research tested two hypotheses: Null (H₀), no difference exists between the CLEMARA Patch and standard therapy; Alternative (H₁), the CLEMARA Patch is more effective. The findings are expected to contribute to the innovation of standardized herbal medicine (SHM) based on Maluku’s local resources, providing a safe, effective, and commercially viable alternative for musculoskeletal pain management.
This study applied a quasi-experimental design with a pretest–posttest control group to evaluate the effectiveness of the CLEMARA Patch, a clove leaf oil–based transdermal patch, in reducing low back pain (LBP).²⁰ The research was conducted at Yos Sudarso Port and Mardika Terminal, Ambon City, from March to September 2025, involving stevedores and drivers as participants.
The study process included clove leaf sorting (July 15), oil distillation (August 8), eugenol isolation at Pattimura University (September 6), patch production at Maluku Husada Health College (September 13), and field implementation (September 15–30).
The patch formula contained 4% pure eugenol, 30 g PVP, 80 g gelatin, 30 g MgCl, 30 g distilled water, 5 g glycerin, 10 g VCO, 5 g PG, 0.2 g PEG, 0.3 g HPMC, and 0.3 g Sensicare, producing nine patch preparations. Each was standardized to 5×7 cm, individually packaged, and sealed for use (Figre 1).21,22,23
Respondents were manual workers with LBP, aged 18–60 years, with an NRS score≥ 2, not on analgesics, and providing informed consent. Exclusion criteria were chronic disease, oil allergy, or open skin wounds.²⁴ A purposive sample of 30 respondents was divided equally into intervention and control groups. The intervention group received the CLEMARA Patch (6–8 hours), while the control group received standard therapy. Pain intensity was assessed using the NRS before, during, and after treatment, alongside monitoring of side effects, comfort, and compliance.
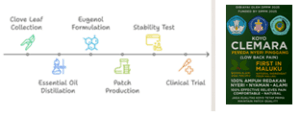
Figure 1. CLEMARA Patch design process
2.1. Statistical Analysis
The research data were analyzed using SPSS version 30. A descriptive analysis was conducted to describe the respondents’ characteristics. Before conducting the inferential test, the assumptions of normality and homogeneity were examined to ensure the validity of the parametric analysis. The Shapiro–Wilk test was used to assess data normality because the sample size was fewer than 50 respondents, and the Levene’s test was applied to evaluate the homogeneity of variances. The results indicated that the data were normally distributed and homogeneous (p > 0.05), thus meeting the assumptions for parametric testing. Therefore, differences in pain scores between the pre- and post-tests within groups were analyzed using the paired sample t-test. The significance level was set at p < 0.05 as the basis for statistical decision-making and to strengthen clinical interpretation. This study received ethical approval from the Research Ethics Committee of STIKes Maluku Husada (No: RK.197.KEPK/STIK/VII/2025). Before participation, all respondents were informed of the study’s objectives, benefits, and potential risks, and then provided consent by signing an informed consent form.
As shown in Table 1, most respondents were in early adulthood (26–35 years), all were male, and the majority had medium–high education. The intervention group consisted entirely of laborers, while the control group consisted of all drivers. Before treatment, in the intervention group 60% of the participants suffered from severe pain; in the control group 53.3% suffered from moderate pain. After intervention, 80% of the intervention group reported no pain, while moderate pain was reported by the control group (46.7%).
3.1. The effect of CLEMARA Patch
The study results in Table 2 indicate that the average pain level in the intervention group before administering the CLEMARA Patch was 7.27 ± 2.120, placing it in the moderate to severe pain category. After using the patch, the average pain level significantly decreased to 1.13 ± 2.416, a difference of 6.14 points. Statistical analysis revealed an Odds Ratio (OR) of 1.95 with a 95% CI of 4.052–8.215, and a P-value of 0.000 (P < 0.05). This demonstrates that using the CLEMARA Patch provides a significant analgesic effect both clinically and statistically. Therefore, the CLEMARA Patch is proven to be effective in reducing low back pain among study participants.
3.2. The Effect of Standard Care on LBP
In Table 3, the control group (standard care) had a mean pain score of 6.80 ± 1.821 before treatment, which was classified as moderate pain. After receiving standard care, the mean pain score slightly decreased to 5.27 ± 2.434, a reduction of 1.53 points. However, statistical analysis showed an Odds Ratio (OR) = 0.464 with a 95% CI = 0.139–3.206 and a p-value of 0.069 (p > 0.05). This suggests that the decrease in pain levels in the control group was not statistically significant. In other words, standard care led to only a small pain reduction and was much less effective than the CLEMARA Patch intervention.
The study results show that most respondents are in the early adulthood age group, a period when physical condition generally reaches its peak. This finding is consistent with Malińska et al. (2021), who stated that early adults, especially drivers and dock workers, are more prone to LBP due to occupational risk factors such as non-ergonomic positions, repetitive movements, and heavy lifting.²5 Prolonged exposure to strenuous physical activity can damage spinal structures and surrounding tissues. In addition, low awareness of ergonomics and inadequate exercise further increase LBP risk. Therefore, education on proper lifting techniques and physical training is crucial to prevent injuries among young adults.
The majority of respondents were male, supporting Jonsdottir et al. (2019), who reported that men face a higher LBP risk compared to women.²⁶ This is linked to occupations dominated by men, such as port laborers and drivers, combined with lifestyle habits that negatively affect back health. Another finding revealed that most respondents had a high level of education (senior high school to university). This aligns with Yosef et al.(2019), who argued that higher education does not guarantee freedom from health problems.²⁷ Poor posture, insufficient physical activity, and psychosocial stressors can still contribute to LBP among educated individuals.
Occupational patterns also played a role, with most respondents working as drivers or port laborers. Khadour et al.(2024) emphasized that such professions increase musculoskeletal strain.²⁸ Drivers spend long hours sitting in static positions, while port laborers frequently lift heavy loads. Both conditions promote muscle tension, poor posture, and repetitive strain injuries, all contributing to LBP.²⁹ Before intervention, respondents reported moderate to severe LBP (pain scores 4–10), indicating significant discomfort affecting daily activities. After applying the CLEMARA Patch and standard care, pain scores dropped to no pain (score 0) or mild pain (scores 1–3). These results mirror Sidiq et al. (2024), who also found significant pain reduction after herbal interventions.³⁰ The presence of eugenol can mechanistically explain the superior effect of the CLEMARA Patch, as the main bioactive compound in clove leaf oil exhibits dual actions: anti-inflammatory and analgesic.
At the molecular level, eugenol inhibits cyclooxygenase (COX) and lipoxygenase (LOX) pathways, thereby reducing the synthesis of pro-inflammatory mediators such as prostaglandins and leukotrienes.³⁵ Moreover, eugenol directly interacts with the transient receptor potential vanilloid 1 (TRPV1) receptor, an ion channel responsible for nociception and thermal pain sensation by blocking calcium influx and desensitizing nociceptors.³⁶ This dual mechanism results in decreased peripheral sensitization and reduced transmission of pain impulses to the central nervous system. Consequently, muscle relaxation and improved microcirculation occur, accelerating tissue recovery.
A comparative analysis showed that the CLEMARA Patch was significantly more effective than standard care, as confirmed by the Paired Sample T-Test. Das et al. (2020) similarly reported that essential oils, including clove oil, reduced LBP more effectively than conventional care.³¹ While pharmacological drugs such as ibuprofen, diclofenac, or naproxen have anti-inflammatory effects, Esmaeili et al. (2022) found their impact limited compared to clove oil, largely due to tolerance and side effects.³² These findings align with previous studies demonstrating that eugenol’s interaction with TRPV1 produces analgesic effects comparable to some synthetic drugs but with a more favorable safety profile.³⁷ In addition to receptor modulation, eugenol scavenges reactive oxygen species (ROS) and upregulates endogenous antioxidant enzymes, further minimizing oxidative stress in inflamed tissues. This multifaceted mechanism supports the observed decline in pain scores following the intervention.
Clinical trials further support these findings. Freiwald et al. (2021) demonstrated that clove oil significantly improved pain outcomes compared to placebo, while Nisar et al. (2021) showed that combining clove oil with physical therapy yielded better results than physical therapy alone.³³,³⁴ Singh et al. (2018)³⁵ confirmed through in vivo studies that eugenol provided stronger analgesic and anti-inflammatory effects in the inflammatory phase compared to diclofenac, though less potent than tramadol at specific doses.
Another strength of eugenol is its safety profile. Unlike NSAIDs and opioids, which carry risks of gastrointestinal or dependency issues, eugenol has minimal side effects and additional antioxidant benefits.³⁶ Clinical evidence even showed that eugenol outperformed articaine in pain control for irreversible pulpitis cases.³⁷ This evidence supports the clinical relevance of eugenol-based interventions such as the CLEMARA Patch, which combine efficacy, safety, and natural bioavailability to achieve sustained pain relief without pharmacological risks.
The CLEMARA Patch, enriched with eugenol from clove leaf oil, proved effective in relieving pain and reducing inflammation, which are the main contributors to LBP. Topical delivery ensures rapid absorption and localized action.³⁸˒³⁹ Through inhibition of inflammatory cascades and modulation of TRPV1 nociceptors, eugenol effectively diminishes both the source and perception of pain. This integrated mechanism underscores the therapeutic potential of the CLEMARA Patch as a safe, natural, and innovative approach for LBP management.
This study was limited by its small sample size (30 respondents, 15 per group), reducing statistical power and representativeness. Long-term effects of the CLEMARA Patch were not assessed, so its repeated-use safety and effectiveness remain unknown. Future research should increase sample size, include more diverse populations, and expand to multiple sites for broader generalizability.
The CLEMARA Patch (clove leaf oil with 4% eugenol) significantly reduced low back pain, achieving an average pain reduction of 6.14 points compared to 1.53 in controls (OR = 195). These results demonstrate strong therapeutic benefits, highlighting the patch as a safe, practical, and innovative non-pharmacological option, with potential for development as a standardized phytopharmaceutical and as the first herbal transdermal product from Maluku.
7. Data Availability
Data generated during this study are available from the corresponding author upon reasonable request.
8. Consent of Patient
All patients were informed about the objectives, procedures, benefits, risks, duration, and timing of the study. They voluntarily agreed to participate and provided written informed consent as proof of approval.
9. Conflict of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
10. Funding
This research utilized funding solely from the Ministry of Higher Education, Science, and Technology (Kemdiktisaintek) of the Republic of Indonesia.
11. Acknowledgments
The authors would like to thank Kemdiktisaintek, the Indonesian Christian University of Maluku, Pattimura University, STIKes Maluku Husada, the Head of the Yos Sudarso Port TKBM, and the Ambon City Transportation Agency for their support and collaboration in this research.
12. Authors’ Contribution
All authors contributed equally to the conceptualization, study design, data collection, analysis, manuscript preparation, editing, and final approval of the draft.
The authors collectively take responsibility for the integrity and accuracy of the research.
Authors affiliations:
- Vernando Yanry Lameky, Universitas Kristen Indonesia Maluku, Ambon, Indonesia, Jln. Jl. Ot Pattimaipauw, Talake - 97115, Indonesia; Email: vernandoyanrylameky@gmail.com; {ORCID:0000-0001-8415-8099}
- Isak Roberth Akollo, Universitas Kristen Indonesia Maluku, Ambon, Indonesia, Jln. Jl. Ot Pattimaipauw, Talake - 97115, Indonesia; Email: roberthakollo@gmail.com; {ORCID:0009-0000-7118-0879}
- Jayanti Djarami, STIKes Maluku Husada, Ambon, Indonesia, Jl. Kebun Cengkeh, Sirimau - 97129, Indonesia; Email: apotekerjayanti@gmail.com
- Rosdiana Tisye Tiwery, Universitas Kristen Indonesia Maluku, Ambon, Indonesia, Jln. Jl. Ot Pattimaipauw, Talake - 97115, Indonesia; Email: diantiwery29@gmail.com
- Ruthmeice Dualemban Ngarbingan, STIKes Maluku Husada, Ambon, Indonesia, Jl. Kebun Cengkeh, Sirimau - 97129, Indonesia; Email: ruthmeicengarbingan@gmail.com
ABSTRACT
Background & Objective: Low back pain (LBP) is a common musculoskeletal problem that reduces quality of life and work productivity. This study aimed to design, develop, and evaluate the effectiveness of the CLEMARA Patch, a clove leaf essential oil-based transdermal patch, in reducing LBP intensity.
Methodology: A quasi-experimental design with a pretest-posttest control group was used, involving 30 respondents divided equally into intervention and control groups. The intervention group received the CLEMARA Patch, which contains 4% pure eugenol, applied to the lumbar area for 6–8 hours, while the control group received standard therapy. Pain levels were assessed using the Numeric Rating Scale (NRS) at baseline, during treatment, and post-test.
Results: The intervention group showed a significant reduction in pain from 7.27 ± 2.120 to 1.13 ± 2.416 (P < 0.05), while the control group experienced only a slight, nonsignificant decrease from 6.80 ± 1.821 to 5.27 ± 2.434 (P > 0.05).
Conclusion: The CLEMARA Patch demonstrated superior effectiveness over standard therapy in reducing LBP, highlighting its potential as a safe, natural, and innovative herbal therapy.
Abbreviations: NRS: Numeric Rating Scale, NSAIDs: Non-Steroidal Anti-Inflammatory Drugs, PVP: PEG: Polyethylene Glycol, Polyvinylpyrrolidone, HPMC: Hydroxypropyl Methylcellulose, PG: Propylene Glycol, VCO: Virgin Coconut Oil, MgCl: Magnesium Chloride, SHM: Standardized Herbal Medicine, GRAS: Generally Recognized As Safe
Keywords: CLEMARA Patch; Clove Leaf Essential Oil; Eugenol; Transdermal Patch; Low Back Pain; Non-pharmacological Therapy
Citation: Lameky VY, Akollo IR, Djarami J, Tiwery RT, Ngarbingan RD. Effectiveness of a skin patch, based on clove leaf essential oil, for low back pain. Anaesth. pain intensive care 2025;29(9):1263-70. DOI: 10.35975/apic.v29i9.3063
Received: August 17, 2025; Revised: September 05, 2025; Accepted: September 06, 2025
1. INTRODUCTION
Low Back Pain (LBP) is a global health problem affecting men and women across various age groups, significantly impacting quality of life and productivity.1 According to the World Health Organization (WHO) in 2022, approximately 1.71 billion people worldwide have musculoskeletal disorders, with LBP ranking third after osteoarthritis (528 million) and rheumatism (335 million), affecting 17.3 million people.2 By 2050, LBP cases are projected to increase to 843 million.3 In Indonesia, Riskesdas (2021) reported 12,914 LBP cases (3.71% of the population), making it the second most common condition after influenza.4 Data from PERDOSSIS (neurology specialist association) across 14 teaching hospitals further identified 4,456 LBP cases from 819 patient visits.5 In Maluku Province, prevalence rose from 3.2% in 2013 to 7.3% in 2018.6
A preliminary study by Lameky et al.7 involving port workers and drivers found that clove leaf oil in roll-on form provided only temporary warmth, lasting 10-15 minutes. Respondents also reported that heavy physical activity and sweating reduced the oil’s absorption, while frequent reliance on over-the-counter painkillers raised concerns. These findings highlight the urgency for effective, practical, and safe alternatives.
LBP management includes pharmacological and non-pharmacological approaches. Non-pharmacological therapies are preferred for their holistic benefits and fewer side effects, including the use of clove leaves.⁸˒⁹ Though often regarded as waste, clove leaves can be distilled into essential oils containing eugenol, β-caryophyllene, and caryophyllene oxide.¹⁰˒¹¹ Prior studies in India, Indonesia, and the United States have shown that clove oil relieves pain more effectively than standard methods.12,13,14 This study therefore sought to design the “CLEMARA Patch,” a clove leaf essential oil-based transdermal patch, leveraging eugenol’s analgesic and anti-inflammatory properties.14
Liquid clove oil poses limitations, such as high volatility, instability, and short duration of action. A transdermal patch offers advantages by ensuring controlled release, longer therapeutic effects, and better stability.15 Patches are also hygienic, practical, and support self-care without dependence on pharmacological drugs.16 From a pharmaceutical perspective, bioadhesive polymers enhance adhesion and transdermal delivery, with potential development as an OTC or phytopharmaceutical product.17
This aligns with WHO recommendations for non-pharmacological LBP therapy,18 Indonesia’s RPJM 2025–2029 (Asta Cita 4 and 5), and the National Research Master Plan (RIRN), supporting self-sufficiency in herbal medicines for musculoskeletal pain.7
Eugenol demonstrates strong analgesic and anti-inflammatory activity. Animal studies show eugenol inhibits acetic-acid-induced pain by up to 92.7% at 100 mg/kg, comparable to indomethacin.18 Its analgesic effects are more evident in the inflammatory phase, linked to inhibiting inflammatory mediators, calcium channels, and TRPV1 receptors. In formulation studies, eugenol has been identified as a natural permeation enhancer with safe and sustained release profiles. Gel preparations with HPMC further confirm its release dynamics, with 3% HPMC yielding optimal release and skin safety. 19
In Indonesia, clove leaf distillation yields 70–85% eugenol, confirming its potential as a local resource.19 WHO recognizes eugenol as Generally Recognized As Safe (GRAS). Eugenol exhibits analgesic, anti-inflammatory, antioxidant, antimicrobial, and neuroprotective properties. Despite these advantages, clinical evidence regarding formulations' long-term effectiveness and stability remains limited. No study has specifically developed a clove leaf essential oil transdermal patch for LBP.
This study, therefore, aimed to design, develop, and evaluate the effectiveness of the CLEMARA Patch in reducing LBP intensity. The research tested two hypotheses: Null (H₀), no difference exists between the CLEMARA Patch and standard therapy; Alternative (H₁), the CLEMARA Patch is more effective. The findings are expected to contribute to the innovation of standardized herbal medicine (SHM) based on Maluku’s local resources, providing a safe, effective, and commercially viable alternative for musculoskeletal pain management.
2. METHODOLOGY
This study applied a quasi-experimental design with a pretest–posttest control group to evaluate the effectiveness of the CLEMARA Patch, a clove leaf oil–based transdermal patch, in reducing low back pain (LBP).²⁰ The research was conducted at Yos Sudarso Port and Mardika Terminal, Ambon City, from March to September 2025, involving stevedores and drivers as participants.
The study process included clove leaf sorting (July 15), oil distillation (August 8), eugenol isolation at Pattimura University (September 6), patch production at Maluku Husada Health College (September 13), and field implementation (September 15–30).
The patch formula contained 4% pure eugenol, 30 g PVP, 80 g gelatin, 30 g MgCl, 30 g distilled water, 5 g glycerin, 10 g VCO, 5 g PG, 0.2 g PEG, 0.3 g HPMC, and 0.3 g Sensicare, producing nine patch preparations. Each was standardized to 5×7 cm, individually packaged, and sealed for use (Figre 1).21,22,23
Respondents were manual workers with LBP, aged 18–60 years, with an NRS score≥ 2, not on analgesics, and providing informed consent. Exclusion criteria were chronic disease, oil allergy, or open skin wounds.²⁴ A purposive sample of 30 respondents was divided equally into intervention and control groups. The intervention group received the CLEMARA Patch (6–8 hours), while the control group received standard therapy. Pain intensity was assessed using the NRS before, during, and after treatment, alongside monitoring of side effects, comfort, and compliance.

Figure 1. CLEMARA Patch design process
2.1. Statistical Analysis
The research data were analyzed using SPSS version 30. A descriptive analysis was conducted to describe the respondents’ characteristics. Before conducting the inferential test, the assumptions of normality and homogeneity were examined to ensure the validity of the parametric analysis. The Shapiro–Wilk test was used to assess data normality because the sample size was fewer than 50 respondents, and the Levene’s test was applied to evaluate the homogeneity of variances. The results indicated that the data were normally distributed and homogeneous (p > 0.05), thus meeting the assumptions for parametric testing. Therefore, differences in pain scores between the pre- and post-tests within groups were analyzed using the paired sample t-test. The significance level was set at p < 0.05 as the basis for statistical decision-making and to strengthen clinical interpretation. This study received ethical approval from the Research Ethics Committee of STIKes Maluku Husada (No: RK.197.KEPK/STIK/VII/2025). Before participation, all respondents were informed of the study’s objectives, benefits, and potential risks, and then provided consent by signing an informed consent form.
3. RESULTS
As shown in Table 1, most respondents were in early adulthood (26–35 years), all were male, and the majority had medium–high education. The intervention group consisted entirely of laborers, while the control group consisted of all drivers. Before treatment, in the intervention group 60% of the participants suffered from severe pain; in the control group 53.3% suffered from moderate pain. After intervention, 80% of the intervention group reported no pain, while moderate pain was reported by the control group (46.7%).
| Table 1: Respondent Characteristics and Low Back Pain Level (n=15) | ||||
| Variable | Intervention Group
n (%) |
Control Group
n (%) |
||
| Age | Late adolescence (17-25 years) | 2 (13.3) | 1 (6.7) | |
| Early adulthood (26-35 years) | 7 (46.7) | 6 (40) | ||
| Late adulthood (36-45 years) | 3 (20) | 4 (26.7) | ||
| Early elderly (46-55 years) | 2 (13.3) | 3 (20) | ||
| Late elderly (56-65 years) | 1 (6.7) | 1 (6.7) | ||
| Gender | Male | 15 (100) | 15 (100) | |
| Female | 0 (0) | 0 (0) | ||
| Education | Low (Elementary-Middle High School) | 5 (33.3) | 6 (40) | |
| High (High School-University) | 10 (66.7) | 9 (60) | ||
| Occupation | Laborers | 15 (100) | 0 (0) | |
| Drivers | 0 (0) | 15 (100) | ||
| Pain level before | No pain (0) | 0 (0) | 0 (0) | |
| Mild pain (1-3) | 1 (6.7) | 2 (13.3) | ||
| Moderate pain (4-6) | 5 (33.3) | 8 (53.3) | ||
| Severe pain (7-10) | 9 (60) | 5 (33) | ||
| Pain level after | No pain (0) | 12 (80) | 0 (0) | |
| Mild pain (1-3) | 2 (13.3) | 3 (20) | ||
| Moderate pain (4-6) | 1 (6.7) | 7 (46.7) | ||
| Severe pain (7-10) | 0 (0) | 5 (33.3) | ||
3.1. The effect of CLEMARA Patch
The study results in Table 2 indicate that the average pain level in the intervention group before administering the CLEMARA Patch was 7.27 ± 2.120, placing it in the moderate to severe pain category. After using the patch, the average pain level significantly decreased to 1.13 ± 2.416, a difference of 6.14 points. Statistical analysis revealed an Odds Ratio (OR) of 1.95 with a 95% CI of 4.052–8.215, and a P-value of 0.000 (P < 0.05). This demonstrates that using the CLEMARA Patch provides a significant analgesic effect both clinically and statistically. Therefore, the CLEMARA Patch is proven to be effective in reducing low back pain among study participants.
| Table 2: The effect of CLEMARA patch administration on low back pain levels | ||||||
| Pain Level | n | Mean ± SD | Standard error | Odds Ratio (95% CI) | P value | |
| Intervention Group (CLEMARA Patch) |
Before | 15 | 7.27 ± 2.120 | 0.547 | 195 (4.052-8.215) | 0.000 |
| After | 15 | 1.13 ± 2.416 | 0.624 | |||
| Difference (δ) | 6.14 | |||||
3.2. The Effect of Standard Care on LBP
In Table 3, the control group (standard care) had a mean pain score of 6.80 ± 1.821 before treatment, which was classified as moderate pain. After receiving standard care, the mean pain score slightly decreased to 5.27 ± 2.434, a reduction of 1.53 points. However, statistical analysis showed an Odds Ratio (OR) = 0.464 with a 95% CI = 0.139–3.206 and a p-value of 0.069 (p > 0.05). This suggests that the decrease in pain levels in the control group was not statistically significant. In other words, standard care led to only a small pain reduction and was much less effective than the CLEMARA Patch intervention.
| Table 3: Effect of standard care on low back pain levels | ||||||
| Pain Level | n | Mean ± SD | Standard Error | Odds Ratio (95% CI) | P value | |
| Control Group (Standard Care) | Before | 15 | 6.80 ± 1.821 | 0.470 | 0.464 (0.139-3.206) | 0.069 |
| After | 15 | 5.27 ± 2.434 | 0.624 | |||
| Difference (δ) | 1.53 | |||||
4.DISCUSSION
The study results show that most respondents are in the early adulthood age group, a period when physical condition generally reaches its peak. This finding is consistent with Malińska et al. (2021), who stated that early adults, especially drivers and dock workers, are more prone to LBP due to occupational risk factors such as non-ergonomic positions, repetitive movements, and heavy lifting.²5 Prolonged exposure to strenuous physical activity can damage spinal structures and surrounding tissues. In addition, low awareness of ergonomics and inadequate exercise further increase LBP risk. Therefore, education on proper lifting techniques and physical training is crucial to prevent injuries among young adults.
The majority of respondents were male, supporting Jonsdottir et al. (2019), who reported that men face a higher LBP risk compared to women.²⁶ This is linked to occupations dominated by men, such as port laborers and drivers, combined with lifestyle habits that negatively affect back health. Another finding revealed that most respondents had a high level of education (senior high school to university). This aligns with Yosef et al.(2019), who argued that higher education does not guarantee freedom from health problems.²⁷ Poor posture, insufficient physical activity, and psychosocial stressors can still contribute to LBP among educated individuals.
Occupational patterns also played a role, with most respondents working as drivers or port laborers. Khadour et al.(2024) emphasized that such professions increase musculoskeletal strain.²⁸ Drivers spend long hours sitting in static positions, while port laborers frequently lift heavy loads. Both conditions promote muscle tension, poor posture, and repetitive strain injuries, all contributing to LBP.²⁹ Before intervention, respondents reported moderate to severe LBP (pain scores 4–10), indicating significant discomfort affecting daily activities. After applying the CLEMARA Patch and standard care, pain scores dropped to no pain (score 0) or mild pain (scores 1–3). These results mirror Sidiq et al. (2024), who also found significant pain reduction after herbal interventions.³⁰ The presence of eugenol can mechanistically explain the superior effect of the CLEMARA Patch, as the main bioactive compound in clove leaf oil exhibits dual actions: anti-inflammatory and analgesic.
At the molecular level, eugenol inhibits cyclooxygenase (COX) and lipoxygenase (LOX) pathways, thereby reducing the synthesis of pro-inflammatory mediators such as prostaglandins and leukotrienes.³⁵ Moreover, eugenol directly interacts with the transient receptor potential vanilloid 1 (TRPV1) receptor, an ion channel responsible for nociception and thermal pain sensation by blocking calcium influx and desensitizing nociceptors.³⁶ This dual mechanism results in decreased peripheral sensitization and reduced transmission of pain impulses to the central nervous system. Consequently, muscle relaxation and improved microcirculation occur, accelerating tissue recovery.
A comparative analysis showed that the CLEMARA Patch was significantly more effective than standard care, as confirmed by the Paired Sample T-Test. Das et al. (2020) similarly reported that essential oils, including clove oil, reduced LBP more effectively than conventional care.³¹ While pharmacological drugs such as ibuprofen, diclofenac, or naproxen have anti-inflammatory effects, Esmaeili et al. (2022) found their impact limited compared to clove oil, largely due to tolerance and side effects.³² These findings align with previous studies demonstrating that eugenol’s interaction with TRPV1 produces analgesic effects comparable to some synthetic drugs but with a more favorable safety profile.³⁷ In addition to receptor modulation, eugenol scavenges reactive oxygen species (ROS) and upregulates endogenous antioxidant enzymes, further minimizing oxidative stress in inflamed tissues. This multifaceted mechanism supports the observed decline in pain scores following the intervention.
Clinical trials further support these findings. Freiwald et al. (2021) demonstrated that clove oil significantly improved pain outcomes compared to placebo, while Nisar et al. (2021) showed that combining clove oil with physical therapy yielded better results than physical therapy alone.³³,³⁴ Singh et al. (2018)³⁵ confirmed through in vivo studies that eugenol provided stronger analgesic and anti-inflammatory effects in the inflammatory phase compared to diclofenac, though less potent than tramadol at specific doses.
Another strength of eugenol is its safety profile. Unlike NSAIDs and opioids, which carry risks of gastrointestinal or dependency issues, eugenol has minimal side effects and additional antioxidant benefits.³⁶ Clinical evidence even showed that eugenol outperformed articaine in pain control for irreversible pulpitis cases.³⁷ This evidence supports the clinical relevance of eugenol-based interventions such as the CLEMARA Patch, which combine efficacy, safety, and natural bioavailability to achieve sustained pain relief without pharmacological risks.
The CLEMARA Patch, enriched with eugenol from clove leaf oil, proved effective in relieving pain and reducing inflammation, which are the main contributors to LBP. Topical delivery ensures rapid absorption and localized action.³⁸˒³⁹ Through inhibition of inflammatory cascades and modulation of TRPV1 nociceptors, eugenol effectively diminishes both the source and perception of pain. This integrated mechanism underscores the therapeutic potential of the CLEMARA Patch as a safe, natural, and innovative approach for LBP management.
5. LIMITATIONS
This study was limited by its small sample size (30 respondents, 15 per group), reducing statistical power and representativeness. Long-term effects of the CLEMARA Patch were not assessed, so its repeated-use safety and effectiveness remain unknown. Future research should increase sample size, include more diverse populations, and expand to multiple sites for broader generalizability.
6. CONCLUSION
The CLEMARA Patch (clove leaf oil with 4% eugenol) significantly reduced low back pain, achieving an average pain reduction of 6.14 points compared to 1.53 in controls (OR = 195). These results demonstrate strong therapeutic benefits, highlighting the patch as a safe, practical, and innovative non-pharmacological option, with potential for development as a standardized phytopharmaceutical and as the first herbal transdermal product from Maluku.
7. Data Availability
Data generated during this study are available from the corresponding author upon reasonable request.
8. Consent of Patient
All patients were informed about the objectives, procedures, benefits, risks, duration, and timing of the study. They voluntarily agreed to participate and provided written informed consent as proof of approval.
9. Conflict of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
10. Funding
This research utilized funding solely from the Ministry of Higher Education, Science, and Technology (Kemdiktisaintek) of the Republic of Indonesia.
11. Acknowledgments
The authors would like to thank Kemdiktisaintek, the Indonesian Christian University of Maluku, Pattimura University, STIKes Maluku Husada, the Head of the Yos Sudarso Port TKBM, and the Ambon City Transportation Agency for their support and collaboration in this research.
12. Authors’ Contribution
All authors contributed equally to the conceptualization, study design, data collection, analysis, manuscript preparation, editing, and final approval of the draft.
The authors collectively take responsibility for the integrity and accuracy of the research.
13. REFERENCES
- Akollo IR, Tasijawa O, Lameky VY. Factors related to low back pain in the working area of Wamlana Public Health Center, Buru District. MolMed. 2023 May 19;16(1):80–87. DOI: 10.30598/molmed.2023.v16.i1.80
- Shashamo BB, Tharwat S, Dai X, Smith A, Bhardwaj P, Meretoja T, et al. Global, regional, and national burden of other musculoskeletal disorders, 1990-2020, and projections to 2050: a systematic analysis of the Global Burden of Disease Study 2021. The Lancet Rheumatology. 2023 Nov 1;5(11):e670–e682. DOI: 10.1016/s2665-9913(23)00232-1
- Cheng M, Xue Y, Cui M, Zeng X, Yang C, Ding F, et al. Global, Regional, and National Burden of Low Back Pain: Findings From the Global Burden of Disease Study 2021 and Projections to 2050. Spine. 2025 Jan 22;50(7):E128–E139. DOI: 10.1097/brs.0000000000005265
- Lameky VY, Goha MM, Tasijawa O. Factors Associated with Low Back Pain in the Working Area of Wamlana Community Health Center, Buru Regency. MHJ. 2024 Aug 14;5(2):1–7. DOI: 10.54639/mhj.v5i1.691
- Alhowimel AS, Alodaibi F, Alshehri MM, Alqahtani BA, Alotaibi M, Alenazi AM. Prevalence and Risk Factors Associated with Low Back Pain in the Saudi Adult Community: A Cross-Sectional Study. IJERPH. 2021 Dec 16;18(24):13288. DOI: 10.3390/ijerph182413288
- Lameky VY, Akollo IR, Tiwery RT. Effectiveness of Clove Leaf (Syzygium aromaticum) and Nutmeg (Myristica fragrans) Essential Oils on Low Back Pain: A Comparative Study in Ambon City. J Kep Flor Night. 2024 Dec 31;7(2):368–374. DOI: 10.52774/jkfn.v7i2.267
- Lameky VY, Pattinasarany DH, Akollo IR, Djarami J, Kolibonso BF. Counseling on Dental and Oral Health, Dental and Oral Care, and Traditional Medicine from Clove Leaves. EJPM. 2023 Nov 29;6(03):355–359. DOI: 10.25134/empowerment.v6i03.8793
- Julianus Sohilait H, Kainama H. Free Radical Scavenging Activity of Essential Oil of Eugenia caryophylata from Amboina Island and Derivatives of Eugenol. Open Chemistry. 2019 July 8;17(1):422–428. DOI: 10.1515/chem-2019-0047
- Nethravathi V, Vijaitha V. Effectiveness of Clove oil massage on Lower Back Pain among Post Natal Mothers at Selected Hospitals, Bangalore. Asia Jour Nurs Educ and Rese. 2015 Jan 1;5(4):467.. DOI: 10.5958/2349-2996.2015.00096.8
- Lating MF, Mahulette AS, Kilkoda AKK. Morphological Diversity of “Tuni” and “Afo” Cloves, Endemic to the Maluku Islands, Indonesia. JTCS. 2024 Nov 4;11(03):335–345. DOI: 10.29244/jtcs.11.03.335-345
- Siringoringo VPM, Dewi N, Bakce D. Analyzing Supply and Demand Response of Indonesian Cloves in International Market. IJOMS. 2023 Mar 25;2(6):2609–2625. DOI: 10.55324/ijoms.v2i6.464
- Susilowati M, Wahyuni S, Setiadi A, Bermawie N. Yield and morphological characteristics of cloves from Semarang plantation, Indonesia. IOP Conf Ser: Earth Environ Sci. 2024 July 1;1377(1):012097. DOI: 10.1088/1755-1315/1377/1/012097
- Han X, Parker TL. Anti-inflammatory activity of clove (Eugenia caryophyllata) essential oil in human dermal fibroblasts. Pharmaceutical Biology. 2017 Jan 1;55(1):1619–1622. DOI: 10.1080/13880209.2017.1314513
- Zyburtowicz K, Ossowicz-Rupniewska P, Muzykiewicz-Szymańska A, Bednarczyk P, Kucharski Ł, Nowak A, et al. Medicinal Anti-Inflammatory Patch Loaded with Lavender Essential Oil. IJMS. 2024 June 4;25(11):6171. DOI: 10.3390/ijms25116171
- Raghav RS, Verma S, Monika M. A Comprehensive Review on Potential Chemical and Herbal Permeation Enhancers Used in Transdermal Drug Delivery Systems. RADDF. 2024 Mar 1;18(1):21–34. DOI: 10.2174/0126673878272043240114123908
- Haro-González JN, Espinosa-Andrews H, Martínez-Velázquez M, Castillo-Herrera GA. Clove Essential Oil (Syzygium aromaticum L. Myrtaceae): Extraction, Chemical Composition, Food Applications, and Essential Bioactivity for Human Health. Molecules. 2021 Oct 22;26(21):6387. DOI: 10.3390/molecules26216387
- Elbestawy MKM, El-Sherbiny GM, Moghannem SA. Antibacterial, Antibiofilm and Anti-Inflammatory Activities of Eugenol Clove Essential Oil against Resistant Helicobacter pylori. Molecules. 2023 Mar 7;28(6):2448. DOI: 10.3390/molecules28062448
- Sharma UK, Kumar R, Pandey A, Pandey AK, Sharma AK, Gupta A. Pharmacological activities of cinnamaldehyde and eugenol: antioxidant, cytotoxic and anti-leishmanial studies. Cell Mol Biol (Noisy-le-grand). 2017 July 31;63(6):73–78. DOI: 10.14715/cmb/2017.63.6.15
- Sharma N, Sharma S, Kaushik R. Formulation and Evaluation of Lornoxicam Transdermal patches using various Permeation Enhancers. Journal of Drug Delivery. 2019 Dec 25;9(4):597–607. DOI: 10.25258/ijddt.9.4.14
- Lameky VY, Nugroho HSW. Book Review of Research and Publication Ethics, written by Santosh Kumar Yadav, Switzerland: Springer Cham, 2023, 255 pp, ISBN 9783031269714, Ebook: $93.08 USD. Health Dyn. 2024 May 28;1(5):144–146. DOI: 10.33846/hd10502
- Upmanyu N, Jain N, Chourasia S, Dangi S, Shukla T. Formulation and evaluation of matrix transdermal patches of meloxicam. J Drug Delivery Ther. 2019 Feb 15;9(1-s):209–213. DOI: 10.22270/jddt.v9i1-s.2326
- Keshari S, Pandey S, Verma A. Formulation and Characterization of Transdermal Patches: A Review. IJPSM. 2024 May 30;9(5):83–90. DOI: 10.47760/ijpsm.2024.v09i05.009
- Fatima A, Apte SS. Formulation and Development of Transdermal Patches. GSC Biol Pharm Sci. 2022 Apr 30;19(1):346–352. 10.30574/gscbps.2022.19.1.0110
- Yoshimoto T, Kokaze A, Ishikawa S, Ochiai H, Muranaga S, Matsudaira K, et al. Presenteeism and Associated Factors Among Nursing Personnel with Low Back Pain: A Cross-Sectional Study. JPR. 2020 Nov 19;13(10137):2979–2986. DOI: 10.2147/jpr.s269529
- Malińska M, Bugajska J, Bartuzi P. Occupational and non-occupational risk factors for neck and lower back pain among computer workers: a cross-sectional study. International Journal of Occupational Safety and Ergonomics. 2021 Apr 22;27(4):1108–1115. DOI: 10.1080/10803548.2021.1899650
- Jonsdottir S, Ahmed H, Tómasson K, Carter B. Factors associated with chronic and acute back pain in Wales, a cross-sectional study. BMC Musculoskelet Disord. 2019 May 15;20(1). DOI: 10.1186/s12891-019-2477-4
- Yosef T, Belachew A, Tefera Y. Magnitude and Contributing Factors of Low Back Pain among Long Distance Truck Drivers at Modjo Dry Port, Ethiopia: A Cross-Sectional Study. Journal of Environmental and Public Health. 2019 Sept 22;2019(1):1–7. DOI: 10.1155/2019/6793090
- Khadour FA, Khadour YA, Alhatem W, Albarroush D, Dao X. Risk factors Associated with Pain Severity in Syrian patients with non-specific low back Pain. BMC Musculoskelet Disord. 2024 Aug 31;25(1). DOI: 10.1186/s12891-024-07828-w
- Unuvar BS, Gercek H, Tufekci O, Torlak MS, Erbas O. The relationship between lower extremity muscle tightness and pain and disability in individuals with non-specific chronic low back pain. WOR. 2024 Sept 11;79(1):323–330. DOI: 10.3233/wor-230547
- Sidiq M, Muzaffar T, Janakiraman B, Janakiraman B, Janakiraman B, Janakiraman B, et al. Effects of pain education on disability, pain, quality of life, and self-efficacy in chronic low back pain: A randomized controlled trial. PLoS ONE. 2024 May 28;19(5):e0294302. DOI: 10.1371/journal.pone.0294302
- Das M, Saha AK, Guha C, Singh M, Roy S. In vitro evaluation of antioxidant and antibacterial properties of supercritical CO2 extracted essential oil from clove bud (Syzygium aromaticum). J Plant Biochem Biotechnol. 2020 May 23;30(2):387–391. DOI: 10.1007/s13562-020-00566-9
- Esmaeili F, Zahmatkeshan M, Yousefpoor Y, Alipanah H, Safari E, Osanloo M. Anti-inflammatory and anti-nociceptive effects of Cinnamon and Clove essential oils nanogels: an in vivo study. BMC Complement Med Ther. 2022 May 20;22(1). DOI: 10.1186/s12906-022-03619-9
- Freiwald J, Magni A, Fanlo-Mazas P, Paulino E, Sequeira De Medeiros L, Moretti B, et al. A Role for Superficial Heat Therapy in the Management of Non-Specific, Mild-to-Moderate Low Back Pain in Current Clinical Practice: A Narrative Review. Life. 2021 Aug 2;11(8):780. DOI: 10.3390/life11080780
- Nisar MF, Khadim M, Rafiq M, Chen J, Yang Y, Wan CC. Pharmacological Properties and Health Benefits of Eugenol: A Comprehensive Review. Oxidative Medicine and Cellular Longevity. 2021 Jan 1;2021(52):1–14. DOI: 10.1155/2021/2497354
- Singh AK, Vinayak M, Kumar S. Recent development in antihyperalgesic effect of phytochemicals: anti-inflammatory and neuro-modulatory actions. Inflamm Res. 2018 May 16;67(8):633–654. DOI: 10.1007/s00011-018-1156-5
- De Grado Gabriel F, Clément B, Florence F, Damien O, Anne-Marie M. Efficacy of Articaine or Eugenol for Pain Relief after Emergency Coronal Pulpotomy in Teeth with Irreversible Pulpitis: A Randomized Clinical Trial. Dentistry Journal. 2023 July 11;11(7):167. DOI: 10.3390/dj11070167
- El Ghallab Y, Ait Haj Said A, Al Jahid A, Zarayby L, Derfoufi S, Jamal Eddine J. Syzygium aromaticum L.: phytochemical investigation and comparison of the scavenging activity of essential oil, extracts and eugenol. ADV TRADIT MED (ADTM). 2019 Dec 6;20(2):153–158. DOI: 10.1007/s13596-019-00416-7
- Liñán-Atero R, Aghababaei F, García SR, Hasiri Z, Ziogkas D, Moreno A, et al. Clove Essential Oil: Chemical Profile, Biological Activities, Encapsulation Strategies, and Food Applications. Antioxidants. 2024 Apr 19;13(4):488. DOI: 10.3390/antiox13040488
- Sheth NS, Mistry RB. Formulation and evaluation of transdermal patches and to study permeation enhancement effect of eugenol. Journal of Applied Pharmaceutical Science. 2011 May 30 (Issue): 96-101. https://japsonline.com/abstract.php?article_id=42&sts=2