Ashani Ratnayake 1 , Sampath Gnanarathne 2 , Gayan Kumarasiri 3 , Ua Isurindi 4*
Authors affiliations:
Background & objective: Postoperative nausea and vomiting (PONV) is a frustrating and uncomfortable experience for many patients after surgery, especially women undergoing laparoscopic gynecological procedures. Despite the widespread use of risk prediction techniques such as the Apfel score, little is known about how well these predictions apply to South Asian populations, especially women from Sri Lanka. This study evaluated prevalence and determinants of postoperative nausea and vomiting following opioid-based anesthesia in laparoscopic gynecological surgery.
Methodology: This project was carried out as a cross-sectional study at the Teaching Hospital of Peradeniya from October 2024 to March 2025. 276 women aged 18 to 65 years who underwent opioid anesthesia for elective laparoscopic gynecological procedures were included in the study. Every patient received the same anesthetic treatment. In this study, PONV symptoms were captured over 48 hours post-surgery, which were further utilized to investigate PONV contributing factors using advanced statistical tools.
Results: About one in four women (25.7%) developed PONV, which was lower than expected based on Apfel score predictions. Significant risk factors included a past history of motion sickness, previous PONV, and higher morphine use. Surgical complexity and duration had no clear link to PONV.
Conclusions: PONV remains a significant issue, but patient-specific factors, more than surgical ones, play the biggest role. Risk prediction tools may need local tailoring for better accuracy.
Abbreviations: IAP: intra-abdominal pressure, PONV: Postoperative nausea and vomiting,
Keywords: Postoperative Nausea and Vomiting (PONV); Laparoscopic Gynecological Surgery; Sri Lankan Cohort; Risk Factors; Opioid Based Anaesthesia
Citation: Ratnayake A, Gnanarathne S, Kumarasiri G, Isurindi U. Prevalence and determinants of postoperative nausea and vomiting following opioid-based anesthesia in laparoscopic gynecological surgery: a cross-sectional study in a Sri Lankan cohort. Anaesth. pain intensive care 2025;29(8):905-911.
DOI: 10.35975/apic.v29i8.3017.
Received: July 18, 2025; Revised: August 05, 2025; Accepted: August 06, 2025
Post-operative nausea and vomiting (PONV) is a common and distressing complication following anesthesia, particularly in gynecological surgeries. The pathophysiology of PONV has been extensively studied and is well understood.¹ Over the years, many drugs have been developed to prevent and treat PONV.¹,² Despite these interventions, PONV remains a major cause of patient discomfort and morbidity in the post-operative period, contributing to dehydration, reduced mobility, prolonged hospital stays, and, in severe cases, complications such as pulmonary aspiration.³
Although laparoscopic gynecological surgery is minimally invasive, it is associated with a significant incidence of PONV, which can negatively impact patient recovery and satisfaction. Identifying and understanding the risk factors for PONV in this context is essential for optimizing anesthesia protocols and improving patient outcomes. Since many of these procedures are performed as day-case surgeries, the development of PONV may result in unnecessary admissions from day-case units.³,⁴
Pre-operative assessment should include evaluation of PONV risk using tools such as the Apfel score, which considers four key factors: female sex, use of opioids, non-smoking status, and a history of PONV.⁵ While global data on PONV prevalence is limited, individual studies suggest that 20–30% of the general population experience PONV, with rates rising to 70–80% in high-risk populations, including patients undergoing laparoscopic procedures.⁶,⁷
Despite its clinical relevance, PONV has not been thoroughly studied in the Sri Lankan population. This study aims to determine the prevalence, associated risk factors, and burden of PONV in patients undergoing laparoscopic gynecological surgery in Sri Lanka.
Given the low smoking rates among Sri Lankan women, females undergoing laparoscopic gynecological procedures who receive opioids have an estimated 61% risk of developing PONV. Moreover, both laparoscopic and gynecological surgeries are independently associated with a high risk of PONV. Our clinical observations suggest that PONV is not as frequently reported in local post-operative wards, highlighting the need for a focused study to assess its prevalence and identify contributing factors within the Sri Lankan patient cohort.
The topic of PONV has been extensively studied in various populations, and probable causative factors have been identified. Risk factors for PONV may be individual, anesthetic, surgical, or post-operative variables, including female gender, increasing age, a history of previous PONV or motion sickness, anesthetic agents, opioid analgesics, longer duration of surgery, uncontrolled pain, dehydration, and hypovolemia.⁸,⁹ Most patient- and surgical technique-related factors are fixed, while some surgery-related and anesthesia-related factors are variable.¹⁰
Most studies have identified female gender as a risk factor, and no study has contradicted this finding.⁵,¹⁴ Non-smoking status has been shown to be an independent PONV risk factor in numerous adult studies, and a history of PONV and/or motion sickness is also a recognized risk factor. Other possible risk factors include better ASA physical status and a history of migraine.
Increasing duration of surgery is an independent PONV risk factor in several well-conducted studies in both adults and children.¹⁰,¹² Although the type of surgery has been identified as a risk factor in numerous reports, its role remains somewhat controversial.5,7,10 Procedures that may increase PONV risk include intra-abdominal, laparoscopic, orthopedic, major gynecological, ENT, thyroid, breast, and plastic surgery, as well as neurosurgery; in children, hernia repair, adeno-tonsillectomy, strabismus, or penile surgery may also increase risk. Large volumes of crystalloid administration during prolonged surgery may lead to gastrointestinal tissue edema, further increasing the incidence of PONV.⁴
Numerous anesthesia-related variables are well-established PONV risk factors, including the use of volatile anesthetics, nitrous oxide, and balanced inhalational versus total IV anesthesia.6,7,10 The use of intraoperative or post-operative opioids and higher perioperative and post-operative doses have also been implicated. Administration of long-acting rather than short-acting opioids is, at best, a possible PONV risk factor.
A study conducted in Sri Lanka reported that the duration of preoperative fasting positively correlated with thirst, hunger, dehydration, headache, and PONV.¹¹ A statistically significant positive correlation was observed between fasting duration and complaints regarding thirst, hunger, dehydration, headache, and PONV.
Another Asian study of 6773 patients found that 1216 (18.0%) experienced PONV.¹² Despite the use of antiemetic prophylaxis, 18.0% of patients receiving intravenous patient-controlled analgesia still developed PONV.
This study was conducted as a cross-sectional study at the Teaching Hospital Peradeniya between October 2024 and March 2025. The primary objective was to determine the incidence of PONV within forty-eight hours following laparoscopic gynecological surgeries performed under opioid-based anesthesia. The study was approved by the institutional ethical committee of Teaching Hospital-Peradeniya and conformed to the ethical principles for medical research involving human subjects as outlined in the Declaration of Helsinki.
Nausea was defined as the subjective sensation of the urge to vomit, often accompanied by autonomic symptoms such as pallor, tachycardia, diaphoresis, and salivation. Vomiting was characterized as a reflexive, forceful oral expulsion of gastrointestinal contents due to strong and sustained contractions of abdominal and thoracic muscles.
All patients received anesthesia as per the hospital’s standardized protocol. This included premedication with oral Metoclopramide 10 mg and Ranitidine 150 mg administered 12 hours and again 2 hours before surgery, unless contraindicated. Anesthesia was induced with intravenous Propofol and maintained with Isoflurane in a low-flow system using 1 L/min of both oxygen and medical air. At induction, intravenous morphine (0.15 to 0.2 mg/kg) was administered, along with IV Dexamethasone 4 mg as part of antiemetic prophylaxis. Towards the end of surgery, IV Ondansetron 4 mg was given. No patients received regular antiemetics in the postoperative period, although IV Ondansetron 4 mg was prescribed on an as-needed basis. Postoperative analgesia was multimodal, including Morphine 0.15 mg/kg, Paracetamol, and Diclofenac suppositories during the first 24 hours. Patients were typically discharged within 24 to 36 hours following surgery.
Eligible participants were recruited during preoperative assessments. Data were collected for 48 hours post-surgery. Preoperative data included demographic information, medical history, and PONV risk factors. Intraoperative data comprised the anesthetic technique, type and dose of opioids used, and duration of surgery. Postoperative data encompassed the incidence and severity of PONV, antiemetic usage, time to recovery, and patient satisfaction.
The study population consisted of female patients aged 18–65 undergoing elective laparoscopic gynecological surgeries under opioid-based anesthesia. Inclusion criteria included ASA physical status I to III. Patients were excluded if opioids were not used, surgeries were converted to open procedures, or if the surgeries were performed on an emergency basis.
Sample size calculation was carried out using G*Power 3.1.0 software, assuming a prevalence of 25%, a power of 80%, and a confidence level of 95%, resulting in a required sample size of 268 participants. Sampling was done consecutively, based on the stated inclusion and exclusion criteria.
Data analysis included descriptive statistics to summarize patient characteristics and the incidence of PONV. Logistic regression analysis was performed to identify factors associated with increased PONV risk. Comparative analyses were also conducted to evaluate the effect of different opioids and antiemetic strategies.
A total of 276 women undergoing laparoscopic gynecological surgery with opioid-based anesthesia were included. The mean age was 39.08 ± 9.49 years, and the median age was 40 years (IQR 32–47), with a range from 18 to 77 years. The mean BMI was 24.0 ± 3.7 kg/m². Preoperative hemoglobin (Hb) averaged 11.8 ± 1.5 g/dL, with anemia (Hb <12 g/dL) present in 36.6% of patients (n = 101). Table 1 summarizes the clinical and surgical details of the cohort. The most common laparoscopic surgery performed was hysterectomy (50.7%), followed by cystectomy (17.8%).
Intraoperative blood loss was less than 750 mL in the majority of patients (94.6%, n = 261), while only one patient (0.4%) experienced blood loss exceeding 750 mL. Regarding intraoperative vasopressin administration, 40.6% of patients (n = 112) received it, whereas 57.6% (n = 159) did not. Intraoperative blood transfusions were administered to 1.4% of patients (n = 4). The mean duration of capnoperitoneum was 51.18 ± 28.36 minutes, with a median of 45 minutes and a range of 8 to 325 minutes. The maximum intra-abdominal pressure (IAP) recorded during surgery averaged 22.12 ± 3.9 mmHg, with a median of 20 mmHg and a range of 15–32 mmHg.
The overall incidence of PONV (any severity) was 25.7%, with the distribution across severity classes shown in Table 2.
The Apfel score predicted moderate-to-high PONV risk for nearly all patients, as all were female with additional risk factors. However, the actual incidence was lower than expected. No patient had a history of smoking, so the maximum Apfel score observed was 3. Figure 1 shows a comparison of expected versus actual PONV based on the Apfel score. Among the cohort, 19.6% (n =54) had a history of motion sickness, 34.8% (n = 96) received postoperative morphine, and 4.0% (n = 11) had a history of PONV. According to our findings, the Apfel score poorly predicted the onset and severity of PONV in this population.
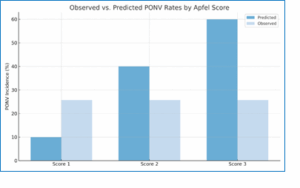
Figure 1: Comparison of expected PONV rates per Apfel score risk levels versus observed PONV incidence in this study population
Bivariate analyses were conducted to identify factors associated with PONV severity, classified into four levels. Chi-square tests and Monte Carlo-adjusted
methods were used for categorical variables, while Kruskal–Wallis tests analyzed continuous predictors. Statistical significance was set at P < 0.05. Some factors were significantly associated with PONV, others showed borderline association, and the rest showed no association.
Significant associations with increasing PONV severity were observed for a past history of motion sickness (P = 0.002), cumulative perioperative morphine consumption (P = 0.002), and past history of PONV (P = 0.045). Borderline significant factors included intrauterine vasopressin administration (P = 0.055), although there was no clear correlation with PONV severity. Conversely, age, BMI, preoperative Hb, duration of surgery, maximum intra-abdominal pressure, presence of comorbidities, surgical complexity, intraoperative blood loss, transfusion requirement, and conversion to open surgery were not significantly associated with PONV severity (all P > 0.05). Table 4 summarizes the factors showing no association with PONV development.
Interestingly, the cumulative morphine dose used during surgery showed a strong association with PONV severity; higher morphine doses were linked to increased severity of PONV.
PONV is a distressing symptom that has been studied widely in the world. There are well recognised risk factors predicting PONV including female sex, non-smoking, opioid use, previous history of PONV and surgical factors like laparoscopic surgery, gynecological surgery and eye and ENT surgeries.1,2,13
It has been reported that gynecological surgeries per se can have a PONV rate of 80-95% if they receive inadequate or no prophylactic antiemetics.14,15,16 As gynecological laparoscopic surgeries involve subset of patients with high risk, the reported incidence ranges from 40-85%.17,18 Many meta-analyses and systematic reviews have been conducted regarding the incidence of PONV, but representation from the South Asian population remains limited. This study highlights that while PONV remains a common complication in women undergoing laparoscopic surgery with opioid-based anesthesia, the incidence in this cohort (25.7%) was lower than that reported in the existing literature. A study conducted in Singapore reported a PONV incidence of 33.3%; however, that study included both males and females.19
The Apfel score is widely used to predict PONV in surgical patients. According to the Apfel score, all patients had a score of 3 (female sex, non-smoking status, and opioid use), with an expected risk of 60–80%. This discrepancy suggests that the Apfel score may overestimate the risk in this specific population. Therefore, alternative methods should be considered for predicting PONV.
Several risk factors were assessed to evaluate their correlation with the occurrence of PONV in this population. Consistent with previous research, a history of motion sickness, a history of PONV, and perioperative opioid use were strong predictors of increased PONV severity.
Notably, preoperative anemia, present in more than one-third of patients, was not associated with an increased PONV risk, indicating that correction of anemia may not directly impact PONV incidence.
Several variables, including patient age, BMI, surgery duration, comorbidities, and surgical complexity, did not significantly influence PONV severity, suggesting that patient-specific and pharmacological factors play a greater role than procedural factors. This supports prioritizing individual risk factors over surgical variables when planning prophylaxis.
Vasopressin is a vasoconstrictor administered intramyometrially to reduce intraoperative bleeding. According to our findings, there is a borderline association between vasopressin use and PONV. Further evaluation with large randomized controlled trials is required before recommending its cautious use.
This research was conducted in a single center, which is a tertiary care hospital. This may limit the generalizability of the findings to other settings such as primary care facilities or rural hospitals. Additionally, the cross-sectional design poses limitations in determining the relationship between causative factors and PONV, as causality cannot be established. Furthermore, self-reporting of nausea may introduce reporting or recall bias. Other factors, such as genetic predisposition or PONV occurring beyond 48 hours, may also influence outcomes, but these were not assessed in this study.
PONV affected one in four women undergoing laparoscopic surgery with opioid-based anesthesia, with a history of motion sickness, previous PONV, and perioperative morphine use emerging as key determinants of increased incidence and severity. The Apfel score overestimated PONV risk in this cohort, indicating limitations in its predictive utility in specific populations. Future research should explore personalized PONV risk assessment tools and alternative analgesic strategies to further reduce this distressing complication.
To validate these findings and assess broader regional differences, future research should include multicenter studies with more diverse populations. A prospective cohort design would further strengthen causal inference. Additionally, exploring pharmacogenetic influences on antiemetic response and opioid metabolism may support personalized strategies to reduce PONV risk.
These findings could be applied more effectively if PONV prevention strategies are tailored based on pharmacogenetic influences on antiemetic response and opioid metabolism.
SG: Conceptualization, Study design, Supervision, Critical revision of manuscript
GK: Patient recruitment, Surgical coordination, Manuscript editing
UAI: Data collection, Statistical analysis, Manuscript writing
11. REFERENCES
Authors affiliations:
- Ashani Ratnayake, Department of Anesthesiology and Critical Care, Faculty of Medicine, University of Peradeniya, Peradeniya, 20400, Sri Lanka; Email: aashaniratnayake@yahoo.com, {ORCID: 0000-0002-7266-1874}
- Sampath Gnanarathne, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Peradeniya, Peradeniya, 20400, Sri Lanka; Email: ssampatht@yahoo.com, {ORCID: 0009-0007-6023-502X}
- Gayan Kumarasiri, Teaching Hospital, Peradeniya, 20400, Sri Lanka; Email: gayanpradeepkumarasiri@gmail.com
- Ua Isurindi, Department of Anesthesiology & Critical Care, Faculty of Medicine, University of Peradeniya, Peradeniya, 20400, Sri Lanka; Email: ashiiuhanovita@gmail.com, {ORCID: 0009-0003-1671-100X}
ABSTRACT
Background & objective: Postoperative nausea and vomiting (PONV) is a frustrating and uncomfortable experience for many patients after surgery, especially women undergoing laparoscopic gynecological procedures. Despite the widespread use of risk prediction techniques such as the Apfel score, little is known about how well these predictions apply to South Asian populations, especially women from Sri Lanka. This study evaluated prevalence and determinants of postoperative nausea and vomiting following opioid-based anesthesia in laparoscopic gynecological surgery.
Methodology: This project was carried out as a cross-sectional study at the Teaching Hospital of Peradeniya from October 2024 to March 2025. 276 women aged 18 to 65 years who underwent opioid anesthesia for elective laparoscopic gynecological procedures were included in the study. Every patient received the same anesthetic treatment. In this study, PONV symptoms were captured over 48 hours post-surgery, which were further utilized to investigate PONV contributing factors using advanced statistical tools.
Results: About one in four women (25.7%) developed PONV, which was lower than expected based on Apfel score predictions. Significant risk factors included a past history of motion sickness, previous PONV, and higher morphine use. Surgical complexity and duration had no clear link to PONV.
Conclusions: PONV remains a significant issue, but patient-specific factors, more than surgical ones, play the biggest role. Risk prediction tools may need local tailoring for better accuracy.
Abbreviations: IAP: intra-abdominal pressure, PONV: Postoperative nausea and vomiting,
Keywords: Postoperative Nausea and Vomiting (PONV); Laparoscopic Gynecological Surgery; Sri Lankan Cohort; Risk Factors; Opioid Based Anaesthesia
Citation: Ratnayake A, Gnanarathne S, Kumarasiri G, Isurindi U. Prevalence and determinants of postoperative nausea and vomiting following opioid-based anesthesia in laparoscopic gynecological surgery: a cross-sectional study in a Sri Lankan cohort. Anaesth. pain intensive care 2025;29(8):905-911.
DOI: 10.35975/apic.v29i8.3017.
Received: July 18, 2025; Revised: August 05, 2025; Accepted: August 06, 2025
1. INTRODUCTION
Post-operative nausea and vomiting (PONV) is a common and distressing complication following anesthesia, particularly in gynecological surgeries. The pathophysiology of PONV has been extensively studied and is well understood.¹ Over the years, many drugs have been developed to prevent and treat PONV.¹,² Despite these interventions, PONV remains a major cause of patient discomfort and morbidity in the post-operative period, contributing to dehydration, reduced mobility, prolonged hospital stays, and, in severe cases, complications such as pulmonary aspiration.³
Although laparoscopic gynecological surgery is minimally invasive, it is associated with a significant incidence of PONV, which can negatively impact patient recovery and satisfaction. Identifying and understanding the risk factors for PONV in this context is essential for optimizing anesthesia protocols and improving patient outcomes. Since many of these procedures are performed as day-case surgeries, the development of PONV may result in unnecessary admissions from day-case units.³,⁴
Pre-operative assessment should include evaluation of PONV risk using tools such as the Apfel score, which considers four key factors: female sex, use of opioids, non-smoking status, and a history of PONV.⁵ While global data on PONV prevalence is limited, individual studies suggest that 20–30% of the general population experience PONV, with rates rising to 70–80% in high-risk populations, including patients undergoing laparoscopic procedures.⁶,⁷
Despite its clinical relevance, PONV has not been thoroughly studied in the Sri Lankan population. This study aims to determine the prevalence, associated risk factors, and burden of PONV in patients undergoing laparoscopic gynecological surgery in Sri Lanka.
Given the low smoking rates among Sri Lankan women, females undergoing laparoscopic gynecological procedures who receive opioids have an estimated 61% risk of developing PONV. Moreover, both laparoscopic and gynecological surgeries are independently associated with a high risk of PONV. Our clinical observations suggest that PONV is not as frequently reported in local post-operative wards, highlighting the need for a focused study to assess its prevalence and identify contributing factors within the Sri Lankan patient cohort.
The topic of PONV has been extensively studied in various populations, and probable causative factors have been identified. Risk factors for PONV may be individual, anesthetic, surgical, or post-operative variables, including female gender, increasing age, a history of previous PONV or motion sickness, anesthetic agents, opioid analgesics, longer duration of surgery, uncontrolled pain, dehydration, and hypovolemia.⁸,⁹ Most patient- and surgical technique-related factors are fixed, while some surgery-related and anesthesia-related factors are variable.¹⁰
Most studies have identified female gender as a risk factor, and no study has contradicted this finding.⁵,¹⁴ Non-smoking status has been shown to be an independent PONV risk factor in numerous adult studies, and a history of PONV and/or motion sickness is also a recognized risk factor. Other possible risk factors include better ASA physical status and a history of migraine.
Increasing duration of surgery is an independent PONV risk factor in several well-conducted studies in both adults and children.¹⁰,¹² Although the type of surgery has been identified as a risk factor in numerous reports, its role remains somewhat controversial.5,7,10 Procedures that may increase PONV risk include intra-abdominal, laparoscopic, orthopedic, major gynecological, ENT, thyroid, breast, and plastic surgery, as well as neurosurgery; in children, hernia repair, adeno-tonsillectomy, strabismus, or penile surgery may also increase risk. Large volumes of crystalloid administration during prolonged surgery may lead to gastrointestinal tissue edema, further increasing the incidence of PONV.⁴
Numerous anesthesia-related variables are well-established PONV risk factors, including the use of volatile anesthetics, nitrous oxide, and balanced inhalational versus total IV anesthesia.6,7,10 The use of intraoperative or post-operative opioids and higher perioperative and post-operative doses have also been implicated. Administration of long-acting rather than short-acting opioids is, at best, a possible PONV risk factor.
A study conducted in Sri Lanka reported that the duration of preoperative fasting positively correlated with thirst, hunger, dehydration, headache, and PONV.¹¹ A statistically significant positive correlation was observed between fasting duration and complaints regarding thirst, hunger, dehydration, headache, and PONV.
Another Asian study of 6773 patients found that 1216 (18.0%) experienced PONV.¹² Despite the use of antiemetic prophylaxis, 18.0% of patients receiving intravenous patient-controlled analgesia still developed PONV.
2. METHODOLOGY
This study was conducted as a cross-sectional study at the Teaching Hospital Peradeniya between October 2024 and March 2025. The primary objective was to determine the incidence of PONV within forty-eight hours following laparoscopic gynecological surgeries performed under opioid-based anesthesia. The study was approved by the institutional ethical committee of Teaching Hospital-Peradeniya and conformed to the ethical principles for medical research involving human subjects as outlined in the Declaration of Helsinki.
Nausea was defined as the subjective sensation of the urge to vomit, often accompanied by autonomic symptoms such as pallor, tachycardia, diaphoresis, and salivation. Vomiting was characterized as a reflexive, forceful oral expulsion of gastrointestinal contents due to strong and sustained contractions of abdominal and thoracic muscles.
All patients received anesthesia as per the hospital’s standardized protocol. This included premedication with oral Metoclopramide 10 mg and Ranitidine 150 mg administered 12 hours and again 2 hours before surgery, unless contraindicated. Anesthesia was induced with intravenous Propofol and maintained with Isoflurane in a low-flow system using 1 L/min of both oxygen and medical air. At induction, intravenous morphine (0.15 to 0.2 mg/kg) was administered, along with IV Dexamethasone 4 mg as part of antiemetic prophylaxis. Towards the end of surgery, IV Ondansetron 4 mg was given. No patients received regular antiemetics in the postoperative period, although IV Ondansetron 4 mg was prescribed on an as-needed basis. Postoperative analgesia was multimodal, including Morphine 0.15 mg/kg, Paracetamol, and Diclofenac suppositories during the first 24 hours. Patients were typically discharged within 24 to 36 hours following surgery.
Eligible participants were recruited during preoperative assessments. Data were collected for 48 hours post-surgery. Preoperative data included demographic information, medical history, and PONV risk factors. Intraoperative data comprised the anesthetic technique, type and dose of opioids used, and duration of surgery. Postoperative data encompassed the incidence and severity of PONV, antiemetic usage, time to recovery, and patient satisfaction.
The study population consisted of female patients aged 18–65 undergoing elective laparoscopic gynecological surgeries under opioid-based anesthesia. Inclusion criteria included ASA physical status I to III. Patients were excluded if opioids were not used, surgeries were converted to open procedures, or if the surgeries were performed on an emergency basis.
Sample size calculation was carried out using G*Power 3.1.0 software, assuming a prevalence of 25%, a power of 80%, and a confidence level of 95%, resulting in a required sample size of 268 participants. Sampling was done consecutively, based on the stated inclusion and exclusion criteria.
Data analysis included descriptive statistics to summarize patient characteristics and the incidence of PONV. Logistic regression analysis was performed to identify factors associated with increased PONV risk. Comparative analyses were also conducted to evaluate the effect of different opioids and antiemetic strategies.
3. RESULTS
A total of 276 women undergoing laparoscopic gynecological surgery with opioid-based anesthesia were included. The mean age was 39.08 ± 9.49 years, and the median age was 40 years (IQR 32–47), with a range from 18 to 77 years. The mean BMI was 24.0 ± 3.7 kg/m². Preoperative hemoglobin (Hb) averaged 11.8 ± 1.5 g/dL, with anemia (Hb <12 g/dL) present in 36.6% of patients (n = 101). Table 1 summarizes the clinical and surgical details of the cohort. The most common laparoscopic surgery performed was hysterectomy (50.7%), followed by cystectomy (17.8%).
| Table 1: Comorbidities, past history of chemotherapy, and types of surgery performed (n = 276). | ||
| Factor | N (%) | |
| Comorbidities |
Diabetes mellitus | 29 (10.5) |
| Hypertension | 38 (13.8) | |
| Dyslipidemia | 9 (3.3) | |
| Hypothyroidism | 9 (3.3) | |
| Anemia | 1 (0.4) | |
| Arthritis | 1 (0.4) | |
| Bronchial Asthma/COPD | 14 (5.1) | |
| Allergic rhinitis | 1 (0.4) | |
| Depression | 3 (1.1) | |
| Ischemic Heart Disease/Heart Failure/arrythmia | 4 (1.4) | |
| neurological | 3 (1.1) | |
| Other (DVT, past history of malignancy, Thalassemia) | 5 (1.9) | |
| Past history of chemotherapy | 6 (2.2) | |
| Type of Surgery |
Laparoscopic cystectomy | 49 (17.8) |
| Diagnostic laparoscopy | 29 (10.5) | |
| Laparoscopic hysterectomy | 140 (50.7) | |
| Laparoscopic Burch colposuspension | 4 (1.4) | |
| Dye test | 38 (13.8) | |
| Laparoscopic LRT | 9 (3.3) | |
| Exploratory laparotomy | 4 (1.4) | |
| Salpingectomy/ Salpingo-oophorectomy |
7 (2.5) | |
| Laparoscopic adhesiolysis | 5 (1.8) | |
| Dilatation & curettage | 4 (1.4) | |
| Myomectomy | 4 (1.4) | |
| other | 10 (3.8) | |
Intraoperative blood loss was less than 750 mL in the majority of patients (94.6%, n = 261), while only one patient (0.4%) experienced blood loss exceeding 750 mL. Regarding intraoperative vasopressin administration, 40.6% of patients (n = 112) received it, whereas 57.6% (n = 159) did not. Intraoperative blood transfusions were administered to 1.4% of patients (n = 4). The mean duration of capnoperitoneum was 51.18 ± 28.36 minutes, with a median of 45 minutes and a range of 8 to 325 minutes. The maximum intra-abdominal pressure (IAP) recorded during surgery averaged 22.12 ± 3.9 mmHg, with a median of 20 mmHg and a range of 15–32 mmHg.
The overall incidence of PONV (any severity) was 25.7%, with the distribution across severity classes shown in Table 2.
| Table 2: Prevalence of PONV severity classes in study population | |
| PONV Class | N (%) |
| No PONV (Class 1) | 205 (74.3) |
| Mild PONV (Class 2) | 38 (13.8) |
| Significant PONV (Class 3) | 26 (9.4) |
| Severe PONV (Class 4) | 7 (2.5) |
The Apfel score predicted moderate-to-high PONV risk for nearly all patients, as all were female with additional risk factors. However, the actual incidence was lower than expected. No patient had a history of smoking, so the maximum Apfel score observed was 3. Figure 1 shows a comparison of expected versus actual PONV based on the Apfel score. Among the cohort, 19.6% (n =54) had a history of motion sickness, 34.8% (n = 96) received postoperative morphine, and 4.0% (n = 11) had a history of PONV. According to our findings, the Apfel score poorly predicted the onset and severity of PONV in this population.

Figure 1: Comparison of expected PONV rates per Apfel score risk levels versus observed PONV incidence in this study population
Bivariate analyses were conducted to identify factors associated with PONV severity, classified into four levels. Chi-square tests and Monte Carlo-adjusted
methods were used for categorical variables, while Kruskal–Wallis tests analyzed continuous predictors. Statistical significance was set at P < 0.05. Some factors were significantly associated with PONV, others showed borderline association, and the rest showed no association.
| Table 3: Summary of the factors that showed a significant association with the development of PONV. | ||||||
| Effect | P value | Interpretation | PONV 1 | PONV 2 | PONV 3 | PONV 4 |
| Past history of PONV | 0.045 | Significant association | 5 (45.5) | 4 (36.4) | 1 (9.1) |
1 (9.1) |
| Past history of motion sickness | 0.002 | Significant association | 34 (64) |
10 (18.5) | 5 (9.3) |
5 (9.3) |
| Cumulative morphine use-perioperatively | 0.002 | Significant association | 110 (71.4) | 29 (18.8) | 9 (5.8) |
6 (3.9) |
Significant associations with increasing PONV severity were observed for a past history of motion sickness (P = 0.002), cumulative perioperative morphine consumption (P = 0.002), and past history of PONV (P = 0.045). Borderline significant factors included intrauterine vasopressin administration (P = 0.055), although there was no clear correlation with PONV severity. Conversely, age, BMI, preoperative Hb, duration of surgery, maximum intra-abdominal pressure, presence of comorbidities, surgical complexity, intraoperative blood loss, transfusion requirement, and conversion to open surgery were not significantly associated with PONV severity (all P > 0.05). Table 4 summarizes the factors showing no association with PONV development.
Interestingly, the cumulative morphine dose used during surgery showed a strong association with PONV severity; higher morphine doses were linked to increased severity of PONV.
| Table 4: Factors having no association with the development of PONV | ||
| Effect | P value | Interpretation |
| Age | 0.787 | No significant association |
| BMI | 0.488 | No significant association |
| Preoperative Hb | 0.088 | No significant association |
| Duration of Surgery | 0.571 | No significant association |
| Max IAP | 0.152 | No significant association |
| Having >= 1 co-morbidities | 0.745 | No significant association |
| Past history of chemotherapy | 0.881 | No significant association |
| Being an advanced surgery | 0.861 | No significant association |
| Blood loss more than 750ml | 1.000 | No significant association |
| Intraoperative transfusion | 0.222 | No significant association |
| Complicated Surgery | 0.897 | No significant association |
| Table 5: Cumulative perioperative morphine use | ||||
| Effect | PONV 1 | PONV 2 | PONV 3 | PONV 4 |
| No morphine was used peri operatively | 95 (77.9) | 9 (7.4) | 17 (13.9) | 1 (0.8) |
| 2Morphine was used during peri-operative period | 110 (71.4) | 29 (18.8) | 9 (5.8) | 6 (3.9) |
4. DISCUSSION
PONV is a distressing symptom that has been studied widely in the world. There are well recognised risk factors predicting PONV including female sex, non-smoking, opioid use, previous history of PONV and surgical factors like laparoscopic surgery, gynecological surgery and eye and ENT surgeries.1,2,13
It has been reported that gynecological surgeries per se can have a PONV rate of 80-95% if they receive inadequate or no prophylactic antiemetics.14,15,16 As gynecological laparoscopic surgeries involve subset of patients with high risk, the reported incidence ranges from 40-85%.17,18 Many meta-analyses and systematic reviews have been conducted regarding the incidence of PONV, but representation from the South Asian population remains limited. This study highlights that while PONV remains a common complication in women undergoing laparoscopic surgery with opioid-based anesthesia, the incidence in this cohort (25.7%) was lower than that reported in the existing literature. A study conducted in Singapore reported a PONV incidence of 33.3%; however, that study included both males and females.19
The Apfel score is widely used to predict PONV in surgical patients. According to the Apfel score, all patients had a score of 3 (female sex, non-smoking status, and opioid use), with an expected risk of 60–80%. This discrepancy suggests that the Apfel score may overestimate the risk in this specific population. Therefore, alternative methods should be considered for predicting PONV.
Several risk factors were assessed to evaluate their correlation with the occurrence of PONV in this population. Consistent with previous research, a history of motion sickness, a history of PONV, and perioperative opioid use were strong predictors of increased PONV severity.
Notably, preoperative anemia, present in more than one-third of patients, was not associated with an increased PONV risk, indicating that correction of anemia may not directly impact PONV incidence.
Several variables, including patient age, BMI, surgery duration, comorbidities, and surgical complexity, did not significantly influence PONV severity, suggesting that patient-specific and pharmacological factors play a greater role than procedural factors. This supports prioritizing individual risk factors over surgical variables when planning prophylaxis.
Vasopressin is a vasoconstrictor administered intramyometrially to reduce intraoperative bleeding. According to our findings, there is a borderline association between vasopressin use and PONV. Further evaluation with large randomized controlled trials is required before recommending its cautious use.
5. LIMITATIONS
This research was conducted in a single center, which is a tertiary care hospital. This may limit the generalizability of the findings to other settings such as primary care facilities or rural hospitals. Additionally, the cross-sectional design poses limitations in determining the relationship between causative factors and PONV, as causality cannot be established. Furthermore, self-reporting of nausea may introduce reporting or recall bias. Other factors, such as genetic predisposition or PONV occurring beyond 48 hours, may also influence outcomes, but these were not assessed in this study.
6. CONCLUSION
PONV affected one in four women undergoing laparoscopic surgery with opioid-based anesthesia, with a history of motion sickness, previous PONV, and perioperative morphine use emerging as key determinants of increased incidence and severity. The Apfel score overestimated PONV risk in this cohort, indicating limitations in its predictive utility in specific populations. Future research should explore personalized PONV risk assessment tools and alternative analgesic strategies to further reduce this distressing complication.
To validate these findings and assess broader regional differences, future research should include multicenter studies with more diverse populations. A prospective cohort design would further strengthen causal inference. Additionally, exploring pharmacogenetic influences on antiemetic response and opioid metabolism may support personalized strategies to reduce PONV risk.
These findings could be applied more effectively if PONV prevention strategies are tailored based on pharmacogenetic influences on antiemetic response and opioid metabolism.
- Data availability
- Conflict of interest
- Funding
- Authors’ contribution
SG: Conceptualization, Study design, Supervision, Critical revision of manuscript
GK: Patient recruitment, Surgical coordination, Manuscript editing
UAI: Data collection, Statistical analysis, Manuscript writing
11. REFERENCES
- Wiesmann, P. Kranke, L. Eberhart Postoperative nausea and vomiting—a narrative review of pathophysiology, pharmacotherapy and clinical management strategies Expert Opin Pharmacother, 16 (2015), pp. 1069-1077 DOI: 10.1517/14656566.2015.1033398
- P. Le, T.J. Gan Update on the management of postoperative nausea and vomiting and postdischarge nausea and vomiting in ambulatory surgery Anesthesiol Clin, 28 (2010), pp. 225-249 DOI: 10.1016/j.anclin.2010.02.003
- S. Smith, E.J. Smith, B.R. Smith Postoperative nausea and vomiting Ann Palliat Med, 1 (2012),94-102 DOI: 10.3978/j.issn.2224-5820.2012.07.05
- Amirshahi M, Behnamfar N, Badakhsh M, Rafiemanesh H, Keikhaie KR, Sheyback M, et al. Prevalence of postoperative nausea and vomiting: A systematic review and meta-analysis. Saudi J Anaesth. 2020 Jan-Mar;14(1):48-56. doi: 10.4103/sja.SJA_401_19. Epub 2020 Jan 6. PMID: 31998020; PMCID: PMC6970369 DOI: 4103/sja.SJA_401_19 .
- Apfel C, Heidrich F, Jukar-Rao S, Jalota L, Hornuss C, Whelan RP, et al Evidence-based analysis of risk factors for postoperative nausea and vomiting Br J Anaesth. 2012;109:742–53 DOI: 1093/bja/aes276
- Gan TJ, Meyer TA, Apfel CC, Chung F, Davis PJ, Habib AS, et al. Society for ambulatory anesthesia guidelines for the management of postoperative nausea and vomiting Anesth Analg. 2007;105:1615–28 DOI: 1213/01.ane.0000295230.55439.f4
- Kovac AL. Update on the management of postoperative nausea and vomiting Drugs. 2013;73:1525–47 DOI: 1007/s40265-013-0110-7
- East JM, Mitchell DI. Postoperative nausea and vomiting in laparoscopic versus open cholecystectomy at two major hospitals in Jamaica. West Indian Med J. 2009 Mar;58(2):130-7. PMID: 21866598.
- Toleska, M., Dimitrovski, A. and Dimitrovska, N.T., 2022. Postoperative nausea and vomiting in opioid-free anesthesia versus opioid based anesthesia in laparoscopic cholecystectomy. Prilozi, 43(3), pp.101-108. DOI: 2478/prilozi-2022-0042
- Gan TJ. Risk factors for postoperative nausea and vomiting. Anesth Analg. 2006 Jun;102(6):1884-98. DOI: 1213/01.ANE.0000219597.16143.4D
- Hapugoda, M., Perera, L. and Nishadi, A.K.D., 2022. Pre-operative Fasting in Elective Adult Surgical Patients at a Tertiary Care Hospital in Sri Lanka. Age (years), 18(29), p.43.DOI: 10.4038/slja.v30i1.8839
- Yi MS, Kang H, Kim MK, Choi GJ, Park YH, Baek CW, et al. Relationship between the incidence and risk factors of postoperative nausea and vomiting in patients with intravenous patient-controlled analgesia. Asian J Surg. 2018 Jul;41(4):301-306. DOI: 1016/j.asjsur.2017.01.005 . Epub 2017 Mar 31. PMID: 28372932.
- Smith CA, Ruth-Sahd L. Reducing the incidence of postoperative nausea and vomiting begins with risk screening: an evaluation of the evidence. J Perianesth Nurs. (2016) 31:158–71. DOI: 1016/j.jopan.2015.03.011
- Apfel CC, Läärä E, Koivuranta M, Greim CA, Roewer N. A simplified risk score for predicting postoperative nausea and vomiting: conclusions from cross-validations between two centers. Anesthesiology. (1999) 91:693–700. DOI: 1097/00000542-199909000-00022
- Koivuranta M, Läärä E, Snåre L, Alahuhta S. A survey of postoperative nausea and vomiting. Anaesthe. (1997) 52:443–9. DOI: 1111/j.1365-2044.1997.117-az0113.x
- Krieser KA, Riley III JB, Baus JE, Hoffman JT, Sullivan JN. PONV Prophylaxis failure disproportionately affects female patients, despite intraoperative computerized decision support guidance. Graduate Med Edu Res J. (2020) 2:6. Guidance. 2020 Sep 29; 2(1).https://digitalcommons.unmc.edu/gmerj/vol2/iss1/6
- https://digitalcommons.unmc.edu/gmerj/vol2/iss1/6Bhakta P, Ghosh BR, Singh U, Govind PS, Gupta A, Kapoor KS, et al. Incidence of postoperative nausea and vomiting following gynecological laparoscopy: A comparison of standard anesthetic technique and propofol infusion. Acta Anaesthesiologica Taiwanica. (2016) 54:108–13. DOI: 1016/j.aat.2016.10.002
- Khan MA, Siddiqi KJ, Khan MS. Prophylactic use of gabapentin to reduce postoperative nausea and vomiting in patients undergoing diagnostic gynecological laparoscopy. Anaesth pain intensive care. (2017) 21:19–24. [FreeFullPaper]
- Leong, Xin Yu Adeline, Kwa, Xian Wen Charlene, Ng, Shu Hui Elizabeth, Koh, Liang Khai Darren, Singapore General Hospital Experience on Ethnicity and the Incidence of Postoperative Nausea and Vomiting after Elective Orthopaedic Surgeries, Journal of Anesthesiology, 2015, 952302, 7 pages, 2015. https://doi.org/10.1155/2015/952302
- Perera B, Fonseka P, Ekanayake R, Lelwala E. Smoking in adults in Sri Lanka: prevalence and attitudes. Asia Pac J Public Health. 2005;17(1):40-5. DOI: 1177/101053950501700110 .