Nahla N. Shehab 1 , Reham M. Fahmy 2 , Ayman S. Abdelrahman 3 , Azza F. Omran 4 , Walaa Y. Elsabeeny 5*
Authors affiliations:
Background & objective: Modified Radical Mastectomy (MRM) is typically performed under general anesthesia that often necessitates opioid administration, which can lead to side effects such as sedation, respiratory depression, and nausea. Fascial plane blocks, including the Erector Spinae Plane Block (ESPB), offer an effective analgesic alternative to opioids. This study aimed to assess the effectiveness of bi-level ESPB with either dexmedetomidine or dexamethasone as adjuvants to bupivacaine, compared with opioid-based analgesia, in patients undergoing MRM.
Methods: This randomized controlled study included female cancer patients scheduled for MRM. Patients were randomized into four groups; Group D (control group) received conventional anesthesia with intraoperative morphine, and the other three groups received bi-level ESPB at T2 and T4. Group A received ESPB with 0.25% bupivacaine, Group B received ESPB with 0.25% bupivacaine and 0.1 mg/kg dexamethasone, and Group C received ESPB with 0.25% bupivacaine and 0.5μg/kg dexmedetomidine.
Results: The time to first rescue morphine was significantly shorter in Group D compared with Groups A, B, and C (P < 0.001). Postoperative morphine consumption was significantly higher in group D than in the other groups (P < 0.001). Intraoperative fentanyl consumption was lower in Group C compared with the rest of the groups (P < 0.001).
Conclusions: In MRM, adding dexmedetomidine as an adjuvant to bupivacaine in ultrasound-guided bi-level ESPB showed a better analgesic profile compared to no adjuvant, dexamethasone, and conventional opioid analgesia with lower pain scores and lower intraoperative opioid consumption. However, dexmedetomidine and dexamethasone, when used as adjuvants, had a comparable effect to no adjuvant with respect to postoperative opioid consumption and the time to first analgesic administration.
Keywords: Dexmedetomidine; Dexamethasone; Modified Radical Mastectomy; Analgesia; Morphine.
Citation: Shehab NN, Fahmy RM, Abdelrahman AS, Omran AF, Elsabeeny WY. Analgesic efficacy of adding adjuvants to local anesthetics in bi-level erector spinae plane block for breast surgeries: a randomized controlled study. Anaesth. Pain Intensive Care 2025;29(6):787-795. DOI: 10.35975/apic.v29i6.2979.
Received: April 16, 2025; Revised: August 02, 2025; Accepted: August 05, 2025
Perioperative analgesia for cancer patients scheduled for modified radical mastectomy is considered a pivotal component of care for such patients.1 Risk of development of chronic post-mastectomy pain syndrome (PMPS) is present following any breast surgical intervention, where a considerable percentage of patients with undertreated acute pain can develop chronic pain.2
Appropriately managing perioperative pain control can reduce both postoperative complications and morbidity by reducing the incidence of the development of post-mastectomy pain syndrome.3 Pharmacological agents such as paracetamol, non-steroidal anti-inflammatory drugs (NSAIDs), opioids, as well as interventional modalities including neuraxial blocks (thoracic epidural analgesia) and locoregional blocks (paravertebral blocks, serratus anterior plane block, pectoral nerve block, and erector spinae plane block) have been considered for perioperative analgesia in breast surgeries.4
ESPB is a unique regional technique that was first described in 2016. It has been widely used as an effective regional block for postoperative analgesia in thoracotomy, with promising results reported since it was first described by Forero and his colleagues.5 Single-shot block may not provide the desired duration of analgesia required6 and inserting a catheter for continuous infusion of local anesthetic is not always feasible; therefore, bi-level ESPB was introduced into practice, aiming to block a wider dermatomal area through enhanced craniocaudal spread.7
Several adjuvants have been added to local anesthetics during regional blocks, including magnesium sulphate, clonidine, and opioids.8 Adding dexamethasone as an adjunct to local anesthetic has been shown to reduce perioperative pain and augment the local anesthetic effect.9 Dexmedetomidine is an alpha-2 adrenergic agonist, known for its sedative, analgesic-sparing, and additive perioperative sympatholytic effects. The use of dexmedetomidine as an adjuvant to local anesthetic drugs in neuraxial and nerve blocks has been shown to extend analgesia.8,10
The current work aims to assess the analgesic efficacy of dexamethasone and dexmedetomidine as adjuvants to bupivacaine in ultrasound (US) guided bi-level ESPB for modified radical mastectomy and its impact on the intensity of acute pain, duration of analgesia, and decreased perioperative opioid consumption.
This prospective randomized controlled single-blinded (outcome assessor) trial was done after agreement of the ethical committee (AP2207-30110) and was registered at clinicaltrials.gov (NCT05591417). The patients were assigned to the study after obtaining written informed consent.
Initially, 93 adult female patients (ASA II–III), aged 18–65 years, with a BMI between 20 and 35 kg/m², and diagnosed with breast cancer scheduled for modified radical mastectomy with axillary evacuation were screened for eligibility. Patients were excluded from the study if they were pregnant or had severe cardiovascular instability, impaired coagulation profiles, abnormal kidney or liver function, bone metastases, infection at the injection site, or known allergies to any of the study medications. Additionally, patients with psychological or mental impairment or on chronic pain medications were also excluded.
Sixty-eight patients meeting the eligibility criteria were randomized into 4 groups through computer random number generation. Group A (17 patients): received ultrasound-guided bi-level ESPB with 15 ml bupivacaine 0.25% at each level. Group B (17 patients): received ultrasound-guided bi-level ESPB with 15 ml bupivacaine 0.25% with 0.1 mg/kg dexamethasone at each level. Group C (15 patients): received ultrasound-guided bi-level ESPB with 15 ml bupivacaine 0.25% with 0.5μg/kg dexmedetomidine at each level. Group D (17 patients): received conventional anesthesia with intraoperative intravenous (IV) morphine 0.1 mg/kg.
2.1. Study Interventions
All patients were clinically assessed at the preoperative assessment clinic. Investigations, including complete blood picture, liver & kidney functions, and coagulation profile, were obtained. In the holding room, the patients’ vital signs were monitored. The block technique and the visual analogue scale (VAS) were explained to all patients, with the scale ranging from 0 cm (no pain) to 10 cm (severe pain). Premedication with IV midazolam 0.02 mg/kg was given.
2.2. Bi-level ESPB technique
After skin sterilization with 10% povidone-iodine, the block was performed on the side of the operation with the patient in the sitting position, 20 minutes before induction of anesthesia, using the technique described by Forero and colleagues. The ultrasound probe (M-Turbo; FUJIFILM SonoSite Inc., Bothell, WA, USA), either linear (6-13 MHz) or curved (2-5 MHz), was placed vertically 1.5-2 cm lateral to the spine after identifying T2 and T4 spinous processes, where blocks were done using an in-plane approach. The needle was introduced through the skin in a cranio-caudal direction toward the transverse process. Two milliliters of normal saline were injected into the fascial plane between the erector spinae muscle and the transverse process, and hydro-dissection of the plane was observed. After confirming the needle position, 15 milliliters of the prepared solution were administered at the level of T2 and similarly at the level of T4. Dermatomal testing was performed by an anesthesiologist who did not participate in the study. If no sensory block was present after 20 minutes, patients were omitted from the study.
General anesthesia was administered to all patients using intravenous (IV) propofol (2-3 mg/kg), fentanyl (2µg/kg), and rocuronium (0.6 mg/kg), followed by endotracheal intubation. Volume-controlled mechanical ventilation was done, and anesthesia was maintained by inhalational anesthesia using isoflurane (1-1.5%) and top-up doses of muscle relaxant as needed. Intraoperative multimodal analgesia was administered in the form of 1gm paracetamol and 30 mg ketorolac IV. Patients were monitored throughout the operation using the standard ASA monitors.
Heart rate (HR) and mean arterial pressure (MAP) were recorded before the induction of general anesthesia, after tracheal intubation, and then every 15 minutes till the end of the operation. During surgery, if there was a reported increase in HR or MAP by >30% from the baseline readings and after exclusion of inadequate anesthesia, a bolus dose of fentanyl 0.5µg/kg was given and recorded. Significant decrease in HR or MAP was addressed according to each patient’s baseline values and medical condition by atropine or ephedrine as needed.
Upon recovery, patients were admitted to the post-anesthesia care unit (PACU), where HR, MAP, pain scores, and sedation scores were recorded. In the ward and for the following 24 hours, monitoring for the same parameters was done at 2, 4, 8, 12, and 24 hours. In the PACU, patients’ sedation status was assessed using the Ramsay Sedation Score (RSS), which ranges from 1 (anxious, restless, or both) to 6 (no response to stimulus). Pain was assessed using the VAS score. Patients with a VAS score < 4 received 30 mg of intravenous ketorolac, while those with a VAS score ≥ 4 were given intravenous morphine as a bolus dose of 3 mg per dose, with the total dose not exceeding 0.1 mg/kg per dose. The total amount of morphine given in 24 hours was recorded in the four groups. Acetaminophen 1g IV was given to all patients every 8 hours. Adverse effects in the form of hematoma, nausea, vomiting, or over-sedation were recorded.
2.4. Sample Size
The G power program was used to calculate a total sample size of 60. Fifteen patients in each group were initially required in order to reject the null hypothesis that the mean 24-hour postoperative morphine consumption is equivalent among the research groups, probability (power) = 0.99999.11 Two patients were added to each group for compensation of any possible attrition during the study. The Type I error probability associated with testing this null hypothesis is 0.05.
2.5. Statistical Analysis
Statistical Package for the Social Sciences (SPSS) version 28 (IBM Corp., Armonk, NY, USA) was used for data coding and statistical analysis. The type of statistical test, parametric or non-parametric, was selected according to the distribution of quantitative variables, which was assessed using the Shapiro–Wilk normality test and histograms. Analysis of variance (ANOVA) was used to compare parametric variables, which were expressed as mean ± standard deviation (SD), among the four groups, and post hoc Tukey testing was performed for intergroup comparisons.
Nonparametric variables were expressed as median and interquartile range (IQR) and were analyzed using the Kruskal–Wallis test, with the Mann–Whitney U test used for further pairwise analysis. The chi-square test was applied to categorical variables, which were expressed as frequencies and percentages. When the expected frequency was less than 5, Exact test was used. Statistical significance was interpreted when the two-tailed P value was less than or equal to 0.05.
2.6. Outcomes
The primary outcome measure was set as the total morphine consumption in the first 24 hours postoperatively. Secondary outcome parameters were perioperative hemodynamics (HR and MAP), total intraoperative fentanyl consumption, time to first rescue analgesia, postoperative pain scores, and any possible complications (hematoma, nausea and/or vomiting, or over sedation) were recorded.
Initially, 93 adult female patients, between 18 and 65 years of age, undergoing modified radical mastectomy were assessed for eligibility. Sixty-eight patients (ASA II–III) were randomized into one of the four study groups.
Patients’ demographic data (age and BMI) and clinical characteristics (diabetes and hypertension) were comparable among the four groups (Table 1).
Intraoperative heart rate values were significantly different among the four groups, starting from 15 minutes until the end of the surgery time (P < 0.001). They were significantly higher in group B compared with Groups A, C, and D from 15 to 135 minutes intraoperatively. Additionally, they were significantly higher in Group D than in Group C, between 15 and 150 minutes (P < 0.05) (Figure 1).
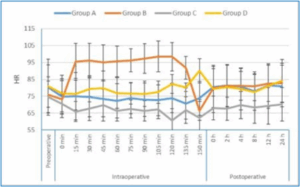
Figure 1: Perioperative heart rate values of the 4 studied groups
Intraoperative MAP showed a significant difference between the four groups, starting from 15 minutes until the end of surgery time (P < 0.001). It was significantly lower in Group C compared with Groups A, B, and D, starting from 15 minutes after induction and continuing until the end of surgery (P < 0.001). MAP, between 30 and 150 minutes, was significantly higher in Group B as compared to Group D, except at 135 minutes (P < 0.05). Additionally, it was significantly lower in Group A than in Group B (between 45 and 120 minutes) (P < 0.001) (Figure 2).
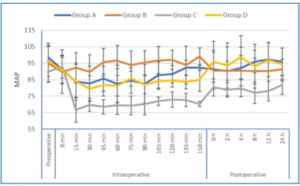
Figure 2: Perioperative mean arterial pressure values of the 4 studied groups
Postoperative heart rate was significantly different among the four groups (P < 0.05). It was markedly lower in Group C than in Groups A, B, and D at the recorded time points during the 24-hour postoperative period (P < 0.05), except at 24 hours compared with Group A and at 4 hours compared with Group D (Figure 1).
Postoperative MAP showed a statistically significant difference among the four groups (P < 0.05). It demonstrated notably lower values at all recorded time points in Group C compared with Groups A, B, and D (P < 0.05) (Figure 2).
Group C didn’t require intraoperative fentanyl, showing a statistically marked difference compared to Group A (60 ± 13.23 µg), Group B (61.67 ± 12.58 µg), and Group D (66.25 ± 7.44 µg); (P < 0.001). Total postoperative morphine consumption was significantly higher for Group D (4.41 ± 2.15 mg) compared with Group A (1.76 ± 2.61 mg), Group B (0.88 ± 1.41 mg), and Group C (0.53 ± 1.18 mg) (P < 0.001) but comparable between the other three groups (P > 0.05) (Table 2).
VAS scores were insignificantly different at 0, 2, and 4 hours among the four groups. Whereas, they were significantly different at 8, 12, and 24 hours (P < 0.05). They were significantly lower at 8 hours in Group C than in Groups B and D (P value 0.047 and 0.009), respectively. At 12 hours, the VAS score was notably lower in Group C than in the other study groups (P < 0.05). However, at 24 hours, it remained lower in Group C compared with both Groups B and D (P < 0.05) (Figure 3).
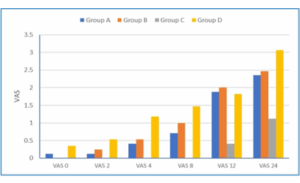
Figure 3: Visual Analogue Scale (VAS) scores of the 4 studied groups
Table 1 shows that the first time to receive morphine was significantly shorter in Group D (8.53 ± 2.79 hr) when compared with Group A (18.50 ± 0.84 hr), Group B (19.00 ± 1.00 hr), and Group C (22.67 ± 1.53 hr), (p <0.001). However, no significant difference was detected between Groups A, B, and C.
Postoperative ketorolac consumption was not significantly different among the four groups (P = 0.861) (Table 1). There was no significant difference in the incidence of PONV among the four groups; however, five patients in Group D experienced at least one episode of PONV, compared with two patients in Group A, none in Group B, and one in Group C (P = 0.067) (Table 1). The RSS in the PACU did not differ significantly among the four groups (Figure 4).
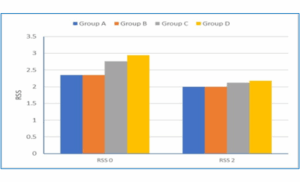
Figure 4: Ramsay Sedation Score (RSS) of the 4 studied groups
Modified radical mastectomy is one of the main surgical approaches for breast cancer. It is commonly operated under general anesthesia with or without a regional block.12 Perioperative pain management for breast cancer surgeries involves pharmacological and interventional modalities.
Pharmacological agents mainly depend on opioid administration, which may be associated with side effects including prolonged sedation, respiratory depression, nausea, vomiting, and ileus. Regional techniques such as thoracic epidural analgesia and paravertebral block became the gold standard techniques for breast surgeries. However, these techniques carry increased risks of complications.13 Facial plane blocks, such as PECS, SAPB, and ESPB, emerged as a reliable alternative analgesic technique that not only provides analgesia but also reduces the need for systemic opioids, minimizes side effects, promotes faster recovery after surgery, and has better patient outcomes.12-14
Administration of ESPB at two levels might enhance the craniocaudal spread of local anesthetics with a wider dermatomal block and potential improvement of the analgesic effect.
Adding additives to local anesthetics during neuraxial and locoregional blocks potentiates the efficacy of local anesthetics and enhances the analgesic efficacy of the blocks.15 Dexamethasone, used as an additive to local anesthetics, prolongs the duration of regional blocks. The mechanism of its action is believed to be through inhibiting the C-fibers' potassium-mediated channels and reducing the release of inflammatory mediators.16 On the other hand, the analgesic effect of adding dexmedetomidine as an additive to local anesthetics is thought to be through its alpha-2 adrenergic agonist effect.17
This study was initiated to investigate the possible merits of additives when added to bi-level ESPB. The study included four groups. Group A received bi-level ESPB with plain bupivacaine, Group B received Bi-level ESPB with 0.1 mg/kg dexamethasone added to bupivacaine 0.25%, Group C received Bi-level ESPB with 0.5 µg/kg dexmedetomidine added to bupivacaine 0.25% and Group D received intraoperative morphine 0.1mg/kg with no intervention.
The current study demonstrated that intraoperative fentanyl consumption and postoperative VAS scores were significantly lower in patients who received bi-level ESPB with dexmedetomidine compared with those who received no adjuvant, dexamethasone, or conventional anesthesia with opioids. However, postoperative morphine consumption was significantly lower in patients who received bi-level ESPB, with or without adjuvants, compared with those who received conventional anesthesia with opioids. Additionally, the time to first morphine administration was significantly prolonged in these patients. Significant differences in perioperative hemodynamic values were observed among the four groups, whereas no significant differences in RSS scores or PONV were reported.
The possible superior analgesic effect of bi-level ESPB was first addressed by Can AKSU and colleagues through performing ESPB on two levels, reporting the efficiency of this block technique compared to conventional anesthesia for breast surgeries.11 Another study that supported the merits of bi-level ESPB was conducted on a patient with rib fractures at multiple levels. They delineated the efficient analgesic effect of bi-level ESPB.18 This was followed by a study that investigated the possible role of extended cranial and caudal extension of local anesthetics on augmenting the analgesic effect of bi-level ESPB during spine surgeries for vertebral correction.19
In a previous study conducted by the authors, they investigated the merits of bi-level ESPB compared to single-level injection and conventional anesthesia. It was reported that bi-level ESPB showed a better analgesic profile compared to the single-level injection.20 In the current study, intraoperative fentanyl consumption and postoperative pain scores were noticed to be lower for the dexmedetomidine group. This was supported by the results of another study that compared dexmedetomidine versus dexamethasone added to levobupivacaine in ESPB during modified radical mastectomy surgeries. They reported that the use of dexmedetomidine resulted in reduced intraoperative analgesic requirements with reduced postoperative pain scores both with movement and at rest. However, they also reported reduced postoperative analgesic consumption with a prolonged duration of analgesia, which, although observed in the current study, did not reach statistical significance.21 This was further supported by a study that compared the additive effects of dexmedetomidine, dexamethasone, and no adjuvant to local anesthetics in an interscalene block for patients undergoing shoulder arthroscopy.
The study reported lower intraoperative opioid consumption in the dexmedetomidine group compared with both the dexamethasone and no-adjuvant groups.22 Adding dexmedetomidine or dexamethasone as adjuvants to local anesthetics in the current study showed comparable results regarding perioperative analgesia. This result was supported by a meta-analysis that compared the role of the two drugs as adjuvants to local anesthetics in peripheral nerve blocks. The results reported that both drugs showed no significant difference regarding block duration, analgesic effect and onset of action.23 In a study investigating the effect of dexmedetomidine (1 µg/kg) as an adjuvant to ropivacaine 0.375% in ESPB during spine surgeries, the results demonstrated a superior analgesic profile for dexmedetomidine compared with no adjuvant in terms of pain scores and opioid consumption. The difference between those findings and the results of the current study might be attributed to the higher dose of dexmedetomidine used.24 Adding dexmedetomidine 1µg/kg versus dexamethasone 10mg as an adjuvant to ropivacaine in ESPB during video-assisted thoracoscopic surgery (VATS) resulted in better analgesia; the superior outcomes reported with dexmedetomidine relative to those in the current study might be due to the different doses used for both dexmedetomidine and dexamethasone.25
Adding dexamethasone as an adjuvant to local anesthetics can reduce the incidence of nausea and vomiting.26,27 Although the current study revealed comparable incidence rates of PONV among patients who received dexamethasone, no patients in the dexamethasone group experienced PONV, whereas five patients in the morphine group did.
The limitations of the current study include its single-center design and relatively small sample size.
Adding dexmedetomidine as an adjuvant to bupivacaine in bi-level ESPB resulted in lower intraoperative analgesic consumption, with lower postoperative VAS scores compared to no adjuvant, dexamethasone, and conventional anesthesia. However, dexmedetomidine and dexamethasone, when used as adjuvants, had a comparable effect to no adjuvant with respect to postoperative opioid consumption and the time to first analgesic administration.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
NS: Manuscript writing, shared analysis of the data_
RM: Manuscript writing, collecting data, initiating the study idea
AS: Shared in writing the manuscript, shared analysis of the data
AF: Study idea; Supervised the manuscript
WE: Writing the manuscript, shared the study idea
Authors affiliations:
- Nahla N Shehab, Assistant Professor of Anaesthesia, Surgical Critical Care and Pain Management, National Cancer Institute, Cairo University, Egypt; Email: nahla.shehab2@gmail.com
- Reham M Fahmy, Lecturer of Anaesthesia, Surgical Critical Care and Pain Management, National Cancer Institute, Cairo University, Egypt; Email: rehammahmoudamr@gmail.com
- Ayman S Abdelrahman, Lecturer of Anaesthesia, Surgical Critical Care and Pain Management, National Cancer Institute, Cairo University, Egypt; Email: ayman.sharawy@nci.cu.edu.eg
- Azza F Omran, Professor of Anaesthesia, Surgical Critical Care and Pain Management, National Cancer Institute, Cairo University, Egypt; Email: azzaomran67@gmail.com
- Walaa Y Elsabeeny, Assistant Professor of Anaesthesia, Surgical Critical Care and Pain Management, National Cancer Institute, Cairo University, Egypt; Email: walaa.elsabeeny@nci.cu.edu.eg; {ORCID:0000-0003-3047-6926}
ABSTRACT
Background & objective: Modified Radical Mastectomy (MRM) is typically performed under general anesthesia that often necessitates opioid administration, which can lead to side effects such as sedation, respiratory depression, and nausea. Fascial plane blocks, including the Erector Spinae Plane Block (ESPB), offer an effective analgesic alternative to opioids. This study aimed to assess the effectiveness of bi-level ESPB with either dexmedetomidine or dexamethasone as adjuvants to bupivacaine, compared with opioid-based analgesia, in patients undergoing MRM.
Methods: This randomized controlled study included female cancer patients scheduled for MRM. Patients were randomized into four groups; Group D (control group) received conventional anesthesia with intraoperative morphine, and the other three groups received bi-level ESPB at T2 and T4. Group A received ESPB with 0.25% bupivacaine, Group B received ESPB with 0.25% bupivacaine and 0.1 mg/kg dexamethasone, and Group C received ESPB with 0.25% bupivacaine and 0.5μg/kg dexmedetomidine.
Results: The time to first rescue morphine was significantly shorter in Group D compared with Groups A, B, and C (P < 0.001). Postoperative morphine consumption was significantly higher in group D than in the other groups (P < 0.001). Intraoperative fentanyl consumption was lower in Group C compared with the rest of the groups (P < 0.001).
Conclusions: In MRM, adding dexmedetomidine as an adjuvant to bupivacaine in ultrasound-guided bi-level ESPB showed a better analgesic profile compared to no adjuvant, dexamethasone, and conventional opioid analgesia with lower pain scores and lower intraoperative opioid consumption. However, dexmedetomidine and dexamethasone, when used as adjuvants, had a comparable effect to no adjuvant with respect to postoperative opioid consumption and the time to first analgesic administration.
Keywords: Dexmedetomidine; Dexamethasone; Modified Radical Mastectomy; Analgesia; Morphine.
Citation: Shehab NN, Fahmy RM, Abdelrahman AS, Omran AF, Elsabeeny WY. Analgesic efficacy of adding adjuvants to local anesthetics in bi-level erector spinae plane block for breast surgeries: a randomized controlled study. Anaesth. Pain Intensive Care 2025;29(6):787-795. DOI: 10.35975/apic.v29i6.2979.
Received: April 16, 2025; Revised: August 02, 2025; Accepted: August 05, 2025
1. INTRODUCTION
Perioperative analgesia for cancer patients scheduled for modified radical mastectomy is considered a pivotal component of care for such patients.1 Risk of development of chronic post-mastectomy pain syndrome (PMPS) is present following any breast surgical intervention, where a considerable percentage of patients with undertreated acute pain can develop chronic pain.2
Appropriately managing perioperative pain control can reduce both postoperative complications and morbidity by reducing the incidence of the development of post-mastectomy pain syndrome.3 Pharmacological agents such as paracetamol, non-steroidal anti-inflammatory drugs (NSAIDs), opioids, as well as interventional modalities including neuraxial blocks (thoracic epidural analgesia) and locoregional blocks (paravertebral blocks, serratus anterior plane block, pectoral nerve block, and erector spinae plane block) have been considered for perioperative analgesia in breast surgeries.4
ESPB is a unique regional technique that was first described in 2016. It has been widely used as an effective regional block for postoperative analgesia in thoracotomy, with promising results reported since it was first described by Forero and his colleagues.5 Single-shot block may not provide the desired duration of analgesia required6 and inserting a catheter for continuous infusion of local anesthetic is not always feasible; therefore, bi-level ESPB was introduced into practice, aiming to block a wider dermatomal area through enhanced craniocaudal spread.7
Several adjuvants have been added to local anesthetics during regional blocks, including magnesium sulphate, clonidine, and opioids.8 Adding dexamethasone as an adjunct to local anesthetic has been shown to reduce perioperative pain and augment the local anesthetic effect.9 Dexmedetomidine is an alpha-2 adrenergic agonist, known for its sedative, analgesic-sparing, and additive perioperative sympatholytic effects. The use of dexmedetomidine as an adjuvant to local anesthetic drugs in neuraxial and nerve blocks has been shown to extend analgesia.8,10
The current work aims to assess the analgesic efficacy of dexamethasone and dexmedetomidine as adjuvants to bupivacaine in ultrasound (US) guided bi-level ESPB for modified radical mastectomy and its impact on the intensity of acute pain, duration of analgesia, and decreased perioperative opioid consumption.
2. METHODOLOGY
This prospective randomized controlled single-blinded (outcome assessor) trial was done after agreement of the ethical committee (AP2207-30110) and was registered at clinicaltrials.gov (NCT05591417). The patients were assigned to the study after obtaining written informed consent.
Initially, 93 adult female patients (ASA II–III), aged 18–65 years, with a BMI between 20 and 35 kg/m², and diagnosed with breast cancer scheduled for modified radical mastectomy with axillary evacuation were screened for eligibility. Patients were excluded from the study if they were pregnant or had severe cardiovascular instability, impaired coagulation profiles, abnormal kidney or liver function, bone metastases, infection at the injection site, or known allergies to any of the study medications. Additionally, patients with psychological or mental impairment or on chronic pain medications were also excluded.
Sixty-eight patients meeting the eligibility criteria were randomized into 4 groups through computer random number generation. Group A (17 patients): received ultrasound-guided bi-level ESPB with 15 ml bupivacaine 0.25% at each level. Group B (17 patients): received ultrasound-guided bi-level ESPB with 15 ml bupivacaine 0.25% with 0.1 mg/kg dexamethasone at each level. Group C (15 patients): received ultrasound-guided bi-level ESPB with 15 ml bupivacaine 0.25% with 0.5μg/kg dexmedetomidine at each level. Group D (17 patients): received conventional anesthesia with intraoperative intravenous (IV) morphine 0.1 mg/kg.
2.1. Study Interventions
All patients were clinically assessed at the preoperative assessment clinic. Investigations, including complete blood picture, liver & kidney functions, and coagulation profile, were obtained. In the holding room, the patients’ vital signs were monitored. The block technique and the visual analogue scale (VAS) were explained to all patients, with the scale ranging from 0 cm (no pain) to 10 cm (severe pain). Premedication with IV midazolam 0.02 mg/kg was given.
2.2. Bi-level ESPB technique
After skin sterilization with 10% povidone-iodine, the block was performed on the side of the operation with the patient in the sitting position, 20 minutes before induction of anesthesia, using the technique described by Forero and colleagues. The ultrasound probe (M-Turbo; FUJIFILM SonoSite Inc., Bothell, WA, USA), either linear (6-13 MHz) or curved (2-5 MHz), was placed vertically 1.5-2 cm lateral to the spine after identifying T2 and T4 spinous processes, where blocks were done using an in-plane approach. The needle was introduced through the skin in a cranio-caudal direction toward the transverse process. Two milliliters of normal saline were injected into the fascial plane between the erector spinae muscle and the transverse process, and hydro-dissection of the plane was observed. After confirming the needle position, 15 milliliters of the prepared solution were administered at the level of T2 and similarly at the level of T4. Dermatomal testing was performed by an anesthesiologist who did not participate in the study. If no sensory block was present after 20 minutes, patients were omitted from the study.
- Group A: received bi-level erector spinae using 15 ml of bupivacaine 0.25% at each level.
- Group B: received bi-level ESPB using 15 ml of bupivacaine 0.25% with 0.1 mg/kg dexamethasone at each level.
- Group C: received bi-level ESPB using 15 ml of bupivacaine 0.25% with 0.5μg/kg dexmedetomidine at each level.
- Group D: received intraoperative intravenous (IV) morphine 0.1 mg/kg
General anesthesia was administered to all patients using intravenous (IV) propofol (2-3 mg/kg), fentanyl (2µg/kg), and rocuronium (0.6 mg/kg), followed by endotracheal intubation. Volume-controlled mechanical ventilation was done, and anesthesia was maintained by inhalational anesthesia using isoflurane (1-1.5%) and top-up doses of muscle relaxant as needed. Intraoperative multimodal analgesia was administered in the form of 1gm paracetamol and 30 mg ketorolac IV. Patients were monitored throughout the operation using the standard ASA monitors.
Heart rate (HR) and mean arterial pressure (MAP) were recorded before the induction of general anesthesia, after tracheal intubation, and then every 15 minutes till the end of the operation. During surgery, if there was a reported increase in HR or MAP by >30% from the baseline readings and after exclusion of inadequate anesthesia, a bolus dose of fentanyl 0.5µg/kg was given and recorded. Significant decrease in HR or MAP was addressed according to each patient’s baseline values and medical condition by atropine or ephedrine as needed.
Upon recovery, patients were admitted to the post-anesthesia care unit (PACU), where HR, MAP, pain scores, and sedation scores were recorded. In the ward and for the following 24 hours, monitoring for the same parameters was done at 2, 4, 8, 12, and 24 hours. In the PACU, patients’ sedation status was assessed using the Ramsay Sedation Score (RSS), which ranges from 1 (anxious, restless, or both) to 6 (no response to stimulus). Pain was assessed using the VAS score. Patients with a VAS score < 4 received 30 mg of intravenous ketorolac, while those with a VAS score ≥ 4 were given intravenous morphine as a bolus dose of 3 mg per dose, with the total dose not exceeding 0.1 mg/kg per dose. The total amount of morphine given in 24 hours was recorded in the four groups. Acetaminophen 1g IV was given to all patients every 8 hours. Adverse effects in the form of hematoma, nausea, vomiting, or over-sedation were recorded.
2.4. Sample Size
The G power program was used to calculate a total sample size of 60. Fifteen patients in each group were initially required in order to reject the null hypothesis that the mean 24-hour postoperative morphine consumption is equivalent among the research groups, probability (power) = 0.99999.11 Two patients were added to each group for compensation of any possible attrition during the study. The Type I error probability associated with testing this null hypothesis is 0.05.
2.5. Statistical Analysis
Statistical Package for the Social Sciences (SPSS) version 28 (IBM Corp., Armonk, NY, USA) was used for data coding and statistical analysis. The type of statistical test, parametric or non-parametric, was selected according to the distribution of quantitative variables, which was assessed using the Shapiro–Wilk normality test and histograms. Analysis of variance (ANOVA) was used to compare parametric variables, which were expressed as mean ± standard deviation (SD), among the four groups, and post hoc Tukey testing was performed for intergroup comparisons.
Nonparametric variables were expressed as median and interquartile range (IQR) and were analyzed using the Kruskal–Wallis test, with the Mann–Whitney U test used for further pairwise analysis. The chi-square test was applied to categorical variables, which were expressed as frequencies and percentages. When the expected frequency was less than 5, Exact test was used. Statistical significance was interpreted when the two-tailed P value was less than or equal to 0.05.
2.6. Outcomes
The primary outcome measure was set as the total morphine consumption in the first 24 hours postoperatively. Secondary outcome parameters were perioperative hemodynamics (HR and MAP), total intraoperative fentanyl consumption, time to first rescue analgesia, postoperative pain scores, and any possible complications (hematoma, nausea and/or vomiting, or over sedation) were recorded.
3. RESULTS
Initially, 93 adult female patients, between 18 and 65 years of age, undergoing modified radical mastectomy were assessed for eligibility. Sixty-eight patients (ASA II–III) were randomized into one of the four study groups.
Patients’ demographic data (age and BMI) and clinical characteristics (diabetes and hypertension) were comparable among the four groups (Table 1).
| Table 1: Demographic data and clinical characteristics of the studied groups | |||||
| Demographic data | Group A
(n = 17) |
Group B
(n = 17) |
Group C
(n = 17) |
Group D
(n = 17) |
P-value |
| Age (years) | 48.24 ± 11.83 | 51.88 ± 11.70 | 50.76 ± 11.08 | 47.82 ± 11.88 | 0.694 |
| BMI (kg/m2) | 30.29 ± 4.01 | 33.12 ± 2.23 | 31.82 ± 3.13 | 32.29 ± 3.75 | 0.105 |
| HTN | 5 (29.4) | 4 (23.5) | 8 (47.1) | 7 (41.2) | 0.462 |
| DM | 5 (29.4) | 4 (23.5) | 6 (35.3) | 5 (29.4) | 0.904 |
| CTH | 5 (29.4) | 8 (47.1) | 9 (52.9) | 8 (47.1) | 0.542 |
| Data presented as mean ± SD or n (%); P < 0.05 is considered statistically significant
BMI: Body mass index, DM: diabetes mellitus, HTN: hypertension, CTH: chemotherapy |
|||||
Intraoperative heart rate values were significantly different among the four groups, starting from 15 minutes until the end of the surgery time (P < 0.001). They were significantly higher in group B compared with Groups A, C, and D from 15 to 135 minutes intraoperatively. Additionally, they were significantly higher in Group D than in Group C, between 15 and 150 minutes (P < 0.05) (Figure 1).

Figure 1: Perioperative heart rate values of the 4 studied groups
Intraoperative MAP showed a significant difference between the four groups, starting from 15 minutes until the end of surgery time (P < 0.001). It was significantly lower in Group C compared with Groups A, B, and D, starting from 15 minutes after induction and continuing until the end of surgery (P < 0.001). MAP, between 30 and 150 minutes, was significantly higher in Group B as compared to Group D, except at 135 minutes (P < 0.05). Additionally, it was significantly lower in Group A than in Group B (between 45 and 120 minutes) (P < 0.001) (Figure 2).

Figure 2: Perioperative mean arterial pressure values of the 4 studied groups
Postoperative heart rate was significantly different among the four groups (P < 0.05). It was markedly lower in Group C than in Groups A, B, and D at the recorded time points during the 24-hour postoperative period (P < 0.05), except at 24 hours compared with Group A and at 4 hours compared with Group D (Figure 1).
Postoperative MAP showed a statistically significant difference among the four groups (P < 0.05). It demonstrated notably lower values at all recorded time points in Group C compared with Groups A, B, and D (P < 0.05) (Figure 2).
Group C didn’t require intraoperative fentanyl, showing a statistically marked difference compared to Group A (60 ± 13.23 µg), Group B (61.67 ± 12.58 µg), and Group D (66.25 ± 7.44 µg); (P < 0.001). Total postoperative morphine consumption was significantly higher for Group D (4.41 ± 2.15 mg) compared with Group A (1.76 ± 2.61 mg), Group B (0.88 ± 1.41 mg), and Group C (0.53 ± 1.18 mg) (P < 0.001) but comparable between the other three groups (P > 0.05) (Table 2).
| Table 2: Intraoperative fentanyl, postoperative morphine, ketorolac consumption, first time to receive morphine and PONV in the studied groups | |||||
| Analgesic use data | Group A
(n = 17) |
Group B
(n = 17) |
Group C
(n = 17) |
Group D
(n = 17) |
P value |
| Total Fentanyl (µg) | 60 ± 13.23 | 61.7 ± 12.58 | 0.0 ± 0.0 | 66.25 ± 7.44 | < 0.001* |
| Total postop morphine (mg) | 1.76 ± 2.61 | 0.88 ± 1.41 | 0.53 ± 1.18 | 4.41 ± 2.15 | < 0.001* |
| Time to 1st. dose morphine (hr) | 18.50 ± 0.84 | 19 ± 1.00 | 22.67 ± 1.53 | 8.53 ± 2.79 | < 0.001* |
| Ketorolac | 2 (11.76) | 3 (17.65) | 1 (5.88) | 3 (17.65) | 0.861 |
| PONV | 2 (11.76) | 0 (0) | 1 (5.88) | 5 (29.41) | 0.067 |
| Data presented as mean ± SD or n (%); P < 0.05 is considered statistically significant | |||||

Figure 3: Visual Analogue Scale (VAS) scores of the 4 studied groups
Table 1 shows that the first time to receive morphine was significantly shorter in Group D (8.53 ± 2.79 hr) when compared with Group A (18.50 ± 0.84 hr), Group B (19.00 ± 1.00 hr), and Group C (22.67 ± 1.53 hr), (p <0.001). However, no significant difference was detected between Groups A, B, and C.
Postoperative ketorolac consumption was not significantly different among the four groups (P = 0.861) (Table 1). There was no significant difference in the incidence of PONV among the four groups; however, five patients in Group D experienced at least one episode of PONV, compared with two patients in Group A, none in Group B, and one in Group C (P = 0.067) (Table 1). The RSS in the PACU did not differ significantly among the four groups (Figure 4).

Figure 4: Ramsay Sedation Score (RSS) of the 4 studied groups
4. DISCUSSION
Modified radical mastectomy is one of the main surgical approaches for breast cancer. It is commonly operated under general anesthesia with or without a regional block.12 Perioperative pain management for breast cancer surgeries involves pharmacological and interventional modalities.
Pharmacological agents mainly depend on opioid administration, which may be associated with side effects including prolonged sedation, respiratory depression, nausea, vomiting, and ileus. Regional techniques such as thoracic epidural analgesia and paravertebral block became the gold standard techniques for breast surgeries. However, these techniques carry increased risks of complications.13 Facial plane blocks, such as PECS, SAPB, and ESPB, emerged as a reliable alternative analgesic technique that not only provides analgesia but also reduces the need for systemic opioids, minimizes side effects, promotes faster recovery after surgery, and has better patient outcomes.12-14
Administration of ESPB at two levels might enhance the craniocaudal spread of local anesthetics with a wider dermatomal block and potential improvement of the analgesic effect.
Adding additives to local anesthetics during neuraxial and locoregional blocks potentiates the efficacy of local anesthetics and enhances the analgesic efficacy of the blocks.15 Dexamethasone, used as an additive to local anesthetics, prolongs the duration of regional blocks. The mechanism of its action is believed to be through inhibiting the C-fibers' potassium-mediated channels and reducing the release of inflammatory mediators.16 On the other hand, the analgesic effect of adding dexmedetomidine as an additive to local anesthetics is thought to be through its alpha-2 adrenergic agonist effect.17
This study was initiated to investigate the possible merits of additives when added to bi-level ESPB. The study included four groups. Group A received bi-level ESPB with plain bupivacaine, Group B received Bi-level ESPB with 0.1 mg/kg dexamethasone added to bupivacaine 0.25%, Group C received Bi-level ESPB with 0.5 µg/kg dexmedetomidine added to bupivacaine 0.25% and Group D received intraoperative morphine 0.1mg/kg with no intervention.
The current study demonstrated that intraoperative fentanyl consumption and postoperative VAS scores were significantly lower in patients who received bi-level ESPB with dexmedetomidine compared with those who received no adjuvant, dexamethasone, or conventional anesthesia with opioids. However, postoperative morphine consumption was significantly lower in patients who received bi-level ESPB, with or without adjuvants, compared with those who received conventional anesthesia with opioids. Additionally, the time to first morphine administration was significantly prolonged in these patients. Significant differences in perioperative hemodynamic values were observed among the four groups, whereas no significant differences in RSS scores or PONV were reported.
The possible superior analgesic effect of bi-level ESPB was first addressed by Can AKSU and colleagues through performing ESPB on two levels, reporting the efficiency of this block technique compared to conventional anesthesia for breast surgeries.11 Another study that supported the merits of bi-level ESPB was conducted on a patient with rib fractures at multiple levels. They delineated the efficient analgesic effect of bi-level ESPB.18 This was followed by a study that investigated the possible role of extended cranial and caudal extension of local anesthetics on augmenting the analgesic effect of bi-level ESPB during spine surgeries for vertebral correction.19
In a previous study conducted by the authors, they investigated the merits of bi-level ESPB compared to single-level injection and conventional anesthesia. It was reported that bi-level ESPB showed a better analgesic profile compared to the single-level injection.20 In the current study, intraoperative fentanyl consumption and postoperative pain scores were noticed to be lower for the dexmedetomidine group. This was supported by the results of another study that compared dexmedetomidine versus dexamethasone added to levobupivacaine in ESPB during modified radical mastectomy surgeries. They reported that the use of dexmedetomidine resulted in reduced intraoperative analgesic requirements with reduced postoperative pain scores both with movement and at rest. However, they also reported reduced postoperative analgesic consumption with a prolonged duration of analgesia, which, although observed in the current study, did not reach statistical significance.21 This was further supported by a study that compared the additive effects of dexmedetomidine, dexamethasone, and no adjuvant to local anesthetics in an interscalene block for patients undergoing shoulder arthroscopy.
The study reported lower intraoperative opioid consumption in the dexmedetomidine group compared with both the dexamethasone and no-adjuvant groups.22 Adding dexmedetomidine or dexamethasone as adjuvants to local anesthetics in the current study showed comparable results regarding perioperative analgesia. This result was supported by a meta-analysis that compared the role of the two drugs as adjuvants to local anesthetics in peripheral nerve blocks. The results reported that both drugs showed no significant difference regarding block duration, analgesic effect and onset of action.23 In a study investigating the effect of dexmedetomidine (1 µg/kg) as an adjuvant to ropivacaine 0.375% in ESPB during spine surgeries, the results demonstrated a superior analgesic profile for dexmedetomidine compared with no adjuvant in terms of pain scores and opioid consumption. The difference between those findings and the results of the current study might be attributed to the higher dose of dexmedetomidine used.24 Adding dexmedetomidine 1µg/kg versus dexamethasone 10mg as an adjuvant to ropivacaine in ESPB during video-assisted thoracoscopic surgery (VATS) resulted in better analgesia; the superior outcomes reported with dexmedetomidine relative to those in the current study might be due to the different doses used for both dexmedetomidine and dexamethasone.25
Adding dexamethasone as an adjuvant to local anesthetics can reduce the incidence of nausea and vomiting.26,27 Although the current study revealed comparable incidence rates of PONV among patients who received dexamethasone, no patients in the dexamethasone group experienced PONV, whereas five patients in the morphine group did.
5. LIMITATIONS
The limitations of the current study include its single-center design and relatively small sample size.
6. CONCLUSION
Adding dexmedetomidine as an adjuvant to bupivacaine in bi-level ESPB resulted in lower intraoperative analgesic consumption, with lower postoperative VAS scores compared to no adjuvant, dexamethasone, and conventional anesthesia. However, dexmedetomidine and dexamethasone, when used as adjuvants, had a comparable effect to no adjuvant with respect to postoperative opioid consumption and the time to first analgesic administration.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
NS: Manuscript writing, shared analysis of the data_
RM: Manuscript writing, collecting data, initiating the study idea
AS: Shared in writing the manuscript, shared analysis of the data
AF: Study idea; Supervised the manuscript
WE: Writing the manuscript, shared the study idea
11. REFERENCES
- Kaur U, Shamshery C, Agarwal A, Prakash N, Valiveru RC, Mishra P. Evaluation of postoperative pain in patients undergoing modified radical mastectomy with pectoralis or serratus-intercostal fascial plane blocks. Korean J Anesthesiol. 2020;73(5):425–33. PMCID: PMC7533170 DOI: 4097/kja.20159
- Poleshuck EL, Katz J, Andrus CH, Hogan LA, Jung BF, Kulick DI, et al. Risk Factors for Chronic Pain Following Breast Cancer Surgery: A Prospective Study. Journal of Pain. 2006;7(9):626–34. PMCID: PMC6983301 DOI: 1016/j.jpain.2006.02.007
- Berger JM, Longhitano Y, Zanza C, Sener SF. Factors affecting the incidence of chronic pain following breast cancer surgery: Preoperative history, anesthetic management, and surgical technique. J Surg Oncol. 2020;122(7):1307–14. DOI: 1002/jso.26176
- Singh NP, Makkar JK, Kuberan A, Guffey R, Uppal V. Efficacy of regional anesthesia techniques for postoperative analgesia in patients undergoing major oncologic breast surgeries: a systematic review and network meta-analysis of randomized controlled trials. Canadian Journal of Anesthesia [Internet]. 2022;69(4):527–49. DOI: 1007/s12630-021-02183-z
- Forero M, Adhikary SD, Lopez H, Tsui C, Chin KJ. The erector spinae plane block a novel analgesic technique in thoracic neuropathic pain. Reg Anesth Pain Med. 2016;41(5):621–7. DOI: 1097/AAP.0000000000000451
- Yu L, Shen XJ, Liu H, Zhou YT, Zhang Q, Zhang ZD, et al. Effect of ultrasound-guided continuous erector spinae plane block on postoperative pain and inflammatory response in patients undergoing modified radical mastectomy for breast cancer: study protocol for a randomised controlled trial. Trials [Internet]. 2024;25(1):1–11. PMCID: PMC10788999 DOI: 1186/s13063-023-07777-0
- Altlparmak B, Korkmaz M, Uysal AAH, Gumus Demirbilek S. Bi-level erector spinae plane block for the control of severe back pain related to vertebral metastasis. BMJ Case Rep. 2019;12(6):2018–20. PMCID: PMC6605916 DOI: 1136/bcr-2018-228129
- Prabhakar A, Lambert T, Kaye RJ, Gaignard SM, Ragusa J, Wheat S, et al. Adjuvants in clinical regional anesthesia practice: A comprehensive review. Best Pract Res Clin Anaesthesiol [Internet]. 2019;33(4):415–23. DOI: 1016/j.bpa.2019.06.001
- Desai N, Kirkham KR, Albrecht E. Local anaesthetic adjuncts for peripheral regional anaesthesia: a narrative review. Anaesthesia. 2021;76(S1):100–9. DOI: 1111/anae.15245
- Emelife PI, Eng MR, Menard BL, Myers AS, Cornett EM, Urman RD, et al. Adjunct medications for peripheral and neuraxial anesthesia. Best Pract Res Clin Anaesthesiol [Internet]. 2018;32(2):83–99. DOI: 1016/j.bpa.2018.06.011
- Aksu C, Kuş A, Yörükoğlu HU, Tor Kiliç C, Gürkan Y. Analgesic effect of the bi-level injection erector spinae plane block after breast surgery: A randomized controlled trial. Agri. 2019;31(3):132–7. DOI: 14744/agri.2019.61687
- Garg R, Bhan S, Vig S. Newer regional analgesia interventions (fascial plane blocks) for breast surgeries: Review of literature. Indian J Anaesth [Internet]. 2018;62(4):254. PMCID: PMC5907430 DOI: 4103/ija.IJA_46_18
- Wahba SS, Kamal SM. Thoracic paravertebral block versus pectoral nerve block for analgesia after breast surgery. Egypt J Anaesth [Internet]. 2014;30(2):129–35. DOI: 10.1016/j.egja.2013.10.006
- Elsabeeny WY, Shehab NN, Wadod MA, Elkady MA. Perioperative analgesic modalities for breast cancer surgeries: A prospective randomized controlled trial. J Pain Res. 2020;13:2885–94. PMCID: PMC7669532 DOI: 2147/JPR.S274808
- Swain A, Nag DS, Sahu S, Samaddar DP. Adjuvants to local anesthetics: Current understanding and future trends. World J Clin Cases. 2017;5(8):307. PMCID: PMC5561500 DOI: 12998/wjcc.v5.i8.307
- Movafegh A, Razazian M, Hajimaohamadi F, Meysamie A. Dexamethasone added to lidocaine prolongs axillary brachial plexus blockade. Anesth Analg. 2006;102(1):263–7. DOI: 1213/01.ane.0000189055.06729.0a
- Urits I, Virgen CG, Alattar H, Jung JW, Berger AA, Kassem H, et al. A Comprehensive Review and Update of the Use of Dexmedetomidine for Regional Blocks. Psychopharmacol Bull. 2020;50(4):121–41. PMCID: PMC7901136
- Liu R, Clark L, Bautista A. Unilateral Bilevel Erector Spinae Plane Catheters for Flail Chest: A Case Report. A A Pract. 2020;14(7):e01211. DOI: 1213/XAA.0000000000001211
- Sinha C, Kumar A, Kumar A, Kumari P, Agrawal P, SK A. Two-level bilateral ultrasound-guided erector spinae plane block for kyphosis corrective surgery - A case report -. Anesth Pain Med (Seoul). 2022;17(3):327–30. PMCID: PMC9346201 DOI: 17085/apm.21107
- Elsabeeny WY, Fahmy RM, Elshamy FH, Shehab NN, Abed SM, Taha WS, et al. Role of bilevel erector spinae with high thoracic block vs conventional unilevel block in analgesia and reduction of pain in axilla in breast cancer surgeries: a randomized controlled trial. Pain Rep [Internet]. 2025 Feb;10(1):e1234. PMCID: PMC11732649 DOI: 1097/PR9.0000000000001234
- Ahmed HI, Ali AR, El Emam NH, Magdy M, Gamal RM. Dexmedetomidine vs dexamethasone as adjuvants to levobupivacaine in ultrasound-guided erector spinae plane block for patients undergoing modified radical mastectomy: a randomized double-blind study. pain intensive care 2022;27(1):65−72. DOI: 10.35975/apic.v27i1.2122
- R. M, J. F, B. T, A. B, D. P, K. G, et al. Comparison of dexmedetomidine and dexamethasone as adjuvants to ultra-sound guided interscalene block in arthroscopic shoulder surgery: A double-blinded randomized placebo-controlled study. Anesth Pain Med [Internet]. 2021;11(3):e117020. Available from: https://sites.kowsarpub.com/aapm/cdn/dl/4eee2664-dcb9-11eb-ac9f-5f026241eea8%0Ahttp://ovidsp.ovid.com/ovidweb.cgi?T = JS&PAGE = reference&D = emed23&NEWS = N&AN = 2013009477
- Song ZG, Pang SY, Wang GY, Zhang Z. Comparison of postoperative analgesic effects in response to either dexamethasone or dexmedetomidine as local anesthetic adjuvants: a systematic review and meta-analysis of randomized controlled trials. J Anesth [Internet]. 2021;35(2):270–87. PMCID: PMC7969577 DOI: 1007/s00540-021-02895-y
- Yi-han W, Rong T, Jun L, Min W, Yan Z, Yi L, et al. Dexmedetomidine combined with ropivacaine for erector spinae plane block after posterior lumbar spine surgery: a randomized controlled trial. BMC Musculoskelet Disord. 2022;23(1):1–9. PMCID: PMC8915521 DOI: 1186/s12891-022-05198-9
- Gao Z, Xiao Y, Wang Q, Li Y. Comparison of dexmedetomidine and dexamethasone as adjuvant for ropivacaine in ultrasound-guided erector spinae plane block for video-assisted thoracoscopic lobectomy surgery: a randomized, double-blind, placebo-controlled trial. Ann Transl Med. 2019;7(22):668–668. PMCID: PMC6944602 DOI: 10.21037/atm.2019.10.74
- Huynh TM, Marret E, Bonnet F. Combination of dexamethasone and local anaesthetic solution in peripheral nerve blocks: A meta-analysis of randomised controlled trials. Eur J Anaesthesiol. 2015 Nov;32(11):751-8. DOI: 10.1097/EJA.0000000000000248
- Gupta B. Role of dexamethasone in peri-operative anesthesia management: A review of literature. Res Pract Anesthesiol Open J. 2017; 2(2): 33- 39. doi: 10.17140/RPAOJ-2-114