Rayan A. Qutob 1*
Author affiliation:
Methoxyflurane is a halogenated ether previously used as a general anesthetic, now reintroduced in subanesthetic doses for analgesia. Although considered safe in low doses, historical data suggest the potential for rare but severe hepatotoxicity. We report a case of methoxyflurane-induced acute liver failure following repeated low-dose exposure, including unsupervised over-the-counter use, which necessitated liver transplantation.
A previously healthy 38-year-old woman with a history of migraine (managed with sumatriptan) and varicose veins developed progressive symptoms of acute hepatitis, including severe epigastric pain, jaundice, and persistent vomiting. These symptoms began three days after she self-administered a third dose of inhaled methoxyflurane, obtained over the counter for post-procedural pain. The initial dose had been administered under supervision during a varicose vein sclerotherapy session, while the subsequent two doses were taken one week apart without medical oversight.
On evaluation, laboratory investigations revealed marked hepatic dysfunction, including hyperbilirubinemia, elevated gamma glutamyl transpeptidase (GGT), and evidence of impaired synthetic function. Coagulopathy was evident, with a significantly prolonged INR. Additional findings included hypoalbuminemia and elevated inflammatory markers. Comprehensive workup excluded viral, autoimmune, and metabolic causes of liver injury. Despite supportive therapy with intravenous fluids and N-acetylcysteine, her liver function continued to decline, and she was referred for liver transplantation. She subsequently underwent orthotopic liver transplantation. Histopathological examination of the explanted liver demonstrated extensive centrilobular necrosis, consistent with drug-induced liver injury likely attributable to methoxyflurane. This case emphasizes that even subanesthetic analgesic doses—especially with repeated or unsupervised use—may trigger immune-mediated hepatic injury analogous to halothane hepatitis. Prompt recognition, exclusion of alternative causes, and early referral for transplantation are essential for optimal outcomes.
Abbreviations: ANA: antinuclear antibody, ASMA: anti-smooth muscle antibody, AMA: anti-mitochondrial antibody, GGT: gamma glutamyl transpeptidase, LKM: liver-kidney microsomal antibody.
Keywords: acute hepatitis; hepatic dysfunction; Methoxyflurane; Toxicity; Transplantation
Citation: Qutob RA. Methoxyflurane-induced hepatotoxicity following repeated analgesic use: a case report. Anaesth. pain intensive care 2025;29(8):1037-1043. DOI: 10.35975/apic.v29i8.2971
Received: June 17, 2025; Revised: July 22, 2025; Accepted: August 02, 2025
Methoxyflurane is a fluorinated hydrocarbon anesthetic that was widely used in the 1960s for general anesthesia. It provided profound analgesia and muscle relaxation, but its high lipid solubility led to prolonged induction and emergence times, as well as extensive metabolism.1,2 Early reports in 1966 linked methoxyflurane to significant nephrotoxicity, characterized by high-output renal failure due to the release of inorganic fluoride.1 Subsequent studies in the 1970s confirmed dose-dependent kidney injury related to methoxyflurane metabolism.2 These safety concerns, along with the introduction of newer volatile anesthetics with more favorable pharmacologic profiles, led to methoxyflurane being withdrawn from anesthetic practice by the late 1970s.1
In addition to nephrotoxicity, methoxyflurane was implicated in rare cases of severe hepatotoxicity. The first published case of methoxyflurane-associated hepatitis appeared in 1966.4], and a 1974 review by Joshi and Conn coined the term “methoxyflurane-associated hepatitis,” suggesting an idiosyncratic, immune-mediated injury analogous to halothane hepatitis.3 However, as methoxyflurane fell out of favor as an anesthetic, discussion of its hepatotoxic potential waned in subsequent decades.
Methoxyflurane has experienced a resurgence since the 1990s in the form of a self-administered inhalational analgesic for short procedures and trauma-related pain. At subanesthetic doses (typically 3 mL per inhaler, marketed as Penthrox®), it provides rapid and effective pain relief with minimal sedation. Millions of administrations have been recorded in prehospital and outpatient settings, particularly in Australia, generally without serious complications. Randomized controlled trials (e.g., the STOP! study) have demonstrated the efficacy and acceptable safety of low-dose methoxyflurane in acute pain management.5 As a result, methoxyflurane has been approved in several countries for use in emergency analgesia.
Despite its favorable safety record in this context, concerns remain that methoxyflurane’s metabolism could, in rare instances, trigger hepatotoxicity—even at analgesic doses. Idiosyncratic liver injury from volatile anesthetics is best exemplified by halothane, which caused fulminant hepatic failure in approximately 1 in 6,000–20,000 exposures (fatal in ~1 in 35,000 single exposures, and up to 1 in 1,000 with repeated exposures).6–8 Susceptible individuals, often middle-aged women with prior exposures, would develop what is now known as “halothane hepatitis,” an immune-mediated hepatic necrosis with a mortality rate of around 50% once liver failure occurs.9 Newer agents such as enflurane, isoflurane, and desflurane undergo limited hepatic metabolism and have only a handful of documented hepatotoxic episodes. Sevoflurane, which undergoes minimal biotransformation into trifluoroacetylated proteins, is considered the safest in this regard.10
Methoxyflurane lies between these extremes. It undergoes extensive metabolism (up to 50–70%) and produces nephrotoxic metabolites such as dichloroacetic acid and inorganic fluoride, and possibly hepatotoxic compounds as well.11,12 Unlike halothane, methoxyflurane lacks a trifluoromethyl group and does not produce trifluoroacetylated protein adducts..], but early reports suggested it could still cause hepatic necrosis in select cases.
Here, we present a case of acute drug-induced liver injury (DILI) attributable to repeated low-dose methoxyflurane used for analgesia. To our knowledge, it is one of very few modern cases linking subanesthetic methoxyflurane use to severe hepatotoxicity. We outline the clinical course, explore the proposed mechanisms, review prior cases, and discuss the differential diagnosis, management—including the role of N-acetylcysteine—and how methoxyflurane compares to other halogenated anesthetics in terms of hepatotoxic risk. Recognizing such rare adverse events is crucial as methoxyflurane use expands globally.
A 38-year-old woman with a history of migraine (managed with sumatriptan) and no known liver disease presented to the emergency department with a five-day history of persistent vomiting, severe epigastric and right upper quadrant pain, and progressive jaundice. She denied alcohol use, herbal or traditional medicines, and recent travel. Three weeks earlier, she had undergone sclerotherapy for varicose veins, during which she received 3 mL of inhaled methoxyflurane (Penthrox®) for analgesia. Over the subsequent two weeks, she independently self-administered two additional doses of methoxyflurane via over-the-counter inhalers for post-procedural pain. Symptoms began three days following the last methoxyflurane exposure.
On examination, she appeared acutely ill, was alert and oriented, with stable vital signs: blood pressure 108/66 mmHg, heart rate 82 bpm, and temperature 36.7°C. She was icteric with dry mucous membranes and displayed epigastric and right upper quadrant tenderness. Murphy’s sign was positive. There was no hepatosplenomegaly, peripheral edema, or encephalopathy. Other systemic examinations were unremarkable.
Initial laboratory tests demonstrated acute liver injury and progressive hepatic synthetic failure: white blood cell count 16 ×10⁹/L; platelet count 60 ×10⁹/L; total bilirubin 98 µmol/L (increased from 73 µmol/L); direct bilirubin 77.3 µmol/L; alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were elevated (values unavailable); gamma-glutamyl transferase (GGT) was markedly elevated at 1160 U/L; alkaline phosphatase (ALP) was 265 U/L. Coagulation parameters were severely deranged, with prothrombin time (PT) of 48 seconds (up from 23), international normalized ratio (INR) of 4.06 (from 1.82), and activated partial thromboplastin time (aPTT) of 62 seconds (from 48). Serum albumin was low at 24 g/L. Serum creatinine was mildly elevated at 157 µmol/L. Inflammatory markers were significantly raised: C-reactive protein (CRP) 222 mg/L, procalcitonin 5.21 ng/mL, and lactate 4.5 mmol/L.
Extensive evaluation for causes of acute liver injury was unremarkable. Viral serologies for hepatitis A, B, C, D, and E were negative. HIV testing was negative. Autoimmune hepatitis panel, including antinuclear antibody (ANA), anti-smooth muscle antibody (ASMA), anti-mitochondrial antibody (AMA), and liver-kidney microsomal antibody (LKM) was all negative. Acetaminophen levels were undetectable. Metabolic tests, including ceruloplasmin, ferritin, and transferrin saturation, were within normal limits, making Wilson’s disease and hemochromatosis unlikely.
Abdominal ultrasound showed an enlarged liver (17.4 cm) with increased echogenicity suggestive of steatosis or inflammation, without focal lesions. The gallbladder was contracted with sludge and mild pericholecystic edema, but no cholelithiasis or biliary dilatation. The portal vein was patent with normal hepatopetal flow (0.9 cm), and no features of Budd–Chiari syndrome or portal hypertension were noted. The spleen, pancreas, kidneys, and urinary bladder were unremarkable. The spleen was normal in size, and there was no ascites. These imaging results suggested an acute hepatocellular injury without signs of chronic liver disease or vascular pathology.
In our patient, abdominal ultrasound showed a mildly enlarged, echogenic liver, but normal portal vein flow and patent hepatic vessels, as shown above.13] . This supported the absence of vascular causes (e.g., portal vein thrombosis) in this case of acute hepatitis.
Despite supportive therapy including intravenous fluids, antiemetics, and N-acetylcysteine infusion, the patient’s liver function deteriorated. She developed worsening coagulopathy and rising bilirubin, meeting criteria for acute liver failure. Given the rapid progression and synthetic dysfunction, the transplant team was urgently consulted. She underwent orthotopic liver transplantation within days of admission.
Histological examination of the explanted liver demonstrated extensive centrilobular (zone 3) hepatocellular necrosis with bridging necrosis and lymphocytic infiltration, consistent with drug-induced liver injury. There was no significant fibrosis, bile duct injury, or chronic liver pathology. No eosinophilia or viral inclusions were observed. The histopathological findings, in the setting of repeated recent methoxyflurane exposure and exclusion of alternative causes, confirmed a diagnosis of methoxyflurane-induced acute liver failure.
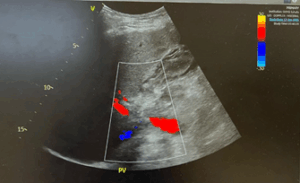
Figure 1: Illustrates the patient’s liver imaging findings. Abdominal ultrasound revealed mild hepatomegaly with diffusely increased echogenicity of the liver parenchyma (consistent with acute inflammation).
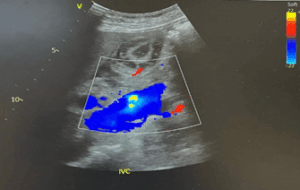
Figure 2: Doppler ultrasound of the portal vein demonstrated normal hepatopetal flow with no evidence of portal vein thrombosis or outflow obstruction.
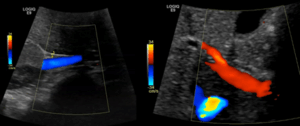
Figure 3: Doppler ultrasound of the portal vein, demonstrating normal hepatopetal blood flow (flow toward the liver indicated by color Doppler).
3.1. Mechanism of Methoxyflurane Hepatotoxicity
The exact mechanism by which methoxyflurane can cause liver injury is not fully elucidated, but it is believed to be idiosyncratic and likely immune-mediated. Halogenated anesthetics as a class are known to cause two forms of hepatic injury: a mild, transient elevation of liver enzymes (Type 1) and a rare, severe hepatotoxic reaction (Type 2) characterized by massive hepatic necrosis.14 Type 2 injury, exemplified by halothane hepatitis, is thought to result from an immune response to reactive metabolites of the anesthetic. Halothane is metabolized by cytochrome P450 2E1 to produce trifluoroacetylated protein adducts in hepatocytes; in susceptible individuals, an immune reaction (with antibodies against these adducts) leads to fulminant hepatitis.10 Methoxyflurane shares some similarities and differences. It undergoes extensive hepatic biotransformation (approximately 50% of absorbed dose) via O-demethylation, yielding metabolites such as dichloroacetic acid (DCAA), methoxyfluoroacetic acid, and inorganic fluoride.15, 16 These metabolites are well known to cause renal toxicity. The role of these or other metabolites in hepatotoxicity is less clear. Importantly, methoxyflurane’s metabolites are not trifluoroacetylated (the compound has a difluorodichloromethyl structure..]), so the classic halothane mechanism via trifluoroacetyl-protein adducts may not fully apply. However, methoxyflurane could still cause immune-mediated liver injury through alternative hapten formation. Some authors have suggested that immune mechanisms (similar to halothane) are involved in methoxyflurane hepatotoxicity.18], given the pattern of injury on re-exposure and the presence of features like fever or rash in some halogenated anesthetic hepatotoxicity cases. In our patient, the injury occurred after multiple exposures, which supports a potential immune sensitization (the first dose priming the immune system, subsequent doses triggering hepatitis). That said, direct toxicity from metabolites cannot be ruled out—methoxyflurane’s unique production of DCAA (not produced by other volatiles) might contribute to hepatocyte necrosis.19,20 Thus, methoxyflurane-induced hepatitis may result from a combination of metabolic and immune-allergic mechanisms, and more research would be needed to delineate this.
3.2. Differential diagnosis
The diagnosis of methoxyflurane-induced hepatotoxicity is one of exclusion. Our patient’s workup systematically ruled out viral hepatitis (A, B, C, EBV, CMV, HSV, and indirectly hepatitis E), autoimmune hepatitis, metabolic liver diseases (such as Wilson’s disease and hemochromatosis), and other drug-related causes (acetaminophen level was undetectable, and no other drugs were taken). The timing of liver injury onset shortly after repeated methoxyflurane exposure, along with progressive clinical deterioration despite cessation, strongly supports a causal relationship. The sclerotherapy agent, sodium tetradecyl sulfate, was considered but is known to have only local effects (chemical phlebitis) and has not been associated with systemic hepatotoxicity. Moreover, it has been used safely in numerous other patients without similar adverse effects, whereas methoxyflurane was the distinct new variable in this case. Ischemic hepatitis was excluded based on the absence of hypotension or hypoxia and imaging showing preserved hepatic blood flow. Biliary obstruction was ruled out by ultrasound, which showed no ductal dilatation or intrahepatic abnormalities. Therefore, an idiosyncratic drug-induced liver injury remains the most likely diagnosis. This case highlights the importance of thorough medication history-taking, including the use of inhalational agents—often overlooked—when evaluating unexplained acute liver injury.21
3.3. Review of Reported Cases
Severe hepatotoxicity from methoxyflurane was primarily documented during the era in which it was used as a general anesthetic. The first published case appeared in 1966, when Klein and Jeffries described acute liver failure following methoxyflurane anesthesia.20 Over the following decade, multiple case reports and small series confirmed that methoxyflurane could, albeit rarely, cause a halothane-like hepatitis syndrome. Lischner et al. (1967) reported fatal hepatic necrosis in a postoperative patient possibly related to intraoperative methoxyflurane use.22 Stefanini and colleagues (1970) documented fatal massive hepatic necrosis after repeated methoxyflurane anesthesia within a short interval.23 Brenner and Kaplan (1971) described recurrent acute hepatitis upon re-exposure, strongly implicating methoxyflurane through a positive rechallenge.24 In 1975, Rubinger et al. reported acute hepatitis following the use of methoxyflurane for obstetric analgesia—an early warning that even subanesthetic doses could pose risk.25 By 1977, the association had gained sufficient recognition that Min et al. published a case report explicitly titled "Methoxyflurane hepatitis," detailing liver failure attributed to the drug.26
In total, approximately 6–7 well-documented cases of severe methoxyflurane hepatotoxicity were published between 1966 and 1977, with at least three fatalities. Common features included female sex, multiple exposures, and a clinical course resembling halothane hepatitis, typically manifesting with jaundice several days after exposure. These reports contributed to growing safety concerns and ultimately to methoxyflurane’s withdrawal from anesthetic use.
After the late 1970s, with methoxyflurane no longer used as an anesthetic, reports of hepatotoxicity became exceedingly rare. In the modern era, methoxyflurane has been reintroduced in subanesthetic doses for analgesia. To our knowledge, only a few contemporary cases have been reported. One such case, described by O’Rourke et al. (2011) in Australia, involved a middle-aged woman who developed acute hepatitis after repeated inhaled methoxyflurane for procedural analgesia.21 She had exclusion of other causes and eventually recovered with supportive care. That report emphasized repeated subanesthetic exposure as the likely trigger and cautioned against frequent use.
Our case reinforces these concerns. Unlike the prior case, our patient developed fulminant liver failure requiring transplantation, highlighting that methoxyflurane hepatotoxicity can range in severity from self-limited hepatitis to life-threatening acute liver failure. The rarity of such events amidst millions of methoxyflurane uses underscores an extremely low incidence, but as with halothane, even rare immune-mediated reactions can result in catastrophic outcomes.
3.4. Clinical Lessons
This case illustrates that, despite a generally favorable safety profile, methoxyflurane can cause idiosyncratic acute hepatitis in rare instances. Repeated dosing within a short time frame may heighten this risk, potentially by triggering an immune-mediated reaction. While some previously reported cases resolved with supportive care alone, our patient progressed to fulminant hepatic failure requiring orthotopic liver transplantation—underscoring that outcomes can vary widely and, in some cases, may be life-threatening.
Clinicians using methoxyflurane for analgesia—particularly in outpatient or emergency settings—should be aware of this rare but serious complication. It would be prudent to avoid frequent, closely spaced dosing in the same patient and to counsel individuals to promptly report symptoms such as malaise, jaundice, or abdominal pain following methoxyflurane use. Importantly, when evaluating a patient with acute hepatitis of unclear origin, a detailed medication history should include inquiry about inhalational agents, which are often overlooked. As this case demonstrates, the causative agent may not be a conventional oral or intravenous medication, but an inhaled compound used for procedural analgesia. The report of this case has some limitations. Being an observation in a single patient, it does not define whether it can ascertain the causality as well as generalize the risk estimates of methoxyflurane-induced hepatotoxicity. Exclusion of common triggers of acute liver failure by an extensive diagnostic workup notwithstanding, the idiosyncratic nature of drug-induced liver injury precludes definite causation. In addition to this, there was the lack of measurement of any serum levels of methoxyflurane or even the metabolites thereof; it could have been a source of objective confirmation of exposure and toxicosis. The second major limitation is that it does not discuss other analgesics, i.e., ketamine, which, although effective, has neuropsychiatric side effects, including hallucinations, dysphoria, and emergence reactions. Such risks are most applicable in outpatient/self-administered settings and should be considered during the evaluation of the safety profiles of analgesic alternatives.
Methoxyflurane, a potent inhaled analgesic, is generally considered safe at subanesthetic doses; however, it can rarely induce severe hepatotoxicity, particularly with repeated exposure. We described a case of methoxyflurane-induced acute liver failure following one procedural administration and two subsequent self-administered doses over a short interval. The patient’s presentation, rapid clinical deterioration, and need for liver transplantation underscore the importance of considering volatile anesthetic exposure in cases of unexplained acute liver injury.
Although idiosyncratic, this form of hepatotoxicity shares clinical and pathological features with halothane hepatitis, likely involving an immune-mediated mechanism triggered by reactive drug metabolites. Management is primarily supportive, with prompt discontinuation of the offending agent and close monitoring. N-acetylcysteine may be beneficial in the early stages of liver injury, and liver transplantation remains the definitive intervention in cases of acute liver failure.
5. Recommendations
Clinicians should use methoxyflurane judiciously, avoid frequent or unsupervised repeat dosing when possible, and counsel patients to report any signs of hepatic injury, such as jaundice, fatigue, or abdominal pain. Ultimately, awareness of this rare but serious adverse effect can facilitate early recognition and timely intervention, helping ensure patient safety while preserving the analgesic benefits of methoxyflurane.
6. Conflict of interest
All authors declare that there was no conflict of interest.
7. Ethical considerations
The authors present this case report in the wider interest of the healthcare personnel as well as for the safety of the patients.
8. Authors’ contribution
Rayan A. Qutob is the sole author of this paper.
Author affiliation:
- Rayan A. Qutob, Department of Internal Medicine, College of Medicine, Imam Mohammad Ibn Saud Islamic University (IMSIU), Riyadh, Saudi Arabia; Email: Dr.Rayan.Qutob@gmail.com
ABSTRACT
Methoxyflurane is a halogenated ether previously used as a general anesthetic, now reintroduced in subanesthetic doses for analgesia. Although considered safe in low doses, historical data suggest the potential for rare but severe hepatotoxicity. We report a case of methoxyflurane-induced acute liver failure following repeated low-dose exposure, including unsupervised over-the-counter use, which necessitated liver transplantation.
A previously healthy 38-year-old woman with a history of migraine (managed with sumatriptan) and varicose veins developed progressive symptoms of acute hepatitis, including severe epigastric pain, jaundice, and persistent vomiting. These symptoms began three days after she self-administered a third dose of inhaled methoxyflurane, obtained over the counter for post-procedural pain. The initial dose had been administered under supervision during a varicose vein sclerotherapy session, while the subsequent two doses were taken one week apart without medical oversight.
On evaluation, laboratory investigations revealed marked hepatic dysfunction, including hyperbilirubinemia, elevated gamma glutamyl transpeptidase (GGT), and evidence of impaired synthetic function. Coagulopathy was evident, with a significantly prolonged INR. Additional findings included hypoalbuminemia and elevated inflammatory markers. Comprehensive workup excluded viral, autoimmune, and metabolic causes of liver injury. Despite supportive therapy with intravenous fluids and N-acetylcysteine, her liver function continued to decline, and she was referred for liver transplantation. She subsequently underwent orthotopic liver transplantation. Histopathological examination of the explanted liver demonstrated extensive centrilobular necrosis, consistent with drug-induced liver injury likely attributable to methoxyflurane. This case emphasizes that even subanesthetic analgesic doses—especially with repeated or unsupervised use—may trigger immune-mediated hepatic injury analogous to halothane hepatitis. Prompt recognition, exclusion of alternative causes, and early referral for transplantation are essential for optimal outcomes.
Abbreviations: ANA: antinuclear antibody, ASMA: anti-smooth muscle antibody, AMA: anti-mitochondrial antibody, GGT: gamma glutamyl transpeptidase, LKM: liver-kidney microsomal antibody.
Keywords: acute hepatitis; hepatic dysfunction; Methoxyflurane; Toxicity; Transplantation
Citation: Qutob RA. Methoxyflurane-induced hepatotoxicity following repeated analgesic use: a case report. Anaesth. pain intensive care 2025;29(8):1037-1043. DOI: 10.35975/apic.v29i8.2971
Received: June 17, 2025; Revised: July 22, 2025; Accepted: August 02, 2025
1. INTRODUCTION
Methoxyflurane is a fluorinated hydrocarbon anesthetic that was widely used in the 1960s for general anesthesia. It provided profound analgesia and muscle relaxation, but its high lipid solubility led to prolonged induction and emergence times, as well as extensive metabolism.1,2 Early reports in 1966 linked methoxyflurane to significant nephrotoxicity, characterized by high-output renal failure due to the release of inorganic fluoride.1 Subsequent studies in the 1970s confirmed dose-dependent kidney injury related to methoxyflurane metabolism.2 These safety concerns, along with the introduction of newer volatile anesthetics with more favorable pharmacologic profiles, led to methoxyflurane being withdrawn from anesthetic practice by the late 1970s.1
In addition to nephrotoxicity, methoxyflurane was implicated in rare cases of severe hepatotoxicity. The first published case of methoxyflurane-associated hepatitis appeared in 1966.4], and a 1974 review by Joshi and Conn coined the term “methoxyflurane-associated hepatitis,” suggesting an idiosyncratic, immune-mediated injury analogous to halothane hepatitis.3 However, as methoxyflurane fell out of favor as an anesthetic, discussion of its hepatotoxic potential waned in subsequent decades.
Methoxyflurane has experienced a resurgence since the 1990s in the form of a self-administered inhalational analgesic for short procedures and trauma-related pain. At subanesthetic doses (typically 3 mL per inhaler, marketed as Penthrox®), it provides rapid and effective pain relief with minimal sedation. Millions of administrations have been recorded in prehospital and outpatient settings, particularly in Australia, generally without serious complications. Randomized controlled trials (e.g., the STOP! study) have demonstrated the efficacy and acceptable safety of low-dose methoxyflurane in acute pain management.5 As a result, methoxyflurane has been approved in several countries for use in emergency analgesia.
Despite its favorable safety record in this context, concerns remain that methoxyflurane’s metabolism could, in rare instances, trigger hepatotoxicity—even at analgesic doses. Idiosyncratic liver injury from volatile anesthetics is best exemplified by halothane, which caused fulminant hepatic failure in approximately 1 in 6,000–20,000 exposures (fatal in ~1 in 35,000 single exposures, and up to 1 in 1,000 with repeated exposures).6–8 Susceptible individuals, often middle-aged women with prior exposures, would develop what is now known as “halothane hepatitis,” an immune-mediated hepatic necrosis with a mortality rate of around 50% once liver failure occurs.9 Newer agents such as enflurane, isoflurane, and desflurane undergo limited hepatic metabolism and have only a handful of documented hepatotoxic episodes. Sevoflurane, which undergoes minimal biotransformation into trifluoroacetylated proteins, is considered the safest in this regard.10
Methoxyflurane lies between these extremes. It undergoes extensive metabolism (up to 50–70%) and produces nephrotoxic metabolites such as dichloroacetic acid and inorganic fluoride, and possibly hepatotoxic compounds as well.11,12 Unlike halothane, methoxyflurane lacks a trifluoromethyl group and does not produce trifluoroacetylated protein adducts..], but early reports suggested it could still cause hepatic necrosis in select cases.
Here, we present a case of acute drug-induced liver injury (DILI) attributable to repeated low-dose methoxyflurane used for analgesia. To our knowledge, it is one of very few modern cases linking subanesthetic methoxyflurane use to severe hepatotoxicity. We outline the clinical course, explore the proposed mechanisms, review prior cases, and discuss the differential diagnosis, management—including the role of N-acetylcysteine—and how methoxyflurane compares to other halogenated anesthetics in terms of hepatotoxic risk. Recognizing such rare adverse events is crucial as methoxyflurane use expands globally.
2. CASE REPORT
A 38-year-old woman with a history of migraine (managed with sumatriptan) and no known liver disease presented to the emergency department with a five-day history of persistent vomiting, severe epigastric and right upper quadrant pain, and progressive jaundice. She denied alcohol use, herbal or traditional medicines, and recent travel. Three weeks earlier, she had undergone sclerotherapy for varicose veins, during which she received 3 mL of inhaled methoxyflurane (Penthrox®) for analgesia. Over the subsequent two weeks, she independently self-administered two additional doses of methoxyflurane via over-the-counter inhalers for post-procedural pain. Symptoms began three days following the last methoxyflurane exposure.
On examination, she appeared acutely ill, was alert and oriented, with stable vital signs: blood pressure 108/66 mmHg, heart rate 82 bpm, and temperature 36.7°C. She was icteric with dry mucous membranes and displayed epigastric and right upper quadrant tenderness. Murphy’s sign was positive. There was no hepatosplenomegaly, peripheral edema, or encephalopathy. Other systemic examinations were unremarkable.
Initial laboratory tests demonstrated acute liver injury and progressive hepatic synthetic failure: white blood cell count 16 ×10⁹/L; platelet count 60 ×10⁹/L; total bilirubin 98 µmol/L (increased from 73 µmol/L); direct bilirubin 77.3 µmol/L; alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were elevated (values unavailable); gamma-glutamyl transferase (GGT) was markedly elevated at 1160 U/L; alkaline phosphatase (ALP) was 265 U/L. Coagulation parameters were severely deranged, with prothrombin time (PT) of 48 seconds (up from 23), international normalized ratio (INR) of 4.06 (from 1.82), and activated partial thromboplastin time (aPTT) of 62 seconds (from 48). Serum albumin was low at 24 g/L. Serum creatinine was mildly elevated at 157 µmol/L. Inflammatory markers were significantly raised: C-reactive protein (CRP) 222 mg/L, procalcitonin 5.21 ng/mL, and lactate 4.5 mmol/L.
Extensive evaluation for causes of acute liver injury was unremarkable. Viral serologies for hepatitis A, B, C, D, and E were negative. HIV testing was negative. Autoimmune hepatitis panel, including antinuclear antibody (ANA), anti-smooth muscle antibody (ASMA), anti-mitochondrial antibody (AMA), and liver-kidney microsomal antibody (LKM) was all negative. Acetaminophen levels were undetectable. Metabolic tests, including ceruloplasmin, ferritin, and transferrin saturation, were within normal limits, making Wilson’s disease and hemochromatosis unlikely.
Abdominal ultrasound showed an enlarged liver (17.4 cm) with increased echogenicity suggestive of steatosis or inflammation, without focal lesions. The gallbladder was contracted with sludge and mild pericholecystic edema, but no cholelithiasis or biliary dilatation. The portal vein was patent with normal hepatopetal flow (0.9 cm), and no features of Budd–Chiari syndrome or portal hypertension were noted. The spleen, pancreas, kidneys, and urinary bladder were unremarkable. The spleen was normal in size, and there was no ascites. These imaging results suggested an acute hepatocellular injury without signs of chronic liver disease or vascular pathology.
In our patient, abdominal ultrasound showed a mildly enlarged, echogenic liver, but normal portal vein flow and patent hepatic vessels, as shown above.13] . This supported the absence of vascular causes (e.g., portal vein thrombosis) in this case of acute hepatitis.
Despite supportive therapy including intravenous fluids, antiemetics, and N-acetylcysteine infusion, the patient’s liver function deteriorated. She developed worsening coagulopathy and rising bilirubin, meeting criteria for acute liver failure. Given the rapid progression and synthetic dysfunction, the transplant team was urgently consulted. She underwent orthotopic liver transplantation within days of admission.
Histological examination of the explanted liver demonstrated extensive centrilobular (zone 3) hepatocellular necrosis with bridging necrosis and lymphocytic infiltration, consistent with drug-induced liver injury. There was no significant fibrosis, bile duct injury, or chronic liver pathology. No eosinophilia or viral inclusions were observed. The histopathological findings, in the setting of repeated recent methoxyflurane exposure and exclusion of alternative causes, confirmed a diagnosis of methoxyflurane-induced acute liver failure.

Figure 1: Illustrates the patient’s liver imaging findings. Abdominal ultrasound revealed mild hepatomegaly with diffusely increased echogenicity of the liver parenchyma (consistent with acute inflammation).

Figure 2: Doppler ultrasound of the portal vein demonstrated normal hepatopetal flow with no evidence of portal vein thrombosis or outflow obstruction.

Figure 3: Doppler ultrasound of the portal vein, demonstrating normal hepatopetal blood flow (flow toward the liver indicated by color Doppler).
3. DISCUSSION
3.1. Mechanism of Methoxyflurane Hepatotoxicity
The exact mechanism by which methoxyflurane can cause liver injury is not fully elucidated, but it is believed to be idiosyncratic and likely immune-mediated. Halogenated anesthetics as a class are known to cause two forms of hepatic injury: a mild, transient elevation of liver enzymes (Type 1) and a rare, severe hepatotoxic reaction (Type 2) characterized by massive hepatic necrosis.14 Type 2 injury, exemplified by halothane hepatitis, is thought to result from an immune response to reactive metabolites of the anesthetic. Halothane is metabolized by cytochrome P450 2E1 to produce trifluoroacetylated protein adducts in hepatocytes; in susceptible individuals, an immune reaction (with antibodies against these adducts) leads to fulminant hepatitis.10 Methoxyflurane shares some similarities and differences. It undergoes extensive hepatic biotransformation (approximately 50% of absorbed dose) via O-demethylation, yielding metabolites such as dichloroacetic acid (DCAA), methoxyfluoroacetic acid, and inorganic fluoride.15, 16 These metabolites are well known to cause renal toxicity. The role of these or other metabolites in hepatotoxicity is less clear. Importantly, methoxyflurane’s metabolites are not trifluoroacetylated (the compound has a difluorodichloromethyl structure..]), so the classic halothane mechanism via trifluoroacetyl-protein adducts may not fully apply. However, methoxyflurane could still cause immune-mediated liver injury through alternative hapten formation. Some authors have suggested that immune mechanisms (similar to halothane) are involved in methoxyflurane hepatotoxicity.18], given the pattern of injury on re-exposure and the presence of features like fever or rash in some halogenated anesthetic hepatotoxicity cases. In our patient, the injury occurred after multiple exposures, which supports a potential immune sensitization (the first dose priming the immune system, subsequent doses triggering hepatitis). That said, direct toxicity from metabolites cannot be ruled out—methoxyflurane’s unique production of DCAA (not produced by other volatiles) might contribute to hepatocyte necrosis.19,20 Thus, methoxyflurane-induced hepatitis may result from a combination of metabolic and immune-allergic mechanisms, and more research would be needed to delineate this.
3.2. Differential diagnosis
The diagnosis of methoxyflurane-induced hepatotoxicity is one of exclusion. Our patient’s workup systematically ruled out viral hepatitis (A, B, C, EBV, CMV, HSV, and indirectly hepatitis E), autoimmune hepatitis, metabolic liver diseases (such as Wilson’s disease and hemochromatosis), and other drug-related causes (acetaminophen level was undetectable, and no other drugs were taken). The timing of liver injury onset shortly after repeated methoxyflurane exposure, along with progressive clinical deterioration despite cessation, strongly supports a causal relationship. The sclerotherapy agent, sodium tetradecyl sulfate, was considered but is known to have only local effects (chemical phlebitis) and has not been associated with systemic hepatotoxicity. Moreover, it has been used safely in numerous other patients without similar adverse effects, whereas methoxyflurane was the distinct new variable in this case. Ischemic hepatitis was excluded based on the absence of hypotension or hypoxia and imaging showing preserved hepatic blood flow. Biliary obstruction was ruled out by ultrasound, which showed no ductal dilatation or intrahepatic abnormalities. Therefore, an idiosyncratic drug-induced liver injury remains the most likely diagnosis. This case highlights the importance of thorough medication history-taking, including the use of inhalational agents—often overlooked—when evaluating unexplained acute liver injury.21
3.3. Review of Reported Cases
Severe hepatotoxicity from methoxyflurane was primarily documented during the era in which it was used as a general anesthetic. The first published case appeared in 1966, when Klein and Jeffries described acute liver failure following methoxyflurane anesthesia.20 Over the following decade, multiple case reports and small series confirmed that methoxyflurane could, albeit rarely, cause a halothane-like hepatitis syndrome. Lischner et al. (1967) reported fatal hepatic necrosis in a postoperative patient possibly related to intraoperative methoxyflurane use.22 Stefanini and colleagues (1970) documented fatal massive hepatic necrosis after repeated methoxyflurane anesthesia within a short interval.23 Brenner and Kaplan (1971) described recurrent acute hepatitis upon re-exposure, strongly implicating methoxyflurane through a positive rechallenge.24 In 1975, Rubinger et al. reported acute hepatitis following the use of methoxyflurane for obstetric analgesia—an early warning that even subanesthetic doses could pose risk.25 By 1977, the association had gained sufficient recognition that Min et al. published a case report explicitly titled "Methoxyflurane hepatitis," detailing liver failure attributed to the drug.26
In total, approximately 6–7 well-documented cases of severe methoxyflurane hepatotoxicity were published between 1966 and 1977, with at least three fatalities. Common features included female sex, multiple exposures, and a clinical course resembling halothane hepatitis, typically manifesting with jaundice several days after exposure. These reports contributed to growing safety concerns and ultimately to methoxyflurane’s withdrawal from anesthetic use.
After the late 1970s, with methoxyflurane no longer used as an anesthetic, reports of hepatotoxicity became exceedingly rare. In the modern era, methoxyflurane has been reintroduced in subanesthetic doses for analgesia. To our knowledge, only a few contemporary cases have been reported. One such case, described by O’Rourke et al. (2011) in Australia, involved a middle-aged woman who developed acute hepatitis after repeated inhaled methoxyflurane for procedural analgesia.21 She had exclusion of other causes and eventually recovered with supportive care. That report emphasized repeated subanesthetic exposure as the likely trigger and cautioned against frequent use.
Our case reinforces these concerns. Unlike the prior case, our patient developed fulminant liver failure requiring transplantation, highlighting that methoxyflurane hepatotoxicity can range in severity from self-limited hepatitis to life-threatening acute liver failure. The rarity of such events amidst millions of methoxyflurane uses underscores an extremely low incidence, but as with halothane, even rare immune-mediated reactions can result in catastrophic outcomes.
3.4. Clinical Lessons
This case illustrates that, despite a generally favorable safety profile, methoxyflurane can cause idiosyncratic acute hepatitis in rare instances. Repeated dosing within a short time frame may heighten this risk, potentially by triggering an immune-mediated reaction. While some previously reported cases resolved with supportive care alone, our patient progressed to fulminant hepatic failure requiring orthotopic liver transplantation—underscoring that outcomes can vary widely and, in some cases, may be life-threatening.
Clinicians using methoxyflurane for analgesia—particularly in outpatient or emergency settings—should be aware of this rare but serious complication. It would be prudent to avoid frequent, closely spaced dosing in the same patient and to counsel individuals to promptly report symptoms such as malaise, jaundice, or abdominal pain following methoxyflurane use. Importantly, when evaluating a patient with acute hepatitis of unclear origin, a detailed medication history should include inquiry about inhalational agents, which are often overlooked. As this case demonstrates, the causative agent may not be a conventional oral or intravenous medication, but an inhaled compound used for procedural analgesia. The report of this case has some limitations. Being an observation in a single patient, it does not define whether it can ascertain the causality as well as generalize the risk estimates of methoxyflurane-induced hepatotoxicity. Exclusion of common triggers of acute liver failure by an extensive diagnostic workup notwithstanding, the idiosyncratic nature of drug-induced liver injury precludes definite causation. In addition to this, there was the lack of measurement of any serum levels of methoxyflurane or even the metabolites thereof; it could have been a source of objective confirmation of exposure and toxicosis. The second major limitation is that it does not discuss other analgesics, i.e., ketamine, which, although effective, has neuropsychiatric side effects, including hallucinations, dysphoria, and emergence reactions. Such risks are most applicable in outpatient/self-administered settings and should be considered during the evaluation of the safety profiles of analgesic alternatives.
4. CONCLUSION
Methoxyflurane, a potent inhaled analgesic, is generally considered safe at subanesthetic doses; however, it can rarely induce severe hepatotoxicity, particularly with repeated exposure. We described a case of methoxyflurane-induced acute liver failure following one procedural administration and two subsequent self-administered doses over a short interval. The patient’s presentation, rapid clinical deterioration, and need for liver transplantation underscore the importance of considering volatile anesthetic exposure in cases of unexplained acute liver injury.
Although idiosyncratic, this form of hepatotoxicity shares clinical and pathological features with halothane hepatitis, likely involving an immune-mediated mechanism triggered by reactive drug metabolites. Management is primarily supportive, with prompt discontinuation of the offending agent and close monitoring. N-acetylcysteine may be beneficial in the early stages of liver injury, and liver transplantation remains the definitive intervention in cases of acute liver failure.
5. Recommendations
Clinicians should use methoxyflurane judiciously, avoid frequent or unsupervised repeat dosing when possible, and counsel patients to report any signs of hepatic injury, such as jaundice, fatigue, or abdominal pain. Ultimately, awareness of this rare but serious adverse effect can facilitate early recognition and timely intervention, helping ensure patient safety while preserving the analgesic benefits of methoxyflurane.
6. Conflict of interest
All authors declare that there was no conflict of interest.
7. Ethical considerations
The authors present this case report in the wider interest of the healthcare personnel as well as for the safety of the patients.
8. Authors’ contribution
Rayan A. Qutob is the sole author of this paper.
9. REFERENCES
- Crandell WB, Pappas SG, Macdonald A. Nephrotoxicity associated with methoxyflurane anesthesia. Anesthesiology. 1966 Sep-Oct;27(5):591-607. DOI: DOI: 10.1097/00000542-196609000-00010. PMID: 5918999.
- Mazze RI, Trudell JR, Cousins MJ. Methoxyflurane metabolism and renal dysfunction: clinical correlation in man. Anesthesiology. 1971 Sep;35(3):247-52. DOI: 10.1097/00000542-197109000-00004 . PMID: 5095537.
- Joshi PH, Conn HO. The syndrome of methoxyflurane-associated hepatitis. Ann Intern Med. 1974;80(3):395–401. DOI: 10.7326/0003-4819-80-3-395 .
- Klein NC, Jeffries GH. Hepatotoxicity after methoxyflurane administration. JAMA 1966;197(12):1037-9. DOI:10.1001/jama.1966.03110120143040
- Coffey F, Wright J, Hartshorn S, Hunt P, Locker T, Mirza K, et al. STOP!: a randomised, double-blind, placebo-controlled study of the efficacy and safety of methoxyflurane for the treatment of acute pain. Emerg Med J. 2014;31(8):613–8. PMCID: PMC4112448 DOI: 10.1136/emermed-2013-202909
- Brown BR Jr. Halothane hepatitis revisited. N Engl J Med. 1985 Nov 21;313(21):1347-8. DOI: 10.1056/NEJM198511213132108 . PMID: 4058525.
- Böttiger LE, Dalén E, Hallén B. Halothane-induced liver damage: an analysis of the material reported to the Swedish Adverse Drug Reaction Committee, 1966-1973. Acta Anaesthesiol Scand. 1976;20(1):40-6. DOI: 10.1111/j.1399-6576.1976.tb05007.x PMID: 1266555.
- Inman WH, Mushin WW. Jaundice after repeated exposure to halothane: an analysis of Reports to the Committee on Safety of Medicines. Br Med J. 1974 Jan 5;1(5896):5-10. PMCID: PMC1632840 DOI: 10.1136/bmj.1.5896.5
- Bunker JP. Final Report of the National Halothane Study. Anesthesiology. 1968 Mar-Apr;29(2):231-2. DOI: 10.1097/00000542-196803000-00001 . PMID: 5635878.
- Motavaf M. Hepatotoxicity of Halogenated Inhalational Anesthetics. Iranian Red Crescent Medical Journal. 2014;
- Finkelstein S, Oliogu E, Yee A, et al. Literature review on the use of methoxyflurane in the management of pain in cancer-related procedures. Support Care Cancer. 2023;31:232. doi:10.1007/s00520-023-07694-7.
- Jones NO. Methoxyflurane nephrotoxicity—a review and a case report. Can Anaes Soc J 1972;19:152-9. DOI:10.1007/BF03005045
- Sonographic Tendencies. Portal vein Doppler [Internet]. 2021 Oct 1. Available from: https://sonographictendencies.com/2021/10/01/portal-vein-doppler/
- Gyorfi MJ, Kim PY. Halothane Toxicity. [Updated 2023 Apr 10]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK545281/
- Mazze RI. Methoxyflurane nephropathy. Environ Health Perspect 1976;15:111. PMCID: PMC1475154 DOI: 10.1289/ehp.7615111
- National Prescribing Service. Methoxyflurane (Penthrox) for analgesia (doctor’s bag listing) [Internet]. Canberra (Australia): National Prescribing Service, Department of Health and Ageing; 2010 [archived 2011 Jul 27; cited 2011 Jun 12].
- National Center for Biotechnology Information. Methoxyflurane [Internet]. Bethesda (MD): National Library of Medicine (US); 2004- [cited 2025 Mar 27]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/Methoxyflurane
- Habibollahi P, Mahboobi N, Esmaeili S, Safari S, Dabbagh A, Alavian SM. Halothane-induced hepatitis: A forgotten issue in developing countries: Halothane-induced hepatitis. Hepat Mon. 2011 Jan;11(1):3-6. PMID: 22087107; PMCID: PMC3206652
- Gottlieb LS, Trey C. The effects of fluorinated anesthetics on the liver and kidneys. Annu Rev Med 1974;25:411-29. DOI: 10.1146/annurev.me.25.020174.002211 . PMID: 4596236.
- Dayan AD. Analgesic use of inhaled methoxyflurane-Evaluation of its safety and efficacy. Hum Exp Toxicol. 2016;35(5):502-13.
- O'Rourke KM, McMaster S, Lust KMC. A case of hepatitis attributable to repeated exposure to methoxyflurane during its use for procedural analgesia. Med J Aust. 2011;194(8):423-424. DOI:10.5694/j.1326-5377.2011.tb03038.x
- Lischner MW, MacNabb GM, Galamhos JT. Fatal hepatic necrosis following surgery: possible relation to methoxyflurane anesthesia. Arch Intern Med 1967;120:724-8.
- Stefanini M, Herland A, Kosyak EP. Fatal massive necrosis of the liver after repeated exposure to methoxyflurane. Anesthesiology 1970;32:374-8. DOI: 10.1097/00000542-197004000-00019
- Brenner Al, Kaplan MM. Recurrent hepatitis due to methoxyflurane anesthesia. N Engl J Med 1971; 284: 961–2. DOI: 10.1056/NEJM197104292841707
- Rubinger D, Davidson JT, Melmed RN. Hepatitis following the use of methoxyflurane in obstetric analgesia. Anesthesiology 1975;43:593-5. DOI: 10.1097/00000542-197511000-00025
- Min KW, Cain GO, Sabel JS, Gyorkey F. Methoxyflurane hepatitis. South Med J 1977;70:1362-4. DOI: 10.1097/00007611-197711000-00037