Noorhan Ameer Khaleel 1 , Mourouge Saadi Abbas1 2 , Liqaa Yahya Mohsen 3
Authors affiliations:
1.Noorhan Ameer Khaleel, Department of Biology, College of Science, University of Babylon, Iraq; Email: Sci112.noorhan.ameer@student.uobabylon.edu.iq
Background & objective: Multidrug-resistant (MDR) Enterobacteriaceae, particularly Escherichia coli and Klebsiella pneumoniae, pose a critical global health threat owing to the limited therapeutic options. Increasing β-lactamase-mediated resistance requires local surveillance to inform treatment and control strategies.
This research was carried out to determine the prevalence, antibiotic resistance patterns, and biofilm production of MDR Enterobacteriaceae in patients with urinary tract infections (UTI) across healthcare facilities in Al-Hilla, Iraq.
Methodology: From September 2024 to April 2025, 215 bacterial isolates were collected from hospitalized and outpatient patients with known UTI in Al-Hilla. Species identification was performed using MacConkey agar, eosin methylene blue (EMB) agar, biochemical tests (IMViC), and the VITEK 2 system. Antibiotic susceptibility testing (16 antibiotics) was performed according to CLSI (2024) disk diffusion guidelines. Biofilm production was assessed using Congo red agar (CRA).
Results: The results of our research show that E. coli (53%, 114/215) and K. pneumoniae (27%, 60/215) were the predominant pathogens. High rates of resistance were observed for ampicillin (E. coli, 99.1%; K. pneumoniae, 98.3%) and ceftazidime (E. coli, 70.2%; K. pneumoniae, 75%). Carbapenems showed low resistance (imipenem: E. coli, 12.3%; K. pneumoniae 20%). Biofilm production occurred in 52.6% of E. coli isolates (strong/moderate: 42.1%) and 68.3% of K. pneumoniae isolates (strong/moderate: 53.3%).
Conclusion: MDR Enterobacteriaceae exhibiting significant β-lactam resistance and biofilm formation are prevalent in Al-Hilla UTI cases, underscoring a critical public health challenge. The correlation between biofilm production and MDR necessitates enhanced stewardship and the development of alternative therapeutic strategies.
Abbreviations: E. coli, Escherichia coli; K. pneumoniae, Klebsiella pneumoniae; MDR, multidrug resistance; ESBL, extended-spectrum β-lactamase; UTI, urinary tract infection.
Keywords: Enterobacteriaceae, Escherichia coli, Klebsiella pneumoniae, Multidrug resistance, Biofilm, Urinary tract infection.
Citation: Khaleel NA, Abbas MS, Mohsen LY. Prevalence and characterization of multidrug-resistant enterobacteriaceae from urinary tract infections in Al-Hilla, Iraq. Anaesth. pain intensive care 2025;29(7):712-218. DOI: 10.35975/apic.v29i7.2955
Received: July 16, 2025; Revised: August 04, 2025; Accepted: August 10, 2025
The Enterobacteriaceae family (phylum Proteobacteria) encompasses diverse gram-negative genera, such as Escherichia, Enterobacter, and Klebsiella. While typically present at low abundance in healthy human intestines,1 several species are prominent causes of difficult-to-treat multidrug-resistant (MDR) nosocomial infections.2 Therapeutic limitations often compel the use of broad-spectrum antibiotics against MDR Enterobacteriaceae.3 β-lactamase production is a key resistance mechanism in these bacteria, contributing significantly to the burden of community-onset and healthcare-associated infections, particularly urinary tract infections (UTIs).4 The alarming global increase in antimicrobial resistance (AMR) among gram-negative bacteria, with Enterobacteriaceae being a primary focus,5 necessitates localized epidemiological studies on β-lactamase prevalence. These investigations are crucial for determining effective empirical therapy and strengthening infection prevention and control protocols.6,7
Recent global surveillance data have revealed alarming trends in multidrug-resistant Enterobacteriaceae infections. In US hospitals from 2013 to 2017, 12.05% of over 1 million Enterobacteriaceae isolates were extended-spectrum β-lactamase (ESBL) producers, 1.21% were carbapenem-resistant, and 7.08% showed multidrug resistance. The rates of ESBL and carbapenem-resistant Enterobacteriaceae increased significantly during this period, with ESBL infections rising at an average slope of 0.0089 per quarter.4 This situation is even more concerning in developing countries. A study from rural southwestern Uganda showed that 19.2% of 2,371 patients with UTI symptoms tested positive for uropathogenic Enterobacteriaceae, 25.05% exhibited multidrug resistance, and 10.77% showed pan-drug resistance. These organisms showed the highest resistance to tetracycline (89.5%) and varying degrees of resistance to other antibiotics, including carbapenem-resistant strains.8
Members of the Enterobacteriaceae family are known to form biofilms, which contribute significantly to their clinical persistence and resistance. These structured assemblages, in which bacteria reside within an extracellular polymeric substance (EPS) matrix,9 demonstrate enhanced tolerance to antimicrobial drugs. The increasing prevalence of drug-resistant pathogens combined with biofilm-mediated protection substantially diminishes antibiotic effectiveness, thereby elevating the treatment complexity.10 This has necessitated the development of new therapeutic platforms effective against biofilm-associated infections.11 This study was conducted to assess the prevalence and diversity of β-lactamases expressed by urinary isolates of E. coli and Klebsiella pneumoniae from hospitals and health centers in Al-Hilla Province.
2.1. Isolation and cultivation of bacteria
This study was conducted between September 2024 and April 2025. A total of 215 urine samples were collected from 102 hospitalized and 72 non-hospitalized UTI patients in the selected health facilities of AL-Hilla, Babylon province, Iraq, Al-Hilla Teaching Hospital, Al-Hashimiya General Hospital, Al-Imam Al-Sadeq Hospital, Merjan Medical City for Internal Medicine and Cardiology, Babel Maternity and Children, and the Al-Kadhya Health Care Center. MacConkey agar and eosin methylene blue agar were prepared according to the manufacturer’s instructions and used as the culture media. Urine samples were cultured in these media and incubated at 35 ± 2ºC for 18 h. Different colonies were subcultured by streaking for purification, and the VITEK 2 system was used for further identification.
2.2. Morphological characterization
2.2.1. Microscopic examination
Microscopic examination was performed using a commercially prepared Gram staining kit (Schuchardt, Germany) to differentiate and observe morphological characteristics of the bacterial specimens.
2.2.2. Biochemical tests
The IMViC test consists of four different tests designed to differentiate gram-negative intestinal bacilli (Enterobacteriaceae). Indole production and methyl red, Voges-Proskauer, and Citrate utilization tests were performed according to the manufacturer’s instructions.
2.2.3. Antibiotic susceptibility test
Specimens were tested for 16 antibiotics using the disk diffusion method (Kirby-Bauer) according to the CLSI guidelines (CLSI, 2024). A sterile cotton swab was dipped into a standardized bacterial suspension adjusted to a McFarland turbidity standard of 0.5. The swab was used to evenly inoculate the entire surface of the Mueller-Hinton agar by streaking in three dimensions to ensure uniform lawn growth. After the surface was dried for 3–5 min, commercial antibiotic discs were aseptically placed on the agar using sterile forceps. Plates were incubated at 35-37°C for 18 h, and antibacterial activity was expressed as the mean inhibition zone diameter in millimeters (mm) (CLSI, 2024). The zone size was measured and classified as sensitive or resistant compared to the standard zone, according to CLSI.
2.2.4. Biofilm formation
All samples taken from hospitalized and non-hospitalized UTI patients were cultured on Congo red agar, and biofilm capacity was assessed using the Congo red agar (CRA) modified method.
A total of 215 specimens that have been collected from hospitalized and non-hospitalized UTI patients, (114/215) 53% of these specimens were identified as E. coli, and (60/215) 27% were identified as K. pneumoniae by culturing on EMB and MacConkey agar. On EMB agar, E. coli gave a green metallic sheen due to the vigorous lactose and/or sucrose fermentation ability of E. coli (4), while Klebsiella, a lactose fermenter, compared to E. coli, does not cause the same level of dye precipitation (less vigorously), so colonies appear pink to purple without green sheen (1) (Figure 1).

Figure 1: A: K. pneumoniae on macConkey agar, B: E. coli on macConkey agar, C: E. coli on EMB agar, D: K. pneumoniae on EMB agar
Biochemical characterization of the urine isolates was conducted using the IMViC test series, designed to distinguish members of the Enterobacteriaceae family, notably E. coli and Klebsiella pneumoniae. This series comprises of four assays: indole production, methyl red, Voges-Proskauer, and citrate utilization. The results indicated the presence of both E. coli and K. pneumoniae in the urine samples from the study. Indole Test: This assay detects tryptophan degradation. A positive result, indicated by red ring formation after adding Kovac's reagent, signifies the production of indole by tryptophanase. E. coli (indole-positive) showed this reaction, whereas K. pneumoniae did not. Methyl Red (MR) Test: This test identifies stable acid end-products from glucose fermentation. A positive MR result (red color) occurred at pH ≤ 4.4, which is characteristic of E. coli. In contrast, K. pneumoniae produces neutral end products (e.g., acetoin), maintaining a pH > 6.0, yielding a negative MR result (yellow-orange). Citrate Utilization Test (Simmons' citrate): This test determines an organism's ability to use citrate as its sole carbon source. K. pneumoniae demonstrated a positive result, as evidenced by bacterial growth and a color change of the medium from green to blue. E. coli was citrate-negative, showing no growth, and leaving the medium green. The combined IMViC profiles are listed in Table 1.
Antibiotic susceptibility tests were performed according to the method outlined by CLSI (CLSI, 2024). The results demonstrated that A significant proportion of the isolates exhibited multidrug resistance (MDR), defined as resistance to at least one agent in three or more antimicrobial categories (Figure 2).
Figure 2: Multidrug resistance pattern in E. coli and K. pneumoniae
All 114 E. coli isolates showed a high resistance rate to all antibiotics, except for imipenem and meropenem, which showed a resistance rate of less than 11%. For Klebsiella isolates, the resistance rates to the antibiotics used were also very high, except for imipenem and meropenem, which showed resistance rates of less than 20% (Table 2)) Figure 3).
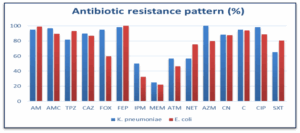
Figure 3: Antibiotic resistance pattern in E. coli and K. pneumoniae
The results from all samples cultured on Congo red agar were compared to the control samples. The biofilm production capability was evaluated for both E. coli and K. pneumoniae. Colonies exhibiting black pigmentation accompanied by a dry, crystalline texture were classified as strongly positive biofilm producers. Weak slime producers typically retain a pink color, although central darkening is occasionally noted. Colonies displaying darkening without characteristic dry crystalline morphology were categorized as indeterminate (Figure 4). The proportions of the producer and non-producer isolates are presented in Table 3.

Figure 4: Biofilm production, A: strong biofilm production, B: moderate biofilm production, C: weak biofilm production, D: non-biofilm producer
Beta-lactam antibiotics were found to have a high level of resistance. These findings indicate the widespread presence of MDR phenotypes among K. pneumoniae and E. coli strains, highlighting the critical need for ongoing surveillance and prudent antibiotic use. Moreover, a high percentage of biofilm production, especially in E. coli isolates, is clearly associated with repeated UTI cases. as biofilms help bacteria to survive longer and resist antibiotics. Obaid and Hasson (2021) reported that 46.6% of K. pneumoniae isolates were strong biofilm producers, 30.6% were non-producers, and the rest of the total number ranged between weak and moderate producers.12
Biofilm formation is a critical virulence factor that enhances antimicrobial resistance in Enterobacteriaceae. Studies have demonstrated that biofilm-producing strains show increased resistance to multiple antibiotics, with minimal inhibitory concentrations 10–10 times higher than those required for planktonic cells.13 Research on cancer patients with UTIs found that 62.7% of coliforms were biofilm formers, and biofilm formation was significantly higher in patients with urinary catheters (83.3%) and diabetes mellitus (77.8%).14
Studies in Iraq have revealed patterns of antimicrobial resistance in the Enterobacteriaceae. Research from Nasiriyah, Iraq, showed the presence of E. coli (44.71%) and K. pneumoniae (38.57%).15 A comprehensive surveillance study from Iraq analyzing 11,592 clinical records found that K. pneumoniae (30.1%) and E. coli were among the most common gram-negative bacteria in adults, with high rates of multidrug resistance.16 In the Al-Hillah River in Babylon Province, Iraq, E. coli isolates showed the highest resistance to β-lactam antibiotics, followed by fosfomycin, and aminoglycosides. This study revealed a high prevalence of multidrug resistance among E. coli isolates from all sampling sites, indicating widespread antimicrobial resistance in the environment.17
Franklin et al (2021) reported that 198 of 203 Klebsiella pneumoniae isolates (97.5%) were regarded as biofilm producers. And 83.3% of Klebsiella spp,18 were biofilm-forming in urine samples tested on Congo agar in Nepal.19 A study done by Kadhum et al. (2021) showed that all E. coli 33 isolates produced biofilms (100%), Out of 33 isolates, 14 were strong biofilm producers. Moderate biofilm-producing isolates were found in 8/33(24.24%) isolates, whereas weak biofilm-producing isolates were found in 11/33 (33.33%) isolates. All K. pneumoniae isolates (100%) produced biofilms. Of the isolates, 5/8 (62.5%) were moderate biofilm producers and the remaining 3/8 (37.5%) were weak biofilm producers.20
In parallel with our study, multiple studies have highlighted antibiotic resistance in urinary pathogens. High resistance rates in E. coli were observed by Jalil et al. (2022) for ceftriaxone (89%), ampicillin (86.6%), levofloxacin (82.9%), and cefotaxime (79.3%),21 whereas Iqbal et al. (2021) reported ≥90% resistance to cefixime and cefuroxime and ≥80% non-susceptibility to numerous other agents, including ciprofloxacin and co-trimoxazole.22 K. pneumoniae isolates in Jalil et al. (2022) exhibited strong resistance to piperacillin (81.6%), levofloxacin (78.9%), ampicillin (76.3%), and cefotaxime (73.7%).21 Both studies noted lower resistance to carbapenems (imipenem: 2.4% for E. coli and 13.2% for K. pneumoniae) and variable resistance to ciprofloxacin (35.4% for E. coli and 36.8% for K. pneumoniae) and cefepime (34.1% for E. coli and 50.0% for K. pneumoniae).21 This widespread resistance contributes to the high MDR prevalence, particularly in E. coli (53%), as reported by Shretha et al. (2021), along with significant MDR rates in E. faecalis (22%).23
ESBL production is one of the most significant resistance mechanisms in the Enterobacteriaceae. A comprehensive study from Isfahan, Iran, involving 1,564 isolates, found that among hospitalized patients, 41.7% of E. coli and 51.5% of K. pneumoniae isolates were ESBL producers. In outpatient samples, ESBL production rates were 25% and 40% for K. pneumoniae.5 This study also identified CTX-M-15 as the predominant β-lactamase enzyme type (79.3%) in community-onset UTIs caused by ESBL-producing K. pneumoniae.24 Research from Northern Thailand demonstrated that approximately 42.5% of E. coli and 30.2% of K. pneumoniae isolates were classified as ESBL producers.25 The prevalence of ESBL increased significantly between 2004 and 2008, plateauing at approximately 30-40% per year, with recurrent UTI episodes showing significantly higher ESBL rates (46.5%) than the first episodes (27.3%).26
Antibiotic resistance rates among bacteria causing UTIs demonstrate considerable inter-study variation attributable to differences in patient demographics and geographic origin. The escalation of resistance is strongly linked to the indiscriminate consumption of antibiotics, frequently without prescriptions. This practice selects resistant mutants that may acquire traits such as reduced drug permeability and enhanced survival. Empirical evidence has confirmed that the resistance profiles of predominant uropathogens have shifted temporally in multiple countries.27
The present study demonstrated a high prevalence of MDR E. coli and Klebsiella pneumoniae among patients with UTI in Al-Hilla, Iraq. A significant proportion of isolates exhibited extensive resistance to β-lactam antibiotics (e.g., ampicillin and ceftazidime) and other antimicrobial classes, although carbapenems (imipenem and meropenem) retained relatively higher efficacy. Notably, biofilm production was observed in E. coli and K. pneumoniae isolates, correlating with enhanced antimicrobial resistance and potential for recurrent infections.
6. Data availability
The numerical data obtained on this research are available from the corresponding author.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
No external or industry funding was involved in the conduct of this study. The authors used only institutional resources in the conduct of the study.
9. Authors’ contribution
Conceptualization, Methodology, Formal Analysis, Writing: NAK
Validation, Investigation, Visualization, Project Administration: MSA
Software, Resources: LYM
Authors affiliations:
1.Noorhan Ameer Khaleel, Department of Biology, College of Science, University of Babylon, Iraq; Email: Sci112.noorhan.ameer@student.uobabylon.edu.iq
- Mourouge Saadi Abbas, Department of Biology, College of Science, University of Babylon, Iraq; Email: Sci.mourouge.saadi@uobabylon.edu.iq
- Liqaa Yahya Mohsen, Department of Biology, College of Science, University of Babylon, Iraq; Email: sci.liqaa.mohsen@uobabylon.edu.iq
ABSTRACT
Background & objective: Multidrug-resistant (MDR) Enterobacteriaceae, particularly Escherichia coli and Klebsiella pneumoniae, pose a critical global health threat owing to the limited therapeutic options. Increasing β-lactamase-mediated resistance requires local surveillance to inform treatment and control strategies.
This research was carried out to determine the prevalence, antibiotic resistance patterns, and biofilm production of MDR Enterobacteriaceae in patients with urinary tract infections (UTI) across healthcare facilities in Al-Hilla, Iraq.
Methodology: From September 2024 to April 2025, 215 bacterial isolates were collected from hospitalized and outpatient patients with known UTI in Al-Hilla. Species identification was performed using MacConkey agar, eosin methylene blue (EMB) agar, biochemical tests (IMViC), and the VITEK 2 system. Antibiotic susceptibility testing (16 antibiotics) was performed according to CLSI (2024) disk diffusion guidelines. Biofilm production was assessed using Congo red agar (CRA).
Results: The results of our research show that E. coli (53%, 114/215) and K. pneumoniae (27%, 60/215) were the predominant pathogens. High rates of resistance were observed for ampicillin (E. coli, 99.1%; K. pneumoniae, 98.3%) and ceftazidime (E. coli, 70.2%; K. pneumoniae, 75%). Carbapenems showed low resistance (imipenem: E. coli, 12.3%; K. pneumoniae 20%). Biofilm production occurred in 52.6% of E. coli isolates (strong/moderate: 42.1%) and 68.3% of K. pneumoniae isolates (strong/moderate: 53.3%).
Conclusion: MDR Enterobacteriaceae exhibiting significant β-lactam resistance and biofilm formation are prevalent in Al-Hilla UTI cases, underscoring a critical public health challenge. The correlation between biofilm production and MDR necessitates enhanced stewardship and the development of alternative therapeutic strategies.
Abbreviations: E. coli, Escherichia coli; K. pneumoniae, Klebsiella pneumoniae; MDR, multidrug resistance; ESBL, extended-spectrum β-lactamase; UTI, urinary tract infection.
Keywords: Enterobacteriaceae, Escherichia coli, Klebsiella pneumoniae, Multidrug resistance, Biofilm, Urinary tract infection.
Citation: Khaleel NA, Abbas MS, Mohsen LY. Prevalence and characterization of multidrug-resistant enterobacteriaceae from urinary tract infections in Al-Hilla, Iraq. Anaesth. pain intensive care 2025;29(7):712-218. DOI: 10.35975/apic.v29i7.2955
Received: July 16, 2025; Revised: August 04, 2025; Accepted: August 10, 2025
1. INTRODUCTION
The Enterobacteriaceae family (phylum Proteobacteria) encompasses diverse gram-negative genera, such as Escherichia, Enterobacter, and Klebsiella. While typically present at low abundance in healthy human intestines,1 several species are prominent causes of difficult-to-treat multidrug-resistant (MDR) nosocomial infections.2 Therapeutic limitations often compel the use of broad-spectrum antibiotics against MDR Enterobacteriaceae.3 β-lactamase production is a key resistance mechanism in these bacteria, contributing significantly to the burden of community-onset and healthcare-associated infections, particularly urinary tract infections (UTIs).4 The alarming global increase in antimicrobial resistance (AMR) among gram-negative bacteria, with Enterobacteriaceae being a primary focus,5 necessitates localized epidemiological studies on β-lactamase prevalence. These investigations are crucial for determining effective empirical therapy and strengthening infection prevention and control protocols.6,7
Recent global surveillance data have revealed alarming trends in multidrug-resistant Enterobacteriaceae infections. In US hospitals from 2013 to 2017, 12.05% of over 1 million Enterobacteriaceae isolates were extended-spectrum β-lactamase (ESBL) producers, 1.21% were carbapenem-resistant, and 7.08% showed multidrug resistance. The rates of ESBL and carbapenem-resistant Enterobacteriaceae increased significantly during this period, with ESBL infections rising at an average slope of 0.0089 per quarter.4 This situation is even more concerning in developing countries. A study from rural southwestern Uganda showed that 19.2% of 2,371 patients with UTI symptoms tested positive for uropathogenic Enterobacteriaceae, 25.05% exhibited multidrug resistance, and 10.77% showed pan-drug resistance. These organisms showed the highest resistance to tetracycline (89.5%) and varying degrees of resistance to other antibiotics, including carbapenem-resistant strains.8
Members of the Enterobacteriaceae family are known to form biofilms, which contribute significantly to their clinical persistence and resistance. These structured assemblages, in which bacteria reside within an extracellular polymeric substance (EPS) matrix,9 demonstrate enhanced tolerance to antimicrobial drugs. The increasing prevalence of drug-resistant pathogens combined with biofilm-mediated protection substantially diminishes antibiotic effectiveness, thereby elevating the treatment complexity.10 This has necessitated the development of new therapeutic platforms effective against biofilm-associated infections.11 This study was conducted to assess the prevalence and diversity of β-lactamases expressed by urinary isolates of E. coli and Klebsiella pneumoniae from hospitals and health centers in Al-Hilla Province.
2. METHODOLOGY
2.1. Isolation and cultivation of bacteria
This study was conducted between September 2024 and April 2025. A total of 215 urine samples were collected from 102 hospitalized and 72 non-hospitalized UTI patients in the selected health facilities of AL-Hilla, Babylon province, Iraq, Al-Hilla Teaching Hospital, Al-Hashimiya General Hospital, Al-Imam Al-Sadeq Hospital, Merjan Medical City for Internal Medicine and Cardiology, Babel Maternity and Children, and the Al-Kadhya Health Care Center. MacConkey agar and eosin methylene blue agar were prepared according to the manufacturer’s instructions and used as the culture media. Urine samples were cultured in these media and incubated at 35 ± 2ºC for 18 h. Different colonies were subcultured by streaking for purification, and the VITEK 2 system was used for further identification.
2.2. Morphological characterization
2.2.1. Microscopic examination
Microscopic examination was performed using a commercially prepared Gram staining kit (Schuchardt, Germany) to differentiate and observe morphological characteristics of the bacterial specimens.
2.2.2. Biochemical tests
The IMViC test consists of four different tests designed to differentiate gram-negative intestinal bacilli (Enterobacteriaceae). Indole production and methyl red, Voges-Proskauer, and Citrate utilization tests were performed according to the manufacturer’s instructions.
2.2.3. Antibiotic susceptibility test
Specimens were tested for 16 antibiotics using the disk diffusion method (Kirby-Bauer) according to the CLSI guidelines (CLSI, 2024). A sterile cotton swab was dipped into a standardized bacterial suspension adjusted to a McFarland turbidity standard of 0.5. The swab was used to evenly inoculate the entire surface of the Mueller-Hinton agar by streaking in three dimensions to ensure uniform lawn growth. After the surface was dried for 3–5 min, commercial antibiotic discs were aseptically placed on the agar using sterile forceps. Plates were incubated at 35-37°C for 18 h, and antibacterial activity was expressed as the mean inhibition zone diameter in millimeters (mm) (CLSI, 2024). The zone size was measured and classified as sensitive or resistant compared to the standard zone, according to CLSI.
2.2.4. Biofilm formation
All samples taken from hospitalized and non-hospitalized UTI patients were cultured on Congo red agar, and biofilm capacity was assessed using the Congo red agar (CRA) modified method.
3. RESULTS
A total of 215 specimens that have been collected from hospitalized and non-hospitalized UTI patients, (114/215) 53% of these specimens were identified as E. coli, and (60/215) 27% were identified as K. pneumoniae by culturing on EMB and MacConkey agar. On EMB agar, E. coli gave a green metallic sheen due to the vigorous lactose and/or sucrose fermentation ability of E. coli (4), while Klebsiella, a lactose fermenter, compared to E. coli, does not cause the same level of dye precipitation (less vigorously), so colonies appear pink to purple without green sheen (1) (Figure 1).

Figure 1: A: K. pneumoniae on macConkey agar, B: E. coli on macConkey agar, C: E. coli on EMB agar, D: K. pneumoniae on EMB agar
Biochemical characterization of the urine isolates was conducted using the IMViC test series, designed to distinguish members of the Enterobacteriaceae family, notably E. coli and Klebsiella pneumoniae. This series comprises of four assays: indole production, methyl red, Voges-Proskauer, and citrate utilization. The results indicated the presence of both E. coli and K. pneumoniae in the urine samples from the study. Indole Test: This assay detects tryptophan degradation. A positive result, indicated by red ring formation after adding Kovac's reagent, signifies the production of indole by tryptophanase. E. coli (indole-positive) showed this reaction, whereas K. pneumoniae did not. Methyl Red (MR) Test: This test identifies stable acid end-products from glucose fermentation. A positive MR result (red color) occurred at pH ≤ 4.4, which is characteristic of E. coli. In contrast, K. pneumoniae produces neutral end products (e.g., acetoin), maintaining a pH > 6.0, yielding a negative MR result (yellow-orange). Citrate Utilization Test (Simmons' citrate): This test determines an organism's ability to use citrate as its sole carbon source. K. pneumoniae demonstrated a positive result, as evidenced by bacterial growth and a color change of the medium from green to blue. E. coli was citrate-negative, showing no growth, and leaving the medium green. The combined IMViC profiles are listed in Table 1.
| Table 1: Biochemical test for E. coli and K. pneumonia | ||
| Biochemical test | E. coli result | K. pneumoniae result |
| Indole test | Positive | Negative |
| Methyl red (MR) | Positive | Negative |
| Simmon’s citrate | Negative | Positive |
| Voges-proskaure (VP) | Positive | |
Antibiotic susceptibility tests were performed according to the method outlined by CLSI (CLSI, 2024). The results demonstrated that A significant proportion of the isolates exhibited multidrug resistance (MDR), defined as resistance to at least one agent in three or more antimicrobial categories (Figure 2).

Figure 2: Multidrug resistance pattern in E. coli and K. pneumoniae
All 114 E. coli isolates showed a high resistance rate to all antibiotics, except for imipenem and meropenem, which showed a resistance rate of less than 11%. For Klebsiella isolates, the resistance rates to the antibiotics used were also very high, except for imipenem and meropenem, which showed resistance rates of less than 20% (Table 2)) Figure 3).
| Table 2: Antibiotic resistance profiles of E. coli and K. pneumoniae isolates | ||
|
Antibiotics |
Resistant E. coli isolates
[n (%)] |
Resistant K. pneumoniae
Isolates [n (%)] |
| Cefoxitin (FOX) | 61 (53.5) | 42 (70) |
| Ceftazidim (CAZ) | 80 (70.17) | 45 (75) |
| Ampicillin (AM) | 113 (99.1) | 59 (98.33) |
| Pipracillin- tazobactam (TPZ) | 103 90.35) | 29 (48.33) |
| Azithromycin (azm) | 93 (81.57) | 52 (86.66) |
| Meropneme (MEM) | 13 (11.40) | 11 (18.33) |
| Imipneme (IPM) | 14 (12.28) | 12 (20) |
| Trimethoprim-sulfamethoxazole (SXT) | 55 (48.24) | 29 (48.33) |
| Amoxicillin-clavulanate (AMC) | 71 (62.28) | 47 (78.33) |
| Cefepime (FEB) | 103 (90.35) | 28 (46.66) |
| Netlimicin (NET) | 33 (28.94) | 24 (0) |
| Aztreonam (ATM) | 23 (20.17) | 22 (36.66) |
| Ciprofloxacin (CIP) | 75 (65.78) | 46 (76.66) |
| Chloramphenicol (C) | 77 (67.54) | 35 (58.33) |
| Gentamicin (CN) | 68 (59.64) | 38 (63.33) |

Figure 3: Antibiotic resistance pattern in E. coli and K. pneumoniae
The results from all samples cultured on Congo red agar were compared to the control samples. The biofilm production capability was evaluated for both E. coli and K. pneumoniae. Colonies exhibiting black pigmentation accompanied by a dry, crystalline texture were classified as strongly positive biofilm producers. Weak slime producers typically retain a pink color, although central darkening is occasionally noted. Colonies displaying darkening without characteristic dry crystalline morphology were categorized as indeterminate (Figure 4). The proportions of the producer and non-producer isolates are presented in Table 3.

Figure 4: Biofilm production, A: strong biofilm production, B: moderate biofilm production, C: weak biofilm production, D: non-biofilm producer
| Table 3: The proportions of the producer and non-producer isolates | ||
| Characteristics of biofilm production | E. coli isolates
[n (%)] |
K. pneumoniae isolates
[n (%)] |
| Strong | 19 (16.66) | 11 (18.33) |
| Moderate | 29 (25.43) | 21(35) |
| Weak | 12 (10.52) | 9 (15) |
| Non-producing | 54 (47.36) | 19 (31.66) |
| Total | 114 | 60 |
4. DISCUSSION
Beta-lactam antibiotics were found to have a high level of resistance. These findings indicate the widespread presence of MDR phenotypes among K. pneumoniae and E. coli strains, highlighting the critical need for ongoing surveillance and prudent antibiotic use. Moreover, a high percentage of biofilm production, especially in E. coli isolates, is clearly associated with repeated UTI cases. as biofilms help bacteria to survive longer and resist antibiotics. Obaid and Hasson (2021) reported that 46.6% of K. pneumoniae isolates were strong biofilm producers, 30.6% were non-producers, and the rest of the total number ranged between weak and moderate producers.12
Biofilm formation is a critical virulence factor that enhances antimicrobial resistance in Enterobacteriaceae. Studies have demonstrated that biofilm-producing strains show increased resistance to multiple antibiotics, with minimal inhibitory concentrations 10–10 times higher than those required for planktonic cells.13 Research on cancer patients with UTIs found that 62.7% of coliforms were biofilm formers, and biofilm formation was significantly higher in patients with urinary catheters (83.3%) and diabetes mellitus (77.8%).14
Studies in Iraq have revealed patterns of antimicrobial resistance in the Enterobacteriaceae. Research from Nasiriyah, Iraq, showed the presence of E. coli (44.71%) and K. pneumoniae (38.57%).15 A comprehensive surveillance study from Iraq analyzing 11,592 clinical records found that K. pneumoniae (30.1%) and E. coli were among the most common gram-negative bacteria in adults, with high rates of multidrug resistance.16 In the Al-Hillah River in Babylon Province, Iraq, E. coli isolates showed the highest resistance to β-lactam antibiotics, followed by fosfomycin, and aminoglycosides. This study revealed a high prevalence of multidrug resistance among E. coli isolates from all sampling sites, indicating widespread antimicrobial resistance in the environment.17
Franklin et al (2021) reported that 198 of 203 Klebsiella pneumoniae isolates (97.5%) were regarded as biofilm producers. And 83.3% of Klebsiella spp,18 were biofilm-forming in urine samples tested on Congo agar in Nepal.19 A study done by Kadhum et al. (2021) showed that all E. coli 33 isolates produced biofilms (100%), Out of 33 isolates, 14 were strong biofilm producers. Moderate biofilm-producing isolates were found in 8/33(24.24%) isolates, whereas weak biofilm-producing isolates were found in 11/33 (33.33%) isolates. All K. pneumoniae isolates (100%) produced biofilms. Of the isolates, 5/8 (62.5%) were moderate biofilm producers and the remaining 3/8 (37.5%) were weak biofilm producers.20
In parallel with our study, multiple studies have highlighted antibiotic resistance in urinary pathogens. High resistance rates in E. coli were observed by Jalil et al. (2022) for ceftriaxone (89%), ampicillin (86.6%), levofloxacin (82.9%), and cefotaxime (79.3%),21 whereas Iqbal et al. (2021) reported ≥90% resistance to cefixime and cefuroxime and ≥80% non-susceptibility to numerous other agents, including ciprofloxacin and co-trimoxazole.22 K. pneumoniae isolates in Jalil et al. (2022) exhibited strong resistance to piperacillin (81.6%), levofloxacin (78.9%), ampicillin (76.3%), and cefotaxime (73.7%).21 Both studies noted lower resistance to carbapenems (imipenem: 2.4% for E. coli and 13.2% for K. pneumoniae) and variable resistance to ciprofloxacin (35.4% for E. coli and 36.8% for K. pneumoniae) and cefepime (34.1% for E. coli and 50.0% for K. pneumoniae).21 This widespread resistance contributes to the high MDR prevalence, particularly in E. coli (53%), as reported by Shretha et al. (2021), along with significant MDR rates in E. faecalis (22%).23
ESBL production is one of the most significant resistance mechanisms in the Enterobacteriaceae. A comprehensive study from Isfahan, Iran, involving 1,564 isolates, found that among hospitalized patients, 41.7% of E. coli and 51.5% of K. pneumoniae isolates were ESBL producers. In outpatient samples, ESBL production rates were 25% and 40% for K. pneumoniae.5 This study also identified CTX-M-15 as the predominant β-lactamase enzyme type (79.3%) in community-onset UTIs caused by ESBL-producing K. pneumoniae.24 Research from Northern Thailand demonstrated that approximately 42.5% of E. coli and 30.2% of K. pneumoniae isolates were classified as ESBL producers.25 The prevalence of ESBL increased significantly between 2004 and 2008, plateauing at approximately 30-40% per year, with recurrent UTI episodes showing significantly higher ESBL rates (46.5%) than the first episodes (27.3%).26
Antibiotic resistance rates among bacteria causing UTIs demonstrate considerable inter-study variation attributable to differences in patient demographics and geographic origin. The escalation of resistance is strongly linked to the indiscriminate consumption of antibiotics, frequently without prescriptions. This practice selects resistant mutants that may acquire traits such as reduced drug permeability and enhanced survival. Empirical evidence has confirmed that the resistance profiles of predominant uropathogens have shifted temporally in multiple countries.27
5. CONCLUSION
The present study demonstrated a high prevalence of MDR E. coli and Klebsiella pneumoniae among patients with UTI in Al-Hilla, Iraq. A significant proportion of isolates exhibited extensive resistance to β-lactam antibiotics (e.g., ampicillin and ceftazidime) and other antimicrobial classes, although carbapenems (imipenem and meropenem) retained relatively higher efficacy. Notably, biofilm production was observed in E. coli and K. pneumoniae isolates, correlating with enhanced antimicrobial resistance and potential for recurrent infections.
6. Data availability
The numerical data obtained on this research are available from the corresponding author.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
No external or industry funding was involved in the conduct of this study. The authors used only institutional resources in the conduct of the study.
9. Authors’ contribution
Conceptualization, Methodology, Formal Analysis, Writing: NAK
Validation, Investigation, Visualization, Project Administration: MSA
Software, Resources: LYM
10. REFERENCES
- Bassetti M, Giacobbe DR, Robba C, Pelosi P, Vena A. Treatment of extended-spectrum β-lactamases infections: what is the current role of new β-lactams/β-lactamase inhibitors? Curr Opin Infect Dis. 2020;33(6):474–81. [PubMed] DOI: 1097/QCO.0000000000000685
- Bader MS, Loeb M, Brooks AA. An update on the management of urinary tract infections in the era of antimicrobial resistance. Postgrad Med. 2017;129(2):242–58. [PubMed] DOI: 1080/00325481.2017.1246055
- Bassetti M, Peghin M, Pecori D. The management of multidrug-resistant Enterobacteriaceae. Curr Opin Infect Dis. 2016;29(6):583–94. [PubMed] DOI: 1097/QCO.0000000000000314
- Gupta V, Ye G, Olesky M, Lawrence K, Murray J, Yu K. Trends in resistant Enterobacteriaceae and Acinetobacter species in hospitalized patients in the United States: 2013-2017. BMC Infect Dis. 2019;19(1):742. [PubMed] DOI: 1186/s12879-019-4387-3
- Moayednia R, Shokri D, Mobasherizadeh S, Baradaran A, Fatemi SM, Merrikhi A. Frequency assessment of β-lactamase enzymes in Escherichia coli and Klebsiella isolates in patients with urinary tract infection. J Res Med Sci. 2014;19(Suppl 1):S41-5. [PubMed]
- Jean SS, Harnod D, Hsueh PR. Global threat of carbapenem-resistant gram-negative bacteria. Front Cell Infect Microbiol. 2022;12:823684. [PubMed] DOI: 3389/fcimb.2022.823684
- Lynch JP 3rd, Clark NM, Zhanel GG. Escalating antimicrobial resistance among Enterobacteriaceae: focus on carbapenemases. Expert Opin Pharmacother. 2021;22(11):1455–73. [PubMed] DOI: 1080/14656566.2021.1904891
- Tuhamize B, Tusubira D, Masembe C, Bessong PO, Byarugaba F, Bazira J. High Prevalence of Multidrug-Resistant Enterobacteriaceae Uropathogens Among Outpatients in Rural Southwestern Uganda. Cureus. 2025;17(1):e78094. [PubMed] DOI: 7759/cureus.78094
- Jamal M, Ahmad W, Andleeb S, Jalil F, Imran M, Nawaz MA, et al. Bacterial biofilm and associated infections. J Chin Med Assoc. 2018;81(1):7–11. [PubMed] DOI: 1016/j.jcma.2017.07.012
- Lila ASA, Rajab AAH, Abdallah MH, Rizvi SMD, Moin A, Khafagy ES, et al. Biofilm Lifestyle in Recurrent Urinary Tract Infections. Life (Basel). 2023;13(1):148. [PubMed] DOI: 3390/life13010148
- Cui F, Li T, Wang D, Yi S, Li J, Li X. Recent advances in carbon-based nanomaterials for combating bacterial biofilm-associated infections. J Hazard Mater. 2022;431:128597. [PubMed] DOI: 1016/j.jhazmat.2022.128597
- Obaid WF, Hasson SO. Pattern of Multi Drug Resistance with Biofilm Formation among Klebsiella pneumonia isolated from Fecal Samples of Diarrheal Iraqi Patients. Ann Rom Soc Cell Biol. 2021;25(3):5350–60. Full Text
- Ramírez-Castillo FY, Guerrero-Barrera AL, Harel J, Avelar-González FJ, Vogeleer P, Arreola-Guerra JM, et al. Biofilm Formation by Escherichia coli Isolated from Urinary Tract Infections from Aguascalientes, Mexico. Microorganisms. 2023;11(12):2858. [PubMed] DOI: 3390/microorganisms11122858
- Vitharana BM, Wahalathant DM, Gunasekara SP, Chathuranga BAG, Gunasekara TDCP. Biofilm formation and antimicrobial resistance of Enterobacteriaceae isolated from cancer patients with urinary tract infections. Sri Lankan J Infect Dis. 2025;15(2):E82. DOI: 4038/sljid.v15i2.8742
- Alhusaynat HQR, Nasser HA. Prevalence and Risk Factors of Urinary Tract Infections Caused by Multidrug-Resistant Enterobacteriaceae in Nasiriyah, Iraq. Thi-Qar Med J. 2024;28(2):346–60. DOI: 32792/jmed.v28i2.590
- Al-Fahad DK, Alpofead JA, Chawsheen MA, Al-Naqshbandi AA, Abas AT. Surveillance of Antimicrobial Resistance in Iraq: A Comprehensive Data Collection Approach. ARO Sci J Koya Univ. 2024;12(2):179–93. DOI: 14500/aro.11689
- Alwash MS, Al-Rafyai HM. Antibiotic Resistance Patterns of Diverse Escherichia coli Phylogenetic Groups Isolated from the Al-Hillah River in Babylon Province, Iraq. ScientificWorldJournal. 2019;2019:5927059. [PubMed] DOI: 1155/2019/5927059
- Franklin-Alming FV, Kaspersen H, Hetland MAK, Bakksjø RJ, Nesse LL, Leangapichart T, et al. Exploring Klebsiella pneumoniae in healthy poultry reveals high genetic diversity, good biofilm-forming abilities and higher prevalence in turkeys than broilers. Front Microbiol. 2021;12:725414. [PubMed] DOI: 3389/fmicb.2021.725414
- Mahato S, Mandal P, Mahato A. Biofilm Production by Uropathogens like Klebsiella spp and Pseudomonas spp and their Antibiotic Susceptibility. Birat J Health Sci. 2020;5(1):902–6. DOI: 3126/bjhs.v5i1.29609
- Kadhum AA, Khudor MH. Phenotypic and molecular characteristics of biofilm and other virulence genes in E. coli and K. pneumoniae isolates from healthy dairy cow, human and environmental sources. Indian J Forensic Med Toxicol. 2021;15(1):2452–8. DOI: 37506/ijfmt.v15i1.13769
- Jalil MB, Al Atbee MYN. The prevalence of multiple drug resistance Escherichia coli and Klebsiella pneumoniae isolated from patients with urinary tract infections. J Clin Lab Anal. 2022;36(9):e24619. [PubMed] DOI: 1002/jcla.24619
- Iqbal Z, Mumtaz MZ, Malik A. Extensive drug-resistance in strains of Escherichia coli and Klebsiella pneumoniae isolated from paediatric urinary tract infections. J Taibah Univ Med Sci. 2021;16(4):565–74. [PubMed] DOI: 1016/j.jtumed.2021.03.004
- Shrestha LB, Baral R, Poudel P, Khanal B. Clinical, etiological and antimicrobial susceptibility profile of pediatric urinary tract infections in a tertiary care hospital of Nepal. BMC Pediatr. 2019;19(1):36. [PubMed] DOI: 1186/s12887-019-1410-1
- Boix-Palop L, Xercavins M, Badía C, Obradors M, Riera M, Freixas N, et al. Emerging extended-spectrum β-lactamase-producing Klebsiella pneumoniae causing community-onset urinary tract infections: a case-control-control study. Int J Antimicrob Agents. 2017;50(2):197–202. [PubMed] DOI: 1016/j.ijantimicag.2017.03.009
- Siriphap A, Kitti T, Khuekankaew A, Boonlao C, Thephinlap C, Thepmalee C, et al. High prevalence of extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae isolates: A 5-year retrospective study at a Tertiary Hospital in Northern Thailand. Front Cell Infect Microbiol. 2022;12:955774. [PubMed] DOI: 3389/fcimb.2022.955774
- Vachvanichsanong P, McNeil EB, Dissaneewate P. Extended-spectrum beta-lactamase Escherichia coli and Klebsiella pneumoniae urinary tract infections. Epidemiol Infect. 2020;149:e12. [PubMed] DOI: 1017/S0950268820003015
- Lomazzi M, Moore M, Johnson A, Balasegaram M, Borisch B. Antimicrobial resistance–moving forward? BMC Public Health. 2019;19(1):858. [PubMed] DOI: 1186/s12889-019-7173-7