Trinh Ke Diep 1 , Pham Huu Lu 2 , Nguyen Quoc Kinh 3 , Luu Quang Thuy 4 , Duong Duc Hung 5
Authors affiliations:
Background & objective: Thoracic surgery is almost always performed under general anesthesia (GA) with controlled ventilation through an endotracheal tube (ETT). This study aimed to compare intraoperative ventilation, blood oxygenation parameters between non-intubated video-assisted thoracic surgery (NI-VATS) using a laryngeal mask airway (LMA) with spontaneous/pressure support ventilation (PSV) versus controlled ventilation through ETT with one-lung ventilation (OLV).
Methodology: 100 patients (ASA I-III) undergoing VATS were randomized into two groups of 50 each: NI-VATS group, to receive spontaneous/pressure support ventilation (PSV) through an LMA of a suitable size, and I-VATS group, to receive controlled ventilation through ETT with OLV. Key ventilation parameters and oxygenation parameters were monitored and compared.
Results: I-VATS group had significantly higher tidal volume and minute ventilation. Conversely, NI-VATS group exhibited significantly lower Ppeak during both two-lung (4.06 ± 1.74 cmH2O vs. 24.18 ± 3.94 cmH2O, P < 0.001) and one-lung ventilation (5.36 ± 1.66 cmH2O vs. 30.42 ± 5.46 cmH2O, P < 0.001). NI-VATS group had higher peak EtCO2 (48.56 ± 4.11 mmHg vs. 40.96 ± 4.71 mmHg, P < 0.001) and PaCO2 during OLV (46.84 ± 6.90 mmHg vs. 38.54 ± 7.48 mmHg, P < 0.001). Regarding oxygenation, NI-VATS group required significantly lower highest FiO2 (P < 0.001) yet maintained higher lowest SpO2 (P < 0.001). During OLV, NI-VATS showed higher PaO2 (P < 0.001), a superior PaO2/FiO2 ratio (P < 0.001), and a lower AaDO2 (P < 0.001).
Conclusion: NI-VATS demonstrated advantages in oxygenation efficiency, requiring lower FiO2 while maintaining better SpO2 and gas exchange during OLV, alongside significantly lower airway pressures.
Abbreviations: ETT: endotracheal tube, GA: general anesthesia, LMA: laryngeal mask airway, NI-VATS: non-intubated video-assisted thoracic surgery, OLV: one-lung ventilation, PSV: pressure support ventilation, TCI: Target controlled infusion, VATS: video-assisted thoracic surgery, VILI: ventilator-induced lung injury
Keywords: ventilation; blood oxygenation; video-assisted; thoracic surgery
Citation: Diep TK, Lu PH, Kinh NQ, Thuy LQ, Hung DD. Comparison of ventilation and blood oxygenation changes in non-intubated versus intubated anesthesia for video-assisted thoracic surgery. Anaesth. pain intensive care 2025;29(7):670-678. DOI: 10.35975/apic.v29i7.2943
Received: June 24, 2025; Revised: August 01, 2025; Accepted: August 01, 2025
General anesthesia with tracheal intubation is widely used in thoracic surgery because it improves surgical safety, but this technique cannot avoid complications of tracheal intubation and medications used. The development of "minimally invasive surgery," including minimally invasive anesthesia, has become a hot topic of research in the field of minimally invasive thoracic surgery. Concomitant with significant advancements in anesthesia management techniques and surgical risk management strategies, non-intubated thoracic surgery (NITS), encompassing procedures from open thoracic surgery (OTS) to uniportal video-assisted thoracic surgery (uniportal VATS), has seen progressively wider adoption globally. This anesthetic methodology permits the maintenance of the patient's physiological spontaneous respiration, obviating the need for a double-lumen endotracheal tube or a specialized endobronchial blocker. As a result, it guarantees the highest level of patient safety while providing a clear surgical field, with patients kept at different states of awareness, ranging from fully awake to conscious sedation or deep sedation. This approach is a safe and practical method used in numerous surgical centers. It reduces complications related to tracheal intubation and anesthetic drugs used, and enhances recovery after thoracic surgery.1,2 While NITS has gained traction globally, many centers typically employ strategies such as high-flow nasal oxygen or awake spontaneous breathing, often supplemented with regional blocks like epidural anesthesia. The best technique is still under investigation, especially in areas with limited resources or where certain drugs are not accessible. Although the need to mitigate intubation-related complications is recognized in Vietnam, a scarcity of systematic studies on alternative techniques persists, largely driven by resource constraints. This research examines a new method for NI-VATS in Vietnam, using a laryngeal mask airway (LMA) to support spontaneous or assisted ventilation. This unique application addresses local challenges such as the unavailability of remifentanil and TCI fentanyl models, allowing for better control of respiration and anesthesia parameters while mitigating the risks associated with traditional intubation and deep sedation with unmonitored spontaneous breathing.
Although a considerable body of literature has documented the application of non-intubated anesthesia in thoracic surgery, to date, we have noted a paucity of studies systematically evaluating the ensuing respiratory alterations, specifically parameters like tidal volume (TV), respiratory rate (RR), and airway pressure, within this clinical setting. This study aims to compare the effects of non-intubated anesthesia using an LMA with spontaneous/assisted ventilation (NI-VATS) versus conventional intubated anesthesia (I-VATS) with one-lung ventilation (OLV) on ventilation and blood oxygenation parameters, as well as immediate postoperative recovery and safety, in patients undergoing video-assisted thoracic surgery. We hypothesize that NI-VATS, by preserving spontaneous respiration and avoiding the complications associated with tracheal intubation, will lead to more physiological ventilation, superior oxygenation, and faster recovery outcomes compared to I-VATS.
Consecutive patients undergoing thoracic surgery were randomized according to whether they underwent VATS at the Center of Anesthesia and Surgical Intensive Care, Viet Duc University Hospital, between August 2022 and May 2024. Patient allocation to either the NI-VATS or I-VATS group was performed using a computer-generated random allocation sequence with a 1:1 ratio.
The minimum sample size for each group was calculated using the two-sample mean comparison formula:
Where: α=0.05 (two-tailed test, Z1−α/2=1.96), statistical power = 80% (Z1−β=0.84). Based on a study by Ke et al. (2020)3 which investigated 160 patients undergoing pulmonary segmentectomy, divided into two groups (81 non-intubated, 79 intubated). PaO₂/FiO₂ ratios were 320.4±150 vs. 245±115.6. Based on those values, the minimal sample size required for each group is n = 49.6. Therefore, a minimum of 50 patients per group was determined for this study.
American Society of Anesthesiologists (ASA) is no more than III, Age from 18 to 60. The respiratory and circulation systems work well (EF above 50%), PaO2 > 60 mmHg, PaCO2 >50 mmHg, and FEV1% above 50% of the expected value), and all essential organs function usually, with BMI < 30 (kg/m2).
Patients with evidence of impairment of the function of other essential organs, unwilling to participate in the study, or a history of significant neuropathy that might affect the assessment in the study, were excluded.
Selected patients were divided into 2 groups: 50 patients to Group 1 (non-intubated video-assisted thoracoscopic surgery - NI-VATS group), and 50 patients to Group 2 (Intubated video-assisted thoracoscopic surgery- I-VATS groups).
Equipment used in anesthesia: Multi-parameter monitor (Nihon Kohden, model BsM-3763, Japan), syringe pump (B. Braun; Perfusor Space Syringe Pump), Anesthesia Machine (Datex Ohmeda; Aespire S5), Neuromuscular monitoring (ToFscan from IDMED), Mallinckrodt double-lumen endobronchial tube (Medtronic, Minneapolis, MN), LMA ProSeal®
We performed pre-operative erector spinae plane blocks (ESPB) with 20 mL ropivacaine 0.5% at the level of the T5 transverse process on the same side of surgery. A peripheral intravenous cannula and radial arterial catheter were inserted before induction for continuous blood pressure monitoring. Target-controlled infusion (TCI) of propofol (model Shinder) was used to induce and maintain anesthesia. A bispectral index was used to keep the target between 40 to 60 in both groups.
In Group 1, no muscle relaxants were used. TCI of propofol and fentanyl 1-2 µg/kg was adjusted to keep the BIS at 40–60. At BIS < 60, an LMA ProSeal® was inserted and attached to the breathing circuit, and respiration was monitored. If spontaneous breathing did not occur, assisted ventilation with synchronized intermittent mechanical ventilation (SIMV) with 4–6 mL/kg was started at 10 times/min, with oxygen (FiO2 50-100%) provided through the LMA at 5 L/min. Patients breathed spontaneously. During the surgery, if SpO2 fell below 90%, EtCO2 ≥ 60 mmHg, or RR 20 cycles/min, pressure support (PS) ventilation with PS 8 cmH2O was started. Propofol was stopped immediately when the chest wall closure started.
Group 2 patients were paralyzed following anesthetic induction, and a double-lumen tube (DLT) was inserted. Position was confirmed, when lung isolation was achieved by deflation of the non-dependent lung. After 10 min, if it was not placed in the correct position, we used a flexible endoscope to place and check. One-lung ventilation of the dependent lung was commenced using protective ventilation strategies with tidal volumes 6-8 mL/kg IBW, RR 10 to 20 times/min, and positive end-expiratory pressure (PEEP) 0-5 cmH2O, keeping peak pressures <30 cmH2O. When the surgery was complete, propofol was stopped, and a mixture of neostigmine and atropine was injected.
Supplemental fentanyl was given to all patients intraoperatively, and doses were recorded. All patients received 1g of paracetamol and 20 mg Nefopam IV. All patients were awakened in the operating room and then transferred to the recovery room, self-ventilating with facemask oxygen. Subsequently, patients were transferred to the Cardiovascular and Thoracic Surgery Department, Viet Duc University Hospital, once the Aldrette Score was 9 to 10.
Surgical Technique: All procedures were performed with a 5 cm skin incision in the right mid-axillary line to access the chest cavity. Thoracoscopic instruments and a video camera were inserted through this incision to visualize the thymus gland. The thymus was carefully dissected and removed entirely. The surgical field was inspected for any bleeding, and chest tubes were placed for drainage before closing the incision.
Variables: Patient demographics: Age, American Society of Anesthesiologists (ASA), gender, weight, height, medical history; Anesthetic time was from the first electronically monitored observation in the anesthetic room until the patient was transferred to the operating room. Surgery time is from the start of skin incision until the end of skin closure. Anesthesia time is from the start of anesthesia to the withdrawal of LMA and DLT.
Data on intraoperative end-tidal carbon dioxide (EtCO2) levels, oxygen saturation (SpO2), fraction of inspired oxygen concentration (FiO2), and Tidal volume (VT), Peak inspiratory pressure (Ppeak) is the highest level of pressure applied to the lungs during inhalation. Surgical rating scales (SRSs) to describe and quantify surgical working conditions notes using a 5-point scale (Optimal conditions, Good conditions, Acceptable conditions, Extremely poor conditions). The visual analog scale (VAS) is a pain rating scale.
Intraoperative physiologic monitoring includes arterial blood gas (ABG), continuous monitoring of electrocardiogram (ECG), heart rate (HR), invasive blood pressure (IBP), pulse oxygen saturation (SpO2), respiratory rate (RR), and pressure of end-tidal carbon dioxide (EtCO2) machine to enable spontaneous breathing. Oxygen (4–5 L/min). Support ventilation (PSV) is used to prevent lung collapse and support breathing at the operation's end. Stop Propofol immediately after skin closure and extubate when spontaneous breathing is above 5 ml/kg (ideal body weight) and BIS is above 80; Postoperative outcomes: Length of hospital stay, postoperative pain scores, complications (e.g., bleeding, infection).
Statistical Analysis: The collected data is processed and analyzed using SPSS software version 16.0. Quantitative variables are presented as mean ± standard or median deviation (quartile) and compared using the t-student or Mann-Whitney U test. Pearson correlation coefficient is used to evaluate the correlation. Qualitative variables, described by frequency (%), are compared using the Chi squared test or Fisher's exact test; relevance is assessed by the Phi coefficient and Cramer's V. Choose P < 0.05 as the level for statistically significant differences.
No patients were excluded from the analysis after being randomized. The results showed no statistically significant differences in demographic factors, e.g., gender, age, height, weight, and BMI between the two groups. The ASA status, smoking rate, and the primary diagnosis and comorbidities were also similar.
Table 2 shows that the types of surgery, surgery times, and lung collapse scores of the two groups are statistically not different.
Time to onset of anesthesia in Group 1 was significantly shorter than in Group 2 (P < 0.001). The dose of fentanyl used in surgery in Group 1 was lower and statistically significant than that of Group 2 (P < 0.001). This may reflect a lower need for opioids in Group 1.
There were no statistically significant differences in time to one lung ventilation (P = 0.4802), total duration of anaesthesia (P = 0.0531 – asymptotic threshold), intraoperative propofol dose (P = 0.2029), or pulmonary collapse point at 10 min (P = 0.2896) and 30 min (P = 0.1752). Similarly, the types of surgery (P > 0.05) and the duration of surgery (P = 0.4654) also did not show a difference between the two groups.
However, another important difference was the time of intubation. Group 1 patients had a much shorter intubation time (5.46 ± 2.05 min) than Group 2 (15.60 ± 3.82 min) (P < 0.001), indicating significantly earlier recovery and spontaneous breathing.
In terms of postoperative parameters, the time to achieve the Aldrete score in Group 1 (4.78 ± 1.62 min) was statistically shorter than that of Group 2 (10.48 ± 3.13 min) (P < 0.001).
In Table 3 and Figure 1, we found many statistically significant differences between Group 1 and Group 2, reflecting certain advantages of each approach.
3.1. Ventilation changes during surgery
The data presented in Figure 1 and Table 3 indicate that the respiratory rate was comparable between the two groups during surgery. However, the tidal volume and minute ventilation in Group 2 are approximately 1.5 times greater than those in Group 1. Additionally, the airway pressure intraoperatively in Group 2 was higher (P < 0.05). Group 1, the highest peak airway pressure in 2 lungs was (4.06 ± 1.74 cmH₂O) and one lung ventilation (5.36 ± 1.66 cmH₂O) were significantly lower, almost 5 times, compared to Group 2 (P < 0.001). In contrast, the Peak EtCO₂ during operation in Group 1 was significantly higher than in Group 2 (P < 0.001). This was also reflected in PaCO₂ during the one lung ventilation phase, when Group 1 had a significantly higher PaCO₂ than Group 2 (P < 0.001). Higher PaCO₂ levels in Group 1 may be related to the characterization of spontaneous ventilation, where CO₂ emissions may not be as effective as positive pressure-controlled ventilation.
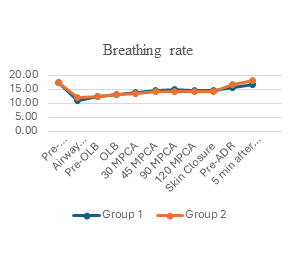
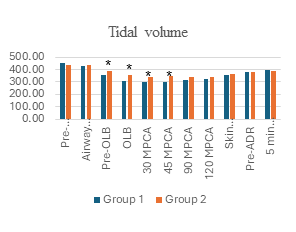
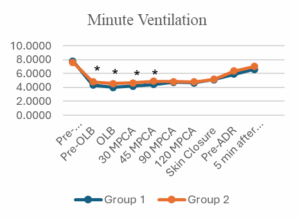
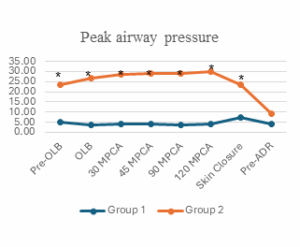
Figure 1: Ventilation and airway pressure changes during surgery
(Abbreviations: OLB, One lung breathing; MPCA, Minutes post-chest cavity access; ADR, Airway device removal; *, P-value < 0.05)
3.2. Oxygenation and gas exchange
Although in-room SpO₂ was no different (P = 0.154). Lowest SpO₂ during operation in Group 1 (97.30 ± 1.69%) was statistically higher than in Group 2 (92.44 ± 6.62%) (P < 0.001). This, along with lower FiO₂ use, showing a more stable and effective ability to maintain blood oxygenation in Group 1. The highest FiO₂ during operation in Group 1 (61.22 ± 4.20%) was also statistically lower than in Group 2 (78.54 ± 16.46%) (P < 0.001).
When considering arterial blood gas before one lung ventilation, the PaO₂/FiO₂ index in Group 1 was significantly higher than in Group 2 (P = 0.048), indicating better initial oxygen exchange. During one lung ventilation phase, this difference became even more pronounced: Group 1 had a significantly higher PaO₂ (P < 0.001) and PaO₂/FiO₂ (P < 0.001) than Group 2.
The alveolar-arterial oxygen pressure differential (AaDO₂) during one lung ventilation in Group 1 was significantly lower than in Group 2 (P < 0.001), indicating better ventilation-perfusion matching (V/Q matching) and fewer shunts in Group 1.
3.3. Postoperative outcomes
After surgery, the PaCO₂ of Group 1 remained significantly higher than that of Group 2 (P = 0.006). At the same time, the postoperative AaDO₂ in Group 1 was significantly lower than that of Group 2 (P < 0.001), further confirming better gas exchange efficiency. Postoperative lactate concentrations in Group 1 were significantly lower than in Group 2 (P < 0.001), suggesting better tissue perfusion and aerobic metabolism in Group 1.
This study compared intraoperative ventilation and oxygenation parameters between non-intubated video-assisted thoracic surgery (NI-VATS) using an LMA with spontaneous/assisted ventilation and conventional intubated VATS (I-VATS) with one-lung ventilation (OLV). Our findings demonstrate several significant advantages in the NI-VATS group, primarily related to lower airway pressures, better oxygenation efficiency with lower FiO2, and faster immediate postoperative recovery, despite higher but clinically manageable CO2 levels.
4.1. Ventilation changes during surgery
A distinctive aspect of our NI-VATS protocol lies in the use of the LMA for airway management during spontaneous/assisted ventilation, as opposed to solely relying on nasal oxygen delivery. While other centers often perform NI-VATS with high-flow nasal oxygen or epidural anesthesia, our approach with the LMA offers several critical advantages, especially relevant in our clinical context. The LMA allowed for precise monitoring of respiratory parameters such as EtCO2 and tidal volume (Vt), which is challenging with mask or nasal oxygen alone. Furthermore, it provided a secure airway, enabling easy transition to assisted ventilation or even intubation through the LMA if needed, without interrupting the surgical field. Additionally, being able to reinflate the lung after surgery easily also offers advantages.
4.2. Breathing rate, Vt, and MV
In our study, the breathing rate was comparable between the two groups during most of the surgery. However, the I-VATS group (Group 2) had significantly higher tidal volumes and, consequently, minute ventilation compared to the NI-VATS group (Group 1). I-VATS group typically aims for and achieves higher Vt and MV, which is intended to help patients avoid hypercapnia. While it is important to recognize that the implementation of OLV can lead to a reduction in tidal volume within the double-lumen group, resulting in increased CO2 levels in the blood, this assertion must be considered within a broader context. In contrast, OLV presents its own set of challenges; controlled ventilation strategies typically strive to maintain an adequate tidal volume, although adjustments may be necessary for the ventilated lung. Interestingly, in our I-VATS group, both tidal volume and minute ventilation were approximately 1.5 times higher than those in the NI-VATS group. This discrepancy may be attributed to the administration of propofol and fentanyl, along with one-lung breathing during surgery.
4.3. Perioperative pH
Before starting OLV, the mean pH in both groups tended towards mild alkalosis (7.46 in Group 1 and 7.47 in Group 2). These values are higher than the average physiological range (7.35-7.45), but not statistically significant (P = 0.18). This can likely be explained by patient anxiety and stress before surgery, leading to an increased respiratory rate, which in turn causes a decrease in PaCO₂ and results in mild respiratory alkalosis. Intraoperative and Postoperative period results show a statistically significant difference in pH between the two groups during and after the surgical process. During OLV, the pH in Group 1 (NI-VATS) was lower than in Group 2 (I-VATS). Postoperatively, this trend continued, although the difference was more minor, with a mean pH in Group 1 of 7.42 compared to 7.43 in Group 2 (P = 0.049). The decrease in pH in Group 1 is directly related to the significantly higher PaCO₂ levels in this group at the same time points. During OLV, the PaCO₂ of Group 1 was 46.84 mmHg, much higher than the 38.54 mmHg of Group 2 (P < 0.001). This state of elevated CO₂ is a characteristic of the spontaneous ventilation method in thoracic surgery and is known as "permissive hypercapnia". The state of hypercapnia and the resulting mild reduction in pH in the NI-VATS group were well-controlled and did not cause adverse effects.
4.4. PaCO2 and EtCO2
The NI-VATS group exhibited significantly higher peak EtCO2 (P < 0.001). PaCO2 during OLV comparable to the I-VATS group (P < 0.001). This permissive hypercapnia is common in patients who are spontaneously ventilating during thoracic surgery and, if maintained within safe limits, as in our NI-VATS group, may be well tolerated. Liu et al.4 in their study of spontaneous pneumothorax, also found higher PetCO2 (likely meant EtCO2) and PaCO2 in their NI-VATS group. The higher PaCO2 levels in the NI-VATS group may be related to the spontaneous ventilation. Postoperatively, PaCO2 in the NI-VATS group remained significantly higher than in the I-VATS group (P = 0.006). There have been reports indicating that an increase in blood CO2 levels up to 100 mmHg can be acceptable.5 A slight rise in CO2 is generally not a concern; in fact, it can sometimes enhance tissue perfusion and shorten recovery time.6 Permissive hypercapnia is contraindicated in patients with elevated intracranial pressure (ICP) or severe cardiopulmonary conditions like pulmonary hypertension, due to risks of CO₂-induced cerebral vasodilation and increased pulmonary vascular resistance. Our study shows that this permissive hypercapnia was clinically manageable and associated with improved tissue perfusion and aerobic metabolism, as indicated by lower postoperative lactate levels (Table 3).
4.5. Airway pressure
Peak airway pressure was significantly lower in the NI-VATS group during both two-lung ventilation (P < 0.001) and in OLV (P < 0.001). While some physiological changes were observed, such as lower peak airway pressures and higher CO2 levels in the NI-VATS group, which might be anticipated with spontaneous ventilation. Our results demonstrate that the peak airway pressure in NI-VATS was nearly five times lower than in I-VATS. This substantial reduction directly translates into a significant clinical benefit, reducing barotrauma and ventilator-induced lung injury (VILI), a concern with conventional OLV.4 Although our study did not specifically measure VILI markers, lower airway pressure is inherently protective.
4.6. Blood oxygenation parameters
4.6.1. FiO2 and Oxygen Saturation (SpO2)
Our findings indicate better oxygenation efficiency in the NI-VATS group. The mean peak FiO2 required during the operation was significantly lower in the NI-VATS group compared to the I-VATS group (P < 0.001). Despite this lower FiO2, the NI-VATS group demonstrated significantly higher lowest SpO2 during the operation (P < 0.001). This suggests a more stable and effective ability to maintain blood oxygenation in the NI-VATS group. Ke HH, et al (2022) reported a 5% incidence of hypoxemia (SpO2 < 90%) during VATS with OLV,4 whereas our NI-VATS group maintained higher SpO2 levels with less supplemental oxygen. The key result demonstrates the enhanced oxygenation efficiency of the LMA-based NI-VATS approach—a notable advantage, given that the I-VATS group had to manage the inherent difficulties of OLV. The maintained spontaneous diaphragmatic motion and negative intrapleural pressure with LMA likely contribute to better ventilation/perfusion (V/Q) matching and reduced intrapulmonary shunting. Therefore, our findings, while aligning with certain physiological principles, provide compelling quantitative data supporting the safety, efficiency, and clinical advantages of LMA-based NI-VATS as a valuable and applicable anesthetic technique for VATS, especially in contexts where conventional intubation carries higher risks or specific pharmacological agents are limited.
4.6.2. PaO2/FiO2 Ratio (P/F Ratio)
The physiological basis for improved oxygenation in NI-VATS, despite OLV (simulated by pathological pneumothorax), is thought to lie in the maintenance of spontaneous diaphragmatic motion and more negative intrapleural pressure during inspiration. This helps maintain functional residual capacity (FRC) to a greater extent than in paralyzed, mechanically ventilated patients, leading to better ventilation/perfusion (V/Q) matching and reduced intrapulmonary shunting.7 Our results support this theory. Prior to OLV, the PaO2/FiO2 ratio in the NI-VATS group (503.20 ± 97.82) was significantly higher than in the I-VATS group (465.23 ± 126.36, P = 0.048). During the OLV phase, this difference became even more pronounced: the NI-VATS group had a significantly higher PaO2 (mean 176.90 ± 60.87 mmHg vs. 121.73 ± 63.32 mmHg, P < 0.001) and PaO2/FiO2 ratio (mean 294.49 ± 99.91 vs. 187.53 ± 107.22, P < 0.001) than the I-VATS group. This is consistent with the review by Fabo et al.7 which highlighted improved V/Q matching and gas exchange as benefits of spontaneous ventilation.
4.6.3. Alveolar-Arterial Oxygen Gradient (AaDO2)
Furthermore, the AaDO2 was significantly lower during one-lung breathing in our NI-VATS group (P < 0.001) and postoperatively (P < 0.001). This suggests more efficient gas exchange and less shunting, consistent with the theoretical advantages of preserved spontaneous ventilation.
Interestingly, postoperative lactate levels were significantly lower in our NI-VATS group (P < 0.001), indicating better tissue perfusion and oxygen delivery, possibly due to the Bohr effect from mild hypercapnia increasing oxygen release to the tissues. Shorter anesthesia induction time (3.75 vs. 15.58 min, P < 0.001), significantly lower intraoperative fentanyl requirement (P < 0.001), significantly shorter extubation time (P < 0.001), and faster achievement of full Aldrete score (P < 0.001) in the NI-VATS group, highlighting the principles of enhanced recovery after surgery.
These findings are consistent with the literature showing that NI-VATS facilitates faster recovery.1 The reduced opioid use may also contribute to a faster return of spontaneous respiration and less postoperative sedation. Comparing our results with other studies, Chen et al. (2012) reported successful intubation-free lung resections using epidural anesthesia and vagal blockade, demonstrating the feasibility of complex procedures without intubation.8 Our study, using LMA and TCI propofol, further supports the adaptability of intubation-free techniques. Huang et al. (2020) found NI-VATS to be safe for mediastinal tumor resection in patients with impaired pulmonary function, with no serious complications in the SV-VATS group.9 While our study focused on patients with generally preserved pulmonary function (ASA I-II), the observed safety profile is consistent with their finding that NI-VATS was well tolerated. Li et al. (2020) also demonstrated the feasibility of intubation-free anesthesia with regional block for thoracoscopic surgery, reinforcing the overall safety and applicability of these approaches.2 The broad applicability and safety of NI-VATS in a variety of thoracic procedures and patient populations become increasingly evident when these studies are considered as a whole.
Our results support NI-VATS as a viable option for patients with preserved respiratory function (ASA I–II, FEV₁ > 50%). The technique demonstrated several clinical advantages, including adequate ventilation, lower airway pressure, better blood oxygenation with lower FiO₂, reduced opioid consumption, and faster recovery after surgery, which have significant clinical implications. However, NI-VATS may not be suitable in some cases, such as a difficult airway, obesity (BMI ≥ 30), or severe comorbidities. The success of this technique relies on meticulous anesthetic management, including using a laryngeal mask and pressure ventilation support in cases of hypoxemia (SpO₂ <90%).
This study has several limitations. First, it was conducted at a single center with a specific patient population (ASA I-II, BMI <30, FEV₁ >50%), which may limit the generalizability of the findings to patients with more severe comorbidities, higher BMI, or significantly impaired lung function. Finally, the study was not blinded to the anesthesiologists providing care, which is inherent in comparing two distinct anesthetic techniques, but could introduce performance bias.
Using NI-VATS, tidal volume and minute ventilation decrease slightly, and CO2 increases as compared to endotracheal intubation. In contrast, airway pressure and blood oxygenation are better compared to the double-lumen intubation method. NI-VATS is also a safe and feasible method that can provide physiological benefits and contribute to the recovery process in appropriately chosen patients. Further research is warranted to evaluate long-term outcomes and expand the application of this technique to a broader range of patients and surgical complexities.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Ethical Issues
The research has the purpose of protecting and improving patients' health and has no other purpose; it was conducted with the patient's consent. They will be treated (if there are unwanted effects) and consulted about research issues, and we will keep confidential the information provided by patients. The study was approved by the ethics committee of Viet Duc University Hospital (March 21st, 2022) and Hanoi Medical University (Ref 659/GCN-HĐĐĐNCYSH-ĐHYHN dated June 7th, 2022).
9. Conflict of interest
All authors declare that there was no conflict of interest.
10. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
11. Authors’ contribution
TKD: conceptualization, writing-original draft, methodology, investigation, data analysis.
PHL: Supervision, methodology, validation, writing-review & editing
NQK: Supervision, conceptualization, methodology, writing-review & editing
LQT, DDH: writing-original draft, data analysis, writing-review
Authors affiliations:
- Trinh Ke Diep, VNU University of Medicine & Pharmacy / Hanoi Medical University, Hanoi, Vietnam; Email: TrinhKeDiep.VDUH@gmail.com
- Pham Huu Lu, Viet Duc University Hospital, 40 Trang Thi str - Ha Noi - Viet Nam; Email: phamhuulucts@gmail.com
- Nguyen Quoc Kinh, VNU University of Medicine and Pharmacy, Thach That district, Hanoi, Vietnam; Email: nguyenquockinh@yahoo.com
- Luu Quang Thuy, VNU University of Medicine and Pharmacy, Thach That district, Hanoi, Vietnam; Email: drluuquangthuy@gmail.com
- Duong Duc Hung, Hanoi Medical University, Hanoi, Vietnam; Email: duongdh38@yahoo.com
ABSTRACT
Background & objective: Thoracic surgery is almost always performed under general anesthesia (GA) with controlled ventilation through an endotracheal tube (ETT). This study aimed to compare intraoperative ventilation, blood oxygenation parameters between non-intubated video-assisted thoracic surgery (NI-VATS) using a laryngeal mask airway (LMA) with spontaneous/pressure support ventilation (PSV) versus controlled ventilation through ETT with one-lung ventilation (OLV).
Methodology: 100 patients (ASA I-III) undergoing VATS were randomized into two groups of 50 each: NI-VATS group, to receive spontaneous/pressure support ventilation (PSV) through an LMA of a suitable size, and I-VATS group, to receive controlled ventilation through ETT with OLV. Key ventilation parameters and oxygenation parameters were monitored and compared.
Results: I-VATS group had significantly higher tidal volume and minute ventilation. Conversely, NI-VATS group exhibited significantly lower Ppeak during both two-lung (4.06 ± 1.74 cmH2O vs. 24.18 ± 3.94 cmH2O, P < 0.001) and one-lung ventilation (5.36 ± 1.66 cmH2O vs. 30.42 ± 5.46 cmH2O, P < 0.001). NI-VATS group had higher peak EtCO2 (48.56 ± 4.11 mmHg vs. 40.96 ± 4.71 mmHg, P < 0.001) and PaCO2 during OLV (46.84 ± 6.90 mmHg vs. 38.54 ± 7.48 mmHg, P < 0.001). Regarding oxygenation, NI-VATS group required significantly lower highest FiO2 (P < 0.001) yet maintained higher lowest SpO2 (P < 0.001). During OLV, NI-VATS showed higher PaO2 (P < 0.001), a superior PaO2/FiO2 ratio (P < 0.001), and a lower AaDO2 (P < 0.001).
Conclusion: NI-VATS demonstrated advantages in oxygenation efficiency, requiring lower FiO2 while maintaining better SpO2 and gas exchange during OLV, alongside significantly lower airway pressures.
Abbreviations: ETT: endotracheal tube, GA: general anesthesia, LMA: laryngeal mask airway, NI-VATS: non-intubated video-assisted thoracic surgery, OLV: one-lung ventilation, PSV: pressure support ventilation, TCI: Target controlled infusion, VATS: video-assisted thoracic surgery, VILI: ventilator-induced lung injury
Keywords: ventilation; blood oxygenation; video-assisted; thoracic surgery
Citation: Diep TK, Lu PH, Kinh NQ, Thuy LQ, Hung DD. Comparison of ventilation and blood oxygenation changes in non-intubated versus intubated anesthesia for video-assisted thoracic surgery. Anaesth. pain intensive care 2025;29(7):670-678. DOI: 10.35975/apic.v29i7.2943
Received: June 24, 2025; Revised: August 01, 2025; Accepted: August 01, 2025
1. INTRODUCTION
General anesthesia with tracheal intubation is widely used in thoracic surgery because it improves surgical safety, but this technique cannot avoid complications of tracheal intubation and medications used. The development of "minimally invasive surgery," including minimally invasive anesthesia, has become a hot topic of research in the field of minimally invasive thoracic surgery. Concomitant with significant advancements in anesthesia management techniques and surgical risk management strategies, non-intubated thoracic surgery (NITS), encompassing procedures from open thoracic surgery (OTS) to uniportal video-assisted thoracic surgery (uniportal VATS), has seen progressively wider adoption globally. This anesthetic methodology permits the maintenance of the patient's physiological spontaneous respiration, obviating the need for a double-lumen endotracheal tube or a specialized endobronchial blocker. As a result, it guarantees the highest level of patient safety while providing a clear surgical field, with patients kept at different states of awareness, ranging from fully awake to conscious sedation or deep sedation. This approach is a safe and practical method used in numerous surgical centers. It reduces complications related to tracheal intubation and anesthetic drugs used, and enhances recovery after thoracic surgery.1,2 While NITS has gained traction globally, many centers typically employ strategies such as high-flow nasal oxygen or awake spontaneous breathing, often supplemented with regional blocks like epidural anesthesia. The best technique is still under investigation, especially in areas with limited resources or where certain drugs are not accessible. Although the need to mitigate intubation-related complications is recognized in Vietnam, a scarcity of systematic studies on alternative techniques persists, largely driven by resource constraints. This research examines a new method for NI-VATS in Vietnam, using a laryngeal mask airway (LMA) to support spontaneous or assisted ventilation. This unique application addresses local challenges such as the unavailability of remifentanil and TCI fentanyl models, allowing for better control of respiration and anesthesia parameters while mitigating the risks associated with traditional intubation and deep sedation with unmonitored spontaneous breathing.
Although a considerable body of literature has documented the application of non-intubated anesthesia in thoracic surgery, to date, we have noted a paucity of studies systematically evaluating the ensuing respiratory alterations, specifically parameters like tidal volume (TV), respiratory rate (RR), and airway pressure, within this clinical setting. This study aims to compare the effects of non-intubated anesthesia using an LMA with spontaneous/assisted ventilation (NI-VATS) versus conventional intubated anesthesia (I-VATS) with one-lung ventilation (OLV) on ventilation and blood oxygenation parameters, as well as immediate postoperative recovery and safety, in patients undergoing video-assisted thoracic surgery. We hypothesize that NI-VATS, by preserving spontaneous respiration and avoiding the complications associated with tracheal intubation, will lead to more physiological ventilation, superior oxygenation, and faster recovery outcomes compared to I-VATS.
2. METHODOLOGY
Consecutive patients undergoing thoracic surgery were randomized according to whether they underwent VATS at the Center of Anesthesia and Surgical Intensive Care, Viet Duc University Hospital, between August 2022 and May 2024. Patient allocation to either the NI-VATS or I-VATS group was performed using a computer-generated random allocation sequence with a 1:1 ratio.
The minimum sample size for each group was calculated using the two-sample mean comparison formula:
Where: α=0.05 (two-tailed test, Z1−α/2=1.96), statistical power = 80% (Z1−β=0.84). Based on a study by Ke et al. (2020)3 which investigated 160 patients undergoing pulmonary segmentectomy, divided into two groups (81 non-intubated, 79 intubated). PaO₂/FiO₂ ratios were 320.4±150 vs. 245±115.6. Based on those values, the minimal sample size required for each group is n = 49.6. Therefore, a minimum of 50 patients per group was determined for this study.
American Society of Anesthesiologists (ASA) is no more than III, Age from 18 to 60. The respiratory and circulation systems work well (EF above 50%), PaO2 > 60 mmHg, PaCO2 >50 mmHg, and FEV1% above 50% of the expected value), and all essential organs function usually, with BMI < 30 (kg/m2).
Patients with evidence of impairment of the function of other essential organs, unwilling to participate in the study, or a history of significant neuropathy that might affect the assessment in the study, were excluded.
Selected patients were divided into 2 groups: 50 patients to Group 1 (non-intubated video-assisted thoracoscopic surgery - NI-VATS group), and 50 patients to Group 2 (Intubated video-assisted thoracoscopic surgery- I-VATS groups).
Equipment used in anesthesia: Multi-parameter monitor (Nihon Kohden, model BsM-3763, Japan), syringe pump (B. Braun; Perfusor Space Syringe Pump), Anesthesia Machine (Datex Ohmeda; Aespire S5), Neuromuscular monitoring (ToFscan from IDMED), Mallinckrodt double-lumen endobronchial tube (Medtronic, Minneapolis, MN), LMA ProSeal®
We performed pre-operative erector spinae plane blocks (ESPB) with 20 mL ropivacaine 0.5% at the level of the T5 transverse process on the same side of surgery. A peripheral intravenous cannula and radial arterial catheter were inserted before induction for continuous blood pressure monitoring. Target-controlled infusion (TCI) of propofol (model Shinder) was used to induce and maintain anesthesia. A bispectral index was used to keep the target between 40 to 60 in both groups.
In Group 1, no muscle relaxants were used. TCI of propofol and fentanyl 1-2 µg/kg was adjusted to keep the BIS at 40–60. At BIS < 60, an LMA ProSeal® was inserted and attached to the breathing circuit, and respiration was monitored. If spontaneous breathing did not occur, assisted ventilation with synchronized intermittent mechanical ventilation (SIMV) with 4–6 mL/kg was started at 10 times/min, with oxygen (FiO2 50-100%) provided through the LMA at 5 L/min. Patients breathed spontaneously. During the surgery, if SpO2 fell below 90%, EtCO2 ≥ 60 mmHg, or RR 20 cycles/min, pressure support (PS) ventilation with PS 8 cmH2O was started. Propofol was stopped immediately when the chest wall closure started.
Group 2 patients were paralyzed following anesthetic induction, and a double-lumen tube (DLT) was inserted. Position was confirmed, when lung isolation was achieved by deflation of the non-dependent lung. After 10 min, if it was not placed in the correct position, we used a flexible endoscope to place and check. One-lung ventilation of the dependent lung was commenced using protective ventilation strategies with tidal volumes 6-8 mL/kg IBW, RR 10 to 20 times/min, and positive end-expiratory pressure (PEEP) 0-5 cmH2O, keeping peak pressures <30 cmH2O. When the surgery was complete, propofol was stopped, and a mixture of neostigmine and atropine was injected.
Supplemental fentanyl was given to all patients intraoperatively, and doses were recorded. All patients received 1g of paracetamol and 20 mg Nefopam IV. All patients were awakened in the operating room and then transferred to the recovery room, self-ventilating with facemask oxygen. Subsequently, patients were transferred to the Cardiovascular and Thoracic Surgery Department, Viet Duc University Hospital, once the Aldrette Score was 9 to 10.
Surgical Technique: All procedures were performed with a 5 cm skin incision in the right mid-axillary line to access the chest cavity. Thoracoscopic instruments and a video camera were inserted through this incision to visualize the thymus gland. The thymus was carefully dissected and removed entirely. The surgical field was inspected for any bleeding, and chest tubes were placed for drainage before closing the incision.
Variables: Patient demographics: Age, American Society of Anesthesiologists (ASA), gender, weight, height, medical history; Anesthetic time was from the first electronically monitored observation in the anesthetic room until the patient was transferred to the operating room. Surgery time is from the start of skin incision until the end of skin closure. Anesthesia time is from the start of anesthesia to the withdrawal of LMA and DLT.
Data on intraoperative end-tidal carbon dioxide (EtCO2) levels, oxygen saturation (SpO2), fraction of inspired oxygen concentration (FiO2), and Tidal volume (VT), Peak inspiratory pressure (Ppeak) is the highest level of pressure applied to the lungs during inhalation. Surgical rating scales (SRSs) to describe and quantify surgical working conditions notes using a 5-point scale (Optimal conditions, Good conditions, Acceptable conditions, Extremely poor conditions). The visual analog scale (VAS) is a pain rating scale.
Intraoperative physiologic monitoring includes arterial blood gas (ABG), continuous monitoring of electrocardiogram (ECG), heart rate (HR), invasive blood pressure (IBP), pulse oxygen saturation (SpO2), respiratory rate (RR), and pressure of end-tidal carbon dioxide (EtCO2) machine to enable spontaneous breathing. Oxygen (4–5 L/min). Support ventilation (PSV) is used to prevent lung collapse and support breathing at the operation's end. Stop Propofol immediately after skin closure and extubate when spontaneous breathing is above 5 ml/kg (ideal body weight) and BIS is above 80; Postoperative outcomes: Length of hospital stay, postoperative pain scores, complications (e.g., bleeding, infection).
Statistical Analysis: The collected data is processed and analyzed using SPSS software version 16.0. Quantitative variables are presented as mean ± standard or median deviation (quartile) and compared using the t-student or Mann-Whitney U test. Pearson correlation coefficient is used to evaluate the correlation. Qualitative variables, described by frequency (%), are compared using the Chi squared test or Fisher's exact test; relevance is assessed by the Phi coefficient and Cramer's V. Choose P < 0.05 as the level for statistically significant differences.
3. RESULTS
No patients were excluded from the analysis after being randomized. The results showed no statistically significant differences in demographic factors, e.g., gender, age, height, weight, and BMI between the two groups. The ASA status, smoking rate, and the primary diagnosis and comorbidities were also similar.
| Table 1: Demographic characteristics of the patients | ||||
| Variables | Group 1
(n = 50) |
Group 2
(n = 50) |
P-value | |
| Gender | Male | 22 (44%) | 28 (56%) | 0.230 |
| Female | 28 (56%) | 22 (44%) | ||
| Age (year) | 43.94 ± 13.63 (19- 60) |
46.94 ± 12.56 (21- 60) |
0.2610 | |
| Height (cm) | 161.44 ± 7.87 | 161.16 ± 7.50 | 0.4279 | |
| Weight (kg) | 56.66 ± 7.81 | 54.36 ± 9.99 | 0.0798 | |
| BMI (kg/m2) | 21.714 ± 2.228 | 20.886 ± 2.743 | 0.052 | |
| ASA I | 27 (54%) | 22 (44%) | 0.317 | |
| ASA II | 23 (46%) | 28 (56%) | ||
| Smoker | 5 (10%) | 11 (22%) | 0.102 | |
| Diagnosis | ||||
| Lung tumor | 18 (36%) | 18 (36%) | 0.773 | |
| Mediastinal tumors | 23 (46%) | 20 (40%) | ||
| Pulmonary cyst | 8 (16%) | 9 (18%) | ||
| Others | 1 (2%) | 3 (6%) | ||
| Comorbidity | ||||
| No systemic disease | 35 (70%) | 28 (56%) | 0.288 | |
| Pulmonary disease | 1 (2%) | 1 (2%) | ||
| Hypertension | 3 (6%) | 4 (8%) | ||
| Diabetes mellitus | 3 (6%) | 1 (2%) | ||
| Others | 8 (16%) | 16 (32%) | ||
| Data presented as n (%) or mean ± SD; P < 0.05 considered as significant | ||||
Table 2 shows that the types of surgery, surgery times, and lung collapse scores of the two groups are statistically not different.
| Table 2: Some intraoperative profiles and postoperative outcomes | |||
| Variables | Group 1
(n = 50) |
Group 2
(n = 50) |
P-value |
| Anesthetic induction duration (min) | 3.75 ± 0.83 | 15.58 ± 7.39 | < 0.001 |
| Time to one lung breathing | 84.42 ± 40.70 | 89.30 ± 42.04 | 0.4802 |
| Anesthetic time (min) | 130.26 ± 45.69 | 145.46 ± 47.50 | 0.0531 |
| Intraoperative fentanyl dose (mg) | 0.11 ± 0,03 | 0.21 ± 0.05 | < 0.001 |
| Intraoperative propofol dose (mg) | 916.94 ± 317,20 | 973.18 ± 355.21 | 0.2029 |
| Lung collapse scores after pleural cavity opening at 10 and 30 min | |||
| 10 min | 6.68 ± 0.51 | 6.74 ± 0.56 | 0.2896 |
| 30 min | 8.64 ± 0.48 | 8.48 ± 0.54 | 0.1752 |
| Surgery type | |||
| Lobectomy | 16 (32%) | (36%) | > 0.05 |
| Segmentectomy | 2 (4%) | 0 | |
| Mediastinal tumor removal | 23 (46%) | 22 (44%) | |
| Bullectomy | 8 (16%) | 9 (18%) | |
| Others | 1 (2%) | 1 (2%) | |
| Surgical duration (min) | 103.88 ± 42.99 | 109.26 ± 43.20 | 0.4654 |
| Extubation time (min) | 5.46 ± 2,05 | 15,60 ± 3.82 | < 0.001 |
| Postoperative | |||
| Aldrette score time (min) | 4.78 ± 1,62 | 10.48 ± 3.13 | < 0.001 |
| Chest tube drainage duration (days) | 4.22 ± 1.92 | 5.28 ± 2.74 | 0.0517 |
| Data presented as n (%) or mean ± SD; P < 0.05 considered as significant | |||
Time to onset of anesthesia in Group 1 was significantly shorter than in Group 2 (P < 0.001). The dose of fentanyl used in surgery in Group 1 was lower and statistically significant than that of Group 2 (P < 0.001). This may reflect a lower need for opioids in Group 1.
There were no statistically significant differences in time to one lung ventilation (P = 0.4802), total duration of anaesthesia (P = 0.0531 – asymptotic threshold), intraoperative propofol dose (P = 0.2029), or pulmonary collapse point at 10 min (P = 0.2896) and 30 min (P = 0.1752). Similarly, the types of surgery (P > 0.05) and the duration of surgery (P = 0.4654) also did not show a difference between the two groups.
However, another important difference was the time of intubation. Group 1 patients had a much shorter intubation time (5.46 ± 2.05 min) than Group 2 (15.60 ± 3.82 min) (P < 0.001), indicating significantly earlier recovery and spontaneous breathing.
In terms of postoperative parameters, the time to achieve the Aldrete score in Group 1 (4.78 ± 1.62 min) was statistically shorter than that of Group 2 (10.48 ± 3.13 min) (P < 0.001).
In Table 3 and Figure 1, we found many statistically significant differences between Group 1 and Group 2, reflecting certain advantages of each approach.
3.1. Ventilation changes during surgery
The data presented in Figure 1 and Table 3 indicate that the respiratory rate was comparable between the two groups during surgery. However, the tidal volume and minute ventilation in Group 2 are approximately 1.5 times greater than those in Group 1. Additionally, the airway pressure intraoperatively in Group 2 was higher (P < 0.05). Group 1, the highest peak airway pressure in 2 lungs was (4.06 ± 1.74 cmH₂O) and one lung ventilation (5.36 ± 1.66 cmH₂O) were significantly lower, almost 5 times, compared to Group 2 (P < 0.001). In contrast, the Peak EtCO₂ during operation in Group 1 was significantly higher than in Group 2 (P < 0.001). This was also reflected in PaCO₂ during the one lung ventilation phase, when Group 1 had a significantly higher PaCO₂ than Group 2 (P < 0.001). Higher PaCO₂ levels in Group 1 may be related to the characterization of spontaneous ventilation, where CO₂ emissions may not be as effective as positive pressure-controlled ventilation.




Figure 1: Ventilation and airway pressure changes during surgery
(Abbreviations: OLB, One lung breathing; MPCA, Minutes post-chest cavity access; ADR, Airway device removal; *, P-value < 0.05)
| Table 3. Ventilation and blood oxygenation | ||||
| Variables | Group 1
(n = 50) |
Group 2
(n = 50) |
P-value | |
| In-room SpO2 (%) | 98.68 ± 0,98 | 98.48 ± 0.97 | 0.154 | |
| Highest peak airway
pressure (cmH2O) |
2 lung ventilation | 4.06 ± 1,74 | 24.18 ± 3.94 | < 0.001 |
| One lung ventilation | 5.36 ± 1.66 | 30.42 ± 5.46 | < 0.001 | |
| Highest intraop FiO2 (%) | 61.22 ± 4.20 | 78.54 ± 16.46 | < 0.001 | |
| Lowest intraop FiO2 (%) | 97.30 ± 1,69 | 92.44 ± 6,62 | < 0.001 | |
| Peak intraop EtCO2 (mmHg) | 48.56 ± 4,11 | 40.96 ± 4.71 | < 0.001 | |
| Arterial blood gas (ABG)
data before one-lung breathing |
pH | 7.46± 0.047 | 7.47± 0.05 | 0.18 |
| PaO2 (mmHg) | 292.26 ± 75,77 | 271.69 ± 75.27 | 0.088 | |
| PaO2/FiO2 | 503.20 ± 97.82 | 465.23 ± 126.36 | 0.048 | |
| PaCO2 (mmHg) | 36.62 ± 6,35 | 35.25 ± 7.23 | 0.168 | |
| AaDO₂ | 87.72 ± 51,99 | 108.57 ± 68.25 | 0.143 | |
| Lactate (mmol/l) | 1.35 ± 0.87 | 1.13 ± 0.75 | 0.083 | |
| Arterial blood gas data
during one-lung breathing |
pH | 7.38 ± 0.05 | 7.44 ± 0.06 | < 0.001 |
| PaO2 (mmHg) | 176.90 ± 60.87 | 121.73 ± 63.32 | < 0.001 | |
| PaO2/FiO2 | 294.49 ± 99.91 | 187.53 ± 107.22 | < 0.001 | |
| PaCO2 (mmHg) | 46.84 ± 6.90 | 38.54 ± 7.48 | < 0.001 | |
| AaDO₂ | 199.03 ± 69.42 | 315.65 ± 141.01 | < 0.001 | |
| Lactate | 1.09 ± 0.55 | 1.17 ± 0.77 | 0.977 | |
| Postoperative arterial blood gas data | pH | 7.42 ± 0.04 | 7.43± 0.03 | 0.049 |
| PaO2 (mmHg) | 184.65 ± 93.49 | 167.35 ± 60.55 | 0.682 | |
| PaO2/FiO2 | 579.77 ± 227.29 | 512.75 ± 157.74 | 0.140 | |
| PaCO2 (mmHg) | 38.15 ± 3.89 | 35.50 ± 5. | 0.006 | |
| AaDO₂ | 32.27 ± 17.82 | 92.25 ± 42.04 | < 0.001 | |
| Lactate | 1.08 ± 0.7 | 1.69 ± 1.03 | < 0.001 | |
| Data presented as mean ± SD; P < 0.05 considered as significant | ||||
Although in-room SpO₂ was no different (P = 0.154). Lowest SpO₂ during operation in Group 1 (97.30 ± 1.69%) was statistically higher than in Group 2 (92.44 ± 6.62%) (P < 0.001). This, along with lower FiO₂ use, showing a more stable and effective ability to maintain blood oxygenation in Group 1. The highest FiO₂ during operation in Group 1 (61.22 ± 4.20%) was also statistically lower than in Group 2 (78.54 ± 16.46%) (P < 0.001).
When considering arterial blood gas before one lung ventilation, the PaO₂/FiO₂ index in Group 1 was significantly higher than in Group 2 (P = 0.048), indicating better initial oxygen exchange. During one lung ventilation phase, this difference became even more pronounced: Group 1 had a significantly higher PaO₂ (P < 0.001) and PaO₂/FiO₂ (P < 0.001) than Group 2.
The alveolar-arterial oxygen pressure differential (AaDO₂) during one lung ventilation in Group 1 was significantly lower than in Group 2 (P < 0.001), indicating better ventilation-perfusion matching (V/Q matching) and fewer shunts in Group 1.
3.3. Postoperative outcomes
After surgery, the PaCO₂ of Group 1 remained significantly higher than that of Group 2 (P = 0.006). At the same time, the postoperative AaDO₂ in Group 1 was significantly lower than that of Group 2 (P < 0.001), further confirming better gas exchange efficiency. Postoperative lactate concentrations in Group 1 were significantly lower than in Group 2 (P < 0.001), suggesting better tissue perfusion and aerobic metabolism in Group 1.
4. DISCUSSION
This study compared intraoperative ventilation and oxygenation parameters between non-intubated video-assisted thoracic surgery (NI-VATS) using an LMA with spontaneous/assisted ventilation and conventional intubated VATS (I-VATS) with one-lung ventilation (OLV). Our findings demonstrate several significant advantages in the NI-VATS group, primarily related to lower airway pressures, better oxygenation efficiency with lower FiO2, and faster immediate postoperative recovery, despite higher but clinically manageable CO2 levels.
4.1. Ventilation changes during surgery
A distinctive aspect of our NI-VATS protocol lies in the use of the LMA for airway management during spontaneous/assisted ventilation, as opposed to solely relying on nasal oxygen delivery. While other centers often perform NI-VATS with high-flow nasal oxygen or epidural anesthesia, our approach with the LMA offers several critical advantages, especially relevant in our clinical context. The LMA allowed for precise monitoring of respiratory parameters such as EtCO2 and tidal volume (Vt), which is challenging with mask or nasal oxygen alone. Furthermore, it provided a secure airway, enabling easy transition to assisted ventilation or even intubation through the LMA if needed, without interrupting the surgical field. Additionally, being able to reinflate the lung after surgery easily also offers advantages.
4.2. Breathing rate, Vt, and MV
In our study, the breathing rate was comparable between the two groups during most of the surgery. However, the I-VATS group (Group 2) had significantly higher tidal volumes and, consequently, minute ventilation compared to the NI-VATS group (Group 1). I-VATS group typically aims for and achieves higher Vt and MV, which is intended to help patients avoid hypercapnia. While it is important to recognize that the implementation of OLV can lead to a reduction in tidal volume within the double-lumen group, resulting in increased CO2 levels in the blood, this assertion must be considered within a broader context. In contrast, OLV presents its own set of challenges; controlled ventilation strategies typically strive to maintain an adequate tidal volume, although adjustments may be necessary for the ventilated lung. Interestingly, in our I-VATS group, both tidal volume and minute ventilation were approximately 1.5 times higher than those in the NI-VATS group. This discrepancy may be attributed to the administration of propofol and fentanyl, along with one-lung breathing during surgery.
4.3. Perioperative pH
Before starting OLV, the mean pH in both groups tended towards mild alkalosis (7.46 in Group 1 and 7.47 in Group 2). These values are higher than the average physiological range (7.35-7.45), but not statistically significant (P = 0.18). This can likely be explained by patient anxiety and stress before surgery, leading to an increased respiratory rate, which in turn causes a decrease in PaCO₂ and results in mild respiratory alkalosis. Intraoperative and Postoperative period results show a statistically significant difference in pH between the two groups during and after the surgical process. During OLV, the pH in Group 1 (NI-VATS) was lower than in Group 2 (I-VATS). Postoperatively, this trend continued, although the difference was more minor, with a mean pH in Group 1 of 7.42 compared to 7.43 in Group 2 (P = 0.049). The decrease in pH in Group 1 is directly related to the significantly higher PaCO₂ levels in this group at the same time points. During OLV, the PaCO₂ of Group 1 was 46.84 mmHg, much higher than the 38.54 mmHg of Group 2 (P < 0.001). This state of elevated CO₂ is a characteristic of the spontaneous ventilation method in thoracic surgery and is known as "permissive hypercapnia". The state of hypercapnia and the resulting mild reduction in pH in the NI-VATS group were well-controlled and did not cause adverse effects.
4.4. PaCO2 and EtCO2
The NI-VATS group exhibited significantly higher peak EtCO2 (P < 0.001). PaCO2 during OLV comparable to the I-VATS group (P < 0.001). This permissive hypercapnia is common in patients who are spontaneously ventilating during thoracic surgery and, if maintained within safe limits, as in our NI-VATS group, may be well tolerated. Liu et al.4 in their study of spontaneous pneumothorax, also found higher PetCO2 (likely meant EtCO2) and PaCO2 in their NI-VATS group. The higher PaCO2 levels in the NI-VATS group may be related to the spontaneous ventilation. Postoperatively, PaCO2 in the NI-VATS group remained significantly higher than in the I-VATS group (P = 0.006). There have been reports indicating that an increase in blood CO2 levels up to 100 mmHg can be acceptable.5 A slight rise in CO2 is generally not a concern; in fact, it can sometimes enhance tissue perfusion and shorten recovery time.6 Permissive hypercapnia is contraindicated in patients with elevated intracranial pressure (ICP) or severe cardiopulmonary conditions like pulmonary hypertension, due to risks of CO₂-induced cerebral vasodilation and increased pulmonary vascular resistance. Our study shows that this permissive hypercapnia was clinically manageable and associated with improved tissue perfusion and aerobic metabolism, as indicated by lower postoperative lactate levels (Table 3).
4.5. Airway pressure
Peak airway pressure was significantly lower in the NI-VATS group during both two-lung ventilation (P < 0.001) and in OLV (P < 0.001). While some physiological changes were observed, such as lower peak airway pressures and higher CO2 levels in the NI-VATS group, which might be anticipated with spontaneous ventilation. Our results demonstrate that the peak airway pressure in NI-VATS was nearly five times lower than in I-VATS. This substantial reduction directly translates into a significant clinical benefit, reducing barotrauma and ventilator-induced lung injury (VILI), a concern with conventional OLV.4 Although our study did not specifically measure VILI markers, lower airway pressure is inherently protective.
4.6. Blood oxygenation parameters
4.6.1. FiO2 and Oxygen Saturation (SpO2)
Our findings indicate better oxygenation efficiency in the NI-VATS group. The mean peak FiO2 required during the operation was significantly lower in the NI-VATS group compared to the I-VATS group (P < 0.001). Despite this lower FiO2, the NI-VATS group demonstrated significantly higher lowest SpO2 during the operation (P < 0.001). This suggests a more stable and effective ability to maintain blood oxygenation in the NI-VATS group. Ke HH, et al (2022) reported a 5% incidence of hypoxemia (SpO2 < 90%) during VATS with OLV,4 whereas our NI-VATS group maintained higher SpO2 levels with less supplemental oxygen. The key result demonstrates the enhanced oxygenation efficiency of the LMA-based NI-VATS approach—a notable advantage, given that the I-VATS group had to manage the inherent difficulties of OLV. The maintained spontaneous diaphragmatic motion and negative intrapleural pressure with LMA likely contribute to better ventilation/perfusion (V/Q) matching and reduced intrapulmonary shunting. Therefore, our findings, while aligning with certain physiological principles, provide compelling quantitative data supporting the safety, efficiency, and clinical advantages of LMA-based NI-VATS as a valuable and applicable anesthetic technique for VATS, especially in contexts where conventional intubation carries higher risks or specific pharmacological agents are limited.
4.6.2. PaO2/FiO2 Ratio (P/F Ratio)
The physiological basis for improved oxygenation in NI-VATS, despite OLV (simulated by pathological pneumothorax), is thought to lie in the maintenance of spontaneous diaphragmatic motion and more negative intrapleural pressure during inspiration. This helps maintain functional residual capacity (FRC) to a greater extent than in paralyzed, mechanically ventilated patients, leading to better ventilation/perfusion (V/Q) matching and reduced intrapulmonary shunting.7 Our results support this theory. Prior to OLV, the PaO2/FiO2 ratio in the NI-VATS group (503.20 ± 97.82) was significantly higher than in the I-VATS group (465.23 ± 126.36, P = 0.048). During the OLV phase, this difference became even more pronounced: the NI-VATS group had a significantly higher PaO2 (mean 176.90 ± 60.87 mmHg vs. 121.73 ± 63.32 mmHg, P < 0.001) and PaO2/FiO2 ratio (mean 294.49 ± 99.91 vs. 187.53 ± 107.22, P < 0.001) than the I-VATS group. This is consistent with the review by Fabo et al.7 which highlighted improved V/Q matching and gas exchange as benefits of spontaneous ventilation.
4.6.3. Alveolar-Arterial Oxygen Gradient (AaDO2)
Furthermore, the AaDO2 was significantly lower during one-lung breathing in our NI-VATS group (P < 0.001) and postoperatively (P < 0.001). This suggests more efficient gas exchange and less shunting, consistent with the theoretical advantages of preserved spontaneous ventilation.
Interestingly, postoperative lactate levels were significantly lower in our NI-VATS group (P < 0.001), indicating better tissue perfusion and oxygen delivery, possibly due to the Bohr effect from mild hypercapnia increasing oxygen release to the tissues. Shorter anesthesia induction time (3.75 vs. 15.58 min, P < 0.001), significantly lower intraoperative fentanyl requirement (P < 0.001), significantly shorter extubation time (P < 0.001), and faster achievement of full Aldrete score (P < 0.001) in the NI-VATS group, highlighting the principles of enhanced recovery after surgery.
These findings are consistent with the literature showing that NI-VATS facilitates faster recovery.1 The reduced opioid use may also contribute to a faster return of spontaneous respiration and less postoperative sedation. Comparing our results with other studies, Chen et al. (2012) reported successful intubation-free lung resections using epidural anesthesia and vagal blockade, demonstrating the feasibility of complex procedures without intubation.8 Our study, using LMA and TCI propofol, further supports the adaptability of intubation-free techniques. Huang et al. (2020) found NI-VATS to be safe for mediastinal tumor resection in patients with impaired pulmonary function, with no serious complications in the SV-VATS group.9 While our study focused on patients with generally preserved pulmonary function (ASA I-II), the observed safety profile is consistent with their finding that NI-VATS was well tolerated. Li et al. (2020) also demonstrated the feasibility of intubation-free anesthesia with regional block for thoracoscopic surgery, reinforcing the overall safety and applicability of these approaches.2 The broad applicability and safety of NI-VATS in a variety of thoracic procedures and patient populations become increasingly evident when these studies are considered as a whole.
5. Clinical Implications
Our results support NI-VATS as a viable option for patients with preserved respiratory function (ASA I–II, FEV₁ > 50%). The technique demonstrated several clinical advantages, including adequate ventilation, lower airway pressure, better blood oxygenation with lower FiO₂, reduced opioid consumption, and faster recovery after surgery, which have significant clinical implications. However, NI-VATS may not be suitable in some cases, such as a difficult airway, obesity (BMI ≥ 30), or severe comorbidities. The success of this technique relies on meticulous anesthetic management, including using a laryngeal mask and pressure ventilation support in cases of hypoxemia (SpO₂ <90%).
6. LIMITATIONS
This study has several limitations. First, it was conducted at a single center with a specific patient population (ASA I-II, BMI <30, FEV₁ >50%), which may limit the generalizability of the findings to patients with more severe comorbidities, higher BMI, or significantly impaired lung function. Finally, the study was not blinded to the anesthesiologists providing care, which is inherent in comparing two distinct anesthetic techniques, but could introduce performance bias.
7. CONCLUSION
Using NI-VATS, tidal volume and minute ventilation decrease slightly, and CO2 increases as compared to endotracheal intubation. In contrast, airway pressure and blood oxygenation are better compared to the double-lumen intubation method. NI-VATS is also a safe and feasible method that can provide physiological benefits and contribute to the recovery process in appropriately chosen patients. Further research is warranted to evaluate long-term outcomes and expand the application of this technique to a broader range of patients and surgical complexities.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Ethical Issues
The research has the purpose of protecting and improving patients' health and has no other purpose; it was conducted with the patient's consent. They will be treated (if there are unwanted effects) and consulted about research issues, and we will keep confidential the information provided by patients. The study was approved by the ethics committee of Viet Duc University Hospital (March 21st, 2022) and Hanoi Medical University (Ref 659/GCN-HĐĐĐNCYSH-ĐHYHN dated June 7th, 2022).
9. Conflict of interest
All authors declare that there was no conflict of interest.
10. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
11. Authors’ contribution
TKD: conceptualization, writing-original draft, methodology, investigation, data analysis.
PHL: Supervision, methodology, validation, writing-review & editing
NQK: Supervision, conceptualization, methodology, writing-review & editing
LQT, DDH: writing-original draft, data analysis, writing-review
12. REFERENCES
- Huang WT, Cheng YJ, Chen JS. Video-assisted thoracoscopic surgery lobectomy for lung cancer in nonintubated anesthesia. Thorac Surg Clin. 2020 Feb;30(1):73-82. [PubMed] DOI: 1016/j.thorsurg.2019.09.002
- Li H, Huang D, Qiao K, Wang Z, Xu S. Feasibility of non-intubated anesthesia and regional block for thoracoscopic surgery under spontaneous respiration: a prospective cohort study. Braz J Med Biol Res. 2020;53(1):e8645. [PubMed] DOI: 1590/1414-431X20198645
- Ke HH, Hsu PK, Tsou MY, Ting CK. Nonintubated video-assisted thoracic surgery with high-flow oxygen therapy shorten hospital stay. J Chin Med Assoc. 2020 Oct;83(10):943-9. [PubMed] DOI: 1097/JCMA.0000000000000408
- Liu J, Liang H, Cui F, Liu H, Zhu C, Liang W, et al. Spontaneous versus mechanical ventilation during video-assisted thoracoscopic surgery for spontaneous pneumothorax: a randomized trial. J Thorac Cardiovasc Surg. 2022 May;163(5):1702-14.e7. [PubMed] DOI: 1016/j.jtcvs.2021.01.093
- Morisaki H, Serita R, Innami Y, Kotake Y, Takeda J. Permissive hypercapnia during thoracic anaesthesia. Acta Anaesthesiol Scand. 1999 Sep;43(8):845-9. [PubMed] DOI: 1034/j.1399-6576.1999.430811.x
- Petran J, Ansems K, Rossaint R, Marx G, Kalvelage C, Kopp R, et al. Effects of hypercapnia versus normocapnia during general anesthesia on outcomes: a systematic review and meta-analysis. Braz J Anesthesiol. 2022 May-Jun;72(3):398-406. [PubMed] DOI: 1016/j.bjane.2020.11.010
- Fabo C, Oszlányi Á, Barta Z, et al. Anesthesiology of the spontaneous ventilation in thoracic surgery: a narrative review. AME Surg J. 2022;2:14. DOI: 21037/asj-21-22
- Chen KC, Cheng YJ, Hung MH, Tseng YD, Chen JS. Nonintubated thoracoscopic lung resection: a 3-year experience with 285 cases in a single institution. J Thorac Dis. 2012 Aug;4(4):347-51. [PubMed] DOI: 3978/j.issn.2072-1439.2012.08.07
- Huang W, Deng H, Lan Y, Wang R, Ge F, Huo Z, et al. Spontaneous ventilation video-assisted thoracic surgery for mediastinal tumor resection in patients with pulmonary function deficiency. Ann Transl Med. 2020 Nov;8(21):1444. [PubMed] DOI: 21037/atm-20-1652