Lakshmi Ambali 1 , Prabhu Thilaak 2 , Rasika Priya M. 3 , Brindha R. 4 , V. A. Sabapathy 5 , Panneerselvam Periasamy 6 , Arbind Kumar Choudhary 7
Authors contribution
Background & objective: Spinal anesthesia is commonly used for lower limb and abdominal surgeries, but its effectiveness is limited by the duration of action of local anesthetics. To prolong analgesia and improve intraoperative conditions, adjuvants such as clonidine and fentanyl are added to local anesthetics. This study aims to compare the efficacy and safety of clonidine and fentanyl as adjuvants to ropivacaine in spinal anesthesia, focusing on sensory and motor blockade duration, postoperative analgesia, hemodynamic changes, and adverse effects.
Methodology: A randomized, double-blind, controlled trial was conducted on 75 patients undergoing elective lower abdominal and lower limb surgeries. Patients were randomly assigned to three groups: Group A: ropivacaine 0.25%, Group B: ropivacaine + clonidine 5 µg, and Group C: ropivacaine + fentanyl 15 µg. The onset and duration of sensory and motor blockade were assessed using the pinprick method and Modified Bromage Scale. Hemodynamic parameters (heart rate, systolic and diastolic blood pressure) were monitored at regular intraoperative intervals. The time to first rescue analgesia and VAS pain scores were recorded at 1, 6, 12, and 24 hours. Adverse effects, including sedation, nausea, hypotension, and bradycardia, were recorded. Statistical analysis was performed using ANOVA and Chi-square tests in SPSS version [X]. P < 0.05 was considered significant.
Results: Group B (clonidine group) had the longest sensory and motor blockade duration, followed by Group C (Fentanyl), with Group A showing the shortest duration (p < 0.0001). Time to first rescue analgesia was significantly prolonged in Group B (528.8 ± 12.2 min) compared to Group C (422.1 ± 13.9 min) and Group A (203.3 ± 16.4 min) (P < 0.0001). Hemodynamic instability was more pronounced in Group B, with a higher incidence of hypotension and bradycardia, whereas Group C had a greater incidence of mild sedation and bradycardia. Sedation was significantly higher in Group B (P = 0.0033), while nausea and pruritus were more frequent in Group A. Group A required significantly more rescue analgesia doses compared to Groups B and C (P < 0.0001).
Conclusions: Clonidine as an adjuvant to ropivacaine in spinal anesthesia significantly prolongs sensory and motor blockade and enhances postoperative analgesia, but is associated with greater sedation and hemodynamic instability. Fentanyl also improves analgesia with a lower risk of hypotension but increases bradycardia incidence.
Abbreviations: DBP: diastolic blood pressure, HR: heart rate, SA: Spinal anesthesia, SBP: systolic blood pressure,
Keywords: Spinal Anesthesia; Ropivacaine; Clonidine; Fentanyl; Postoperative Analgesia; Hemodynamic Stability; Adverse Effects; Regional Anesthesia
Citation: Ambali L, Thilaak P, Priya MR, Brindha R, Sabapathy VA, Periasamy P, Choudhary AK. Comparative study of clonidine and fentanyl as adjuvants to ropivacaine in spinal anesthesia: impact on analgesic efficacy, hemodynamic stability, and adverse effects. Anaesth. pain intensive care 2025;29(6):528-534; DOI: 10.35975/apic.v29i6.2903
Received: March 05, 2025; Revised: May 06, 2025; Accepted: May 15, 2025
Spinal anesthesia (SA) is a widely used regional anesthetic technique, particularly in lower limb and lower abdominal surgeries. It provides effective sensory and motor blockade, excellent intraoperative analgesia, and a lower risk of systemic complications compared to general anesthesia (Routray et al., 2017). However, the limited duration of analgesia and early regression of the anesthetic effect remain challenges in clinical practice.1 To address this, adjuvants such as clonidine and fentanyl are frequently added to local anesthetics to enhance the quality of anesthesia, prolong its duration, and improve postoperative pain relief (Sharan et al., 2016). Ropivacaine, a long-acting amide local anesthetic, is preferred for SA due to its favorable safety profile, lower cardiotoxicity, and reduced motor blockade compared to bupivacaine (Mahendru et al., 2013). However, like other local anesthetics, its effect duration is limited.2,3 The use of adjuvants such as clonidine (α2-adrenergic agonist) and fentanyl (opioid receptor agonist) can modulate spinal nociceptive pathways, providing prolonged analgesia and better hemodynamic stability (Schug et al., 2006). Clonidine acts by enhancing the inhibitory effects on nociceptive transmission in the dorsal horn, while fentanyl enhances analgesia through opioid receptor activation in the spinal cord. Both agents improve pain control but differ in sedation levels, hemodynamic effects, and adverse event profiles (Schwartz et al., 2022).
The choice of an ideal adjuvant remains a topic of debate, as both clonidine and fentanyl have advantages and limitations. Clonidine has been associated with prolonged sensory and motor block, effective postoperative analgesia, and sedation, making it beneficial in long-duration procedures. However, it may also lead to bradycardia, hypotension, and excessive sedation, necessitating careful perioperative monitoring. Fentanyl, on the other hand, is well known for potentiating intraoperative and early postoperative analgesia with minimal hemodynamic effects. However, it has been associated with a higher incidence of pruritus, nausea, and respiratory depression, which can impact patient comfort (Sharan et al., 2016).
Despite the frequent use of adjuvants in SA, there is no consensus on whether clonidine or fentanyl offers superior efficacy and safety in combination with ropivacaine. Existing studies provide conflicting evidence regarding their comparative benefits, and head-to-head trials evaluating their effects on sensory and motor blockade, postoperative analgesia, hemodynamic stability, and adverse effects are still limited.4,5
This study aims to provide a comprehensive comparison of clonidine and fentanyl as adjuvants to ropivacaine in SA, focusing on their impact on the onset and duration of sensory and motor blockade, postoperative pain relief, sedation, and adverse effects. By systematically evaluating these parameters, the study seeks to offer clinically relevant insights that can guide anesthesiologists in optimizing SA protocols while minimizing complications.
This study was conducted as a randomized, double-blind, controlled trial at Vinayaka Mission’s Kirupananda Variyar Medical College & Hospitals (VMKVMC&H) after obtaining approval from the Institutional Ethics Committee (IEC) (Approval No. VMKVMC&H/IEC/24/287). Written informed consent was obtained from all participants before enrollment. The study adhered to the ethical principles outlined in the Declaration of Helsinki (2013) and Good Clinical Practice (GCP) guidelines.
The study included patients scheduled for elective lower abdominal and lower limb surgeries under SA. A total of 75 patients were enrolled based on inclusion and exclusion criteria. Patients were aged 15 to 49 years and classified as American Society of Anesthesiologists (ASA) Physical Status I-II. Patients with known hypersensitivity to ropivacaine, clonidine, or fentanyl, severe cardiovascular, hepatic, renal, or neurological disorders, coagulopathy, pregnancy, or refusal to participate were excluded from the study.
Patients were randomly assigned to three groups using a computer-generated randomization sequence, ensuring equal allocation. The allocation sequence was concealed in opaque, sealed envelopes, which were opened just before administering SA. The study followed a double-blind design, where the anesthetist preparing the drug solutions was not involved in data collection, and the anesthesiologist administering SA and the patient were blinded to group assignment.
All patients were preloaded with 500 mL of Ringer’s lactate to prevent hypotension. Standard monitoring, including electrocardiography (ECG), non-invasive blood pressure (NIBP), and pulse oximetry (SpO₂), was applied. SA was performed under strict aseptic precautions in the L3-L4 or L4-L5 interspace using a 25G Quincke spinal needle with the patient in a sitting position. Once cerebrospinal fluid (CSF) flow was confirmed, the assigned drug combination was administered.
Group A (Control) received 15 mg of ropivacaine, Group B (clonidine) received 15 mg of ropivacaine + 30 μg of clonidine, and Group C (Fentanyl) received 15 mg of ropivacaine + 25 μg of fentanyl. After the injection, the patients were positioned in supine posture with oxygen supplementation at 3 L/min via nasal cannula.
Sensory and motor blockade parameters were recorded at baseline, then at 2-minute intervals until the highest sensory block level was achieved. Sensory block was assessed using the pinprick method, while motor blockade was evaluated using the Modified Bromage Scale. The duration of sensory blockade was measured from the time of administration until two-segment regression, and the duration of motor block was recorded until the Bromage Score returned to zero.
Hemodynamic parameters, including heart rate (HR), systolic blood pressure (SBP), and diastolic blood pressure (DBP), were recorded at baseline and intraoperatively at 0, 5, 10, 15, 30, 45, and 60 minutes. Any episodes of hypotension (SBP < 90 mmHg) and bradycardia (HR < 50 bpm) were documented and managed appropriately.
Postoperative analgesia was assessed by recording the time to the first rescue analgesia, defined as the interval from SA administration until the patient first requested additional pain relief. The number of rescue analgesia doses required within 24 hours was also recorded. Pain intensity was measured using the Visual Analog Scale (VAS) at 1, 6, 12, and 24 hours postoperatively.
Sedation was assessed using the Ramsay Sedation Scale at 10, 30, and 60 minutes after SA administration. Patients were monitored for adverse effects, including nausea, vomiting, hypotension, bradycardia, respiratory depression, shivering, and pruritus. Any complications were appropriately managed.
2.1. Statistical analysis
All collected data were analyzed using SPSS software version [X]. Continuous variables were presented as mean ± standard deviation (SD) and compared using a one-way ANOVA test followed by post hoc analysis for intergroup comparisons. Categorical variables were expressed as percentages and analyzed using the Chi-square test or Fisher’s exact test, as appropriate. P < 0.05 was considered statistically significant.
Figure 1 depicts the CONSORT flowchart illustrating the patient enrolment, randomization, allocation, follow-up, and analysis for this study. The flowchart visually represents the study methodology, ensuring clarity in participant distribution across the three groups.
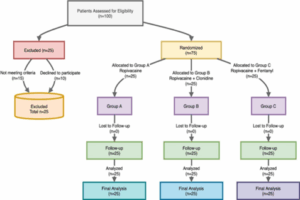
Figure 1: The CONSORT flow diagram of the study
Table 1 presents the mean and standard deviation of age, weight, ASA grade distribution, HR, SBP, and DBP across Groups A, B, and C. A statistical comparison using ANOVA for continuous variables and the Chi-square test for categorical variables was performed to determine any significant differences.
The demographic and baseline hemodynamic parameters were found to be statistically comparable among all three groups, indicating that any further differences in outcomes can be attributed to the effect of anesthetic regimens rather than baseline differences. No significant variation was observed in age, weight, ASA grade distribution, HR, SBP, and DBP across the groups (P > 0.05 for all variables).
To evaluate the efficacy of different anesthetic regimens, the onset, duration, and peak level of sensory and motor blockade were analyzed across Group A, Group B, and Group C. The table below summarizes the mean ± SD values for each parameter, along with statistical comparisons.
Onset of Sensory and Motor Block: The time required to achieve sensory and motor blockade was comparable among all three groups (P > 0.05, NS).
Peak Sensory Level: The most frequently achieved dermatomal level was T10 in all groups.
Duration of Sensory Block: Group B (clonidine + Ropivacaine) had the longest sensory block (P < 0.0001, significant), followed by Group C (Fentanyl + Ropivacaine), with Group A (Ropivacaine alone) showing the shortest duration.
Duration of Motor Block: Group B had the longest motor block duration, followed by Group C, with Group
A showing the shortest effect (P < 0.0001, significant). The mean sensory block onset time across the three groups;
Group A, B, and C, exhibited similar onset times, indicating no significant difference in the initiation of sensory blockade. The onset time of sensory blockade was comparable across all groups. The addition of clonidine (Group B) and Fentanyl (Group C) did not significantly alter the onset of sensory block compared to Group A.
Motor Block Onset and Recovery Time in 3 Groups:
Group B exhibited the fastest onset and longest duration of motor block, followed by Group C, while Group A had the shortest duration.
Motor onset time was slightly faster in Group B (clonidine) compared to Groups A and C.
Motor block duration was significantly prolonged in Group B, indicating that clonidine contributes to extended motor blockade.
Group A had the shortest motor block duration, suggesting that Ropivacaine alone produces a shorter motor blockade.
Criteria for Motor Blockade Assessment
The Modified Bromage Scale is used to assess the degree of motor blockade following SA. The table below outlines the criteria used in this study.
Grade 0: No motor impairment, indicating full recovery.
Grade 1: Mild weakness, but still able to move some joints.
Grade 2: Significant impairment, only slight movement possible.
Grade 3: Complete loss of motor function in the assessed limb.
All three groups exhibited a gradual decline in HR over time. Group B (clonidine) had a more pronounced reduction in HR, suggesting its additional hemodynamic stability effects. Group C (Fentanyl) maintained a relatively stable HR, whereas Group A (Ropivacaine alone) showed moderate variation. The trend highlights the potential bradycardic effect of clonidine, requiring careful monitoring.
Group B (clonidine) showed a greater reduction in SBP and DBP, indicating its vasodilatory effect. Group A (Ropivacaine alone) and Group C (Fentanyl) exhibited relatively stable blood pressure trends. DBP followed a similar trend to SBP, confirming consistent hemodynamic changes in each group. These variations suggest that clonidine has a significant impact on blood pressure regulation, potentially requiring additional hemodynamic support in susceptible patients.
Hypotension was observed more frequently in Group B (clonidine: 24.0%) and Group C (Fentanyl: 20.0%), compared to Group A (Ropivacaine alone: 12.0%). Bradycardia was most frequently observed in Group C (20.0%); whereas Groups A (4.0%) and B (8.0%) had lower incidences. Chi-square test for hypotension (P = 0.541) suggests no significant difference among the groups. Chi-square test for bradycardia (P = 0.162) suggests no significant difference in bradycardia frequency across groups.
Although not statistically significant, Group B (clonidine) showed a higher tendency for hypotension, requiring closer hemodynamic monitoring. Group C (Fentanyl) exhibited the highest incidence of bradycardia, indicating a potential depressant effect on HR.
Group B (clonidine) had the longest pain relief duration, followed by Group C (Fentanyl). Group A (Ropivacaine alone) required the earliest rescue analgesia, indicating a shorter duration of pain relief. The addition of adjuvants (clonidine or Fentanyl) significantly prolonged analgesia, reducing the need for early intervention.
Sedation was significantly higher in Group B, affecting more than half of the patients, while Group C had a moderate incidence, and Group A had the least. Hypotension and bradycardia occurred more frequently in Groups B and C, with Group C showing the highest rate of bradycardia. Though these differences were not statistically significant, they highlight the potential hemodynamic impact of clonidine and fentanyl as adjuvants. Table 5 presents the distribution of sedation scores across Group A (Ropivacaine alone), Group B (Ropivacaine + Clonidine), and Group C (Ropivacaine + Fentanyl).
Nausea and pruritus were reported more often in Group A, whereas their occurrence was lower in the other groups. Shivering and respiratory depression were infrequent across all groups without notable differences. The findings suggest that clonidine is associated with more sedation and hypotension, while fentanyl may contribute to a higher incidence of bradycardia, emphasizing the need for individualized anesthetic planning and careful perioperative monitoring.
Group B exhibited the highest sedation levels, with over half of the patients showing drowsiness (56%), confirming clonidine’s significant sedative effect. Groups A and C had lower sedation scores, with most patients remaining awake (84-80%). The difference in sedation was statistically significant, highlighting clonidine’s role in enhanced sedation compared to ropivacaine alone or fentanyl.
The use of adjuvants in SA has been widely studied to improve analgesic quality, prolong the duration of sensory and motor blockade, and optimize patient outcomes. This study compared the effects of ropivacaine alone (Group A), ropivacaine + clonidine (Group B), and ropivacaine + fentanyl (Group C), evaluating their impact on sensory and motor blockade, hemodynamic stability, analgesic efficacy, and adverse effects.
The time required to achieve sensory and motor blockade was comparable among all three groups (P > 0.05), indicating that the addition of adjuvants did not significantly alter the onset time of anesthesia. However, the duration of both sensory and motor block was significantly longer in Group B (clonidine) compared to Groups A and C (P < 0.0001). These findings are consistent with previous studies, which have reported that clonidine, an α2-adrenergic agonist, prolongs anesthesia by inhibiting norepinephrine release and reducing afferent pain signal transmission (Sharan et al., 2016). Similarly, Group C (Fentanyl) also showed an extended duration of sensory and motor block compared to Group A, though not as pronounced as in Group B.6,7 This effect can be attributed to fentanyl’s synergistic action on opioid receptors in the spinal cord, enhancing analgesic potency (Routray et al., 2017).
Hemodynamic variations were observed among the groups, particularly in HR and blood pressure trends during the intraoperative period. Group B exhibited a more pronounced reduction in HR compared to Groups A and C, suggesting an additional bradycardic effect associated with clonidine. This aligns with prior research demonstrating clonidine’s sympatholytic action, leading to reduced HR and cardiac output (Schug et al., 2006). Similarly, mean SBP and DBP was significantly lower in Group B, reinforcing clonidine’s vasodilatory properties. These findings emphasize the need for close hemodynamic monitoring when using clonidine as an adjuvant.
In contrast, Group C (Fentanyl) exhibited a more stable HR trend but had the highest incidence of bradycardia (20%), which may be linked to fentanyl’s central depressant effects on vagal tone. These findings align with those of Mahendru et al. (2013), who noted a higher incidence of bradycardia when fentanyl was used as an adjuvant in neuraxial anesthesia.8,9
Postoperative Analgesia and Pain Scores
A key advantage of using adjuvants in SA is the prolongation of postoperative analgesia, reducing the need for rescue analgesics. Group B showed the longest time to first rescue analgesia (528.8 ± 12.2 min), significantly longer than Group C (422.1 ± 13.9 min) and Group A (203.3 ± 16.4 min, p < 0.0001). Similarly, Group B required the least number of additional analgesic doses within 24 hours, further supporting the superior analgesic effect of clonidine. These findings correlate with those of Sharan et al. (2016), who found that clonidine significantly prolonged postoperative analgesia compared to fentanyl or local anesthetics alone.
Although VAS pain scores were comparable across groups at 1, 6, 12, and 24 hours postoperatively, the prolonged analgesia duration in Groups B and C highlights the opioid-sparing potential of these adjuvants, which may reduce the need for systemic opioids in the postoperative period.10,11
Adverse Effects
The incidence of hypotension and bradycardia was higher in Groups B and C compared to Group A. While these differences were not statistically significant (P > 0.05), they suggest that both clonidine and fentanyl may contribute to hemodynamic instability. Hypotension was more frequent in Group B (24%) and Group C (20%), aligning with previous findings on clonidine-induced vasodilation and fentanyl’s depressive effects on sympathetic tone (Schwartz et al., 2022).
Sedation was significantly higher in Group B (56%, P = 0.0033), confirming clonidine’s central sedative effects. In contrast, Group A had the least sedation (16%), and Group C had moderate levels (20%), consistent with fentanyl’s mild sedative properties. The heightened sedation with clonidine necessitates careful postoperative monitoring, particularly in elderly patients or those at risk for respiratory depression (Schug et al., 2006).
Other adverse effects, such as nausea and pruritus, were more frequent in Group A (24%), whereas their occurrence was lower in the other groups. These findings suggest that the addition of adjuvants may reduce nausea and pruritus, possibly by altering opioid receptor activity in the spinal cord.12,13,145
This study confirms that the addition of clonidine or fentanyl to ropivacaine enhances the quality of spinal anesthesia by prolonging sensory and motor block, improving postoperative analgesia, and reducing systemic analgesic requirements. However, clonidine is associated with significant sedation and hypotension, while fentanyl increases the risk of bradycardia. These findings support the selective use of adjuvants based on individual patient profiles and surgical requirements, ensuring optimized pain control while minimizing complications.
7. Data availability
The numerical data generated during this research are available from the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
LA: Concept and study design, data collection, manuscript drafting, and statistical analysis.
PT: Study supervision, methodology validation, and manuscript review.
RPM: Literature review, manuscript editing, and data interpretation.
BR: Critical revision of the manuscript, ethical approval coordination, and patient recruitment.
VAS: Corresponding author, overseeing manuscript finalization and study coordination.
PP: Statistical analysis, data validation, and reference management.
Authors contribution
- Lakshmi Ambali, Postgraduate Trainee, Department of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College & Hospitals, VMRF, Salem, India; Email: lakshmiambali@gmail.com; ORCID: {0009-0008-3158-3997}
- Prabhu Thilaak, MD, Professor of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College & Hospitals, VMRF, Salem, India; Email: prabhuthilaak@gmail.com; ORCID: {0009-0003-7452-2602}
- Rasika Priya M. MD, Associate Professor of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College & Hospitals, VMRF, Salem, India; Email: rasikamadhan1985@gmail.com; ORCID: {0009-0005-7763-5850}
- Brindha R., MD, DA, HOD, Professor of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College & Hospitals, VMRF, Salem, India; Email: mskbrins63@gmail.com; ORCID: {0000-0002-9776-2700}
- V.A. Sabapathy, MD, Professor of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College & Hospitals, VMRF, Salem, India; Email: sabapathyv.appavoo@gmail.com; ORCID: {0000-0002-3417-0222}
- Panneerselvam Periasamy, Assistant Professor of Physiology, Government Erode Medical College and Hospital, Erode, Tamil Nadu, India; Email: pannphysio@gmail.com; ORCID: {0000-0002-3358-313X}
- Arbind Kumar Choudhary
ABSTRACT
Background & objective: Spinal anesthesia is commonly used for lower limb and abdominal surgeries, but its effectiveness is limited by the duration of action of local anesthetics. To prolong analgesia and improve intraoperative conditions, adjuvants such as clonidine and fentanyl are added to local anesthetics. This study aims to compare the efficacy and safety of clonidine and fentanyl as adjuvants to ropivacaine in spinal anesthesia, focusing on sensory and motor blockade duration, postoperative analgesia, hemodynamic changes, and adverse effects.
Methodology: A randomized, double-blind, controlled trial was conducted on 75 patients undergoing elective lower abdominal and lower limb surgeries. Patients were randomly assigned to three groups: Group A: ropivacaine 0.25%, Group B: ropivacaine + clonidine 5 µg, and Group C: ropivacaine + fentanyl 15 µg. The onset and duration of sensory and motor blockade were assessed using the pinprick method and Modified Bromage Scale. Hemodynamic parameters (heart rate, systolic and diastolic blood pressure) were monitored at regular intraoperative intervals. The time to first rescue analgesia and VAS pain scores were recorded at 1, 6, 12, and 24 hours. Adverse effects, including sedation, nausea, hypotension, and bradycardia, were recorded. Statistical analysis was performed using ANOVA and Chi-square tests in SPSS version [X]. P < 0.05 was considered significant.
Results: Group B (clonidine group) had the longest sensory and motor blockade duration, followed by Group C (Fentanyl), with Group A showing the shortest duration (p < 0.0001). Time to first rescue analgesia was significantly prolonged in Group B (528.8 ± 12.2 min) compared to Group C (422.1 ± 13.9 min) and Group A (203.3 ± 16.4 min) (P < 0.0001). Hemodynamic instability was more pronounced in Group B, with a higher incidence of hypotension and bradycardia, whereas Group C had a greater incidence of mild sedation and bradycardia. Sedation was significantly higher in Group B (P = 0.0033), while nausea and pruritus were more frequent in Group A. Group A required significantly more rescue analgesia doses compared to Groups B and C (P < 0.0001).
Conclusions: Clonidine as an adjuvant to ropivacaine in spinal anesthesia significantly prolongs sensory and motor blockade and enhances postoperative analgesia, but is associated with greater sedation and hemodynamic instability. Fentanyl also improves analgesia with a lower risk of hypotension but increases bradycardia incidence.
Abbreviations: DBP: diastolic blood pressure, HR: heart rate, SA: Spinal anesthesia, SBP: systolic blood pressure,
Keywords: Spinal Anesthesia; Ropivacaine; Clonidine; Fentanyl; Postoperative Analgesia; Hemodynamic Stability; Adverse Effects; Regional Anesthesia
Citation: Ambali L, Thilaak P, Priya MR, Brindha R, Sabapathy VA, Periasamy P, Choudhary AK. Comparative study of clonidine and fentanyl as adjuvants to ropivacaine in spinal anesthesia: impact on analgesic efficacy, hemodynamic stability, and adverse effects. Anaesth. pain intensive care 2025;29(6):528-534; DOI: 10.35975/apic.v29i6.2903
Received: March 05, 2025; Revised: May 06, 2025; Accepted: May 15, 2025
1. INTRODUCTION
Spinal anesthesia (SA) is a widely used regional anesthetic technique, particularly in lower limb and lower abdominal surgeries. It provides effective sensory and motor blockade, excellent intraoperative analgesia, and a lower risk of systemic complications compared to general anesthesia (Routray et al., 2017). However, the limited duration of analgesia and early regression of the anesthetic effect remain challenges in clinical practice.1 To address this, adjuvants such as clonidine and fentanyl are frequently added to local anesthetics to enhance the quality of anesthesia, prolong its duration, and improve postoperative pain relief (Sharan et al., 2016). Ropivacaine, a long-acting amide local anesthetic, is preferred for SA due to its favorable safety profile, lower cardiotoxicity, and reduced motor blockade compared to bupivacaine (Mahendru et al., 2013). However, like other local anesthetics, its effect duration is limited.2,3 The use of adjuvants such as clonidine (α2-adrenergic agonist) and fentanyl (opioid receptor agonist) can modulate spinal nociceptive pathways, providing prolonged analgesia and better hemodynamic stability (Schug et al., 2006). Clonidine acts by enhancing the inhibitory effects on nociceptive transmission in the dorsal horn, while fentanyl enhances analgesia through opioid receptor activation in the spinal cord. Both agents improve pain control but differ in sedation levels, hemodynamic effects, and adverse event profiles (Schwartz et al., 2022).
The choice of an ideal adjuvant remains a topic of debate, as both clonidine and fentanyl have advantages and limitations. Clonidine has been associated with prolonged sensory and motor block, effective postoperative analgesia, and sedation, making it beneficial in long-duration procedures. However, it may also lead to bradycardia, hypotension, and excessive sedation, necessitating careful perioperative monitoring. Fentanyl, on the other hand, is well known for potentiating intraoperative and early postoperative analgesia with minimal hemodynamic effects. However, it has been associated with a higher incidence of pruritus, nausea, and respiratory depression, which can impact patient comfort (Sharan et al., 2016).
Despite the frequent use of adjuvants in SA, there is no consensus on whether clonidine or fentanyl offers superior efficacy and safety in combination with ropivacaine. Existing studies provide conflicting evidence regarding their comparative benefits, and head-to-head trials evaluating their effects on sensory and motor blockade, postoperative analgesia, hemodynamic stability, and adverse effects are still limited.4,5
This study aims to provide a comprehensive comparison of clonidine and fentanyl as adjuvants to ropivacaine in SA, focusing on their impact on the onset and duration of sensory and motor blockade, postoperative pain relief, sedation, and adverse effects. By systematically evaluating these parameters, the study seeks to offer clinically relevant insights that can guide anesthesiologists in optimizing SA protocols while minimizing complications.
2. METHODOLOGY
This study was conducted as a randomized, double-blind, controlled trial at Vinayaka Mission’s Kirupananda Variyar Medical College & Hospitals (VMKVMC&H) after obtaining approval from the Institutional Ethics Committee (IEC) (Approval No. VMKVMC&H/IEC/24/287). Written informed consent was obtained from all participants before enrollment. The study adhered to the ethical principles outlined in the Declaration of Helsinki (2013) and Good Clinical Practice (GCP) guidelines.
The study included patients scheduled for elective lower abdominal and lower limb surgeries under SA. A total of 75 patients were enrolled based on inclusion and exclusion criteria. Patients were aged 15 to 49 years and classified as American Society of Anesthesiologists (ASA) Physical Status I-II. Patients with known hypersensitivity to ropivacaine, clonidine, or fentanyl, severe cardiovascular, hepatic, renal, or neurological disorders, coagulopathy, pregnancy, or refusal to participate were excluded from the study.
Patients were randomly assigned to three groups using a computer-generated randomization sequence, ensuring equal allocation. The allocation sequence was concealed in opaque, sealed envelopes, which were opened just before administering SA. The study followed a double-blind design, where the anesthetist preparing the drug solutions was not involved in data collection, and the anesthesiologist administering SA and the patient were blinded to group assignment.
All patients were preloaded with 500 mL of Ringer’s lactate to prevent hypotension. Standard monitoring, including electrocardiography (ECG), non-invasive blood pressure (NIBP), and pulse oximetry (SpO₂), was applied. SA was performed under strict aseptic precautions in the L3-L4 or L4-L5 interspace using a 25G Quincke spinal needle with the patient in a sitting position. Once cerebrospinal fluid (CSF) flow was confirmed, the assigned drug combination was administered.
Group A (Control) received 15 mg of ropivacaine, Group B (clonidine) received 15 mg of ropivacaine + 30 μg of clonidine, and Group C (Fentanyl) received 15 mg of ropivacaine + 25 μg of fentanyl. After the injection, the patients were positioned in supine posture with oxygen supplementation at 3 L/min via nasal cannula.
Sensory and motor blockade parameters were recorded at baseline, then at 2-minute intervals until the highest sensory block level was achieved. Sensory block was assessed using the pinprick method, while motor blockade was evaluated using the Modified Bromage Scale. The duration of sensory blockade was measured from the time of administration until two-segment regression, and the duration of motor block was recorded until the Bromage Score returned to zero.
Hemodynamic parameters, including heart rate (HR), systolic blood pressure (SBP), and diastolic blood pressure (DBP), were recorded at baseline and intraoperatively at 0, 5, 10, 15, 30, 45, and 60 minutes. Any episodes of hypotension (SBP < 90 mmHg) and bradycardia (HR < 50 bpm) were documented and managed appropriately.
Postoperative analgesia was assessed by recording the time to the first rescue analgesia, defined as the interval from SA administration until the patient first requested additional pain relief. The number of rescue analgesia doses required within 24 hours was also recorded. Pain intensity was measured using the Visual Analog Scale (VAS) at 1, 6, 12, and 24 hours postoperatively.
Sedation was assessed using the Ramsay Sedation Scale at 10, 30, and 60 minutes after SA administration. Patients were monitored for adverse effects, including nausea, vomiting, hypotension, bradycardia, respiratory depression, shivering, and pruritus. Any complications were appropriately managed.
2.1. Statistical analysis
All collected data were analyzed using SPSS software version [X]. Continuous variables were presented as mean ± standard deviation (SD) and compared using a one-way ANOVA test followed by post hoc analysis for intergroup comparisons. Categorical variables were expressed as percentages and analyzed using the Chi-square test or Fisher’s exact test, as appropriate. P < 0.05 was considered statistically significant.
Figure 1 depicts the CONSORT flowchart illustrating the patient enrolment, randomization, allocation, follow-up, and analysis for this study. The flowchart visually represents the study methodology, ensuring clarity in participant distribution across the three groups.

Figure 1: The CONSORT flow diagram of the study
3. RESULTS
Table 1 presents the mean and standard deviation of age, weight, ASA grade distribution, HR, SBP, and DBP across Groups A, B, and C. A statistical comparison using ANOVA for continuous variables and the Chi-square test for categorical variables was performed to determine any significant differences.
| Table 1: Demographic profile and baseline hemodynamic | ||||
| Variable | Group A | Group B | Group C | P-value |
| Age (years) | 34.20 ± 7.73 | 31.96 ± 6.48 | 35.20 ± 10.01 | 0.3649 |
| Weight (kg) | 65.92 ± 11.52 | 72.88 ± 14.40 | 66.16 ± 10.15 | 0.0782 |
| ASA Grade | ||||
| 1 | 9 (36) | 9 (36) | 14 (56) | 0.2559 |
| 2 | 16 (64) | 16 (64) | 11 (44) | |
| Heart Rate (beats/min) | 73.99 ± 8.41 | 72.36 ± 8.96 | 73.96 ± 7.86 | 0.7381 |
| Systolic BP (mmHg) | 124.38 ± 8.16 | 125.59 ± 6.93 | 121.61 ± 7.36 | 0.1659 |
| Diastolic BP (mmHg) | 76.42 ± 9.20 | 75.67 ± 8.29 | 74.04 ± 8.78 | 0.6208 |
| Data presented as mean ± SD or n (%);
P > 0.05 is considered significant; ASA = American Society of Anesthesiologists |
||||
The demographic and baseline hemodynamic parameters were found to be statistically comparable among all three groups, indicating that any further differences in outcomes can be attributed to the effect of anesthetic regimens rather than baseline differences. No significant variation was observed in age, weight, ASA grade distribution, HR, SBP, and DBP across the groups (P > 0.05 for all variables).
To evaluate the efficacy of different anesthetic regimens, the onset, duration, and peak level of sensory and motor blockade were analyzed across Group A, Group B, and Group C. The table below summarizes the mean ± SD values for each parameter, along with statistical comparisons.
| Table 2: Sensory and motor block characteristics | ||||
| Parameter | Group A | Group B | Group C | P -value |
| Onset of Sensory Block (min) | 4.27 ± 0.93 | 4.31 ± 0.86 | 4.34 ± 0.82 | 0.9556 |
| Max Sensory Level Achieved | T10 | T10 | T10 | - |
| Duration of Sensory Block (min) | 209.11 ± 15.58 | 335.96 ± 16.59 | 282.54 ± 17.34 | <0.0001* |
| Onset of Motor Block (min) | 5.30 ± 0.87 | 4.77 ± 1.16 | 4.93 ± 1.10 | 0.1899 |
| Duration of Motor Block (min) | 172.02 ± 15.77 | 315.11 ± 15.57 | 249.83 ± 14.76 | <0.0001* |
| Data presented as n (%); P < 0.05 considered as significant | ||||
Onset of Sensory and Motor Block: The time required to achieve sensory and motor blockade was comparable among all three groups (P > 0.05, NS).
Peak Sensory Level: The most frequently achieved dermatomal level was T10 in all groups.
Duration of Sensory Block: Group B (clonidine + Ropivacaine) had the longest sensory block (P < 0.0001, significant), followed by Group C (Fentanyl + Ropivacaine), with Group A (Ropivacaine alone) showing the shortest duration.
Duration of Motor Block: Group B had the longest motor block duration, followed by Group C, with Group
A showing the shortest effect (P < 0.0001, significant). The mean sensory block onset time across the three groups;
Group A, B, and C, exhibited similar onset times, indicating no significant difference in the initiation of sensory blockade. The onset time of sensory blockade was comparable across all groups. The addition of clonidine (Group B) and Fentanyl (Group C) did not significantly alter the onset of sensory block compared to Group A.
Motor Block Onset and Recovery Time in 3 Groups:
Group B exhibited the fastest onset and longest duration of motor block, followed by Group C, while Group A had the shortest duration.
Motor onset time was slightly faster in Group B (clonidine) compared to Groups A and C.
Motor block duration was significantly prolonged in Group B, indicating that clonidine contributes to extended motor blockade.
Group A had the shortest motor block duration, suggesting that Ropivacaine alone produces a shorter motor blockade.
Criteria for Motor Blockade Assessment
The Modified Bromage Scale is used to assess the degree of motor blockade following SA. The table below outlines the criteria used in this study.
Grade 0: No motor impairment, indicating full recovery.
Grade 1: Mild weakness, but still able to move some joints.
Grade 2: Significant impairment, only slight movement possible.
Grade 3: Complete loss of motor function in the assessed limb.
All three groups exhibited a gradual decline in HR over time. Group B (clonidine) had a more pronounced reduction in HR, suggesting its additional hemodynamic stability effects. Group C (Fentanyl) maintained a relatively stable HR, whereas Group A (Ropivacaine alone) showed moderate variation. The trend highlights the potential bradycardic effect of clonidine, requiring careful monitoring.
Group B (clonidine) showed a greater reduction in SBP and DBP, indicating its vasodilatory effect. Group A (Ropivacaine alone) and Group C (Fentanyl) exhibited relatively stable blood pressure trends. DBP followed a similar trend to SBP, confirming consistent hemodynamic changes in each group. These variations suggest that clonidine has a significant impact on blood pressure regulation, potentially requiring additional hemodynamic support in susceptible patients.
Hypotension was observed more frequently in Group B (clonidine: 24.0%) and Group C (Fentanyl: 20.0%), compared to Group A (Ropivacaine alone: 12.0%). Bradycardia was most frequently observed in Group C (20.0%); whereas Groups A (4.0%) and B (8.0%) had lower incidences. Chi-square test for hypotension (P = 0.541) suggests no significant difference among the groups. Chi-square test for bradycardia (P = 0.162) suggests no significant difference in bradycardia frequency across groups.
| Table 3: Incidence of hypotension and bradycardia across groups | ||||
| Groups | Hypotension
n (%) |
Bradycardia
n (%) |
P-value
(Hypotension) |
P-value
(Bradycardia) |
| Group A | 3 (12.0) | 1 (4.0) | 0.541 | 0.162 |
| Group B | 6 (24.0) | 2 (8.0) | 0.541 | 0.162 |
| Group C | 5 (20.0) | 5 (20.0) | 0.541 | 0.162 |
| Data presented as n (%); P < 0.05 considered as significant | ||||
Although not statistically significant, Group B (clonidine) showed a higher tendency for hypotension, requiring closer hemodynamic monitoring. Group C (Fentanyl) exhibited the highest incidence of bradycardia, indicating a potential depressant effect on HR.
Group B (clonidine) had the longest pain relief duration, followed by Group C (Fentanyl). Group A (Ropivacaine alone) required the earliest rescue analgesia, indicating a shorter duration of pain relief. The addition of adjuvants (clonidine or Fentanyl) significantly prolonged analgesia, reducing the need for early intervention.
Sedation was significantly higher in Group B, affecting more than half of the patients, while Group C had a moderate incidence, and Group A had the least. Hypotension and bradycardia occurred more frequently in Groups B and C, with Group C showing the highest rate of bradycardia. Though these differences were not statistically significant, they highlight the potential hemodynamic impact of clonidine and fentanyl as adjuvants. Table 5 presents the distribution of sedation scores across Group A (Ropivacaine alone), Group B (Ropivacaine + Clonidine), and Group C (Ropivacaine + Fentanyl).
| Table 5: Ramsay Sedation Scale scores | ||
| Group | Sedation Score 0
(Awake) |
Sedation Score 1
(Drowsy) |
| Group A | 21 (84.0) | 4 (16.0) |
| Group B | 11 (44.0) | 14 (56.0) |
| Group C | 20 (80.0) | 5 (20.0) |
| Data presented as n (%) | ||
Nausea and pruritus were reported more often in Group A, whereas their occurrence was lower in the other groups. Shivering and respiratory depression were infrequent across all groups without notable differences. The findings suggest that clonidine is associated with more sedation and hypotension, while fentanyl may contribute to a higher incidence of bradycardia, emphasizing the need for individualized anesthetic planning and careful perioperative monitoring.
Group B exhibited the highest sedation levels, with over half of the patients showing drowsiness (56%), confirming clonidine’s significant sedative effect. Groups A and C had lower sedation scores, with most patients remaining awake (84-80%). The difference in sedation was statistically significant, highlighting clonidine’s role in enhanced sedation compared to ropivacaine alone or fentanyl.
4. DISCUSSION
The use of adjuvants in SA has been widely studied to improve analgesic quality, prolong the duration of sensory and motor blockade, and optimize patient outcomes. This study compared the effects of ropivacaine alone (Group A), ropivacaine + clonidine (Group B), and ropivacaine + fentanyl (Group C), evaluating their impact on sensory and motor blockade, hemodynamic stability, analgesic efficacy, and adverse effects.
The time required to achieve sensory and motor blockade was comparable among all three groups (P > 0.05), indicating that the addition of adjuvants did not significantly alter the onset time of anesthesia. However, the duration of both sensory and motor block was significantly longer in Group B (clonidine) compared to Groups A and C (P < 0.0001). These findings are consistent with previous studies, which have reported that clonidine, an α2-adrenergic agonist, prolongs anesthesia by inhibiting norepinephrine release and reducing afferent pain signal transmission (Sharan et al., 2016). Similarly, Group C (Fentanyl) also showed an extended duration of sensory and motor block compared to Group A, though not as pronounced as in Group B.6,7 This effect can be attributed to fentanyl’s synergistic action on opioid receptors in the spinal cord, enhancing analgesic potency (Routray et al., 2017).
Hemodynamic variations were observed among the groups, particularly in HR and blood pressure trends during the intraoperative period. Group B exhibited a more pronounced reduction in HR compared to Groups A and C, suggesting an additional bradycardic effect associated with clonidine. This aligns with prior research demonstrating clonidine’s sympatholytic action, leading to reduced HR and cardiac output (Schug et al., 2006). Similarly, mean SBP and DBP was significantly lower in Group B, reinforcing clonidine’s vasodilatory properties. These findings emphasize the need for close hemodynamic monitoring when using clonidine as an adjuvant.
In contrast, Group C (Fentanyl) exhibited a more stable HR trend but had the highest incidence of bradycardia (20%), which may be linked to fentanyl’s central depressant effects on vagal tone. These findings align with those of Mahendru et al. (2013), who noted a higher incidence of bradycardia when fentanyl was used as an adjuvant in neuraxial anesthesia.8,9
Postoperative Analgesia and Pain Scores
A key advantage of using adjuvants in SA is the prolongation of postoperative analgesia, reducing the need for rescue analgesics. Group B showed the longest time to first rescue analgesia (528.8 ± 12.2 min), significantly longer than Group C (422.1 ± 13.9 min) and Group A (203.3 ± 16.4 min, p < 0.0001). Similarly, Group B required the least number of additional analgesic doses within 24 hours, further supporting the superior analgesic effect of clonidine. These findings correlate with those of Sharan et al. (2016), who found that clonidine significantly prolonged postoperative analgesia compared to fentanyl or local anesthetics alone.
Although VAS pain scores were comparable across groups at 1, 6, 12, and 24 hours postoperatively, the prolonged analgesia duration in Groups B and C highlights the opioid-sparing potential of these adjuvants, which may reduce the need for systemic opioids in the postoperative period.10,11
Adverse Effects
The incidence of hypotension and bradycardia was higher in Groups B and C compared to Group A. While these differences were not statistically significant (P > 0.05), they suggest that both clonidine and fentanyl may contribute to hemodynamic instability. Hypotension was more frequent in Group B (24%) and Group C (20%), aligning with previous findings on clonidine-induced vasodilation and fentanyl’s depressive effects on sympathetic tone (Schwartz et al., 2022).
Sedation was significantly higher in Group B (56%, P = 0.0033), confirming clonidine’s central sedative effects. In contrast, Group A had the least sedation (16%), and Group C had moderate levels (20%), consistent with fentanyl’s mild sedative properties. The heightened sedation with clonidine necessitates careful postoperative monitoring, particularly in elderly patients or those at risk for respiratory depression (Schug et al., 2006).
Other adverse effects, such as nausea and pruritus, were more frequent in Group A (24%), whereas their occurrence was lower in the other groups. These findings suggest that the addition of adjuvants may reduce nausea and pruritus, possibly by altering opioid receptor activity in the spinal cord.12,13,145
| Table 4: Comparative incidence of adverse effects in two groups | |||||||||||
| Group | Nausea | Hypotension | Bradycardia | Respiratory Depression | Shivering | Pruritus | Sedation | P-value (Nausea) | P-value (Hypotension) | P-value (Sedation) | |
| Group A | 6 (24.0) | 3 (12.0) | 1 (4.0) | 1 (4.0) | 4 (16.0) | 6 (24.0) | 4 (16.0) | 0.1578 | 0.5407 | 0.0033* | |
| Group B | 2 (8.0) | 6 (24.0) | 2 (8.0) | 2 (8.0) | 5 (20.0) | 4 (16.0) | 14 (56.0) | 0.1578 | 0.5407 | 0.0033* | |
| Group C | 2 (8.0) | 5 (20.0) | 5 (20.0) | 2 (8.0) | 4 (16.0) | 4 (16.0) | 5 (20.0) | 0.1578 | 0.5407 | 0.0033* | |
| Data presented as n (%); P < 0.05 considered as significant | |||||||||||
5. Clinical Implications
- Clonidine significantly prolongs both sensory and motor block durations, making it a valuable adjuvant for surgeries requiring prolonged anesthesia. However, its sedative and hypotensive effects warrant caution in high-risk patients.
- Fentanyl also enhances analgesia duration but is associated with a higher incidence of bradycardia, necessitating vigilant intraoperative monitoring.
- Both adjuvants reduce the need for systemic opioids postoperatively, supporting their use in opioid-sparing anesthesia protocols.
- Hemodynamic fluctuations were more common in Groups B and C, reinforcing the need for patient selection and appropriate perioperative hemodynamic management.
6. CONCLUSION
This study confirms that the addition of clonidine or fentanyl to ropivacaine enhances the quality of spinal anesthesia by prolonging sensory and motor block, improving postoperative analgesia, and reducing systemic analgesic requirements. However, clonidine is associated with significant sedation and hypotension, while fentanyl increases the risk of bradycardia. These findings support the selective use of adjuvants based on individual patient profiles and surgical requirements, ensuring optimized pain control while minimizing complications.
7. Data availability
The numerical data generated during this research are available from the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
LA: Concept and study design, data collection, manuscript drafting, and statistical analysis.
PT: Study supervision, methodology validation, and manuscript review.
RPM: Literature review, manuscript editing, and data interpretation.
BR: Critical revision of the manuscript, ethical approval coordination, and patient recruitment.
VAS: Corresponding author, overseeing manuscript finalization and study coordination.
PP: Statistical analysis, data validation, and reference management.
11. REFERENCES
- Routray SS, Raut K, Pradhan A, Dash A, Soren M. Comparison of intrathecal clonidine and fentanyl as adjuvants to hyperbaric bupivacaine in subarachnoid block for lower limb orthopedic surgery. Anesth Essays Res. 2017;11(3):589-93. [PubMed] DOI: 4103/aer.AER_91_17
- Mahendru V, Tewari A, Katyal S, Grewal A, Singh MR, Katyal R. A comparison of intrathecal dexmedetomidine, clonidine, and fentanyl as adjuvants to hyperbaric bupivacaine for lower limb surgery: A double-blind controlled study. J Anaesthesiol Clin Pharmacol. 2013;29(4):496-502. [PubMed] DOI: 4103/0970-9185.119151
- Schwartz RH, Hernandez S, Noor N, Topfer J, Farrell K, Singh N, et al. A comprehensive review of the use of alpha-2 agonists in spinal anesthetics. Pain Physician. 2022;25(2):E193-E201. [PubMed]
- Schug SA, Saunders D, Kurowski I, Paech MJ. Neuraxial drug administration: A review of treatment options for anaesthesia and analgesia. CNS Drugs. 2006;20(11):917-33. [PubMed] DOI: 2165/00023210-200620110-00005
- Sharan R, Verma R, Dhawan A, Kumar J. Comparison of clonidine and fentanyl as adjuvants to ropivacaine in spinal anaesthesia. Anesth Essays Res. 2016;10(3):526-31. [PubMed] DOI: 4103/0259-1162.180781
- Varghese LA, Taksande K. A comparison between intrathecal dexmedetomidine with hyperbaric bupivacaine and intrathecal fentanyl with hyperbaric bupivacaine in lower abdominal surgeries: A prospective double-blinded study. J Datta Meghe Inst Med Sci Univ. 2017;12:99-109. Full Text
- Anusha T. A comparative study between intrathecal isobaric ropivacaine 0.75% (15 mg) plus dexmedetomidine (10 mcg) and isobaric ropivacaine 0.75% (15 mg) plus clonidine (30 mcg) for elective lower abdominal and lower limb surgeries. Int J Med Anesth. 2021;4:185-8. DOI: 33545/26643766.2021.v4.i1c.220
- Soori R, Bhat G, Sayeed SA, Kandavar S. A comparison of intrathecal dexmedetomidine and fentanyl as adjuvant to spinal bupivacaine in lower abdominal and lower limb surgeries—a double-blind randomized study. J Evol Med Dent Sci. 2020;9:1706-12.
- Hagedorn JM, Bremer N, Deer TR, Pope JE. Intrathecal analgesics: Choices and optimization of delivery. In: Neuraxial Therapeutics: A Comprehensive Guide. Vol. 3. Berlin: Springer; 2024. p. 637-43.
- Abd-Sayed A, Fiala K, Weisbein J, Chopra P, Lam C, Kalia H, et al. Intrathecal drug delivery systems survey: Trends in utilization in pain practice. J Pain Res. 2022;3:1305-14. [PubMed] DOI: 2147/JPR.S344409
- Roberts DJ, Nagpal SK, Kubelik D, Brandys T, Stelfox HT, Lalu MM, et al. Association between neuraxial anaesthesia or general anaesthesia for lower limb revascularisation surgery in adults and clinical outcomes: Population-based comparative effectiveness study. BMJ. 2020;371:m4104. [PubMed] DOI: 1136/bmj.m4104
- Lee JK, Park JH, Hyun SJ, Hodel D, Hausmann ON. Regional anesthesia for lumbar spine surgery: Can it be a standard in the future? Neurospine. 2021;18:733-40. [PubMed] DOI: 14245/ns.2142584.292
- Whizar-Lugo VM, Íñiguez-López KL, Cárdenas-Maytorena AC, Ramírez-Puerta CD. Local anesthetics. In: Topics in Local Anesthetics. Vol. 30. London: IntechOpen; 2020. p. 1-18.
- Luck JF, Fettes PD, Wildsmith JA. Spinal anaesthesia for elective surgery: A comparison of hyperbaric solutions of racemic bupivacaine, levobupivacaine, and ropivacaine. Br J Anaesth. 2008;101(5):705-10. [PubMed] DOI: 1093/bja/aen250