Mutia Farina 1 , Ratna Farida 2 , Riyadh Firdaus 3 , Yohannes George 4 , Dimas Rahmatisa 5 , Syifa Nadobudskaya 6
Authors affiliations:
Background & objective: Sella region tumors are among the most prevalent of all central nervous system tumors. Endoscopic transsphenoidal surgery (ETSS) for this condition can cause hypothalamic damage, potentially resulting in complications such as central diabetes insipidus (DI) after surgery. Copeptin is cleaved from the same precursor as arginine vasopressin and is released in equimolar amounts with arginine vasopressin from the posterior pituitary. This study aimed to determine the effects of fluid restriction on copeptin secretion in patients with Sella region tumors following ETSS.
Methodology: This randomized, unblinded clinical trial was conducted at the National Brain Centre Hospital during 2022. We recruited patients aged 18 years and diagnosed with Sella region tumors who underwent endoscopic transsphenoidal surgery. Copeptin was examined post-anesthesia, 3rd day, and 5th day after surgery. The incidence of postoperative diabetic insipidus was observed by monitoring the patient's health for 5 days following surgery.
Results: A total of 23 patients received their usual amount of fluids, and 25 patients had their intake of fluids limited to two-thirds of their basal requirements. Copeptin levels were higher in the fluid restriction group on days three and five, measuring 3.53 (1.34) and 4.8 (6.12) pmol/L, respectively, than in the non-restricted group, which measured 3.2 (1.74) and 2.8 (1.51) pmol/L. On day 3, the restriction group had a lower percentage of postoperative DI than the non-restricted group (18.8% (n = 9) versus 31.3% (n = 15); nonetheless, there was no statistically significant difference seen (P = 0.082). The percentage of postoperative DI on day five was also found to be similar, with a P value of 0.157 for 14.6% (n = 7) and 25% (n = 12) in the restricted and non-restricted groups, respectively.
Conclusions: Fluid restriction can enhance copeptin levels, particularly on the 5th day of treatment, and lower the risk of diabetic Insipidus in patients with Sella region tumors following endoscopic transsphenoidal surgery.
Abbreviations: AVP: arginine vasopressin, DI: Diabetes insipidus, ETSS: Endoscopic transsphenoidal surgery,
Keywords: AVP; Biomarker; Copeptin; Diabetes insipidus, Endocrine; ETSS, Fluid restriction
Citation: Farina M, Farida R, Firdaus R, George Y, Rahmatisa D, Nadobudskaya S. Anaesth. pain intensive care 2025;29(6):476-82. DOI: 10.35975/apic.v29i6.2890
Received: June 01, 2025; Revised: July 09, 2025; Accepted: July 26, 2025
Brain tumors occupy 85-90% of all central nervous system tumors, with those in the Sella region being among the most prevalent.1 The only proven treatment for tumors in this area is surgical removal or reduction of the tumor. Currently, trans-sphenoidal endoscopic surgery (ETSS) is the primary approach.2
However, this procedure can cause hypothalamic damage, potentially resulting in complications such as central diabetes insipidus (DI) after surgery.3 Mendel Castle-Kirszbaum and colleagues found that 10% of patients who undergo ETSS develop central DI.4 Data from the National Brain Centre Hospital between 2021 and 2022 reported an incidence of 19-22 cases of DI per 100 individuals following ETSS.5
Transient DI after surgery occurs due to damage to the magnocellular nerve, which connects the supraoptic and paraventricular nuclei to the posterior pituitary, leading to a reduction in the secretion of arginine vasopressin (AVP). Arginine vasopressin is a peptide hormone secreted by the hypothalamus that plays a key role in regulating fluid balance by exerting an anti-diuretic effect. The hypothalamus not only secretes AVP but also secretes copeptin, which is released concurrently with AVP.6,7 Copeptin is widely recognized as a serological marker for AVP, it has not been previously utilized to assess fluid intake in post-ETSS patients with DI. Fluid restriction in patients undergoing ETSS is believed to stimulate AVP secretion by increasing plasma osmolality. Takeuchi et al. demonstrated that fluid restriction in post-ETSS patients was effective in preventing hyponatremia and had minimal effects on hypernatremia.8 However, the effect of fluid restriction on the incidence of DI in post-ETSS patients has not been fully explored. This study is thus conceived to elucidate the mechanistic and clinical impact of postoperative fluid restriction on plasma copeptin levels and the incidence of central diabetes insipidus (CDI) following endoscopic endonasal transsphenoidal surgery (ETSS) for Sellar region tumors. In targeting copeptin—a highly stable and clinically validated surrogate biomarker for arginine vasopressin (AVP)—this research leverages an accessible molecular endpoint to probe the dynamic integrity of hypothalamic–neurohypophysis axis function under varying osmotic stress conditions.
Postoperative CDI remains a common yet underappreciated complication with potential for significant morbidity, and its onset often reflects a delicate disruption in osmoregulatory neuroendocrine pathways, particularly within the hypothalamus and posterior pituitary, structures inherently vulnerable during ETSS. By modulating fluid intake postoperatively, we hypothesize that mild hyperosmolar stimuli can restore or enhance endogenous AVP (and thus copeptin) secretion through baroreceptor and osmoreceptor-mediated feedback loops—potentially averting the onset or progression of CDI.
More broadly, this study seeks to bridge a critical translational gap by linking surgical neuroendocrine pathophysiology with biomarker-guided fluid management strategies. In the current absence of standardized guidelines, our findings may inform individualized perioperative care paradigms grounded in real-time biochemical feedback, thereby reducing iatrogenic complications, optimizing fluid homeostasis, and improving patient safety and recovery trajectories. This integrative approach positions fluid restriction not merely as a supportive care measure, but as a strategic intervention with mechanistic rationale and prognostic relevance.
This study was an unblinded, randomized controlled clinical trial that aimed to compare the effects of fluid intake restriction versus basal fluid intake on copeptin levels and the incidence of DI in patients with Sella region tumours undergoing ETSS. The research was conducted at the National Brain Centre Hospital in Jakarta from May 2023 to January 2024. The diagnosis of postoperative DI was based on the criteria of the National Brain Centre Hospital, which was defined as urine output ≥40 mL/kg/day, with or without alterations in serum electrolyte levels.
Inclusion criteria consisted of patients over the age of 18 diagnosed with a Sella region tumour who underwent endoscopic endonasal transsphenoid surgery. Exclusion criteria included patients with preoperative DI and those with stage 2-5 renal dysfunction according to Kidney Disease: Improving Global Outcomes (KDIGO) criteria for chronic kidney disease. Patients who experienced complications other than DI on days 0 and 1 after surgery or who died before the completion of monitoring and data collection within 5 days post-surgery were excluded from the study.
The minimum sample size was calculated to be 24 patients per group. Simple randomization was used for subject selection, conducted through manual draw. A raffle box contained 24 pieces of paper labelled "fluid intake restriction" and 25 labelled "basal fluid intake." Nurses in the post anaesthesia care unit (PACU) room randomly selected a piece of paper for each subject, assigning the corresponding treatment. Subjects were aware of the treatment they received.
The intravenous fluid administered was Wida Bes fluid, and oral fluids were mineral water provided by the National Brain Centre Hospital. Basal fluid requirements were calculated using the formula of 30-40 mL/kg body weight per 24 hours. The assigned fluid intake regimen continued for 5 days during hospitalization, along with monitoring of consciousness, vital signs, and 24-hour urine volume. Urine volume was measured for 5 days, and blood samples were collected on the 3rd and 5th postoperative days to measure copeptin and serum electrolyte levels.
During the study, the clinical condition of the subjects was evaluated using the Early Warning Score physiological parameters. If complications other than DI occurred, subjects were treated according to protocols and standard operating procedures at the National Brain Centre Hospital. If DI developed (urine output ≥40 mL/kg/day with or without changes in serum electrolytes), the management was to check electrolytes immediately, admit the patient to the intensive care unit, rehydrate 2/3 of the water deficit with oral fluids, the remaining 1/3 with intravenous fluids, and administer oral Desmopressin with an initial dose of 2x0.2 mg.
The data analysis to be conducted is univariate analysis which aims to provide an overview of the research subjects. Shapiro-Wilk is used for the normality test as the sample size is <50. A bivariate analysis is performed to evaluate the hypothesis about the effects of fluid restriction on the incidence of diabetic insipidus and copeptin levels. Copeptin data that is normally distributed will be evaluated using the Dependent T-test, whereas non-normally distributed data will be analysed using the Mann Whitney method. DI outcome data were analysed using Chi-square or Fisher. The level of significance used is P < 0.05.
The study has obtained ethical approval from the Research Ethics Committee of the Faculty of Medicine, University of Indonesia, the Research Ethics Committee of RSCM, and the Research Ethics Committee of the National Brain Centre Hospital. Before the study began, the research subjects were given information about the purpose, benefits, procedures, risks, and potential complications of the study. All subjects who were willing to participate would sign a consent form. This study is voluntary, without any coercion, and will not change the standard of care provided.
Of the 50 patients initially enrolled, three were excluded before randomization due to intraoperative and early postoperative complications. Two patients experienced massive intraoperative bleeding, which necessitated deviation from the standardized surgical protocol. The third patient required re-operation (re-ETSS) on postoperative day 1 due to acute tumor bleeding. These cases were excluded to maintain the internal validity of the study and ensure uniform exposure to the intended intervention protocol. Therefore, 48 patients were eligible and randomized for final analysis.
Figure 1 shows that from the 48 patients who met the study's inclusion criteria, none were excluded. These 48 patients were then randomized using the researcher's randomizer, with 25 patients assigned to the fluid restriction group and 23 patients assigned to the group without fluid restriction. Therefore, the data that can be analysed includes 25 subjects from the fluid restriction group and 23 subjects from the group without fluid restriction.
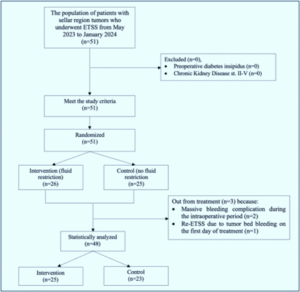
Figure 1: A flowchart illustrating the selection and randomization process
Table 1 shows that the distribution of gender, average age, weight, and height in both study groups was found to be not significantly different (P = 1.00, P > 0.05). The most common diagnosis in both groups was MAH. The average copeptin levels pre-operative did not differ significantly between the two groups, with values of 2.6 (1.62) versus 2.6 (1.71), respectively.
In general, there was an increase in copeptin levels in the fluid restriction group. Table 2 shows that copeptin levels on day 3 and day 5 were higher in the fluid restriction group, with values of 3.5 (1.115) and 3.7 (1.89), respectively, compared to the non-restriction group, which had values of 2.6 (1.65) and 2.1 (1.72).
The copeptin level on day 5 between the fluid restriction and non-restriction groups was significantly different, with a P = 0.011, indicating statistical significance.
Based on Table 3, it was found that the incidence of DI was lower in patients who underwent fluid restriction compared to those who did not receive fluid restriction.
But statistically, according to the results of the bivariate analysis using the Chi-Square test, a p-value of 0.082 was obtained, which is greater than 0.05 (Table 4). This indicates that there is no significant effect of fluid restriction on the occurrence of postoperative DI on day 3. The bivariate analysis of the therapy group for postoperative DI on day 5 yielded a p-value of 0.157, which is likewise greater than 0.05. Thus, fluid restriction had no significant impact on the incidence of postoperative diabetic insipidus on day 5.
In general, sodium levels before surgery in both test groups were not statistically substantially different, at 137 mmol/L versus 139 mmol/L. On day 3, the restriction group had lower sodium levels than the non-restricted group, with 138 (6) against 141 (5) (P = 0.034, P < 0.05). On day 5, sodium levels did not change substantially between test groups (P = 0.105, P < 0.05).
In this study, copeptin levels on day 3 and day 5 were found to be higher in the fluid restriction group, with values of 3.5 (1.115) and 3.7 (1.89) pmol/L, respectively, compared to the non-restriction group, which had values
of 2.6 (1.65) and 2.1 (1.72) pmol/L. Both of these values are within the physiological range for plasma copeptin, which is 1.0 to 13.8 pmol/L. According to the theoretical basis of this study, AVP and copeptin are released in proportional amounts from the same precursor, with the main stimuli being osmotic (increased osmolality) and non-osmotic stimuli such as hypovolemia and reduced blood pressure.9 Fluid restriction stimulates the release of AVP and copeptin from the posterior pituitary into the systemic circulation as a response to hemodynamic and osmotic stimuli, helping to maintain vascular tone (via V1a-R) and fluid homeostasis (via V2-R).9 A study involving 55 patients with DI found that fluid loss increased plasma copeptin levels from 4.6 ± 1.7 pmol/L to 9.2 ± 5.2 pmol/L. Serum osmolality reduction using hypotonic saline decreased plasma copeptin levels from 6.2 ± 2.4 pmol/L to 2.4 ± 2.1 pmol/L.
On the other hand, when comparing copeptin levels from day 0 to day 5, a decrease from 2.5 pmol/L to 2.1 pmol/L was observed in the non-restriction group. This is thought to be related to vasogenic edema caused by postoperative trauma, exacerbated by excessive fluid intake, which may disrupt AVP regulation. Consistent with previous studies, there was a reduction in copeptin levels from 4.9 pmol/L to 3.2 pmol/L after an oral fluid intake of 200-300 mL.7 Research by Lemetais showed similar results, where increased water intake over a relatively short intervention period (6 weeks) reduced circulating copeptin by 24.7%. In this study, initial copeptin values for groups A (50-80% fluid intake), B (81-120% fluid intake), and C (121-200% fluid intake) were 5.28 [3.3;7.5] pmol/L, 5.23 [3.5;7.6] pmol/L, and 4.20 [3.2;5.9] pmol/L, respectively. After the intervention, copeptin levels dropped to 3.93 and 3.90 pmol/L in groups A and B, respectively, with Group C showing a slightly lower value of 4.20 pmol/L. These findings may be attributed to the higher total fluid intake in Groups A and B during the intervention (2.8 and 2.7 L/day, respectively, compared to 2.4 L/day in Group C) and the higher contribution of drinking water.10
Clinically, central DI is diagnosed when there is a hypotonic polyuria syndrome (>50 mL/kg/24 hours, < 800 mOsm/kg) with normal copeptin levels (< 21.4 pmol/L) and arginine-stimulated copeptin < 3.8 pmol/L, or when hypotonic polyuria syndrome is accompanied by hypernatremia and copeptin levels <4.9 pmol/L.11 In this study, there was no statistically significant difference in the changes in copeptin levels from day 0 to day 3, and from day 3 to day 5, in both the fluid restriction and non-restriction groups. However, the relationship between higher copeptin levels and the incidence of DI can be seen in Table 4, which shows that the incidence of postoperative DI on day 3 was lower in the fluid restriction group compared to the non-restriction group. Fluid restriction may be an effective management strategy for reduce postoperative DI. This suggests that limiting fluid intake may help increase circulating copeptin levels in everyday conditions. The increase in copeptin levels associated with fluid restriction is thought to contribute to the reduced incidence of DI.
In general, DI is a rare disease with a prevalence of approximately 1 in 25,000 people. This disorder can occur at any age, and its prevalence is similar between men and women. DI is characterized by a syndrome of hypotonic polyuria (> 50 mL/kg/24 hours, < 800 mOsm/kg) and polydipsia (> 3 L/day). It can be caused by primary conditions (central or renal) or secondary conditions (primary polydipsia). Central DI is caused by inadequate secretion of AVP in the hypothalamic-neurohypophysis system in response to osmotic stimuli.11
Postoperative trauma can lead to vasogenic oedema due to damage to the blood-brain barrier, followed by endothelial injury and activation of glial cells. These glial cells produce various inflammatory mediators such as bradykinin, serotonin, histamine, arachidonic acid, nitric oxide, and leukotrienes, which cause fluid extravasation into the extracellular space.3 Damage to the blood-brain barrier can also disrupt osmotic gradient stability. Disruption of this gradient, along with the administration of isotonic crystalloid fluids that lower plasma osmolality, can lead to fluid shift into brain tissue, exacerbating oedema and further affecting AVP secretion.12 Fluid restriction is expected to stimulate the release of AVP and copeptin to regulate fluid homeostasis and prevent secondary insult due to fluid shift into the extracellular space.
In the Chi-square test (Table 4), the proportion of postoperative DI on day 3 was found to be lower in the fluid restriction group, at 18.8% (n=9) compared to 31.3% (n = 15) in the non-restriction group. However, this finding was not statistically significant (P = 0.082). The same result was observed on day 5, with proportions of 14.6% (n = 7) in the restriction group and 25% (n = 12) in the non-restriction group, with a P-value of 0.157. When compared to previous data from the National Brain Centre Hospital in 2021-2022, where the incidence of DI was reported to be 19-22 cases per 100 people, the incidence of DI in this study was found to be lower in patients who underwent fluid restriction compared to those who did not (Table 3).
The study by Canelo et al. found that one-third of patients undergoing transsphenoidal surgery experience water and electrolyte imbalances.13 This secondary effect is due to dysfunction of the posterior pituitary or infundibulum. The most common electrolyte disturbances observed in these patients are serum sodium imbalances, with both hypernatremia and hyponatremia potentially occurring in such cases. Hypernatremia is typically caused by DI and usually occurs in the early postoperative period. On the other hand, hyponatremia may be a result of the Syndrome of Inappropriate Antidiuretic Hormone (SIADH), which typically manifests a few days after surgery.14 A study conducted in North Sumatra examined the effects of transsphenoidal surgery on electrolyte balance, finding an increase in the average blood sodium levels from 137.6 meq/L before surgery to 139 meq/L on the first postoperative day.15 In this study, it was noted that the sodium levels before surgery were not statistically significantly different between the two test groups, with values of 137 mmol/L versus 139 mmol/L. The sodium levels in the study participants tended to be slightly elevated, possibly related to daily sodium intake. The average daily sodium intake in Indonesia is 2700–3500 mg, well above the recommended maximum of 2000 mg/day, while potassium intake averages 946 mg/day, which is much lower than the WHO recommendation of a minimum of 3500 mg/day.16,17 Similarly, a 2021 study in Jakarta found that the average daily sodium intake exceeded 2 g/day, with the highest intake observed in teenagers (6.74 g salt/day or 2694 mg sodium/day), followed by adults (5.86 g salt/day or 2345 mg sodium/day), and children (5.82 g salt/day or 2329 mg sodium/day).18
After treatment, sodium levels were significantly higher on day 3 in the restriction group (P = 0.036) compared to the non-restriction group (P = 0.003), although sodium levels were lower in the restriction group compared to the non-restriction group (138 ± 6 mmol/L vs. 141 ± 5 mmol/L, P = 0.034, p<0.05). On day 5, sodium levels showed a significant decrease in both groups (restriction, P = 0.036; non-restriction, P = 0.002). The greater increase in sodium levels in the non-restriction group is thought to result from increased plasma volume, which lowers plasma osmolality and inhibits the release of AVP and copeptin. With lower AVP levels, the kidneys are less able to excrete water, resulting in more diluted urine. However, urine specific gravity did not show a significant difference between the two groups. The significant decrease in sodium on day 5 is thought to be related to the management of DI, as per the protocol followed for some subjects in both the restriction and non-restriction groups. In the non-restriction group, two subjects received desmopressin therapy (0.1 mg twice or thrice daily), four subjects received fluid restriction therapy, and three subjects received a combination of desmopressin (0.1 mg twice daily) and fluid restriction. This treatment was most commonly administered on day 3 of the study. A large cohort study showed that hyponatremia occurred more frequently immediately after desmopressin was given.19 Desmopressin is a synthetic analogue of the AVP hormone that acts on the selective vasopressin V2 receptors in the kidney's collecting ducts and distal tubules, increasing water permeability. This leads to a decrease in urine volume and an increase in urine osmolality. A study by Takeuchi found that fluid restriction could prevent hyponatremia,20 which is consistent with research by Kazempour on post-transsphenoidal surgery patients.21
The results of this study showed that restricting fluid intake to two-thirds of basal requirements, with a maximum oral intake of 1000 mL over 24 hours, significantly increased copeptin levels on days 3 and 5 post-ETSS compared to patients without fluid restriction. Fluid restriction and the resulting increase in copeptin are believed to have a clinical impact on the occurrence of DI after ETSS. The incidence of DI was found to be lower on days 3 and 5 in the fluid-restricted group. Fluid restriction was shown to reduce the likelihood of developing DI by 0.847 times on day 3 and by 3.7 times on day 5. This approach also helped maintain fluid and sodium balance, which is disrupted due to posterior pituitary dysfunction in patients after ETSS.
Fluid intake restriction can increase copeptin levels, particularly on the fifth day of care in patients with Sella region tumors following ETSS. Additionally, in post-ETSS patients, fluid restriction is more effective than unrestricted fluid intake in preventing the occurrence of postoperative DI. This approach also helped maintain fluid and sodium balance.
6. Data availability
The numerical data generated during this research are available with the authors.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Acknowledgments
The author would like to express sincere gratitude to the Faculty of Medicine, University of Indonesia, RSCM, and the National Brain Centre Hospital for providing the necessary support and resources to collect the research data. Special thanks are also extended to all the participants for their invaluable contributions to the study. The author is deeply appreciative of the supervisors for their insightful suggestions during data analysis and their guidance in interpreting the results. Additionally, the author would like to thank the staff of the Department of Anesthesiology and Intensive Therapy at RSCM, as well as the nursing and medical staff at the National Brain Centre Hospital, for their dedicated support throughout the course of the study.
10. Authors’ contribution
MF: Conceived and designed the experiments; performed the experiments; analyzed the data; wrote the first draft of the manuscript.
RF: Contributed to the experimental design; helped analyze the data; critically revised the manuscript.
YG, RF, DR: supervised the project; reviewed and approved the final manuscript.
Authors affiliations:
- Mutia Farina, Anesthesiologist, Intensive Therapy, Department of Anesthesiology and Intensive Care, University of Indonesia, Jakarta, Indonesia; Email: mutia.farina.md@gmail.com
- Ratna Farida, Anesthesiologist, PhD, Intensive Therapy, Department of Anesthesiology and Intensive Care, University of Indonesia, Jakarta, Indonesia; Email: fida_soenarto@yahii.com
- Riyadh Firdaus, Anesthesiologist, PhD, Intensive Therapy, Department of Anesthesiology and Intensive Care, University of Indonesia, Jakarta, Indonesia; Email: putribudiutami@gmail.com
- Yohannes George, Anesthesiologist, PhD, Intensive Therapy, Department of Anesthesiology and Intensive Care, University of Indonesia, Jakarta, Indonesia; Email: yohanesjorge@gmail.com
- Dimas Rahmatisa, Anesthesiologist, Intensive Therapy, Department of Anesthesiology and Intensive Care, National Brain Centre Hospital, Jakarta, Indonesia; Email: dimasr@gmail.com
- Syifa Nadobudskaya, MD, Research Assistant, Pembangunan Nasional Veteran Jakarta University, Jakarta, Indonesia.
ABSTRACT
Background & objective: Sella region tumors are among the most prevalent of all central nervous system tumors. Endoscopic transsphenoidal surgery (ETSS) for this condition can cause hypothalamic damage, potentially resulting in complications such as central diabetes insipidus (DI) after surgery. Copeptin is cleaved from the same precursor as arginine vasopressin and is released in equimolar amounts with arginine vasopressin from the posterior pituitary. This study aimed to determine the effects of fluid restriction on copeptin secretion in patients with Sella region tumors following ETSS.
Methodology: This randomized, unblinded clinical trial was conducted at the National Brain Centre Hospital during 2022. We recruited patients aged 18 years and diagnosed with Sella region tumors who underwent endoscopic transsphenoidal surgery. Copeptin was examined post-anesthesia, 3rd day, and 5th day after surgery. The incidence of postoperative diabetic insipidus was observed by monitoring the patient's health for 5 days following surgery.
Results: A total of 23 patients received their usual amount of fluids, and 25 patients had their intake of fluids limited to two-thirds of their basal requirements. Copeptin levels were higher in the fluid restriction group on days three and five, measuring 3.53 (1.34) and 4.8 (6.12) pmol/L, respectively, than in the non-restricted group, which measured 3.2 (1.74) and 2.8 (1.51) pmol/L. On day 3, the restriction group had a lower percentage of postoperative DI than the non-restricted group (18.8% (n = 9) versus 31.3% (n = 15); nonetheless, there was no statistically significant difference seen (P = 0.082). The percentage of postoperative DI on day five was also found to be similar, with a P value of 0.157 for 14.6% (n = 7) and 25% (n = 12) in the restricted and non-restricted groups, respectively.
Conclusions: Fluid restriction can enhance copeptin levels, particularly on the 5th day of treatment, and lower the risk of diabetic Insipidus in patients with Sella region tumors following endoscopic transsphenoidal surgery.
Abbreviations: AVP: arginine vasopressin, DI: Diabetes insipidus, ETSS: Endoscopic transsphenoidal surgery,
Keywords: AVP; Biomarker; Copeptin; Diabetes insipidus, Endocrine; ETSS, Fluid restriction
Citation: Farina M, Farida R, Firdaus R, George Y, Rahmatisa D, Nadobudskaya S. Anaesth. pain intensive care 2025;29(6):476-82. DOI: 10.35975/apic.v29i6.2890
Received: June 01, 2025; Revised: July 09, 2025; Accepted: July 26, 2025
1. INTRODUCTION
Brain tumors occupy 85-90% of all central nervous system tumors, with those in the Sella region being among the most prevalent.1 The only proven treatment for tumors in this area is surgical removal or reduction of the tumor. Currently, trans-sphenoidal endoscopic surgery (ETSS) is the primary approach.2
However, this procedure can cause hypothalamic damage, potentially resulting in complications such as central diabetes insipidus (DI) after surgery.3 Mendel Castle-Kirszbaum and colleagues found that 10% of patients who undergo ETSS develop central DI.4 Data from the National Brain Centre Hospital between 2021 and 2022 reported an incidence of 19-22 cases of DI per 100 individuals following ETSS.5
Transient DI after surgery occurs due to damage to the magnocellular nerve, which connects the supraoptic and paraventricular nuclei to the posterior pituitary, leading to a reduction in the secretion of arginine vasopressin (AVP). Arginine vasopressin is a peptide hormone secreted by the hypothalamus that plays a key role in regulating fluid balance by exerting an anti-diuretic effect. The hypothalamus not only secretes AVP but also secretes copeptin, which is released concurrently with AVP.6,7 Copeptin is widely recognized as a serological marker for AVP, it has not been previously utilized to assess fluid intake in post-ETSS patients with DI. Fluid restriction in patients undergoing ETSS is believed to stimulate AVP secretion by increasing plasma osmolality. Takeuchi et al. demonstrated that fluid restriction in post-ETSS patients was effective in preventing hyponatremia and had minimal effects on hypernatremia.8 However, the effect of fluid restriction on the incidence of DI in post-ETSS patients has not been fully explored. This study is thus conceived to elucidate the mechanistic and clinical impact of postoperative fluid restriction on plasma copeptin levels and the incidence of central diabetes insipidus (CDI) following endoscopic endonasal transsphenoidal surgery (ETSS) for Sellar region tumors. In targeting copeptin—a highly stable and clinically validated surrogate biomarker for arginine vasopressin (AVP)—this research leverages an accessible molecular endpoint to probe the dynamic integrity of hypothalamic–neurohypophysis axis function under varying osmotic stress conditions.
Postoperative CDI remains a common yet underappreciated complication with potential for significant morbidity, and its onset often reflects a delicate disruption in osmoregulatory neuroendocrine pathways, particularly within the hypothalamus and posterior pituitary, structures inherently vulnerable during ETSS. By modulating fluid intake postoperatively, we hypothesize that mild hyperosmolar stimuli can restore or enhance endogenous AVP (and thus copeptin) secretion through baroreceptor and osmoreceptor-mediated feedback loops—potentially averting the onset or progression of CDI.
More broadly, this study seeks to bridge a critical translational gap by linking surgical neuroendocrine pathophysiology with biomarker-guided fluid management strategies. In the current absence of standardized guidelines, our findings may inform individualized perioperative care paradigms grounded in real-time biochemical feedback, thereby reducing iatrogenic complications, optimizing fluid homeostasis, and improving patient safety and recovery trajectories. This integrative approach positions fluid restriction not merely as a supportive care measure, but as a strategic intervention with mechanistic rationale and prognostic relevance.
2. METHODOLOGY
This study was an unblinded, randomized controlled clinical trial that aimed to compare the effects of fluid intake restriction versus basal fluid intake on copeptin levels and the incidence of DI in patients with Sella region tumours undergoing ETSS. The research was conducted at the National Brain Centre Hospital in Jakarta from May 2023 to January 2024. The diagnosis of postoperative DI was based on the criteria of the National Brain Centre Hospital, which was defined as urine output ≥40 mL/kg/day, with or without alterations in serum electrolyte levels.
Inclusion criteria consisted of patients over the age of 18 diagnosed with a Sella region tumour who underwent endoscopic endonasal transsphenoid surgery. Exclusion criteria included patients with preoperative DI and those with stage 2-5 renal dysfunction according to Kidney Disease: Improving Global Outcomes (KDIGO) criteria for chronic kidney disease. Patients who experienced complications other than DI on days 0 and 1 after surgery or who died before the completion of monitoring and data collection within 5 days post-surgery were excluded from the study.
The minimum sample size was calculated to be 24 patients per group. Simple randomization was used for subject selection, conducted through manual draw. A raffle box contained 24 pieces of paper labelled "fluid intake restriction" and 25 labelled "basal fluid intake." Nurses in the post anaesthesia care unit (PACU) room randomly selected a piece of paper for each subject, assigning the corresponding treatment. Subjects were aware of the treatment they received.
The intravenous fluid administered was Wida Bes fluid, and oral fluids were mineral water provided by the National Brain Centre Hospital. Basal fluid requirements were calculated using the formula of 30-40 mL/kg body weight per 24 hours. The assigned fluid intake regimen continued for 5 days during hospitalization, along with monitoring of consciousness, vital signs, and 24-hour urine volume. Urine volume was measured for 5 days, and blood samples were collected on the 3rd and 5th postoperative days to measure copeptin and serum electrolyte levels.
During the study, the clinical condition of the subjects was evaluated using the Early Warning Score physiological parameters. If complications other than DI occurred, subjects were treated according to protocols and standard operating procedures at the National Brain Centre Hospital. If DI developed (urine output ≥40 mL/kg/day with or without changes in serum electrolytes), the management was to check electrolytes immediately, admit the patient to the intensive care unit, rehydrate 2/3 of the water deficit with oral fluids, the remaining 1/3 with intravenous fluids, and administer oral Desmopressin with an initial dose of 2x0.2 mg.
The data analysis to be conducted is univariate analysis which aims to provide an overview of the research subjects. Shapiro-Wilk is used for the normality test as the sample size is <50. A bivariate analysis is performed to evaluate the hypothesis about the effects of fluid restriction on the incidence of diabetic insipidus and copeptin levels. Copeptin data that is normally distributed will be evaluated using the Dependent T-test, whereas non-normally distributed data will be analysed using the Mann Whitney method. DI outcome data were analysed using Chi-square or Fisher. The level of significance used is P < 0.05.
The study has obtained ethical approval from the Research Ethics Committee of the Faculty of Medicine, University of Indonesia, the Research Ethics Committee of RSCM, and the Research Ethics Committee of the National Brain Centre Hospital. Before the study began, the research subjects were given information about the purpose, benefits, procedures, risks, and potential complications of the study. All subjects who were willing to participate would sign a consent form. This study is voluntary, without any coercion, and will not change the standard of care provided.
3. RESULTS
Of the 50 patients initially enrolled, three were excluded before randomization due to intraoperative and early postoperative complications. Two patients experienced massive intraoperative bleeding, which necessitated deviation from the standardized surgical protocol. The third patient required re-operation (re-ETSS) on postoperative day 1 due to acute tumor bleeding. These cases were excluded to maintain the internal validity of the study and ensure uniform exposure to the intended intervention protocol. Therefore, 48 patients were eligible and randomized for final analysis.
Figure 1 shows that from the 48 patients who met the study's inclusion criteria, none were excluded. These 48 patients were then randomized using the researcher's randomizer, with 25 patients assigned to the fluid restriction group and 23 patients assigned to the group without fluid restriction. Therefore, the data that can be analysed includes 25 subjects from the fluid restriction group and 23 subjects from the group without fluid restriction.

Figure 1: A flowchart illustrating the selection and randomization process
Table 1 shows that the distribution of gender, average age, weight, and height in both study groups was found to be not significantly different (P = 1.00, P > 0.05). The most common diagnosis in both groups was MAH. The average copeptin levels pre-operative did not differ significantly between the two groups, with values of 2.6 (1.62) versus 2.6 (1.71), respectively.
| Table 1: Baseline demographic data of study participants | |||
| Variable | Fluid-restricted Group
(n = 25) |
Non-restricted Group
(n = 23) |
P-value |
| Gender (M/F) | 14 / 11 | 12 / 11 | 1.000 |
| Age (years) | 46.5 ± 10.0 | 41.8 ± 10.0 | 0.115 |
| Weight (kg) | 68 (50–85) | 65 (47–110) | 0.812 |
| Height (cm) | 161 (155–176) | 163 (148–175) | 0.541 |
| Main Diagnosis (MAH) | 100% | 95.7% | – |
| Copeptin Day 0 (pmol/L) | 2.6 ± 1.62 | 2.6 ± 1.71 | 0.992 |
| Data presented as number, Median (Range), percentage or Mean ± SD; P < 0.05 considered as significant. | |||
In general, there was an increase in copeptin levels in the fluid restriction group. Table 2 shows that copeptin levels on day 3 and day 5 were higher in the fluid restriction group, with values of 3.5 (1.115) and 3.7 (1.89), respectively, compared to the non-restriction group, which had values of 2.6 (1.65) and 2.1 (1.72).
| Table 2: Copeptin levels over time | |||
| Time | Fluid-restricted Group
(n = 25) |
Non-restricted Group
(n = 23) |
P-value |
| Day 0 | 2.6 ± 1.62 | 2.6 ± 1.71 | 0.992 |
| Day 3 | 3.5 ± 1.12 | 2.6 ± 1.65 | 0.095 |
| Day 5 | 3.7 ± 1.89 | 2.1 ± 1.72 | 0.011 |
| Data presented as Mean ± SD; Copeptin level on Day 5 was significantly higher in the fluid restriction group (p < 0.05). | |||
The copeptin level on day 5 between the fluid restriction and non-restriction groups was significantly different, with a P = 0.011, indicating statistical significance.
Based on Table 3, it was found that the incidence of DI was lower in patients who underwent fluid restriction compared to those who did not receive fluid restriction.
| Table 3: Incidence of diabetes insipidus (DI) | |||
| Postoperative Day | Fluid-restricted Group
(n = 25) |
Non-restricted Group
(n = 23) |
P-value |
| Day 3 | 9 (18.8%) | 15 (31.3%) | 0.082 |
| Day 5 | 7 (14.6%) | 12 (25.0%) | 0.157 |
| Data presented as Mean ± SD; Copeptin level on Day 5 was significantly higher in the fluid restriction group (p < 0.05). | |||
But statistically, according to the results of the bivariate analysis using the Chi-Square test, a p-value of 0.082 was obtained, which is greater than 0.05 (Table 4). This indicates that there is no significant effect of fluid restriction on the occurrence of postoperative DI on day 3. The bivariate analysis of the therapy group for postoperative DI on day 5 yielded a p-value of 0.157, which is likewise greater than 0.05. Thus, fluid restriction had no significant impact on the incidence of postoperative diabetic insipidus on day 5.
| Table 4: Bivariate analysis based on fluid administration groups on the incidence of diabetes insipidus | ||||
| Variables | Diabetes insipidus Post-op Day 3 | P-value | Diabetes
insipidus Post-op Day 5 |
P-value |
| Restricted Group | 9 (18.8) | 0.082a | 7 (14.6) | 0.157a |
| Non-restricted Group | 15 (31.3) | 12 (25) | ||
| Data given as n (%); aChi-square Test; *P-value < 0.05 is significant. | ||||
In general, sodium levels before surgery in both test groups were not statistically substantially different, at 137 mmol/L versus 139 mmol/L. On day 3, the restriction group had lower sodium levels than the non-restricted group, with 138 (6) against 141 (5) (P = 0.034, P < 0.05). On day 5, sodium levels did not change substantially between test groups (P = 0.105, P < 0.05).
4. DISCUSSION
In this study, copeptin levels on day 3 and day 5 were found to be higher in the fluid restriction group, with values of 3.5 (1.115) and 3.7 (1.89) pmol/L, respectively, compared to the non-restriction group, which had values
of 2.6 (1.65) and 2.1 (1.72) pmol/L. Both of these values are within the physiological range for plasma copeptin, which is 1.0 to 13.8 pmol/L. According to the theoretical basis of this study, AVP and copeptin are released in proportional amounts from the same precursor, with the main stimuli being osmotic (increased osmolality) and non-osmotic stimuli such as hypovolemia and reduced blood pressure.9 Fluid restriction stimulates the release of AVP and copeptin from the posterior pituitary into the systemic circulation as a response to hemodynamic and osmotic stimuli, helping to maintain vascular tone (via V1a-R) and fluid homeostasis (via V2-R).9 A study involving 55 patients with DI found that fluid loss increased plasma copeptin levels from 4.6 ± 1.7 pmol/L to 9.2 ± 5.2 pmol/L. Serum osmolality reduction using hypotonic saline decreased plasma copeptin levels from 6.2 ± 2.4 pmol/L to 2.4 ± 2.1 pmol/L.
On the other hand, when comparing copeptin levels from day 0 to day 5, a decrease from 2.5 pmol/L to 2.1 pmol/L was observed in the non-restriction group. This is thought to be related to vasogenic edema caused by postoperative trauma, exacerbated by excessive fluid intake, which may disrupt AVP regulation. Consistent with previous studies, there was a reduction in copeptin levels from 4.9 pmol/L to 3.2 pmol/L after an oral fluid intake of 200-300 mL.7 Research by Lemetais showed similar results, where increased water intake over a relatively short intervention period (6 weeks) reduced circulating copeptin by 24.7%. In this study, initial copeptin values for groups A (50-80% fluid intake), B (81-120% fluid intake), and C (121-200% fluid intake) were 5.28 [3.3;7.5] pmol/L, 5.23 [3.5;7.6] pmol/L, and 4.20 [3.2;5.9] pmol/L, respectively. After the intervention, copeptin levels dropped to 3.93 and 3.90 pmol/L in groups A and B, respectively, with Group C showing a slightly lower value of 4.20 pmol/L. These findings may be attributed to the higher total fluid intake in Groups A and B during the intervention (2.8 and 2.7 L/day, respectively, compared to 2.4 L/day in Group C) and the higher contribution of drinking water.10
Clinically, central DI is diagnosed when there is a hypotonic polyuria syndrome (>50 mL/kg/24 hours, < 800 mOsm/kg) with normal copeptin levels (< 21.4 pmol/L) and arginine-stimulated copeptin < 3.8 pmol/L, or when hypotonic polyuria syndrome is accompanied by hypernatremia and copeptin levels <4.9 pmol/L.11 In this study, there was no statistically significant difference in the changes in copeptin levels from day 0 to day 3, and from day 3 to day 5, in both the fluid restriction and non-restriction groups. However, the relationship between higher copeptin levels and the incidence of DI can be seen in Table 4, which shows that the incidence of postoperative DI on day 3 was lower in the fluid restriction group compared to the non-restriction group. Fluid restriction may be an effective management strategy for reduce postoperative DI. This suggests that limiting fluid intake may help increase circulating copeptin levels in everyday conditions. The increase in copeptin levels associated with fluid restriction is thought to contribute to the reduced incidence of DI.
In general, DI is a rare disease with a prevalence of approximately 1 in 25,000 people. This disorder can occur at any age, and its prevalence is similar between men and women. DI is characterized by a syndrome of hypotonic polyuria (> 50 mL/kg/24 hours, < 800 mOsm/kg) and polydipsia (> 3 L/day). It can be caused by primary conditions (central or renal) or secondary conditions (primary polydipsia). Central DI is caused by inadequate secretion of AVP in the hypothalamic-neurohypophysis system in response to osmotic stimuli.11
Postoperative trauma can lead to vasogenic oedema due to damage to the blood-brain barrier, followed by endothelial injury and activation of glial cells. These glial cells produce various inflammatory mediators such as bradykinin, serotonin, histamine, arachidonic acid, nitric oxide, and leukotrienes, which cause fluid extravasation into the extracellular space.3 Damage to the blood-brain barrier can also disrupt osmotic gradient stability. Disruption of this gradient, along with the administration of isotonic crystalloid fluids that lower plasma osmolality, can lead to fluid shift into brain tissue, exacerbating oedema and further affecting AVP secretion.12 Fluid restriction is expected to stimulate the release of AVP and copeptin to regulate fluid homeostasis and prevent secondary insult due to fluid shift into the extracellular space.
In the Chi-square test (Table 4), the proportion of postoperative DI on day 3 was found to be lower in the fluid restriction group, at 18.8% (n=9) compared to 31.3% (n = 15) in the non-restriction group. However, this finding was not statistically significant (P = 0.082). The same result was observed on day 5, with proportions of 14.6% (n = 7) in the restriction group and 25% (n = 12) in the non-restriction group, with a P-value of 0.157. When compared to previous data from the National Brain Centre Hospital in 2021-2022, where the incidence of DI was reported to be 19-22 cases per 100 people, the incidence of DI in this study was found to be lower in patients who underwent fluid restriction compared to those who did not (Table 3).
The study by Canelo et al. found that one-third of patients undergoing transsphenoidal surgery experience water and electrolyte imbalances.13 This secondary effect is due to dysfunction of the posterior pituitary or infundibulum. The most common electrolyte disturbances observed in these patients are serum sodium imbalances, with both hypernatremia and hyponatremia potentially occurring in such cases. Hypernatremia is typically caused by DI and usually occurs in the early postoperative period. On the other hand, hyponatremia may be a result of the Syndrome of Inappropriate Antidiuretic Hormone (SIADH), which typically manifests a few days after surgery.14 A study conducted in North Sumatra examined the effects of transsphenoidal surgery on electrolyte balance, finding an increase in the average blood sodium levels from 137.6 meq/L before surgery to 139 meq/L on the first postoperative day.15 In this study, it was noted that the sodium levels before surgery were not statistically significantly different between the two test groups, with values of 137 mmol/L versus 139 mmol/L. The sodium levels in the study participants tended to be slightly elevated, possibly related to daily sodium intake. The average daily sodium intake in Indonesia is 2700–3500 mg, well above the recommended maximum of 2000 mg/day, while potassium intake averages 946 mg/day, which is much lower than the WHO recommendation of a minimum of 3500 mg/day.16,17 Similarly, a 2021 study in Jakarta found that the average daily sodium intake exceeded 2 g/day, with the highest intake observed in teenagers (6.74 g salt/day or 2694 mg sodium/day), followed by adults (5.86 g salt/day or 2345 mg sodium/day), and children (5.82 g salt/day or 2329 mg sodium/day).18
After treatment, sodium levels were significantly higher on day 3 in the restriction group (P = 0.036) compared to the non-restriction group (P = 0.003), although sodium levels were lower in the restriction group compared to the non-restriction group (138 ± 6 mmol/L vs. 141 ± 5 mmol/L, P = 0.034, p<0.05). On day 5, sodium levels showed a significant decrease in both groups (restriction, P = 0.036; non-restriction, P = 0.002). The greater increase in sodium levels in the non-restriction group is thought to result from increased plasma volume, which lowers plasma osmolality and inhibits the release of AVP and copeptin. With lower AVP levels, the kidneys are less able to excrete water, resulting in more diluted urine. However, urine specific gravity did not show a significant difference between the two groups. The significant decrease in sodium on day 5 is thought to be related to the management of DI, as per the protocol followed for some subjects in both the restriction and non-restriction groups. In the non-restriction group, two subjects received desmopressin therapy (0.1 mg twice or thrice daily), four subjects received fluid restriction therapy, and three subjects received a combination of desmopressin (0.1 mg twice daily) and fluid restriction. This treatment was most commonly administered on day 3 of the study. A large cohort study showed that hyponatremia occurred more frequently immediately after desmopressin was given.19 Desmopressin is a synthetic analogue of the AVP hormone that acts on the selective vasopressin V2 receptors in the kidney's collecting ducts and distal tubules, increasing water permeability. This leads to a decrease in urine volume and an increase in urine osmolality. A study by Takeuchi found that fluid restriction could prevent hyponatremia,20 which is consistent with research by Kazempour on post-transsphenoidal surgery patients.21
The results of this study showed that restricting fluid intake to two-thirds of basal requirements, with a maximum oral intake of 1000 mL over 24 hours, significantly increased copeptin levels on days 3 and 5 post-ETSS compared to patients without fluid restriction. Fluid restriction and the resulting increase in copeptin are believed to have a clinical impact on the occurrence of DI after ETSS. The incidence of DI was found to be lower on days 3 and 5 in the fluid-restricted group. Fluid restriction was shown to reduce the likelihood of developing DI by 0.847 times on day 3 and by 3.7 times on day 5. This approach also helped maintain fluid and sodium balance, which is disrupted due to posterior pituitary dysfunction in patients after ETSS.
5. CONCLUSIONS
Fluid intake restriction can increase copeptin levels, particularly on the fifth day of care in patients with Sella region tumors following ETSS. Additionally, in post-ETSS patients, fluid restriction is more effective than unrestricted fluid intake in preventing the occurrence of postoperative DI. This approach also helped maintain fluid and sodium balance.
6. Data availability
The numerical data generated during this research are available with the authors.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Acknowledgments
The author would like to express sincere gratitude to the Faculty of Medicine, University of Indonesia, RSCM, and the National Brain Centre Hospital for providing the necessary support and resources to collect the research data. Special thanks are also extended to all the participants for their invaluable contributions to the study. The author is deeply appreciative of the supervisors for their insightful suggestions during data analysis and their guidance in interpreting the results. Additionally, the author would like to thank the staff of the Department of Anesthesiology and Intensive Therapy at RSCM, as well as the nursing and medical staff at the National Brain Centre Hospital, for their dedicated support throughout the course of the study.
10. Authors’ contribution
MF: Conceived and designed the experiments; performed the experiments; analyzed the data; wrote the first draft of the manuscript.
RF: Contributed to the experimental design; helped analyze the data; critically revised the manuscript.
YG, RF, DR: supervised the project; reviewed and approved the final manuscript.
11. REFERENCES
- Kementerian Kesehatan Republik Indonesia. Pedoman Nasional Pelayanan Kedokteran Tumor Otak. Jakarta: Kementerian Kesehatan RI.
- Cappabianca P, Cavallo LM, Esposito F, de Divitiis E. Endoscopic endonasal transsphenoidal surgery: procedure, endoscopic equipment and instrumentation. Childs Nerv Syst. 2004;20(11):796-801. [PubMed] DOI: 1007/s00381-004-0933-3
- Schreckinger M, Szerlip N, Mittal S. Diabetes insipidus following resection of pituitary tumors. Clin Neurol Neurosurg. 2013;115(2):121-6. [PubMed] DOI: 1016/j.clineuro.2012.08.009
- Castle-Kirszbaum M, Fuller P, Wang YY, King J, Goldschlager T. Diabetes insipidus after endoscopic transsphenoidal surgery: multicenter experience and development of the SALT score. Pituitary. 2021;24(6):867-77. [PubMed] DOI: 1007/s11102-021-01159-y
- RS Pusat Otak Nasional. Rekapitulasi poli rawat jalan Klinik Pituitari RS Pusat Otak Nasional Prof. DR. Mahar Mardjono. 2021.
- Christ-Crain M, Fenske W. Copeptin in the diagnosis of vasopressin-dependent disorders of fluid homeostasis. Nat Rev Endocrinol. 2016;12(3):168-76. [PubMed] DOI: 1038/nrendo.2015.224
- Refardt J, Winzeler B, Christ-Crain M. Copeptin and its role in the diagnosis of diabetes insipidus and the syndrome of inappropriate antidiuresis. Clin Endocrinol (Oxf). 2019;91(1):22-32. [PubMed] DOI: 1111/cen.13991
- Takeuchi K, Nagatani T, Okumura E, Wakabayashi T. A novel method for managing water and electrolyte balance after transsphenoidal surgery: preliminary study of moderate water intake restriction. Nagoya J Med Sci. 2014;76(1-2). [PubMed]
- Martino M, Arnaldi G. Copeptin and Stress. Endocrines. 2021;2(4):384-404. DOI: 3390/endocrines2040035
- Lemetais G, Melander O, Vecchio M, Bottin JH, Enhörning S, Perrier ET. Effect of increased water intake on plasma copeptin in healthy adults. Eur J Nutr. 2018;57(5):1883-90. [PubMed] DOI: 1007/s00394-017-1471-6
- Christ-Crain M, Winzeler B, Refardt J. Diagnosis and management of diabetes insipidus for the internist: an update. J Intern Med. 2021;290(1):73-87. [PubMed] DOI: 1111/joim.13261
- Tommasino C. Fluids and the neurosurgical patient. Anesthesiol Clin North Am. 2002;20(2):329-46. [PubMed] DOI: 1016/s0889-8537(01)00013-x
- Canelo Moreno JM, Dios Fuentes E, Venegas Moreno E, Remón Ruíz PJ, Muñoz Gómez C, Piñar Gutiérrez A, et al. Postoperative water and electrolyte disturbances after extended endoscopic endonasal transsphenoidal surgery. Front Endocrinol (Lausanne). 2022;13. [PubMed] DOI: 3389/fendo.2022.963707
- Blair ET, Clemmer JS, Harkey HL, Hester RL, Pruett WA. Physiologic Mechanisms of Water and Electrolyte Disturbances After Transsphenoidal Pituitary Surgery. World Neurosurg. 2017;107:429-36. [PubMed] DOI: 1016/j.wneu.2017.07.175
- Deni Nasution M, Mouza A, Ari Irsyad M. Transsphenoid surgery outcomes in pituitary adenoma patients at Sumatera Utara University Hospital in 2022–2023: A prospective study. Interdiscip Neurosurg. 2024;36. DOI: 1016/j.inat.2023.101901
- Sari DW, Noguchi-Watanabe M, Sasaki S, Sahar J, Yamamoto-Mitani N. Estimation of sodium and potassium intakes assessed by two 24-hour urine collections in a city of Indonesia. Br J Nutr. 2021;126(10):1537-48. [PubMed] DOI: 1017/S0007114521000271
- Prihatini S, Permaesih D, Diana R. ASUPAN NATRIUM PENDUDUK INDONESIA: Analisis Data Survei Konsumsi Makanan Individu(SKMI) 2014 . Gizi Indon. 2016;39(1):15-24.
- Andarwulan N, Madanijah S, Briawan D, Anwar K, Bararah A, Saraswati, et al. Food consumption pattern and the intake of sugar, salt, and fat in the South Jakarta City—Indonesia. Nutrients. 2021;13(4). [PubMed] DOI: 3390/nu13041289
- Fralick M, Schneeweiss S, Wallis CJD, Jung EH, Kesselheim AS. Desmopressin and the risk of hyponatremia: A population-based cohort study. PLoS Med. 2019;16(10). [PubMed] DOI: 1371/journal.pmed.1002930
- Narayen G, Mandal S. Vasopressin receptor antagonists and their role in clinical medicine. Indian J Endocrinol Metab. 2012;16(2):183-91. [PubMed] DOI: 4103/2230-8210.93734
- Kazempour M, Simani L, Sadeghi M, Nejad FM, Saber A, Futuhi F. Prevention of Hyponatremia After Transsphenoidal Surgery: A Systematic Review. Nephrourol Mon. 2022;14(4). DOI: 5812/numonthly-128929