Kalis Waren 1 , Iwan Fuadi 2 , Ayu Puji Lestari 3
Authors affiliations:
Background & objective: The use of muscle relaxants is a routine in most of the pediatric surgeries. At the end of the surgery, the residual effect of the muscle relaxants needs to be reversed. In most of the countries the reversal agents used are neostigmine plus atropine. In the previous few decades sugammadex has been introduced for this purpose. We reviewed the available research regarding efficacy and safety of sugammadex compared with neostigmine in reversing rocuronium-induced neuromuscular blockade (NMB) among pediatric patients.
Methodology: Using specific keywords, we comprehensively went through the potential articles, comparing efficacy and safety of sugammadex with neostigmine in reversing rocuronium-induced neuromuscular blockade (NMB) among pediatric patients. We searched Europe PMC, Cochrane Library, Scopus, Medline, and ClinicalTrials.gov sources until September 22, 2022. Outcomes were expressed in risk ratio (RR) and mean difference (MD).
Results: A total of 17 randomized controlled trials (RCTs) were included. Our pooled analysis revealed that the use of sugammadex as NMB agent in pediatric population was associated with faster time to reach train of four (TOF) > 0.9 [MD -10.32 min (95% CI -12.42, -8.23), P < 0.00001] and shorter time of extubation [MD -9.40 min (95% CI -11.84, -6.96), P < 0.00001] compared with neostigmine + atropine. Regarding safety, incidence of post-operative nausea vomiting (PONV) [RR 0.44 (95% CI 0.25 – 0.78), P = 0.005] and tachycardia [RR 0.13 (95% CI 0.04 – 0.42), P = 0.0006] was significantly lower among children receiving sugammadex than those who received neostigmine + atropine. Incidence of other adverse events did not differ significantly between the two groups.
Conclusions: Based upon the results of our research, we conclude that sugammadex possesses higher efficacy and better safety than neostigmine to reverse rocuronium-induced NMB among pediatric populations.
Abbreviations: NMB: neuromuscular blockade, NMBA: neuromuscular blocking agents, PMC: PubMed Central, RCT: randomized controlled trial, TOF: train of four
Keywords: Sugammadex; Neostigmine; Neuromuscular Blockade; Pediatric; Meta-Analysis
Citation: Waren K, Fuadi I, Lestari AP. Sugammadex versus neostigmine for neuromuscular blockade reversal after surgery in pediatric patients: a systematic review and meta-analysis of randomized clinical trials. Anaesth. pain intensive care 2025;29(5):393-404. DOI: 10.35975/apic.v29i5.2869
Received: May 09, 2024; Revised: October 26, 2024; Accepted: January 01, 2025
In modern anesthesia, neuromuscular blocking agents (NMBAs) are extensively used for muscle relaxation to facilitate endotracheal intubation and mechanical ventilation during surgical procedures.1,2 However, NMBA may reside and serve as primary contributors to postoperative pulmonary and respiratory problems, hypoxia, upper airway obstruction, and reduced oxygen saturation, potentially elevating the frequency of tracheal re-intubation in critical care settings.3 Due to this reason, another agent is needed to accelerate the reverse effect of NMBAs.4 One of the most common to reverse NMBAs are cholinesterase inhibitor.4
Among cholinesterase inhibitor agents, neostigmine commonly used as this agent is the most potent and selective one.4 However, neostigmine has some downsides such as multi-systemic side effect and adverse effect such as bradycardia, QT-lengthening, bronchoconstriction, hypersalivation, and increased motility.4 To overcome this issue, anticholinergic agent was needed in a mixture with neostigmine, such as sugamadex.5,6 Suggamadex is a γ-cyclodextrin compound that counteracts non-depolarizing neuromuscular blockade (NMB) caused by aminosteroids like rocuronium and vecuronium.5,6
Recent years, usage of sugammadex and supporting evidence of this drug in adult patient have been established, while in pediatric patients this agent has been frequently used in recent years by anesthesiologist.6,7 Sugammadex, being the inaugural non-competitive antagonist for NMB reversal, possesses a distinct mechanism that differs from that of acetylcholinesterase inhibitors.8 It swiftly encapsulates rocuronium or vecuronium by one-to-one molecule binding, delivering rapid and predictable reversal effects of NMB, while effectively reducing the occurrence of residual block.8
In adult patients, sugammadex has been demonstrated to be a safe and superior drug for NMB reversal in comparison to neostigmine, but in the pediatric population the evidence is few. In attempt to enlighten the effect of sugammadex in pediatric population, some meta-analyses had been carried out by Won JW et al.9 and Liu G et al.10, thus concluding that sugammadex is effective and able reversing NMB in short time with low incidences of adverse events. Nonetheless, these meta-analyses are limited by a small number of included studies (ten or fewer) and a small participant pool (fewer than 1,000), with some studies exhibiting a high risk of bias, resulting in low-quality evidence. Therefore, further meta-analysis that include research papers with higher quality and larger sample sizes is necessary for validation. To provide a more comprehensive understanding of sugammadex efficacy and safety in reversing rocuronium-induced NMB in pediatric patients, we conducted an updated meta-analysis of RCTs.
A systematic review and meta-analysis were performed using data from randomized controlled trials (RCTs).
2.1. Studies Selection
Records were included in this study if they met the inclusion criteria established by the Population, Intervention, Comparison, Outcomes and Study (PICOS) framework:
(1) non-randomized interventional studies
(2) observational studies (cohort or case-control), cross-sectional, and case-series
(3) studies on adult population (ages >18 years)
(4) studies which compare sugammadex with placebo or other intervention besides neostigmine
(5) studies lacking a comparison group
(6) unpublished study or abstract; and
(7) nonprimary research.
2.2 Search Strategy and Studies Selection
We evaluated five electronic databases for pertinent research that were published in any language until September 22nd, 2022. These databases include Europe PMC, Scopus, MEDLINE, Cochrane Database (CENTRAL), and ClinicalTrials.gov with following keywords were used for literature searching: "(sugammadex OR bridion OR Org 25969) AND (neostigmine OR prostigmine OR bloxiverz) AND (pediatric OR paediatric OR children) AND (trial OR clinical-trial OR RCT)".
Initially, to determine acceptable research for inclusion, both authors reviewed all retrieved records based on titles and abstracts. A supplementary assessment of references from identified relevant research was performed to identify additional potential papers. Redundant articles were eliminated. The last step involved comprehensive screening of the chosen records according to the established inclusion and exclusion criteria. Any identified disparities were addressed through dialogue. The screening software Covidence was employed to streamline the screening process and eliminate duplicates. The study utilized the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline.11
2.3. Data Extraction and Quality Assessment
Data extraction was conducted separately by two authors. The subsequent items were retrieved from each article: first author, year of study, country, and study type, type of surgery, dosage of neuromuscular blocking agent (NMBA), sugammadex dose, neostigmine dose, time of reversal agent administration, number of samples in each study arms, age, gender, American Society of Anesthesiologist (ASA) score, as well as the outcome of interest.
We have divided the outcomes of interest in this study into the primary and secondary outcomes. The primary outcomes in this study were time to reach train of four (TOF) >0.9 and/or time of extubation. Time to reach TOF >0.9 was defined as time from the start of study drug administration to a TOF ratio of >0.9. Train of four (TOF), also known as peripheral nerve stimulator is an equipment to measure degree of neuromuscular blockade (NMB) which works with the nerve-muscle acceleromyometry principle, usually was placed on the ulnar nerve of all patients to trigger stimulation of the adductor pollicis muscle.12 Time of extubation was defined as the time from stoppage of anaesthetic inhalation until the patient fulfilled global and respiratory criteria for safe extubation. Meanwhile the secondary outcome in this study was any adverse events which occur in both groups. Adverse events assessed in this study may include any of the followings: post-operative nausea and vomiting (PONV), shivering, bradycardia tachycardia, hypotension, bronchospasm/laryngospasm, oxygen desaturation, and respiratory depression.
The evaluation of research quality was performed separately by two authors utilizing the Risk of Bias version 2 (RoB v2) framework from Cochrane Collaborations.13 This instrument assessed methodological quality across five domains: (a) randomization procedure; (b) modifications from planned treatments; (c) incomplete endpoint data; (d) endpoint assessment; and (e) selection of results that were reported. The RCTs were categorized as “low-risk” of bias (low risk across all domains), “high-risk” (high risk in one or more domains), or “some-concern risk” (some concern regarding bias in one or more key areas).
2.4 Statistical Analysis
A meta-analysis was conducted utilizing Review Manager 5.4 software from the Cochrane Collaboration. We aggregated the continuous variable outcomes in this investigation employing the Inverse-Variance methodology with random-effects models, irrespective of heterogeneity, to derive the mean difference (MD) and its standard deviations (SD). The outcomes of the dichotomous variable were aggregated using the Mantel-Haenszel approach with random-effects models to derive the risk ratio (RR) and its 95% confidence interval (95% CI). The Cochrane Q test was employed to assess the heterogeneity of the results, defined by the I-squared statistic (I2, Inconsistency). An I2 score exceeding 50% indicated statistically substantial heterogeneity between studies.14 Data submitted as medians with interquartile ranges or as medians with minimum-to-maximum ranges were transformed into means and standard deviations for meta-analysis pooling utilizing the formula by Wan X et al. (2014).15 Sensitivity analysis through leave-one-out method was performed for outcomes with significant heterogeneity. Funnel plot analysis was employed to evaluate the qualitative risk of publication bias.
3.1 Study Selection and Characteristics
The preliminary database search produced 219 studies, of which 78 were deemed eligible following the screening of titles and abstracts and the elimination of duplicates. Following the full-text screening, 61 articles were subsequently excluded because of the following reasons: twenty three papers were done in adult populations (age >18 years), fourteen papers lacked a comparison/control group, eleven papers were not RCTs, six papers did not utilize neostigmine as the comparison group, five were review articles, and two papers did not report the specified outcomes. The final analysis encompassed a total of 17 studies.16-32 which included a total of 1,213 pediatric who undergo surgical procedures with neuromuscular blockade for the analysis (Figure 1). Sample sizes ranged from 24 to 276.
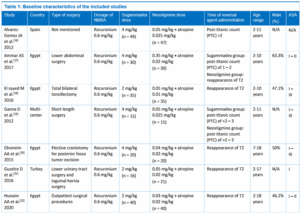
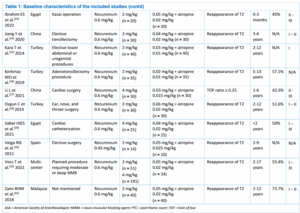
Six trials were from Egypt, four trials were from Turkey, two trials were done in Spain, China, and multi-center, each respectively, and the remaining one trial were from Malaysia. The types of surgery were varied from tonsillectomy to cardiac or gastrointestinal surgery, and even craniotomy procedure. Almost all of the included studies use rocuronium at a dose of 0.6 mg/kg as neuromuscular blocking agent. More than half of the included studies use 2 mg/kg sugammadex, while the rest of trials use the sugammadex at 4 mg/kg dose. The dose of neostigmine used in the included trials ranged from 0.02 mg/kg to 0.35 mg/kg, combined with atropine. These reversal agents were mostly administered at the time of reappearance of T2. Table 1 gives out the details of each included research.
3.2 Quality of Study Assessment
From the employment of RoB v2 tool, it was revealed that eleven17,20-23,25,26,28,29,31,32 out of seventeen included trials have low risk of bias in all five domains which were assessed. The remaining six trials20,22,23,28,31,34 were judged to have some concern risk of bias, one trial23 did not mention details for randomization method and whether there is allocation concealment after randomization, two trials22,31 did not have enough blinding during measurement of outcomes which may cause some bias from the assessors, three trials20,28,34 did not describe the details of randomization process and have lack of blinding during measurement of outcomes. The summary risk of bias assessment is presented in Table 2.
3.3 Primary Outcomes
3.3.1. Time to Reach TOF > 0.9
Sixteen trials (n = 1,125) reported the time to reach TOF > 0.9 outcome. Our pooled analysis revealed that NMB reversal using sugammadex was associated with faster time to reach TOF ratio of > 0.9, when compared with using neostigmine + atropine [Mean Difference -10.32 min (95% CI -12.42, -8.23), P < 0.00001, I2 = 98%, random-effect model] (Figure 2A). Sensitivity analysis through leave-one-out method still showed statistically observed heterogeneity (Supplementary Figure 1).
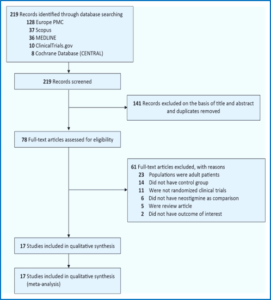
Figure 1: Studies selection flow chart
3.3.2. Time to Extubation
Twelve trials (n = 783) reported the time of extubation outcome. Our pooled analysis revealed that neuromuscular blockade reversal using sugammadex was associated with shorter time of extubation when compared with using neostigmine + atropine [Mean Difference -9.40 min (95% CI -11.84, -6.96), P < 0.00001, I2 = 99%, random-effect model] (Figure 2B). Sensitivity analysis through leave-one-out method still showed statistically significant results without significant changes in the observed heterogeneity (Supplementary Figure 2).
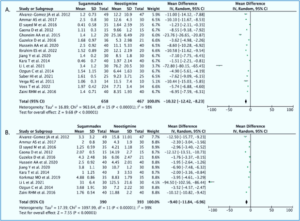
Figure 2: Forest plot that demonstrates the time to reach TOF >0.9 (A) and time of extubation (B) outcome when using sugammadex or neostigmine for neuromuscular blockade reversal in pediatric populations.
vomiting (PONV) when compared with neostigmine + atropine group [RR 0.44 (95% CI 0.25 – 0.78), P = 0.005, I2 = 24%, random-effect model]. Pooled analysis from two trials also revealed that using sugammadex for neuromuscular blockade reversal was associated with lower risk of tachycardia after surgery when compared with neostigmine + atropine [RR 0.13 (95% CI 0.04 – 0.42), P = 0.0006, I2 = 0%, random-effect model]. Meanwhile, the incidence of other adverse events, such as shivering, bradycardia, hypotension, bronchospasm/laryngospasm, oxygen desaturation, and respiratory depression after surgery did not differ significantly between sugammadex and neostigmine + atropine group. The details for each of these adverse events can be seen in Table 3.
3.4. Secondary Outcome
3.4.1. Adverse Events
Pooled analysis from twelve trials revealed that reversal of neuromuscular blockade using sugammadex was associated with lower risk of post-operative nausea and European countries, thereby potentially constraining the generalizability of the findings from this study. Third, the included studies primarily evaluate short-term recovery and immediate post-operative effects with lack of data regarding long-term impact of sugammadex in pediatric patients. Thus, studies with longer time of follow-up duration are still needed to evaluate delayed adverse effects that may arise from the use of sugammadex. Ultimately, several included trials exhibited a "some-concern" risk of bias, particularly regarding the randomization sequence and outcome measurement, necessitating cautious interpretation of the study's findings.
3.5. Publication Bias
We employed Funnel plot analysis to evaluate publication bias in the primary outcome. This analysis revealed a fairly symmetrical inverted curve for the time to achieve TOF >0.9 and the extubation outcome, suggesting the absence of publication bias (Figure 3A and 3B).
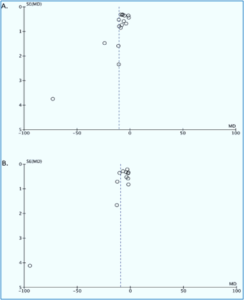
Figure 3: Funnel plot analysis for the primary outcome: time to reach TOF > 0.9 (A), and time of extubation (B).
Sugammadex, a γ-cyclodextrin drug is used to reverse non-depolarizing neuromuscular blockade caused by aminosteroids such as rocuronium and vecuronium. It works by encapsulating them through one-to-one molecular binding, ensuring fast and predictable reversal, while efficiently reducing the risk of residual blockade.5,6,8 Sugammadex has been introduced for use in pediatric population due to the unique physiological characteristics of children, such as the immaturity of their neuromuscular junction receptors, which alters the pharmacodynamic and pharmacokinetic profiles of NMBAs in children compared to adults. The traditional cholinesterase inhibitors, like neostigmine, act by inhibiting the breakdown of acetylcholine, rather than directly targeting NMBA, which can lead to systemic side effects and adverse reactions.4 This highlights the need for further research to optimize sugammadex application particularly in pediatric settings.
Various meta-analyses have been done to analyze efficacy and safety of sugammadex compared to neostigmine as the traditional agent for NMBAs, and found its superiority in providing faster recovery in children. However, these studies have limited sample size (253 patients and 575 patients) and the number of studies included.9,10 Our study try to show an updated finding based on recent studies. Advantage of this study is that it includes larger studies consisting of 17 RCTs with over 1,200 pediatrics patients.
This meta-analysis finds sugammadex could be used in varied types of surgery to help reversing the effect of NMBAs, including in tonsillectomy to cardiac or gastrointestinal surgery, and even craniotomy procedure. Sugammadex could be used in a dose of 2 mg/kg or 4 mg/kg, 2 mg/kg being more prevalent. These doses demonstrated that sugammadex is capable of reducing the duration to reach TOF ratio of > 0.9 in 10.3 min and extubation time in 9.4 min compared to neostigmine plus-atropine as the primary outcome of this study. This result corroborated and reinforced the findings of previous meta-analyses. It also found that sugammadex could reverse the effect of moderate NMB (T2 or T3 reappearance) or profound neuromuscular blockade post-titanic count (PTC) < 2 or 2–3, in pediatric patients, but explanation whether sugammadex sodium was equally effective in mild or in intense NMB, is not described. Another advantage of this study is the analysis of funnel plot showed a relatively symmetrical inverted plot for time to reach TOF > 0.9 and time to extubation outcome, indicating no publication bias.
Secondary outcomes of this study showed that pediatric patients’ tolerance to sugammadex is better in terms of PONV incidence. if compared to neostigmine + atropine. This finding is in line with previous reviews.9,10 Moreover, this study found that sugammadex was associated with a lower risk of tachycardia after surgery when compared with neostigmine + atropine. However, different results were found between studies in terms of bradycardia. This study found there was no difference in the incidence of bradycardia, in line with Zhou S et al. (2023), that found the incidence of bradycardia was not differ with control group.33 Yet, this finding is contrary with previous review by Liu G et al. (2017), and a retrospective analysis by Arends J et al. (2020), that bradycardia is less common with sugammadex than neostigmine.10,34 In terms of other adverse effects, such as shivering, hypotension, bronchospasm/laryngospasm, oxygen desaturation, and respiratory depression after surgery, this study found no significant difference with neostigmine + atropine group.
This study has limitations. Firstly, significant heterogeneity was identified in several outcomes of interest, potentially attributable to variations in the type of surgery performed, differences in the dosage of neostigmine + atropine administered to patients, and diverse participant characteristics. This significant variability suggests that additional research is needed to understand how sugammadex performs across different conditions and patient demographics. Secondly, most of the studies included originated from Egypt and Turkey, with lack of contributions from the American.
Compared with neostigmine, sugammadex may reverse rocuronium-induced neuromuscular blockade more rapidly and safely in pediatric patients. Further multi-center RCTs with large sample sizes should be conducted to help confirm the efficacy and safety of sugammadex in this special population.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
Authors affiliations:
- Kalis Waren, Department of Anesthesiology & Intensive Therapy, Faculty of Medicine, Padjajaran University, Bandung, Indonesia; Email: kaliswaren1996@gmail.com; {ORCID:0000-0002-6903-305X}
- Iwan Fuadi, Department of Anaesthesiology and Intensive Therapy, Faculty of Medicine, Padjajaran University, Bandung, Indonesia; Email: iwan.fuadi@unpad.ac.id; {ORCID:0009-0006-0634-2134}
- Ayu Puji Lestari, Department of Anaesthesiology and Intensive Therapy, Faculty of Medicine, Padjajaran University, Bandung, Indonesia; Email: ayupujilestari988@gmail.com
ABSTRACT
Background & objective: The use of muscle relaxants is a routine in most of the pediatric surgeries. At the end of the surgery, the residual effect of the muscle relaxants needs to be reversed. In most of the countries the reversal agents used are neostigmine plus atropine. In the previous few decades sugammadex has been introduced for this purpose. We reviewed the available research regarding efficacy and safety of sugammadex compared with neostigmine in reversing rocuronium-induced neuromuscular blockade (NMB) among pediatric patients.
Methodology: Using specific keywords, we comprehensively went through the potential articles, comparing efficacy and safety of sugammadex with neostigmine in reversing rocuronium-induced neuromuscular blockade (NMB) among pediatric patients. We searched Europe PMC, Cochrane Library, Scopus, Medline, and ClinicalTrials.gov sources until September 22, 2022. Outcomes were expressed in risk ratio (RR) and mean difference (MD).
Results: A total of 17 randomized controlled trials (RCTs) were included. Our pooled analysis revealed that the use of sugammadex as NMB agent in pediatric population was associated with faster time to reach train of four (TOF) > 0.9 [MD -10.32 min (95% CI -12.42, -8.23), P < 0.00001] and shorter time of extubation [MD -9.40 min (95% CI -11.84, -6.96), P < 0.00001] compared with neostigmine + atropine. Regarding safety, incidence of post-operative nausea vomiting (PONV) [RR 0.44 (95% CI 0.25 – 0.78), P = 0.005] and tachycardia [RR 0.13 (95% CI 0.04 – 0.42), P = 0.0006] was significantly lower among children receiving sugammadex than those who received neostigmine + atropine. Incidence of other adverse events did not differ significantly between the two groups.
Conclusions: Based upon the results of our research, we conclude that sugammadex possesses higher efficacy and better safety than neostigmine to reverse rocuronium-induced NMB among pediatric populations.
Abbreviations: NMB: neuromuscular blockade, NMBA: neuromuscular blocking agents, PMC: PubMed Central, RCT: randomized controlled trial, TOF: train of four
Keywords: Sugammadex; Neostigmine; Neuromuscular Blockade; Pediatric; Meta-Analysis
Citation: Waren K, Fuadi I, Lestari AP. Sugammadex versus neostigmine for neuromuscular blockade reversal after surgery in pediatric patients: a systematic review and meta-analysis of randomized clinical trials. Anaesth. pain intensive care 2025;29(5):393-404. DOI: 10.35975/apic.v29i5.2869
Received: May 09, 2024; Revised: October 26, 2024; Accepted: January 01, 2025
1. INTRODUCTION
In modern anesthesia, neuromuscular blocking agents (NMBAs) are extensively used for muscle relaxation to facilitate endotracheal intubation and mechanical ventilation during surgical procedures.1,2 However, NMBA may reside and serve as primary contributors to postoperative pulmonary and respiratory problems, hypoxia, upper airway obstruction, and reduced oxygen saturation, potentially elevating the frequency of tracheal re-intubation in critical care settings.3 Due to this reason, another agent is needed to accelerate the reverse effect of NMBAs.4 One of the most common to reverse NMBAs are cholinesterase inhibitor.4
Among cholinesterase inhibitor agents, neostigmine commonly used as this agent is the most potent and selective one.4 However, neostigmine has some downsides such as multi-systemic side effect and adverse effect such as bradycardia, QT-lengthening, bronchoconstriction, hypersalivation, and increased motility.4 To overcome this issue, anticholinergic agent was needed in a mixture with neostigmine, such as sugamadex.5,6 Suggamadex is a γ-cyclodextrin compound that counteracts non-depolarizing neuromuscular blockade (NMB) caused by aminosteroids like rocuronium and vecuronium.5,6
Recent years, usage of sugammadex and supporting evidence of this drug in adult patient have been established, while in pediatric patients this agent has been frequently used in recent years by anesthesiologist.6,7 Sugammadex, being the inaugural non-competitive antagonist for NMB reversal, possesses a distinct mechanism that differs from that of acetylcholinesterase inhibitors.8 It swiftly encapsulates rocuronium or vecuronium by one-to-one molecule binding, delivering rapid and predictable reversal effects of NMB, while effectively reducing the occurrence of residual block.8
In adult patients, sugammadex has been demonstrated to be a safe and superior drug for NMB reversal in comparison to neostigmine, but in the pediatric population the evidence is few. In attempt to enlighten the effect of sugammadex in pediatric population, some meta-analyses had been carried out by Won JW et al.9 and Liu G et al.10, thus concluding that sugammadex is effective and able reversing NMB in short time with low incidences of adverse events. Nonetheless, these meta-analyses are limited by a small number of included studies (ten or fewer) and a small participant pool (fewer than 1,000), with some studies exhibiting a high risk of bias, resulting in low-quality evidence. Therefore, further meta-analysis that include research papers with higher quality and larger sample sizes is necessary for validation. To provide a more comprehensive understanding of sugammadex efficacy and safety in reversing rocuronium-induced NMB in pediatric patients, we conducted an updated meta-analysis of RCTs.
2. METHODOLOGY
A systematic review and meta-analysis were performed using data from randomized controlled trials (RCTs).
2.1. Studies Selection
Records were included in this study if they met the inclusion criteria established by the Population, Intervention, Comparison, Outcomes and Study (PICOS) framework:
- P–Population: pediatric patients (age < 18 years old) undergoing surgical procedure with general anesthesia
- I–Intervention: receiving sugammadex with any dose for reversal of NMB after surgical procedure
- C–Control: receiving neostigmine with any dose for reversal of NMB after the surgery was done
- O–Outcome: reporting the primary outcomes (time to reach train of four (TOF) > 0.9 and/or time of extubation), with or without secondary outcome (adverse events) in the sugammadex and neostigmine group
- S–Study design: RCTs
(1) non-randomized interventional studies
(2) observational studies (cohort or case-control), cross-sectional, and case-series
(3) studies on adult population (ages >18 years)
(4) studies which compare sugammadex with placebo or other intervention besides neostigmine
(5) studies lacking a comparison group
(6) unpublished study or abstract; and
(7) nonprimary research.
2.2 Search Strategy and Studies Selection
We evaluated five electronic databases for pertinent research that were published in any language until September 22nd, 2022. These databases include Europe PMC, Scopus, MEDLINE, Cochrane Database (CENTRAL), and ClinicalTrials.gov with following keywords were used for literature searching: "(sugammadex OR bridion OR Org 25969) AND (neostigmine OR prostigmine OR bloxiverz) AND (pediatric OR paediatric OR children) AND (trial OR clinical-trial OR RCT)".
Initially, to determine acceptable research for inclusion, both authors reviewed all retrieved records based on titles and abstracts. A supplementary assessment of references from identified relevant research was performed to identify additional potential papers. Redundant articles were eliminated. The last step involved comprehensive screening of the chosen records according to the established inclusion and exclusion criteria. Any identified disparities were addressed through dialogue. The screening software Covidence was employed to streamline the screening process and eliminate duplicates. The study utilized the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline.11
2.3. Data Extraction and Quality Assessment
Data extraction was conducted separately by two authors. The subsequent items were retrieved from each article: first author, year of study, country, and study type, type of surgery, dosage of neuromuscular blocking agent (NMBA), sugammadex dose, neostigmine dose, time of reversal agent administration, number of samples in each study arms, age, gender, American Society of Anesthesiologist (ASA) score, as well as the outcome of interest.
We have divided the outcomes of interest in this study into the primary and secondary outcomes. The primary outcomes in this study were time to reach train of four (TOF) >0.9 and/or time of extubation. Time to reach TOF >0.9 was defined as time from the start of study drug administration to a TOF ratio of >0.9. Train of four (TOF), also known as peripheral nerve stimulator is an equipment to measure degree of neuromuscular blockade (NMB) which works with the nerve-muscle acceleromyometry principle, usually was placed on the ulnar nerve of all patients to trigger stimulation of the adductor pollicis muscle.12 Time of extubation was defined as the time from stoppage of anaesthetic inhalation until the patient fulfilled global and respiratory criteria for safe extubation. Meanwhile the secondary outcome in this study was any adverse events which occur in both groups. Adverse events assessed in this study may include any of the followings: post-operative nausea and vomiting (PONV), shivering, bradycardia tachycardia, hypotension, bronchospasm/laryngospasm, oxygen desaturation, and respiratory depression.
The evaluation of research quality was performed separately by two authors utilizing the Risk of Bias version 2 (RoB v2) framework from Cochrane Collaborations.13 This instrument assessed methodological quality across five domains: (a) randomization procedure; (b) modifications from planned treatments; (c) incomplete endpoint data; (d) endpoint assessment; and (e) selection of results that were reported. The RCTs were categorized as “low-risk” of bias (low risk across all domains), “high-risk” (high risk in one or more domains), or “some-concern risk” (some concern regarding bias in one or more key areas).
2.4 Statistical Analysis
A meta-analysis was conducted utilizing Review Manager 5.4 software from the Cochrane Collaboration. We aggregated the continuous variable outcomes in this investigation employing the Inverse-Variance methodology with random-effects models, irrespective of heterogeneity, to derive the mean difference (MD) and its standard deviations (SD). The outcomes of the dichotomous variable were aggregated using the Mantel-Haenszel approach with random-effects models to derive the risk ratio (RR) and its 95% confidence interval (95% CI). The Cochrane Q test was employed to assess the heterogeneity of the results, defined by the I-squared statistic (I2, Inconsistency). An I2 score exceeding 50% indicated statistically substantial heterogeneity between studies.14 Data submitted as medians with interquartile ranges or as medians with minimum-to-maximum ranges were transformed into means and standard deviations for meta-analysis pooling utilizing the formula by Wan X et al. (2014).15 Sensitivity analysis through leave-one-out method was performed for outcomes with significant heterogeneity. Funnel plot analysis was employed to evaluate the qualitative risk of publication bias.
3. RESULTS
3.1 Study Selection and Characteristics
The preliminary database search produced 219 studies, of which 78 were deemed eligible following the screening of titles and abstracts and the elimination of duplicates. Following the full-text screening, 61 articles were subsequently excluded because of the following reasons: twenty three papers were done in adult populations (age >18 years), fourteen papers lacked a comparison/control group, eleven papers were not RCTs, six papers did not utilize neostigmine as the comparison group, five were review articles, and two papers did not report the specified outcomes. The final analysis encompassed a total of 17 studies.16-32 which included a total of 1,213 pediatric who undergo surgical procedures with neuromuscular blockade for the analysis (Figure 1). Sample sizes ranged from 24 to 276.
Six trials were from Egypt, four trials were from Turkey, two trials were done in Spain, China, and multi-center, each respectively, and the remaining one trial were from Malaysia. The types of surgery were varied from tonsillectomy to cardiac or gastrointestinal surgery, and even craniotomy procedure. Almost all of the included studies use rocuronium at a dose of 0.6 mg/kg as neuromuscular blocking agent. More than half of the included studies use 2 mg/kg sugammadex, while the rest of trials use the sugammadex at 4 mg/kg dose. The dose of neostigmine used in the included trials ranged from 0.02 mg/kg to 0.35 mg/kg, combined with atropine. These reversal agents were mostly administered at the time of reappearance of T2. Table 1 gives out the details of each included research.


3.2 Quality of Study Assessment
From the employment of RoB v2 tool, it was revealed that eleven17,20-23,25,26,28,29,31,32 out of seventeen included trials have low risk of bias in all five domains which were assessed. The remaining six trials20,22,23,28,31,34 were judged to have some concern risk of bias, one trial23 did not mention details for randomization method and whether there is allocation concealment after randomization, two trials22,31 did not have enough blinding during measurement of outcomes which may cause some bias from the assessors, three trials20,28,34 did not describe the details of randomization process and have lack of blinding during measurement of outcomes. The summary risk of bias assessment is presented in Table 2.
3.3 Primary Outcomes
3.3.1. Time to Reach TOF > 0.9
Sixteen trials (n = 1,125) reported the time to reach TOF > 0.9 outcome. Our pooled analysis revealed that NMB reversal using sugammadex was associated with faster time to reach TOF ratio of > 0.9, when compared with using neostigmine + atropine [Mean Difference -10.32 min (95% CI -12.42, -8.23), P < 0.00001, I2 = 98%, random-effect model] (Figure 2A). Sensitivity analysis through leave-one-out method still showed statistically observed heterogeneity (Supplementary Figure 1).

Figure 1: Studies selection flow chart
3.3.2. Time to Extubation
Twelve trials (n = 783) reported the time of extubation outcome. Our pooled analysis revealed that neuromuscular blockade reversal using sugammadex was associated with shorter time of extubation when compared with using neostigmine + atropine [Mean Difference -9.40 min (95% CI -11.84, -6.96), P < 0.00001, I2 = 99%, random-effect model] (Figure 2B). Sensitivity analysis through leave-one-out method still showed statistically significant results without significant changes in the observed heterogeneity (Supplementary Figure 2).

Figure 2: Forest plot that demonstrates the time to reach TOF >0.9 (A) and time of extubation (B) outcome when using sugammadex or neostigmine for neuromuscular blockade reversal in pediatric populations.
vomiting (PONV) when compared with neostigmine + atropine group [RR 0.44 (95% CI 0.25 – 0.78), P = 0.005, I2 = 24%, random-effect model]. Pooled analysis from two trials also revealed that using sugammadex for neuromuscular blockade reversal was associated with lower risk of tachycardia after surgery when compared with neostigmine + atropine [RR 0.13 (95% CI 0.04 – 0.42), P = 0.0006, I2 = 0%, random-effect model]. Meanwhile, the incidence of other adverse events, such as shivering, bradycardia, hypotension, bronchospasm/laryngospasm, oxygen desaturation, and respiratory depression after surgery did not differ significantly between sugammadex and neostigmine + atropine group. The details for each of these adverse events can be seen in Table 3.
| Table 3: Secondary outcome of adverse events in both group of interventions | ||||||
| Adverse events | Number of studies | Sugammadex group
|
Neostigmine group
|
Risk ratio
(95% CI) |
I2
(%) |
P-value |
| PONV | 12 | 53/577 (9.2) | 59/372 (15.8) | 0.44 (0.25 – 0.78) | 24 | 0.005 |
| Shivering | 2 | 7/50 (14) | 9/50 (18) | 0.78 (0.31 – 1.92) | 0 | 0.59 |
| Bradycardia | 6 | 7/391 (1.8) | 16/181 (8.8) | 0.31 (0.06 – 1.75) | 48 | 0.19 |
| Tachycardia | 2 | 3/50 (6) | 24/50 (48) | 0.13 (0.04 – 0.42) | 0 | 0.0006 |
| Hypotension | 3 | 6/95 (6.3) | 3/95 (3.2) | 1.71 (0.47 – 6.20) | 0 | 0.42 |
| Bronchospasm/ Laryngospasm |
3 | 1/114 (0.9) | 4/112 (3.5) | 0.46 (0.08 – 2.67) | 0 | 0.39 |
| Oxygen desaturation | 4 | 3/135 (2.2) | 5/135 (3.7) | 0.66 (0.17 – 2.50) | 0 | 0.54 |
| Respiratory depression | 4 | 0/121 (0) | 1/126 (0.8) | 0.33 (0.01 – 7.91) | 0 | 0.50 |
| Data presented as n (%);
CI = confidence interval; PONV = post-operative nausea and vomiting |
||||||
3.4. Secondary Outcome
3.4.1. Adverse Events
Pooled analysis from twelve trials revealed that reversal of neuromuscular blockade using sugammadex was associated with lower risk of post-operative nausea and European countries, thereby potentially constraining the generalizability of the findings from this study. Third, the included studies primarily evaluate short-term recovery and immediate post-operative effects with lack of data regarding long-term impact of sugammadex in pediatric patients. Thus, studies with longer time of follow-up duration are still needed to evaluate delayed adverse effects that may arise from the use of sugammadex. Ultimately, several included trials exhibited a "some-concern" risk of bias, particularly regarding the randomization sequence and outcome measurement, necessitating cautious interpretation of the study's findings.
3.5. Publication Bias
We employed Funnel plot analysis to evaluate publication bias in the primary outcome. This analysis revealed a fairly symmetrical inverted curve for the time to achieve TOF >0.9 and the extubation outcome, suggesting the absence of publication bias (Figure 3A and 3B).

Figure 3: Funnel plot analysis for the primary outcome: time to reach TOF > 0.9 (A), and time of extubation (B).
4. DISCUSSION
Sugammadex, a γ-cyclodextrin drug is used to reverse non-depolarizing neuromuscular blockade caused by aminosteroids such as rocuronium and vecuronium. It works by encapsulating them through one-to-one molecular binding, ensuring fast and predictable reversal, while efficiently reducing the risk of residual blockade.5,6,8 Sugammadex has been introduced for use in pediatric population due to the unique physiological characteristics of children, such as the immaturity of their neuromuscular junction receptors, which alters the pharmacodynamic and pharmacokinetic profiles of NMBAs in children compared to adults. The traditional cholinesterase inhibitors, like neostigmine, act by inhibiting the breakdown of acetylcholine, rather than directly targeting NMBA, which can lead to systemic side effects and adverse reactions.4 This highlights the need for further research to optimize sugammadex application particularly in pediatric settings.
Various meta-analyses have been done to analyze efficacy and safety of sugammadex compared to neostigmine as the traditional agent for NMBAs, and found its superiority in providing faster recovery in children. However, these studies have limited sample size (253 patients and 575 patients) and the number of studies included.9,10 Our study try to show an updated finding based on recent studies. Advantage of this study is that it includes larger studies consisting of 17 RCTs with over 1,200 pediatrics patients.
This meta-analysis finds sugammadex could be used in varied types of surgery to help reversing the effect of NMBAs, including in tonsillectomy to cardiac or gastrointestinal surgery, and even craniotomy procedure. Sugammadex could be used in a dose of 2 mg/kg or 4 mg/kg, 2 mg/kg being more prevalent. These doses demonstrated that sugammadex is capable of reducing the duration to reach TOF ratio of > 0.9 in 10.3 min and extubation time in 9.4 min compared to neostigmine plus-atropine as the primary outcome of this study. This result corroborated and reinforced the findings of previous meta-analyses. It also found that sugammadex could reverse the effect of moderate NMB (T2 or T3 reappearance) or profound neuromuscular blockade post-titanic count (PTC) < 2 or 2–3, in pediatric patients, but explanation whether sugammadex sodium was equally effective in mild or in intense NMB, is not described. Another advantage of this study is the analysis of funnel plot showed a relatively symmetrical inverted plot for time to reach TOF > 0.9 and time to extubation outcome, indicating no publication bias.
Secondary outcomes of this study showed that pediatric patients’ tolerance to sugammadex is better in terms of PONV incidence. if compared to neostigmine + atropine. This finding is in line with previous reviews.9,10 Moreover, this study found that sugammadex was associated with a lower risk of tachycardia after surgery when compared with neostigmine + atropine. However, different results were found between studies in terms of bradycardia. This study found there was no difference in the incidence of bradycardia, in line with Zhou S et al. (2023), that found the incidence of bradycardia was not differ with control group.33 Yet, this finding is contrary with previous review by Liu G et al. (2017), and a retrospective analysis by Arends J et al. (2020), that bradycardia is less common with sugammadex than neostigmine.10,34 In terms of other adverse effects, such as shivering, hypotension, bronchospasm/laryngospasm, oxygen desaturation, and respiratory depression after surgery, this study found no significant difference with neostigmine + atropine group.
5. LIMITATIONS
This study has limitations. Firstly, significant heterogeneity was identified in several outcomes of interest, potentially attributable to variations in the type of surgery performed, differences in the dosage of neostigmine + atropine administered to patients, and diverse participant characteristics. This significant variability suggests that additional research is needed to understand how sugammadex performs across different conditions and patient demographics. Secondly, most of the studies included originated from Egypt and Turkey, with lack of contributions from the American.
6. CONCLUSION
Compared with neostigmine, sugammadex may reverse rocuronium-induced neuromuscular blockade more rapidly and safely in pediatric patients. Further multi-center RCTs with large sample sizes should be conducted to help confirm the efficacy and safety of sugammadex in this special population.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
11. REFERENCES
- Penzo B, Petrò L, DeGasperi A. Neuromuscular blocking agents: review on agents (NMBA and antagonists) and monitoring. In: Chiumello, D. (eds) Pract Trends Anesth Intensive Care 2019; 2020. p. 41–62. DOI: 1007/978-3-030-43873-9_4
- Murphy GS. Neuromuscular Monitoring in the Perioperative Period. Anesth Analg. 2018;126(2):464-468. [PubMed] DOI: 1213/ANE.0000000000002387
- Murphy GS, Szokol JW, Avram MJ, Greenberg SB, Shear T, Vender JS, et al. Postoperative residual neuromuscular blockade is associated with impaired clinical recovery. Anesth Analg. 2013;117(1):133-41. [PubMed] DOI: 1213/ANE.0b013e3182742e75
- Abad-Gurumeta A, Ripollés-Melchor J, Casans-Francés R, Espinosa A, Martínez-Hurtado E, Fernández-Pérez C, et al. A systematic review of sugammadex vs neostigmine for reversal of neuromuscular blockade. Anaesthesia. 2015;70(12):1441-52. [PubMed] DOI: 1111/anae.13277
- Bom A, Bradley M, Cameron K, Clark JK, Van Egmond J, Feilden H, et al. A novel concept of reversing neuromuscular block: chemical encapsulation of rocuronium bromide by a cyclodextrin-based synthetic host. Angew Chem Int Ed Engl. 2002;41(2):266-70. [PubMed] DOI: 1002/1521-3773(20020118)41:2<265::aid-anie265>3.0.co;2-q
- Ledowski T. Sugammadex: what do we know and what do we still need to know? A review of the recent (2013 to 2014) literature. Anaesth Intensive Care. 2015;43(1):14-22. [PubMed] DOI: 1177/0310057X1504300104
- Faulk DJ, Austin TM, Thomas JJ, Strupp K, Macrae AW, Yaster M. A Survey of the Society for Pediatric Anesthesia on the Use, Monitoring, and Antagonism of Neuromuscular Blockade. Anesth Analg. 2021;132(6):1518-1526. [PubMed] DOI: 1213/ANE.0000000000005386
- Caldwell JE. Sugammadex: past, present, and future. Adv Anesth. 2011;29(1):19–37. DOI: 1016/j.aan.2011.07.007
- Won YJ, Lim BG, Lee DK, Kim H, Kong MH, Lee IO. Sugammadex for reversal of rocuronium-induced neuromuscular blockade in pediatric patients: A systematic review and meta-analysis. Medicine (Baltimore). 2016;95(34):e4678. [PubMed] DOI: 1097/MD.0000000000004678
- Liu G, Wang R, Yan Y, Fan L, Xue J, Wang T. The efficacy and safety of sugammadex for reversing postoperative residual neuromuscular blockade in pediatric patients: A systematic review. Sci Rep. 2017;7(1):5724. [PubMed] DOI: 1038/s41598-017-06159-2
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [PubMed] DOI: 1136/bmj.n71
- Capron F, Alla F, Hottier C, Meistelman C, Fuchs-Buder T. Can acceleromyography detect low levels of residual paralysis? A probability approach to detect a mechanomyographic train-of-four ratio of 0.9. Anesthesiology. 2004;100(5):1119-24. [PubMed] DOI: 1097/00000542-200405000-00013
- Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. [PubMed] DOI: 1136/bmj.l4898
- Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557-60. [PubMed] DOI: 1136/bmj.327.7414.557
- Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. 2014;14:135. [PubMed] DOI: 1186/1471-2288-14-135
- Alvarez-Gomez JA, Baron M, Ruiz GV, Lopez RL, Meija JB, Asensi PD, et al. Efficacy and safety of the reversal with sugammadex from deep rocuronium-induced neuromuscular blockade in children; 2012.
- Ammar AS, Mahmoud KM, Kasemy ZA. A comparison of sugammadex and neostigmine for reversal of rocuronium-induced neuromuscular blockade in children. Acta Anaesthesiol Scand. 2017;61(4):374-380. [PubMed] DOI: 1111/aas.12868
- El sayed M, Hasan S. Does sugammadex facilitate recovery after outpatient tonsillectomy in children? Egypt J Anesth. 2016;32(4):447-450. DOI: 1016/j.egja.2016.08.021
- Gaona D, Carceles M, Veiga G, Lopez R, Mejia J, Asensi P. Efficacy and safety of the reversal with sugammadex in deep neuromuscular blockade induced by rocuronium in pediatrics. Br J Anaesth. 2012;108(S2):308–309.
- Ghoneim AA, El Beltagy MA. Comparative study between sugammadex and neostigmine in neurosurgical anesthesia in pediatric patients. Saudi J Anaesth. 2015;9(3):247-52. [PubMed] DOI: 4103/1658-354X.154696
- Güzelce D, Kendigelen P, Tütüncü A, Kaya G, Altintas F. Comparison of sugammadex and neostigmine in terms of time to extubation in pediatrics. Med Bull Haseki. 2016;54:207–211. DOI: 4274/haseki.3091
- Hussein AA, Hegazy MM, Elramely MA, Mohamed AG. Sugammadex versus neostigmine in pediatric cancer patients undergoing outpatient surgical procedures. Int J Res Pharm Sci. 2020;11(2):2741–2746. Full Text
- Ibrahim ES, Elkhadry SW. Sugammadex versus neostigmine for reversal of rocuronium-induced neuromuscular blockade in infants: A prospective randomized pilot study. Perioper Care Oper Room Manag. 2022;26:100224. DOI: 1016/j.pcorm.2021.100224
- Jiang Y, Wei R, Zhang R, Zheng J. Efficacy and safety of sugammadex in reversing rocuronium-induced neuromuscular blockade in children post elective tonsillectomy. Pharm Clin Res. 2020;02:139–141. DOI: 13664/j.cnki.pcr.2020.02.015
- Kara T, Ozbagriacik O, Turk HS, Isil CT, Gokuc O, Unsal O, et al. Sugammadex versus neostigmina em pacientes pediátricos: estudo prospectivo e randomizado. Rev Bras Anestesiol. 2014;64(6):400-5. [PubMed] DOI: 1016/j.bjan.2014.03.001
- Korkmaz MO, Sayhan H, Guven M. Does sugammadex decrease the severity of agitation and complications in pediatric patients undergoing adenotonsillectomy? Saudi Med J. 2019;40(9):907-913. [PubMed] DOI: 15537/smj.2019.9.24485
- Li L, Jiang Y, Zhang W. Sugammadex for Fast-Track Surgery in Children Undergoing Cardiac Surgery: A Randomized Controlled Study. J Cardiothorac Vasc Anesth. 2021;35(5):1388-1392. [PubMed] DOI: 1053/j.jvca.2020.08.069
- Ozgün C, Cakan T, Baltacı B, Başar H. Comparison of reversal and adverse effects of sugammadex and combination of - Anticholinergic-Anticholinesterase agents in pediatric patients. J Res Med Sci. 2014;19(8):762-8. [PubMed]
- Saber HIES, Mousa SA, AbouRezk AR, Zaglool A. Recovery Profile of Sugammadex versus Neostigmine in Pediatric Patients Undergoing Cardiac Catheterization: A Randomized Double-Blind Study. Anesth Essays Res. 2021;15(3):272-278. [PubMed] DOI: 4103/aer.aer_139_21
- Veiga R, Carceles B, Dominguez S, Lopez F, Orozco M, Alvarez-Gomez J. Sugammadex reversal efficacy and security vs neostigmine in the rocuronium-induced neuromuscular blockade in paediatric patients. Eur J Anaesthesiol. 2011;28:153. DOI: 1097/00003643-201106001-00490
- Voss T, Wang A, DeAngelis M, Speek M, Saldien V, Hammer GB, et al. Sugammadex for reversal of neuromuscular blockade in pediatric patients: Results from a phase IV randomized study. Paediatr Anaesth. 2022;32(3):436-445. [PubMed] DOI: 1111/pan.14370
- Mohamad Zaini RH, Penny Tevaraj JM, Wan Hassan WN, Iberahim MI, Shukeri WM. Comparison between the efficacy of neostigmine versus sugammadex for reversal of rocuronium induced neuromuscular blockade in paediatric patients. Anesth Analg. 2016;123:329. DOI: 1213/01.ane.0000492650.20862.e4
- Zhou S, Hu H, Ru J. Efficacy and safety of sugammadex sodium in reversing rocuronium-induced neuromuscular blockade in children: An updated systematic review and meta-analysis. Heliyon. 2023;9(8):e18356. [PubMed] DOI: 1016/j.heliyon.2023.e18356
- Arends J, Hubbard R, Shafy SZ, Hakim M, Kim SS, Tumin D, et al. Heart Rate Changes Following the Administration of Sugammadex to Infants and Children With Comorbid Cardiac, Cardiovascular, and Congenital Heart Diseases. Cardiol Res. 2020;11(5):274-279. [PubMed] DOI: 14740/cr1045