Ekramy M. Elmorsy 1, Basil D. M. Alruwaili 2, Hatem R. H. Alrwaili 3, Anwar S. J. Alrawaili 4, Abdulelah M. M. Alanazi 5, Fahad N. K. Alanazi 6, Ibraheem D. H. Alanazi 7
Authors affiliations:
Background & objectives: Tuberculosis (TB) continues to be a significant global health issue, as an increasing number of individuals are presenting with severe, life-threatening conditions that require admission to intensive care units (ICUs). The management of critical care environments is increasingly complicated by the rise of drug-resistant tuberculosis, co-infection with HIV, and late-stage diagnoses. Despite its increasing significance, there is a paucity of focused research on the critical care aspects of tuberculosis.
This review aims to systematically examine the current data and clinical guidelines pertaining to the treatment of tuberculosis in critical care settings. The focus is on diagnostic challenges, treatment choices, supportive methods, and outcome forecasts relevant to critically ill TB patients.
Methodology: A structured literature evaluation was conducted, focusing on publications from 2000 to 2025, using databases such as [PubMed], Scopus, and Web of Science. This study examines the management of tuberculosis in intensive and critical care settings, focusing on multidrug-resistant tuberculosis (MDR-TB), the role of mechanical ventilation, and the implications of sepsis in affected patients. A review of clinical recommendations, cohort studies, retrospective analyses, and relevant case series facilitated the summarization of the database for ICU care techniques in tuberculosis patients.
Results: Critically ill tuberculosis patients primarily exhibit severe respiratory failure, miliary spread, tuberculosis sepsis, or central nervous system involvement. Suboptimal outcomes stem from diagnostic delays caused by ambiguous clinical features and the limitations of conventional microbiological diagnostics. In ICU cohorts with tuberculosis, mortality rates range from 30% to 70%, with elevated rates observed in patients requiring mechanical ventilation or experiencing multi-organ failure. Delayed initiation of antitubercular treatment (ATT) in the ICU often results in altered pharmacokinetics, that hinder medication distribution. Drug-resistant tuberculosis continues to pose significant challenges regarding therapy selection and infection control. Supportive care interventions, including mechanical ventilation, fluid resuscitation, and nutritional support, are critically important yet often underappreciated.
Conclusion: Tuberculosis in the ICU represents a distinct clinical entity characterized by elevated mortality rates, diagnostic delays, and complex therapeutic decisions. Enhancing outcomes relies on early detection, prompt initiation of antitubercular therapy, customized pharmaceutical interventions, and multidisciplinary supportive care. Prospective studies are essential to establish ICU-specific tuberculosis care guidelines, especially for individuals with drug resistance and co-infections.
Abbreviations: APACHE II: Acute Physiology and Chronic Health Evaluation II, ARDS: acute respiratory distress syndrome ATT: antitubercular treatment, CRP: C-reactive protein, DR-TB: drug-resistant tuberculosis, ECMO: extracorporeal membrane oxygenation, ICU: intensive care units, MDR-TB: multidrug-resistant tuberculosis, TB: Tuberculosis, XDR extensively drug-resistant, TDM: Therapeutic antibiotic monitoring
Keywords: Tuberculosis; Critical Care; Intensive Care Unit (ICU); Antitubercular Therapy; Drug-Resistant TB
Citation: Ekramy M. Elmorsy, Basil D. M. Alruwaili, Hatem R. H. Alrwaili, Anwar S. J. Alrawaili, Abdulelah M. M. Alanazi, Fahad N. K. Alanazi, and Ibraheem D. H. Alanazi. Critical care management of tuberculosis: challenges, evidence, and evolving strategies. Anaesth. pain intensive care 2025;29(5):405-417. DOI: 10.35975/apic.v29i5.2867
Received: June 11, 2025; Revised: June 28, 2025; Accepted: July 11, 2025
Tuberculosis (TB) continues to pose a significant global health challenge, with projections of 10.6 million new cases and 1.6 million deaths in 2021. While the majority of tuberculosis cases are managed in outpatient settings, certain patients experience severe complications necessitating admission to an intensive care unit (ICU).1 Stated death rates for TB range from 50% to 60%, with approximately 3–4% of hospitalized patients requiring intensive care unit (ICU) treatment.2 Critically ill tuberculosis patients frequently present with sudden respiratory failure, septic shock, or multi-organ dysfunction, conditions often worsened by delayed diagnosis and the initiation of appropriate treatment.3 The limitations of conventional microbiological testing, coupled with the nonspecific clinical manifestations of tuberculosis in critically ill patients, exacerbate diagnostic challenges.4 In critically ill patients, the pharmacokinetics of antitubercular medications may be significantly altered, leading to subtherapeutic drug levels and complicating treatment.5
The emergence of extensively drug-resistant (XDR) and multidrug-resistant (MDR), TB bacteria introduces additional challenges to the management of TB in critical care settings. Such situations necessitate close monitoring and supportive treatment due to the requirement for contemporary, potentially hazardous, and inadequately researched medication regimens.6 Despite the high mortality and complex treatment associated with tuberculosis in the ICU, there is a lack of targeted research and evidence-based guidelines to address this issue. This review aims to summarize current information on the critical care management of tuberculosis, highlighting areas that require further research, challenges in diagnosis, therapeutic strategies, and supportive care measures.
A systematic literature review covering the years 2000 to 2025 was conducted. We conducted a search in [PubMed], Scopus, and Web of Science using the following phrases and Boolean operators: ("tuberculosis" OR "TB") AND ("intensive care" OR "critical care" OR "ICU") AND ("mechanical ventilation" OR "sepsis" OR "MDR-TB"). Studies were required to meet the following criteria for inclusion: The articles were composed in English, focused on adult patients (≥18 years) with tuberculosis necessitating ICU admission, and examined critical care-specific clinical outcomes, management strategies, and related issues. The study analyzed cohort studies, retrospective analyses, case series, and clinical guidelines. Research focusing exclusively on children, animals, or in vitro studies, as well as articles not pertaining to intensive or critical care, were excluded from consideration. Two reviewers conducted independent examinations of titles and abstracts. Full texts of relevant articles were reviewed to confirm eligibility. All parties reached a consensus to address the issues.
The emergence of extensively drug-resistant (XDR) and multidrug-resistant (MDR) tuberculosis strains significantly complicates the critical care management of the disease. Such situations necessitate close monitoring and supportive treatment due to the requirement for contemporary, potentially hazardous, and inadequately researched medication regimens.6 In critically ill patients, the pharmacokinetics of antitubercular medications may be significantly altered, leading to subtherapeutic drug levels and complicating treatment.10 Despite the high mortality and complex treatment associated with tuberculosis in the ICU, there is a lack of targeted research and evidence-based recommendations to address this issue. This review aims to summarize current information on the critical care management of tuberculosis, highlighting areas that require further research, challenges in diagnosis, therapeutic strategies, and supportive care measures.
Tuberculosis (TB) in the intensive care unit (ICU) complicates early identification and treatment due to its presentation of various ambiguous clinical symptoms.3 Acute respiratory failure, typically associated with severe pulmonary disease, is the primary reason for ICU admission among TB patients.11 Radiographic findings typically reveal bilateral infiltrates and cavitary lesions. Common complications such as bacterial superinfection, chronic obstructive pulmonary disease, and cancer further restrict respiratory function.12 A significant number of these patients exhibited acute respiratory distress syndrome (ARDS), and post-mortem analyses indicate that confluent tuberculous bronchopneumonia may resemble ARDS.13
Extrapulmonary tuberculosis, characterized by symptoms such as central nervous system involvement (e.g., tuberculous meningitis), lymphadenopathy, pleural effusion, and miliary spread, constitutes a notable proportion of ICU admissions.14 Miliary tuberculosis is characterized by extensive hematogenous dissemination, presenting with nonspecific systemic symptoms such as fever, weight loss, and hepatosplenomegaly.15 Disseminated tuberculosis is more prevalent among immunocompromised individuals, particularly those with HIV co-infection, and may present atypically, often complicating the diagnostic process.16
Patients with critical illness tuberculosis frequently exhibit systemic symptoms such as weight loss, anorexia, night sweats, and persistent fever.3 Examples of organ dysfunctions that demonstrate the systemic nature of the disease include hepatic impairment, renal failure, and adrenal insufficiency. Altered mental state and seizures, as neurological symptoms, may indicate central nervous system involvement.17 Paradoxical reactions complicate diagnosis and treatment, as they involve clinical decline despite appropriate therapeutic interventions.18
In endemic regions or among high-risk populations, the diverse presentations of tuberculosis in the ICU necessitate heightened clinical suspicion. Enhancing outcomes relies on timely recognition of antitubercular therapy and prompt initiation of treatment.19 The effective management of these complex cases relies on comprehensive evaluation, which includes assessing extrapulmonary involvement and potential co-infections.
4.2. Diagnostic Challenges in ICU for TB
Diagnosing TB in critically ill patients in the ICU presents significant challenges due to unusual clinical presentations, constraints of conventional diagnostic techniques, and the urgency needed to begin suitable treatment.20 Since critically ill patients usually have ambiguous symptoms like fever, respiratory problems, or altered mental state that could be ascribed to various diseases, TB diagnosis is more challenging.21 Furthermore, diagnosis might be delayed due to comorbidities and the effects of immunosuppression, which might conceal typical TB symptoms. Complicating the diagnosis, immunocompromised patients—including those on immunosuppressive therapy or HIV—may present unique TB symptoms and run a higher risk of spreading disease.22 Early diagnosis is even more crucial; for critically sick patients, appropriate therapy starts with fast and accurate diagnostic instruments. Higher morbidity and death can result from delays.20
Tools for conventional diagnosis have some restrictions. Getting quality sputum samples from patients under intubation or vegetative state is challenging. While induced sputum or bronchoalveolar lavage may be required in unstable patients, they can be harmful.23 Though the gold standard is still Smear Microscopy and Culture, it takes time—often weeks—which is untenable in acute situations. Especially in paucibacillary conditions, smear microscopy has poor sensitivity.24 While Xpert MTB/RIF molecular diagnostics offers fast results, in those with low bacterial loads or in extrapulmonary TB its sensitivity may be reduced.25 Moreover, overlapping findings of several pulmonary illnesses make reading chest radiographs in ICU patients difficult. Advanced imaging modalities as CT scans provide more detail, so unstable patients might not be able to receive them.12 Particularly in endemic areas or among high-risk groups, resolving the diagnostic challenges of tuberculosis in the ICU demands for a high index of suspicion. Fast molecular diagnostics, together with the integration of clinical, radiographic, and laboratory findings, define timeliness in diagnosis and therapy.12,23-25
4.3. Antitubercular Therapy in Critical Illness
The management of tuberculosis (TB) in critically ill patients presents unique challenges due to altered pharmacokinetics, restricted medication formulations, and the necessity for initiating effective therapy.10 Standard first-line antitubercular medications—rifampicin, isoniazid, pyrazinamide, and ethambutol—may exhibit subtherapeutic levels in this population, necessitating tailored treatments to ensure optimal outcomes.
In critical illness, there are significant alterations in drug absorption, distribution, metabolism, and excretion.26 Factors such as gastrointestinal hypomotility, vasopressor usage, and fluid changes may compromise enteral absorption and alter drug distribution volumes. Research indicates that critically ill patients with nasogastric tubes frequently exhibit reduced plasma concentrations of first-line antitubercular medications, particularly rifampicin, leading to insufficient therapeutic levels and potentially poorer outcomes.27
The enteral route, frequently utilized for medication administration in the ICU, exhibits reduced reliability in critically ill patients.10 Intravenous (IV) formulations of antitubercular medications, while often limited in availability, provide more predictable pharmacokinetics. In such cases, especially with drug-resistant TB, alternative strategies such as the administration of intravenous formulations of medications like moxifloxacin and amikacin should be considered.28
Therapeutic antibiotic monitoring (TDM) can effectively control tuberculosis in critically ill patients by ensuring appropriate antibiotic exposure and minimizing toxicity. However, its implementation is limited by financial constraints and the requirement for specific laboratory facilities. For medications with narrow therapeutic indices, it can guide dosage adjustments to achieve therapeutic objectives in settings where therapeutic drug monitoring is applicable.29,30 Hence, Effective management of tuberculosis in critically ill patients necessitates an understanding of altered pharmacokinetics in this demographic, Careful selection of dosage forms tailored to clinical status, and prompt initiation of treatment.
In critically ill tuberculosis patients, targeted application informed by clinical indicators can enhance therapeutic drug monitoring (TDM). Therapeutic drug monitoring (TDM) is indicated for patients who do not respond to treatment, exhibit signs of malabsorption (such as those on vasopressors, continuous enteral feeding, or with gastrointestinal complications), present with substantial liver or kidney impairment, or are at risk of drug-drug interactions that could alter drug efficacy. Certain algorithms initiate antitubercular therapy by assessing organ function and potential drug interactions. Therapeutic drug monitoring for essential medications such as rifampicin and isoniazid is conducted between days 3 and 5. Monitoring of peak levels for rifampicin and isoniazid should occur 2 hours and 1 to 2 hours post-dose, respectively. Target serum concentrations should be 8-24 μg/mL for rifampicin and 3-6 μg/mL for isoniazid. Evaluation of the results should take into account the patient's clinical progress, pharmacological history, and levels of albumin and bilirubin. TDM dose adjustments must be tailored to individual patients. If clinical failure or significant physiological changes continue, it may be necessary to collect samples again. TDM is still limited in numerous low-resource contexts. Administering it to high-risk ICU patients can enhance the efficacy and safety of therapies.
4.4. Critical Supportive Care Strategies for TB patients
Effective supportive care is essential for managing tuberculosis (TB) patients in the intensive care unit (ICU), as it addresses disease-specific complications alongside the challenges associated with critical illness.31 Acute respiratory failure, frequently necessitating mechanical ventilation, is a common reason for ICU admission in tuberculosis patients. To reduce ventilator-induced lung injury, it is essential to implement standard lung-protective ventilation strategies, which include utilizing low tidal volumes and appropriate positive end-expiratory pressure (PEEP).32 In cases of severe hypoxemia, additional treatment options may include neuromuscular blockade and the prone position. While research on the efficacy of extracorporeal membrane oxygenation (ECMO) in tuberculosis is limited, it is considered a last-resort option for patients with resistant cases.
A significant number of critically ill tuberculosis patients present with multiorgan failure and septic shock. Early goal-directed therapy primarily involves fluid resuscitation and the administration of vasopressors.33 In cases of acute kidney injury, renal replacement therapy should be initiated promptly.34 Due to the hepatotoxic properties of numerous antitubercular medications, it is crucial to monitor for drug-induced hepatotoxicity.35 Corticosteroids have demonstrated mortality benefits in specific forms of tuberculosis, such as pericarditis and tuberculous meningitis. Their application in different forms of tuberculosis remains contentious and should be tailored according to clinical assessment.36
Malnutrition serves as both a risk factor for and a consequence of tuberculosis (TB). Nutritional assessment and assistance are fundamental components of treatment, which may include enteral or parenteral nutrition when appropriate.37 Research presents conflicting evidence; however, micronutrient supplements, particularly vitamins A, D, and E, may offer benefits.38
Severe illness can exert significant psychological impacts. Incorporating psychological support, such as counseling and, when feasible, early mobilization, contributes to recovery and improves overall outcomes.39 The management of critically ill tuberculosis patients requires a multidisciplinary approach that encompasses respiratory and hemodynamic support, dietary management, infection control, and psychosocial care.40 The prognosis and recovery outcomes can be significantly influenced by tailoring support plans to meet individual patient needs.41
4.5. Drug-Resistant TB in the ICU
Drug-resistant tuberculosis (DR-TB), encompassing multidrug-resistant (MDR-TB) and extensively drug-resistant (XDR-TB) strains, poses significant challenges in the ICU due to complex resistance patterns, restricted treatment options, and elevated mortality rates.42 The success rates of therapy for MDR-TB patients vary from 50% to 77%, indicating a substantial global burden of DR-TB. Cases of XDR-TB demonstrate significantly reduced success rates, approximately 39%. Patients with DR-TB often present with severe respiratory failure, septic shock, and multi-organ dysfunction, necessitating comprehensive critical care in ICU settings.43
Accurate and rapid identification of DR-TB in critically ill patients is essential, despite the challenges involved.42 Conventional culture and drug sensitivity testing are time-consuming processes that hinder timely treatment.44 Xpert MTB/RIF and other molecular diagnostics enable rapid identification of rifampicin resistance; however, they may not detect resistance to additional medications, necessitating comprehensive resistance profiling.45
In the ICU, the management of DR-TB requires tailored, multi-drug therapies informed by resistance patterns.42 The World Health Organization recommends a minimum treatment duration of 20 months for MDR/XDR-TB, utilizing a combination of four or more effective medications. Newer agents, such as delamanid and bedaquiline, have demonstrated potential in enhancing outcomes; however, monitoring for side effects is essential. Therapeutic drug monitoring may mitigate toxicity and optimize dosage.46,47 In addition to antibiotic treatment, supportive care is essential. This addresses hemodynamic support in septic shock, mechanical ventilation for respiratory failure, and renal replacement therapy for acute kidney injury. Nutritional support and management of comorbidities are essential components of treatment.33-36
The primary objective is to halt the transmission of nosocomial DR-TB. To mitigate the risk of transmission within healthcare settings, it is essential to implement airborne precautions, utilize negative-pressure isolation rooms, and ensure appropriate personal protective equipment for healthcare professionals is employed.48 In an ICU setting, DR-TB poses significant therapeutic and diagnostic challenges. Enhancing patient outcomes and preventing transmission within hospital settings requires a multidisciplinary strategy that integrates rapid diagnosis, customized treatment, robust supportive care, and stringent infection control protocols.49
The diagnosis and treatment of severely ill patients with tuberculosis and co-infections such as HIV or COVID-19 present significant challenges. In patients with HIV, the presence of fever, respiratory issues, and decreased lymphocyte counts can complicate the diagnosis of tuberculosis, particularly when the infection disseminates beyond the pulmonary system. The initiation of antiretroviral therapy (ART) in the ICU warrants careful consideration. Administering it too rapidly, particularly during the initial two weeks of antitubercular therapy, may lead to immune reconstitution inflammatory syndrome (IRIS), potentially exacerbating the patient's condition and resulting in organ failure. Initiating ART after 8 weeks in individuals with low CD4 counts (<50 cells/μL) is associated with an elevated risk of mortality. Establish the timing for each individual according to the severity of their illness and the robustness of their immune system.16,20 The coexistence of tuberculosis (TB) and COVID-19 complicates diagnosis and treatment due to overlapping radiographic features, and the use of corticosteroids or immunomodulators may reactivate latent TB. Rifampicin may interact pharmacokinetically and pharmacodynamically with COVID-19 medications such as remdesivir and nirmatrelvir/ritonavir, potentially reducing their efficacy or increasing their risk.52 Critically ill patients with multiple comorbidities frequently exhibit poorer outcomes. Multidisciplinary care, continuous monitoring, and treatment adjustments are essential, to prevent polypharmacy and immunological dysregulation.40
The main lines of management of TB cases in ICU are summarized in Figure 1.
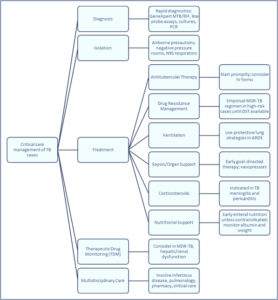
Figure 1: Summary of management lines of TB cases in ICU
4.6. High-Risk Groups in Intensive Care Management of TB (2020–2025)
Tuberculosis (TB) remains a significant global health concern, particularly among high-risk populations requiring intensive care unit (ICU) management. Recent studies from 2020 to 2025 have highlighted several vulnerable groups and the complexities involved in their care50.
5.1. Clinical Severity Scores
The Acute Physiology and Chronic Health Evaluation II (APACHE II) score is a validated predictor of mortality in tuberculosis patients with respiratory failure.56 A retrospective analysis involving 153 patients with pulmonary tuberculosis and respiratory failure found that higher APACHE II scores were independently associated with an increased risk of death. The study developed a predictive model that integrates APACHE II scores, arterial oxygen pressure (PaO₂), serum albumin, and C-reactive protein (CRP) levels, achieving an area under the curve (AUC) of 0.818, with sensitivity at 83.3% and specificity at 73.1%.57
5.2. Laboratory Indicators
Various laboratory factors have been linked to outcomes in patients with critical illness tuberculosis. Reduced levels of PaO₂ associated with ICU admission are correlated with increased mortality rates.58 Serum albumin levels indicate that poor outcomes associated with hypoalbuminemia reflect both nutritional status and systemic inflammation.59 Elevated CRP levels correlate with increased inflammatory response and, consequently, a higher risk of mortality.60 The predictive model outlined for assessing mortality risk in TB patients with respiratory failure is entirely reliant on these markers.
5.3. Pharmacogenomics and Treatment Response
Treatment outcomes in individuals with tuberculous meningitis (TBM) are significantly affected by genetic polymorphisms in the N-acetyltransferase 2 (NAT2) gene and the dosage of isoniazid (INH).61 High-dose INH (600 mg/day) was beneficial for patients with intermediate acetylator (IA) NAT2 genotypes, as evidenced by a study involving 119 TBM patients, which demonstrated lower rates of disability and mortality compared to those receiving standard doses. Key predictive factors identified through multivariate analysis included INH dosage, cranial nerve palsy, age, and headache.62
5.4. Mortality Rates and Outcomes
Mortality rates for tuberculosis patients in intensive care units continue to be elevated. In a study of 153 individuals with pulmonary tuberculosis and respiratory failure, the mortality rate was 39.22%.61 Factors contributing to high mortality include delayed diagnosis, advanced disease stage at presentation, and the presence of concomitant diseases.60,61
5.5. Implications for Clinical Treatment
Clinical decisions may be guided by the identification of prognostic indicators such as APACHE II scores, PaO₂, serum albumin, CRP levels, and genetic factors. Early identification of high-risk patients facilitates prompt interventions, including aggressive supportive care, tailored medication, and assessment of supplementary treatments.63 The incorporation of pharmacogenomic information, specifically NAT2 genotyping, may enhance therapeutic strategies for patients with TBM.61 The management of TB patients in ICU settings requires a comprehensive approach that incorporates clinical severity, laboratory markers, genetic factors, and individualized treatment plans to optimize outcomes.40
This review addresses a significant and contemporary issue: the management of tuberculosis (TB) in ICUs, emphasizing drug-resistant strains and co-infections such as HIV. The systematic approach and extensive array of topics are noteworthy, encompassing diagnostic challenges, treatment options, and supportive care strategies. The review provides a comprehensive overview of the clinical challenges associated with managing tuberculosis in the intensive care unit, synthesizing literature from 2000 to 2025 and incorporating various study types, including guidelines, cohort studies, and retrospective analyses. The emphasis on MDR-TB and the impact of serious illness on drug efficacy provides valuable clinical insights.
However, certain issues within the review diminish its utility. The absence of a quantitative synthesis of the data limits the strength of its conclusions. The diversity of study types may complicate the application of results to other contexts, and the absence of stratification by location or healthcare resource setting diminishes the utility of the recommendations in particular scenarios. The paper discusses the prolonged duration of diagnosis and treatment but lacks detailed examination of emerging diagnostic technologies, such as molecular tests, which may hold significant relevance in intensive care units. A further issue is the lack of specific pharmacological adjustments and therapeutic monitoring protocols for severely ill TB patients.
The review contains several significant gaps that diminish its overall utility. The discussion omits high-risk populations such as pediatric and elderly ICU patients, and it fails to address the application of prognostic tools or scoring systems for clinical decision-making. Discussing supportive treatment is essential; however, it would be more beneficial to incorporate established ICU protocols for tuberculosis-related complications such as sepsis or acute respiratory distress syndrome into the conversation. Future research should focus on developing guidelines tailored specifically for ICUs. Consideration must be given to the issues of medication resistance, host immunosuppression, and the variability of resources across different health systems globally.
The management of tuberculosis (TB) in intensive care presents significant clinical challenges due to elevated mortality rates, diagnostic delays, and complex comorbidities. Patients with respiratory failure, HIV co-infection, drug-resistant tuberculosis, or sepsis—specifically critically ill tuberculosis patients—exhibit an elevated risk of mortality, which typically ranges from 30% to 50%. Recent research from 2020 to 2025 identifies low arterial oxygen levels, hypoalbuminemia, elevated C-reactive protein, and high APACHE II scores as significant predictors of adverse outcomes. Delays in diagnosis, stemming from the limited application of rapid diagnostic techniques in ICU settings, remain a significant barrier to timely treatment. Coexisting illnesses such as malnutrition, hepatic or renal failure, and COVID-19 complicate treatment and worsen prognosis. Recent research in pharmacogenomics, particularly involving NAT2 genotyping in tuberculosis meningitis, highlights the potential for personalized treatment strategies to improve outcomes. Prioritizing early recognition, tailored therapeutic approaches, comprehensive comorbidity management, and investment in diagnostics and precision medicine is essential for reducing mortality and enhancing recovery in the intensive care management of tuberculosis.
10. Conflict of interest
All authors declare that there was no conflict of interest.
11. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
12. Authors’ contribution
EME: Conceptualization, Methodology, Writing – Original Draft, Supervision
BDMA: Literature Review, Data Curation, Writing – Review & Editing
HRHA: Investigation, Formal Analysis, Writing – Review & Editing
ASJA: Validation, Visualization, Writing – Review & Editing
AMMA: Resources, Literature Search, Writing – Review & Editing
FNKA: Project Administration, Critical Review, Writing – Review & Editing
IDHA: Supervision, Final Approval of the Manuscript, Writing – Review & Editing
Authors affiliations:
- Ekramy M. Elmorsy, Center for Health Research, Northern Border University, Arar, Saudi Arabia; Email: ekramy.elmorsy@nbu.edu.sa
- Basil D. M. Alruwaili, Faculty of Medicine, Northern Border University, Arar, Saudi Arabia; Email: Basil.dd10@gmail.com
- Hatem R. H. Alrwaili, Faculty of Medicine, Northern Border University, Arar, Saudi Arabia; Email: hatemrh1122@gmail.com
- Anwar S. J. Alrawaili, Faculty of Medicine, Northern Border University, Arar, Saudi Arabia; Email: A.12a.12714@gmail.com
- Abdulelah M. M. Alanazi, Faculty of Medicine, Northern Border University, Arar, Saudi Arabia; Email: alanazi.0249@gmail.com
- Fahad N. K. Alanazi, Faculty of Medicine, Northern Border University, Arar, Saudi Arabia; Email: fnfn32273@gmail.com
- Ibraheem D. H. Alanazi, Faculty of Medicine, Northern Border University, Arar, Saudi Arabia; Email: Ibraheem5050@hotmail.com
ABSTRACT
Background & objectives: Tuberculosis (TB) continues to be a significant global health issue, as an increasing number of individuals are presenting with severe, life-threatening conditions that require admission to intensive care units (ICUs). The management of critical care environments is increasingly complicated by the rise of drug-resistant tuberculosis, co-infection with HIV, and late-stage diagnoses. Despite its increasing significance, there is a paucity of focused research on the critical care aspects of tuberculosis.
This review aims to systematically examine the current data and clinical guidelines pertaining to the treatment of tuberculosis in critical care settings. The focus is on diagnostic challenges, treatment choices, supportive methods, and outcome forecasts relevant to critically ill TB patients.
Methodology: A structured literature evaluation was conducted, focusing on publications from 2000 to 2025, using databases such as [PubMed], Scopus, and Web of Science. This study examines the management of tuberculosis in intensive and critical care settings, focusing on multidrug-resistant tuberculosis (MDR-TB), the role of mechanical ventilation, and the implications of sepsis in affected patients. A review of clinical recommendations, cohort studies, retrospective analyses, and relevant case series facilitated the summarization of the database for ICU care techniques in tuberculosis patients.
Results: Critically ill tuberculosis patients primarily exhibit severe respiratory failure, miliary spread, tuberculosis sepsis, or central nervous system involvement. Suboptimal outcomes stem from diagnostic delays caused by ambiguous clinical features and the limitations of conventional microbiological diagnostics. In ICU cohorts with tuberculosis, mortality rates range from 30% to 70%, with elevated rates observed in patients requiring mechanical ventilation or experiencing multi-organ failure. Delayed initiation of antitubercular treatment (ATT) in the ICU often results in altered pharmacokinetics, that hinder medication distribution. Drug-resistant tuberculosis continues to pose significant challenges regarding therapy selection and infection control. Supportive care interventions, including mechanical ventilation, fluid resuscitation, and nutritional support, are critically important yet often underappreciated.
Conclusion: Tuberculosis in the ICU represents a distinct clinical entity characterized by elevated mortality rates, diagnostic delays, and complex therapeutic decisions. Enhancing outcomes relies on early detection, prompt initiation of antitubercular therapy, customized pharmaceutical interventions, and multidisciplinary supportive care. Prospective studies are essential to establish ICU-specific tuberculosis care guidelines, especially for individuals with drug resistance and co-infections.
Abbreviations: APACHE II: Acute Physiology and Chronic Health Evaluation II, ARDS: acute respiratory distress syndrome ATT: antitubercular treatment, CRP: C-reactive protein, DR-TB: drug-resistant tuberculosis, ECMO: extracorporeal membrane oxygenation, ICU: intensive care units, MDR-TB: multidrug-resistant tuberculosis, TB: Tuberculosis, XDR extensively drug-resistant, TDM: Therapeutic antibiotic monitoring
Keywords: Tuberculosis; Critical Care; Intensive Care Unit (ICU); Antitubercular Therapy; Drug-Resistant TB
Citation: Ekramy M. Elmorsy, Basil D. M. Alruwaili, Hatem R. H. Alrwaili, Anwar S. J. Alrawaili, Abdulelah M. M. Alanazi, Fahad N. K. Alanazi, and Ibraheem D. H. Alanazi. Critical care management of tuberculosis: challenges, evidence, and evolving strategies. Anaesth. pain intensive care 2025;29(5):405-417. DOI: 10.35975/apic.v29i5.2867
Received: June 11, 2025; Revised: June 28, 2025; Accepted: July 11, 2025
1. INTRODUCTION
Tuberculosis (TB) continues to pose a significant global health challenge, with projections of 10.6 million new cases and 1.6 million deaths in 2021. While the majority of tuberculosis cases are managed in outpatient settings, certain patients experience severe complications necessitating admission to an intensive care unit (ICU).1 Stated death rates for TB range from 50% to 60%, with approximately 3–4% of hospitalized patients requiring intensive care unit (ICU) treatment.2 Critically ill tuberculosis patients frequently present with sudden respiratory failure, septic shock, or multi-organ dysfunction, conditions often worsened by delayed diagnosis and the initiation of appropriate treatment.3 The limitations of conventional microbiological testing, coupled with the nonspecific clinical manifestations of tuberculosis in critically ill patients, exacerbate diagnostic challenges.4 In critically ill patients, the pharmacokinetics of antitubercular medications may be significantly altered, leading to subtherapeutic drug levels and complicating treatment.5
The emergence of extensively drug-resistant (XDR) and multidrug-resistant (MDR), TB bacteria introduces additional challenges to the management of TB in critical care settings. Such situations necessitate close monitoring and supportive treatment due to the requirement for contemporary, potentially hazardous, and inadequately researched medication regimens.6 Despite the high mortality and complex treatment associated with tuberculosis in the ICU, there is a lack of targeted research and evidence-based guidelines to address this issue. This review aims to summarize current information on the critical care management of tuberculosis, highlighting areas that require further research, challenges in diagnosis, therapeutic strategies, and supportive care measures.
2. METHODOLOGY
A systematic literature review covering the years 2000 to 2025 was conducted. We conducted a search in [PubMed], Scopus, and Web of Science using the following phrases and Boolean operators: ("tuberculosis" OR "TB") AND ("intensive care" OR "critical care" OR "ICU") AND ("mechanical ventilation" OR "sepsis" OR "MDR-TB"). Studies were required to meet the following criteria for inclusion: The articles were composed in English, focused on adult patients (≥18 years) with tuberculosis necessitating ICU admission, and examined critical care-specific clinical outcomes, management strategies, and related issues. The study analyzed cohort studies, retrospective analyses, case series, and clinical guidelines. Research focusing exclusively on children, animals, or in vitro studies, as well as articles not pertaining to intensive or critical care, were excluded from consideration. Two reviewers conducted independent examinations of titles and abstracts. Full texts of relevant articles were reviewed to confirm eligibility. All parties reached a consensus to address the issues.
3. Epidemiology of Tuberculosis in ICU
In 2023, tuberculosis (TB) is anticipated to result in 10.8 million new cases and 1.25 million deaths, underscoring its status as a significant global health concern.7 While the majority of tuberculosis cases are managed in outpatient settings, certain patients experience severe complications necessitating admission to an intensive care unit (ICU).2 Some cohorts have demonstrated a prevalence of culture-confirmed pulmonary tuberculosis requiring ICU hospitalization of nearly 8.0%. Critically ill tuberculosis patients frequently present with abrupt respiratory failure, septic shock, or multi-organ dysfunction, conditions often worsened by delayed diagnosis and initiation of appropriate treatment3. The average mortality rate for TB patients admitted to the ICU is approximately 52.9%, with in-hospital death rates varying significantly between 27% and 95%.8 Advanced age, central nervous system involvement, the requirement for mechanical ventilation, and the presence of comorbidities contribute to an increased mortality rate.9The emergence of extensively drug-resistant (XDR) and multidrug-resistant (MDR) tuberculosis strains significantly complicates the critical care management of the disease. Such situations necessitate close monitoring and supportive treatment due to the requirement for contemporary, potentially hazardous, and inadequately researched medication regimens.6 In critically ill patients, the pharmacokinetics of antitubercular medications may be significantly altered, leading to subtherapeutic drug levels and complicating treatment.10 Despite the high mortality and complex treatment associated with tuberculosis in the ICU, there is a lack of targeted research and evidence-based recommendations to address this issue. This review aims to summarize current information on the critical care management of tuberculosis, highlighting areas that require further research, challenges in diagnosis, therapeutic strategies, and supportive care measures.
4. Clinical Presentation
4.1. Clinical Presentation of Tuberculosis in Critically Ill PatientsTuberculosis (TB) in the intensive care unit (ICU) complicates early identification and treatment due to its presentation of various ambiguous clinical symptoms.3 Acute respiratory failure, typically associated with severe pulmonary disease, is the primary reason for ICU admission among TB patients.11 Radiographic findings typically reveal bilateral infiltrates and cavitary lesions. Common complications such as bacterial superinfection, chronic obstructive pulmonary disease, and cancer further restrict respiratory function.12 A significant number of these patients exhibited acute respiratory distress syndrome (ARDS), and post-mortem analyses indicate that confluent tuberculous bronchopneumonia may resemble ARDS.13
Extrapulmonary tuberculosis, characterized by symptoms such as central nervous system involvement (e.g., tuberculous meningitis), lymphadenopathy, pleural effusion, and miliary spread, constitutes a notable proportion of ICU admissions.14 Miliary tuberculosis is characterized by extensive hematogenous dissemination, presenting with nonspecific systemic symptoms such as fever, weight loss, and hepatosplenomegaly.15 Disseminated tuberculosis is more prevalent among immunocompromised individuals, particularly those with HIV co-infection, and may present atypically, often complicating the diagnostic process.16
Patients with critical illness tuberculosis frequently exhibit systemic symptoms such as weight loss, anorexia, night sweats, and persistent fever.3 Examples of organ dysfunctions that demonstrate the systemic nature of the disease include hepatic impairment, renal failure, and adrenal insufficiency. Altered mental state and seizures, as neurological symptoms, may indicate central nervous system involvement.17 Paradoxical reactions complicate diagnosis and treatment, as they involve clinical decline despite appropriate therapeutic interventions.18
In endemic regions or among high-risk populations, the diverse presentations of tuberculosis in the ICU necessitate heightened clinical suspicion. Enhancing outcomes relies on timely recognition of antitubercular therapy and prompt initiation of treatment.19 The effective management of these complex cases relies on comprehensive evaluation, which includes assessing extrapulmonary involvement and potential co-infections.
4.2. Diagnostic Challenges in ICU for TB
Diagnosing TB in critically ill patients in the ICU presents significant challenges due to unusual clinical presentations, constraints of conventional diagnostic techniques, and the urgency needed to begin suitable treatment.20 Since critically ill patients usually have ambiguous symptoms like fever, respiratory problems, or altered mental state that could be ascribed to various diseases, TB diagnosis is more challenging.21 Furthermore, diagnosis might be delayed due to comorbidities and the effects of immunosuppression, which might conceal typical TB symptoms. Complicating the diagnosis, immunocompromised patients—including those on immunosuppressive therapy or HIV—may present unique TB symptoms and run a higher risk of spreading disease.22 Early diagnosis is even more crucial; for critically sick patients, appropriate therapy starts with fast and accurate diagnostic instruments. Higher morbidity and death can result from delays.20
Tools for conventional diagnosis have some restrictions. Getting quality sputum samples from patients under intubation or vegetative state is challenging. While induced sputum or bronchoalveolar lavage may be required in unstable patients, they can be harmful.23 Though the gold standard is still Smear Microscopy and Culture, it takes time—often weeks—which is untenable in acute situations. Especially in paucibacillary conditions, smear microscopy has poor sensitivity.24 While Xpert MTB/RIF molecular diagnostics offers fast results, in those with low bacterial loads or in extrapulmonary TB its sensitivity may be reduced.25 Moreover, overlapping findings of several pulmonary illnesses make reading chest radiographs in ICU patients difficult. Advanced imaging modalities as CT scans provide more detail, so unstable patients might not be able to receive them.12 Particularly in endemic areas or among high-risk groups, resolving the diagnostic challenges of tuberculosis in the ICU demands for a high index of suspicion. Fast molecular diagnostics, together with the integration of clinical, radiographic, and laboratory findings, define timeliness in diagnosis and therapy.12,23-25
4.3. Antitubercular Therapy in Critical Illness
The management of tuberculosis (TB) in critically ill patients presents unique challenges due to altered pharmacokinetics, restricted medication formulations, and the necessity for initiating effective therapy.10 Standard first-line antitubercular medications—rifampicin, isoniazid, pyrazinamide, and ethambutol—may exhibit subtherapeutic levels in this population, necessitating tailored treatments to ensure optimal outcomes.
In critical illness, there are significant alterations in drug absorption, distribution, metabolism, and excretion.26 Factors such as gastrointestinal hypomotility, vasopressor usage, and fluid changes may compromise enteral absorption and alter drug distribution volumes. Research indicates that critically ill patients with nasogastric tubes frequently exhibit reduced plasma concentrations of first-line antitubercular medications, particularly rifampicin, leading to insufficient therapeutic levels and potentially poorer outcomes.27
The enteral route, frequently utilized for medication administration in the ICU, exhibits reduced reliability in critically ill patients.10 Intravenous (IV) formulations of antitubercular medications, while often limited in availability, provide more predictable pharmacokinetics. In such cases, especially with drug-resistant TB, alternative strategies such as the administration of intravenous formulations of medications like moxifloxacin and amikacin should be considered.28
Therapeutic antibiotic monitoring (TDM) can effectively control tuberculosis in critically ill patients by ensuring appropriate antibiotic exposure and minimizing toxicity. However, its implementation is limited by financial constraints and the requirement for specific laboratory facilities. For medications with narrow therapeutic indices, it can guide dosage adjustments to achieve therapeutic objectives in settings where therapeutic drug monitoring is applicable.29,30 Hence, Effective management of tuberculosis in critically ill patients necessitates an understanding of altered pharmacokinetics in this demographic, Careful selection of dosage forms tailored to clinical status, and prompt initiation of treatment.
In critically ill tuberculosis patients, targeted application informed by clinical indicators can enhance therapeutic drug monitoring (TDM). Therapeutic drug monitoring (TDM) is indicated for patients who do not respond to treatment, exhibit signs of malabsorption (such as those on vasopressors, continuous enteral feeding, or with gastrointestinal complications), present with substantial liver or kidney impairment, or are at risk of drug-drug interactions that could alter drug efficacy. Certain algorithms initiate antitubercular therapy by assessing organ function and potential drug interactions. Therapeutic drug monitoring for essential medications such as rifampicin and isoniazid is conducted between days 3 and 5. Monitoring of peak levels for rifampicin and isoniazid should occur 2 hours and 1 to 2 hours post-dose, respectively. Target serum concentrations should be 8-24 μg/mL for rifampicin and 3-6 μg/mL for isoniazid. Evaluation of the results should take into account the patient's clinical progress, pharmacological history, and levels of albumin and bilirubin. TDM dose adjustments must be tailored to individual patients. If clinical failure or significant physiological changes continue, it may be necessary to collect samples again. TDM is still limited in numerous low-resource contexts. Administering it to high-risk ICU patients can enhance the efficacy and safety of therapies.
4.4. Critical Supportive Care Strategies for TB patients
Effective supportive care is essential for managing tuberculosis (TB) patients in the intensive care unit (ICU), as it addresses disease-specific complications alongside the challenges associated with critical illness.31 Acute respiratory failure, frequently necessitating mechanical ventilation, is a common reason for ICU admission in tuberculosis patients. To reduce ventilator-induced lung injury, it is essential to implement standard lung-protective ventilation strategies, which include utilizing low tidal volumes and appropriate positive end-expiratory pressure (PEEP).32 In cases of severe hypoxemia, additional treatment options may include neuromuscular blockade and the prone position. While research on the efficacy of extracorporeal membrane oxygenation (ECMO) in tuberculosis is limited, it is considered a last-resort option for patients with resistant cases.
A significant number of critically ill tuberculosis patients present with multiorgan failure and septic shock. Early goal-directed therapy primarily involves fluid resuscitation and the administration of vasopressors.33 In cases of acute kidney injury, renal replacement therapy should be initiated promptly.34 Due to the hepatotoxic properties of numerous antitubercular medications, it is crucial to monitor for drug-induced hepatotoxicity.35 Corticosteroids have demonstrated mortality benefits in specific forms of tuberculosis, such as pericarditis and tuberculous meningitis. Their application in different forms of tuberculosis remains contentious and should be tailored according to clinical assessment.36
Malnutrition serves as both a risk factor for and a consequence of tuberculosis (TB). Nutritional assessment and assistance are fundamental components of treatment, which may include enteral or parenteral nutrition when appropriate.37 Research presents conflicting evidence; however, micronutrient supplements, particularly vitamins A, D, and E, may offer benefits.38
Severe illness can exert significant psychological impacts. Incorporating psychological support, such as counseling and, when feasible, early mobilization, contributes to recovery and improves overall outcomes.39 The management of critically ill tuberculosis patients requires a multidisciplinary approach that encompasses respiratory and hemodynamic support, dietary management, infection control, and psychosocial care.40 The prognosis and recovery outcomes can be significantly influenced by tailoring support plans to meet individual patient needs.41
4.5. Drug-Resistant TB in the ICU
Drug-resistant tuberculosis (DR-TB), encompassing multidrug-resistant (MDR-TB) and extensively drug-resistant (XDR-TB) strains, poses significant challenges in the ICU due to complex resistance patterns, restricted treatment options, and elevated mortality rates.42 The success rates of therapy for MDR-TB patients vary from 50% to 77%, indicating a substantial global burden of DR-TB. Cases of XDR-TB demonstrate significantly reduced success rates, approximately 39%. Patients with DR-TB often present with severe respiratory failure, septic shock, and multi-organ dysfunction, necessitating comprehensive critical care in ICU settings.43
Accurate and rapid identification of DR-TB in critically ill patients is essential, despite the challenges involved.42 Conventional culture and drug sensitivity testing are time-consuming processes that hinder timely treatment.44 Xpert MTB/RIF and other molecular diagnostics enable rapid identification of rifampicin resistance; however, they may not detect resistance to additional medications, necessitating comprehensive resistance profiling.45
In the ICU, the management of DR-TB requires tailored, multi-drug therapies informed by resistance patterns.42 The World Health Organization recommends a minimum treatment duration of 20 months for MDR/XDR-TB, utilizing a combination of four or more effective medications. Newer agents, such as delamanid and bedaquiline, have demonstrated potential in enhancing outcomes; however, monitoring for side effects is essential. Therapeutic drug monitoring may mitigate toxicity and optimize dosage.46,47 In addition to antibiotic treatment, supportive care is essential. This addresses hemodynamic support in septic shock, mechanical ventilation for respiratory failure, and renal replacement therapy for acute kidney injury. Nutritional support and management of comorbidities are essential components of treatment.33-36
The primary objective is to halt the transmission of nosocomial DR-TB. To mitigate the risk of transmission within healthcare settings, it is essential to implement airborne precautions, utilize negative-pressure isolation rooms, and ensure appropriate personal protective equipment for healthcare professionals is employed.48 In an ICU setting, DR-TB poses significant therapeutic and diagnostic challenges. Enhancing patient outcomes and preventing transmission within hospital settings requires a multidisciplinary strategy that integrates rapid diagnosis, customized treatment, robust supportive care, and stringent infection control protocols.49
The diagnosis and treatment of severely ill patients with tuberculosis and co-infections such as HIV or COVID-19 present significant challenges. In patients with HIV, the presence of fever, respiratory issues, and decreased lymphocyte counts can complicate the diagnosis of tuberculosis, particularly when the infection disseminates beyond the pulmonary system. The initiation of antiretroviral therapy (ART) in the ICU warrants careful consideration. Administering it too rapidly, particularly during the initial two weeks of antitubercular therapy, may lead to immune reconstitution inflammatory syndrome (IRIS), potentially exacerbating the patient's condition and resulting in organ failure. Initiating ART after 8 weeks in individuals with low CD4 counts (<50 cells/μL) is associated with an elevated risk of mortality. Establish the timing for each individual according to the severity of their illness and the robustness of their immune system.16,20 The coexistence of tuberculosis (TB) and COVID-19 complicates diagnosis and treatment due to overlapping radiographic features, and the use of corticosteroids or immunomodulators may reactivate latent TB. Rifampicin may interact pharmacokinetically and pharmacodynamically with COVID-19 medications such as remdesivir and nirmatrelvir/ritonavir, potentially reducing their efficacy or increasing their risk.52 Critically ill patients with multiple comorbidities frequently exhibit poorer outcomes. Multidisciplinary care, continuous monitoring, and treatment adjustments are essential, to prevent polypharmacy and immunological dysregulation.40
The main lines of management of TB cases in ICU are summarized in Figure 1.

Figure 1: Summary of management lines of TB cases in ICU
4.6. High-Risk Groups in Intensive Care Management of TB (2020–2025)
Tuberculosis (TB) remains a significant global health concern, particularly among high-risk populations requiring intensive care unit (ICU) management. Recent studies from 2020 to 2025 have highlighted several vulnerable groups and the complexities involved in their care50.
| Table 1: TB-associated vulnerable groups and the complexities | |||
| Topic | Key Points | Implications | Ref |
| HIV and Drug-Resistant TB (DR-TB) | - Co-infection complicates treatment and increases mortality- Underweight HIV-DR-TB patients had the lowest cure rate (23.2%) in the Eastern Cape study- HIV co-infection linked with poorer outcomes | Emphasizes the need for integrated nutritional and HIV interventions in DR-TB management | 16 |
| Cardiovascular Disease (CVD) Risk | - Acute TB phase increases cardiovascular events- Incidence rates: 3.5 (US), 2.7 (UK)- TB patients show higher CVD risk even after adjusting for comorbidities | TB is an independent risk factor for cardiovascular events, requiring cardiac monitoring | 51 |
| Nutritional Status and Malnutrition | - Malnutrition both a risk factor and consequence of TB- BMI <18.5 linked to higher mortality- Nutritional supplements can aid weight gain | Nutritional support is crucial in TB treatment plans, particularly for underweight patients | 37 |
| COVID-19 and TB Co-infection | - TB patients face higher COVID-19 complications- Increased emergency visits (HR 1.244) and critical care (HR 1.404)- Risk of breakthrough COVID-19 infections higher | TB patients should be prioritized for COVID-19 vaccination and close monitoring | 52 |
| Renal and Hepatic Comorbidities | - TB drugs may cause liver and kidney toxicity- Hepatic: Adjust regimens in cirrhosis to reduce hepatotoxicity- Renal: Avoid nephrotoxic drugs (e.g., aminoglycosides) in renal failure | Requires individualized drug regimens and regular liver/kidney function monitoring | 53 |
| Hypersensitivity to Anti-TB Drugs | - 2.9% of TB patients had hypersensitivity reactions- Most common: skin rash- Risk factors: female, age <65, liver disease, allergy history | Vigilant monitoring and alternative drug options needed in ICU settings | 54 |
| Socioeconomic and Demographic Factors | - TB prevalent in low-income, high-density, marginalized areas- Leads to delayed diagnosis, poor treatment adherence, high transmission | Socioeconomic factors must be addressed to improve TB control and outcomes | 55 |
5. Prognostic Indicators and Outcomes
Patients with TB admitted to ICUs exhibit significant morbidity and mortality rates. Recent research from 2020 to 2025 has identified several prognostic factors that affect outcomes in this cohort.95.1. Clinical Severity Scores
The Acute Physiology and Chronic Health Evaluation II (APACHE II) score is a validated predictor of mortality in tuberculosis patients with respiratory failure.56 A retrospective analysis involving 153 patients with pulmonary tuberculosis and respiratory failure found that higher APACHE II scores were independently associated with an increased risk of death. The study developed a predictive model that integrates APACHE II scores, arterial oxygen pressure (PaO₂), serum albumin, and C-reactive protein (CRP) levels, achieving an area under the curve (AUC) of 0.818, with sensitivity at 83.3% and specificity at 73.1%.57
5.2. Laboratory Indicators
Various laboratory factors have been linked to outcomes in patients with critical illness tuberculosis. Reduced levels of PaO₂ associated with ICU admission are correlated with increased mortality rates.58 Serum albumin levels indicate that poor outcomes associated with hypoalbuminemia reflect both nutritional status and systemic inflammation.59 Elevated CRP levels correlate with increased inflammatory response and, consequently, a higher risk of mortality.60 The predictive model outlined for assessing mortality risk in TB patients with respiratory failure is entirely reliant on these markers.
5.3. Pharmacogenomics and Treatment Response
Treatment outcomes in individuals with tuberculous meningitis (TBM) are significantly affected by genetic polymorphisms in the N-acetyltransferase 2 (NAT2) gene and the dosage of isoniazid (INH).61 High-dose INH (600 mg/day) was beneficial for patients with intermediate acetylator (IA) NAT2 genotypes, as evidenced by a study involving 119 TBM patients, which demonstrated lower rates of disability and mortality compared to those receiving standard doses. Key predictive factors identified through multivariate analysis included INH dosage, cranial nerve palsy, age, and headache.62
5.4. Mortality Rates and Outcomes
Mortality rates for tuberculosis patients in intensive care units continue to be elevated. In a study of 153 individuals with pulmonary tuberculosis and respiratory failure, the mortality rate was 39.22%.61 Factors contributing to high mortality include delayed diagnosis, advanced disease stage at presentation, and the presence of concomitant diseases.60,61
5.5. Implications for Clinical Treatment
Clinical decisions may be guided by the identification of prognostic indicators such as APACHE II scores, PaO₂, serum albumin, CRP levels, and genetic factors. Early identification of high-risk patients facilitates prompt interventions, including aggressive supportive care, tailored medication, and assessment of supplementary treatments.63 The incorporation of pharmacogenomic information, specifically NAT2 genotyping, may enhance therapeutic strategies for patients with TBM.61 The management of TB patients in ICU settings requires a comprehensive approach that incorporates clinical severity, laboratory markers, genetic factors, and individualized treatment plans to optimize outcomes.40
6. ICU OUTCOMES OF CASES OF TB
Outcome of TB cases cared in ICU differs markedly according to TB subtypes. MDR‑TB, especially with HIV co-infection, carries substantial ICU mortality (~38%), though pediatric outcomes are notably better.10,42,43 HIV co-infection, even without MDR‑TB, approximately doubles mortality compared to HIV-negative TB.16 TB meningitis in ICU is especially lethal-mortality ranges from 30% (in-hospital) to > 65% at one year, worsening further with HIV and MDR‑TB.36,61 Pulmonary TB leading to respiratory failure is associated with high ICU mortality (~60–70%), frequently compounded by ARDS and multi-organ dysfunction..32 While Miliary TB, although less frequent, can progress rapidly in ICU settings, especially without prompt treatment, leading to MOF and high fatality.15 ICU outcomes of the different TB cases subtypes with the main risk factors and expected survival are summarized in Table 2.| Table 2: ICU outcomes of the different TB subtypes with the main risk factors and expected survival | ||
| TB Subtype | ICU Mortality / Survival | Key Risk Factors / Notes |
| MDR‑TB (including HIV‑co-infection) 10,42,43 | Adults: ~38% mortality; success rate ~50–57%. Children fare better (mortality ~11%). |
Higher mortality in adults; better outcomes with fluoroquinolones, aminoglycosides, Group IV drugs. HIV presence worsens prognosis. |
| TB‑HIV co‑infection16,22 | Pooled mortality ~16.7% overall; TB‑HIV mortality 16–35% vs TB-only ~4–9%. | Poor outcomes linked to advanced age, bedridden status, extrapulmonary TB, low CD4 (<50 cells/mm³), advanced WHO stage, OIs, and lack of prophylaxis. |
| TB meningitis (TBM)36,61 | General TBM mortality ~20%; ICU cases up to 65% at 1 year. In-hospital ~30%; HIV+ mortality ~53% vs ~22% in HIV−. |
Risk factors: HIV, advanced BMRC grade II/III, positive CSF culture, age >40, low GCS, MDR‑TB. CNS involvement, need for mechanical ventilation, and neurosurgical intervention are common and portend worse outcomes. |
| Pulmonary/Disseminated TB with respiratory failure32 | ICU mortality 60–69% for ventilated TB; significantly higher than non-TB ARDS (56%) & pneumonia (36%). | Common complications: ARDS, multi-organ failure (MOF), nosocomial infections, pneumothorax, renal failure. Risk factors include older age, high APACHE‑II scores, delayed recognition, and extensive pulmonary involvement. |
| Miliary TB15 | Mortality ~25% overall; ICU-associated MOF-related death up to ~100% if untreated. | High risk of ARDS and MOF; often associated with HIV or immunosuppression. |
7. ICU management of TB cases in children and elderly
Diagnosing and treating critically ill pediatric and geriatric tuberculosis patients necessitates specialized attention. Early diagnosis of disseminated or extrapulmonary tuberculosis in children is challenging due to nonspecific symptoms and a low bacillary load. Weight-based dosing is essential because of pharmacokinetic alterations associated with age. The absence of pediatric formulations complicates drug administration. Pediatric ICUs must take into account growth, nutrition, and development in the provision of supportive care.64,65 In elderly tuberculosis patients, there is a higher prevalence of unusual symptoms, comorbidities, polypharmacy, and organ dysfunction. These factors elevate the risk of pharmaceutical toxicity, encompassing neurotoxicity and hepatotoxicity. Immunosenescence and frailty may obscure clinical signs and postpone diagnosis. Older ICU patients require geriatric assessments, ongoing monitoring for adverse drug reactions, and careful adjustments to medications considering renal and hepatic function. Comprehending the distinct characteristics of high-risk TB patients requiring intensive care is essential for achieving optimal outcomes.66,678. Future Directions and Research Gaps
Despite ongoing advances in diagnostics and treatment, tuberculosis (TB) remains a significant cause of critical illness and ICU admission, particularly in low- and middle-income countries. Several important future directions and research gaps have been identified in the 2020–2025 literature that warrant further exploration (Table 3).| Table 3: Future directions and research gaps in intensive care management of TB | |||
| Priority Rank | Topic | Gap | Future Direction |
| 1 | Early Diagnostic Tools for ICU Settings | Traditional diagnostic methods (e.g., sputum microscopy, culture) are slow and often impractical in ICU patients who are intubated or unable to expectorate. | Develop and validate point-of-care molecular diagnostics (e.g., ultra-sensitive GeneXpert, TB-LAMP). Advance non-sputum-based tools (e.g., urine LAM, blood biomarkers) for ICU use. |
| 2 | Validated Prognostic Scoring Systems | No TB-specific ICU scoring system fully reflects TB pathophysiology and host factors, though general scores (e.g., APACHE II, SOFA) are used. | Design and validate TB-specific prognostic models including immune markers, imaging, comorbidities, and organ dysfunction indicators. |
| 3 | Optimal Management of TB-Associated Sepsis and ARDS | Limited evidence guides treatment of TB-related sepsis, ARDS, or MODS in ICU patients. | Conduct clinical trials on adjunctive therapies (e.g., corticosteroids, immunomodulators, statins, anti-TNF agents) to establish treatment protocols. |
| 4 | Drug-Resistant TB (DR-TB) in Critical Care | Scarce data on outcomes and drug management (e.g., bedaquiline, pretomanid) in ICU patients with organ dysfunction. | Perform pharmacokinetic and safety studies on newer and second-line TB drugs in critically ill populations to guide dosing and improve outcomes. |
| 5 | Integrated Management of TB with Comorbid Conditions | Lack of integrated care pathways for ICU TB patients with comorbidities (e.g., HIV, diabetes, liver disease, malnutrition). | Develop multidisciplinary care models involving infectious disease, nutrition, endocrinology, and hepatology. Study bundled interventions for multi-risk patients. |
| – | Precision Medicine and Pharmacogenomics | Current ICU TB management does not account for individual variability in drug metabolism (e.g., NAT2 genotype effects on isoniazid). | Incorporate pharmacogenomic profiling into ICU TB treatment planning, especially for TB meningitis and DR-TB, to personalize therapies and reduce toxicity. |
| – | Long-Term Outcomes and Post-ICU Care | Insufficient data on long-term functional, cognitive, and respiratory outcomes of ICU-treated TB survivors. | Launch prospective studies to evaluate quality of life, rehabilitation needs, and TB recurrence after ICU discharge for better follow-up and planning. |
| – | Equity and Access in Low-Resource Settings | Research and interventions are mostly urban or high-income-based, limiting relevance to settings with highest TB burden. | Focus implementation science on delivering ICU TB care in resource-limited areas using telemedicine, mobile diagnostics, and task-shifting models. |
This review addresses a significant and contemporary issue: the management of tuberculosis (TB) in ICUs, emphasizing drug-resistant strains and co-infections such as HIV. The systematic approach and extensive array of topics are noteworthy, encompassing diagnostic challenges, treatment options, and supportive care strategies. The review provides a comprehensive overview of the clinical challenges associated with managing tuberculosis in the intensive care unit, synthesizing literature from 2000 to 2025 and incorporating various study types, including guidelines, cohort studies, and retrospective analyses. The emphasis on MDR-TB and the impact of serious illness on drug efficacy provides valuable clinical insights.
However, certain issues within the review diminish its utility. The absence of a quantitative synthesis of the data limits the strength of its conclusions. The diversity of study types may complicate the application of results to other contexts, and the absence of stratification by location or healthcare resource setting diminishes the utility of the recommendations in particular scenarios. The paper discusses the prolonged duration of diagnosis and treatment but lacks detailed examination of emerging diagnostic technologies, such as molecular tests, which may hold significant relevance in intensive care units. A further issue is the lack of specific pharmacological adjustments and therapeutic monitoring protocols for severely ill TB patients.
The review contains several significant gaps that diminish its overall utility. The discussion omits high-risk populations such as pediatric and elderly ICU patients, and it fails to address the application of prognostic tools or scoring systems for clinical decision-making. Discussing supportive treatment is essential; however, it would be more beneficial to incorporate established ICU protocols for tuberculosis-related complications such as sepsis or acute respiratory distress syndrome into the conversation. Future research should focus on developing guidelines tailored specifically for ICUs. Consideration must be given to the issues of medication resistance, host immunosuppression, and the variability of resources across different health systems globally.
9. CONCLUSION
The management of tuberculosis (TB) in intensive care presents significant clinical challenges due to elevated mortality rates, diagnostic delays, and complex comorbidities. Patients with respiratory failure, HIV co-infection, drug-resistant tuberculosis, or sepsis—specifically critically ill tuberculosis patients—exhibit an elevated risk of mortality, which typically ranges from 30% to 50%. Recent research from 2020 to 2025 identifies low arterial oxygen levels, hypoalbuminemia, elevated C-reactive protein, and high APACHE II scores as significant predictors of adverse outcomes. Delays in diagnosis, stemming from the limited application of rapid diagnostic techniques in ICU settings, remain a significant barrier to timely treatment. Coexisting illnesses such as malnutrition, hepatic or renal failure, and COVID-19 complicate treatment and worsen prognosis. Recent research in pharmacogenomics, particularly involving NAT2 genotyping in tuberculosis meningitis, highlights the potential for personalized treatment strategies to improve outcomes. Prioritizing early recognition, tailored therapeutic approaches, comprehensive comorbidity management, and investment in diagnostics and precision medicine is essential for reducing mortality and enhancing recovery in the intensive care management of tuberculosis.
10. Conflict of interest
All authors declare that there was no conflict of interest.
11. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
12. Authors’ contribution
EME: Conceptualization, Methodology, Writing – Original Draft, Supervision
BDMA: Literature Review, Data Curation, Writing – Review & Editing
HRHA: Investigation, Formal Analysis, Writing – Review & Editing
ASJA: Validation, Visualization, Writing – Review & Editing
AMMA: Resources, Literature Search, Writing – Review & Editing
FNKA: Project Administration, Critical Review, Writing – Review & Editing
IDHA: Supervision, Final Approval of the Manuscript, Writing – Review & Editing
13. REFERENCES
- Cords O, Martinez L, Warren JL, O'Marr JM, Walter KS, Cohen T, et al. Incidence and prevalence of tuberculosis in incarcerated populations: a systematic review and meta-analysis. Lancet Public Health. 2021;6(5):e300-8. [PubMed] DOI: 1016/S2468-2667(21)00025-6
- Martinez L, Warren JL, Harries AD, Croda J, Espinal MA, Olarte RAL, et al. Global, regional, and national estimates of tuberculosis incidence and case detection among incarcerated individuals from 2000 to 2019: a systematic analysis. Lancet Public Health. 2023;8(7):e511-9. [PubMed] DOI: 1016/S2468-2667(23)00097-X
- Thomas L, Chacko B, Jupudi S, Mathuram A, George T, Gunasekaran K, et al. Clinical Profile and Outcome of Critically Ill Patients with Tuberculosis. Indian J Crit Care Med. 2021;25(1):21-8. [PubMed] DOI: 5005/jp-journals-10071-23503
- Muthu V, Dhooria S, Agarwal R, Prasad KT, Aggarwal AN, Behera D, et al. Profile of Patients with Active Tuberculosis Admitted to a Respiratory Intensive Care Unit in a Tertiary Care Center of North India. Indian J Crit Care Med. 2018;22(2):63-6. [PubMed] DOI: 4103/ijccm.IJCCM_491_17
- Muthu V, Dhooria S, Aggarwal AN, Behera D, Sehgal IS, Agarwal R. Acute Respiratory Distress Syndrome Due To Tuberculosis in a Respiratory ICU Over a 16-Year Period. Crit Care Med. 2017 Oct;45(10):e1087-e1090. [PubMed]DOI: 1097/CCM.0000000000002479
- Chowdhury K, Ahmad R, Sinha S, Dutta S, Haque M. Multidrug-Resistant TB (MDR-TB) and Extensively Drug-Resistant TB (XDR-TB) Among Children: Where We Stand Now. Cureus. 2023;15(2):e35154. [PubMed] DOI: 7759/cureus.35154
- Singh M, Sarhan MO, Damiba NNL, Singh AK, Villabona-Rueda A, Nino-Meza OJ, et al. Proapoptotic Bcl-2 inhibitor as potential host directed therapy for pulmonary tuberculosis. Nat Commun. 2025;16(1):3003. [PubMed] DOI: 1038/s41467-025-58190-x
- Anton C, Lemos CX, Machado FD, Bernardi RM, Freitas AA, Silva DR. Tuberculosis in the intensive care unit: alternative treatment regimens and association with mortality. Trop Med Int Health. 2021;26(1):111-4. [PubMed] DOI: 1111/tmi.13511
- Wang JL, Yin QY, Han C, Liu FL, Wang MS. Risk factors for death in tuberculosis patients requiring ICU care. Epidemiol Infect. 2021;149:e22. [PubMed] DOI: 1017/S0950268820003106
- Rao PS, Moore CC, Mbonde AA, Nuwagira E, Orikiriza P, Nyehangane D, et al. Population Pharmacokinetics and Significant Under-Dosing of Anti-Tuberculosis Medications in People with HIV and Critical Illness. Antibiotics. 2021;10(6):739. [PubMed] DOI: 3390/antibiotics10060739
- Galvin J, Tiberi S, Akkerman O, Kerstjens HAM, Kunst H, Kurhasani X, et al. Pulmonary tuberculosis in intensive care setting, with a focus on the use of severity scores, a multinational collaborative systematic review. Pulmonology. 2022;28(4):297-309. [PubMed] DOI: 1016/j.pulmoe.2022.01.016
- Nel M, Franckling-Smith Z, Pillay T, Andronikou S, Zar HJ. Chest Imaging for Pulmonary TB-An Update. Pathogens. 2022;11(2):161. [PubMed] DOI: 3390/pathogens11020161
- Sudarsan TI, Thomas L, Samprathi A, Chacko B, Mathuram A, George T, et al. Tuberculous ARDS is associated with worse outcome when compared with non-tuberculous infectious ARDS. J Crit Care. 2021;61:138-43. [PubMed] DOI: 1016/j.jcrc.2020.10.015
- Tan DTM, See KC. Diagnosis and management of severe pulmonary and extrapulmonary tuberculosis in critically ill patients: A mini review for clinicians. World J Crit Care Med. 2024;13(2):91435. [PubMed] DOI: 5492/wjccm.v13.i2.91435
- Sharma SK, Mohan A. Miliary Tuberculosis. Microbiol Spectr. 2017;5(2):TNMI7-0013-2016. [PubMed] DOI: 1128/microbiolspec.TNMI7-0013-2016
- Meintjes G, Maartens G. HIV-Associated Tuberculosis. N Engl J Med. 2024;391(4):343-55. [PubMed] DOI: 1056/NEJMra2308181
- Migliori GB, Ong CWM, Petrone L, D'Ambrosio L, Centis R, Goletti D. The definition of tuberculosis infection based on the spectrum of tuberculosis disease. Breathe. 2021;17(3):210079. [PubMed] DOI: 1183/20734735.0079-2021
- Hermans SM, Akkerman OW, Meintjes G, Grobusch MP. Post-tuberculosis treatment paradoxical reactions. Infection. 2024;52(5):2083-95. [PubMed] DOI: 1007/s15010-024-02310-
- Starke JR, Erkens C, Ritz N, Kitai I. Strengthening Tuberculosis Services for Children and Adolescents in Low Endemic Settings. Pathogens. 2022;11(2):158. [PubMed] DOI: 3390/pathogens11020158
- So C, Ling L, Wong WT, Zhang JZ, Ho CM, Ng PY, et al. Population study on diagnosis, treatment and outcomes of critically ill patients with tuberculosis (2008-2018). Thorax. 2023;78(7):674-81. [PubMed] DOI: 1136/thorax-2022-218868
- Divala TH, Lewis J, Bulterys MA, Lutje V, Corbett EL, Schumacher SG, et al. Missed opportunities for diagnosis and treatment in patients with TB symptoms: a systematic review. Public Health Action. 2022;12(1):10-7. [PubMed] DOI: 5588/pha.21.0022
- Kacprzak A, Oniszh K, Podlasin R, Marczak M, Cielniak I, Augustynowicz-Kopeć E, et al. Atypical Pulmonary Tuberculosis as the First Manifestation of Advanced HIV Disease-Diagnostic Difficulties. Diagnostics. 2022;12(8):1886. [PubMed] DOI: 3390/diagnostics12081886
- Savage HR, Rickman HM, Burke RM, Odland ML, Savio M, Ringwald B, et al. Accuracy of upper respiratory tract samples to diagnose Mycobacterium tuberculosis: a systematic review and meta-analysis. Lancet Microbe. 2023;4(10):e811-21. [PubMed] DOI: 1016/S2666-5247(23)00190-8
- Rahmati S, Bahrampour A, Nasehi M, Mirzazadeh A, Ghaderi H, Shahesmaeili A. An Evaluation of The Diagnostic Value of Sputum Smears Microscopy and Pcr Relative to Sputum Culture in The Diagnosis of Pulmonary Tuberculosis: A Systematic Review and Meta-Analysis in Iran. Med J Islam Repub Iran. 2022;36:112. [PubMed] DOI: 47176/mjiri.36.112
- Sava C, Sava M, Drăgan AM, Iuhas A, Niulaș L, Marinău CP, et al. The Use of Xpert MTB/RIF Ultra Testing for Early Diagnosis of Tuberculosis: A Retrospective Study from a Single-Center Database. Genes. 2023;14(6):1231. [PubMed] DOI: 3390/genes14061231
- Chacko B, Chaudhry D, Peter JV, Khilnani GC, Saxena P, Sehgal IS, et al. ISCCM Position Statement on the Approach to and Management of Critically Ill Patients with Tuberculosis. Indian J Crit Care Med. 2024;28(Suppl 2):S67-91. [PubMed] DOI: 5005/jp-journals-10071-24783
- Beraldi-Magalhaes F, Parker SL, Sanches C, Garcia LS, Souza Carvalho BK, Costa AA, et al. Is the Pharmacokinetics of First-Line Anti-TB Drugs a Cause of High Mortality Rates in TB Patients Admitted to the ICU? A Non-Compartmental Pharmacokinetic Analysis. Trop Med Infect Dis. 2023;8(6):312. [PubMed] DOI: 3390/tropicalmed8060312
- Sturkenboom MGG, Märtson AG, Svensson EM, Sloan DJ, Dooley KE, van den Elsen SHJ, et al. Population Pharmacokinetics and Bayesian Dose Adjustment to Advance TDM of Anti-TB Drugs. Clin Pharmacokinet. 2021;60(6):685-710. [PubMed] DOI: 1007/s40262-021-00997-0
- Sarkar M, Sarkar J. Therapeutic drug monitoring in tuberculosis. Eur J Clin Pharmacol. 2024;80(11):1659-84. [PubMed] DOI: 1007/s00228-024-03749-8
- Metarfi Y, Chellal W, Ben Khadda Z, Hoummani H, Amara B, Achour S. Therapeutic drug monitoring in anti-tuberculosis treatment: a systematic review. J Antimicrob Chemother. 2025. [PubMed] DOI: 1093/jac/dkaf126
- Chaudhry D, Tyagi D. Tuberculosis in Intensive Care Unit. Indian J Crit Care Med. 2021;25(Suppl 2):S150-4. [PubMed] DOI: 5005/jp-journals-10071-23872
- Hashemian SM, Khoundabi B, Bahrami A, Jamaati H, Varahram M, Saljoughi L, et al. Optimal Positive End-expiratory Pressure Levels in Tuberculosis-associated Acute Respiratory Distress Syndrome. Int J Mycobacteriol. 2024;13(3):247-51. [PubMed] DOI: 4103/ijmy.ijmy_136_24
- Jaffet MF, Abubakar M, Ibrahim Y, Ogbuneke U, Wahoo W. Disseminated Tuberculosis Resulting in Septic Shock in an Immunocompetent Patient. Cureus. 2022;14(8):e28025. [PubMed] DOI: 7759/cureus.28025
- Du ZX, Chang FQ, Wang ZJ, Zhou DM, Li Y, Yang JH. A risk prediction model for acute kidney injury in patients with pulmonary tuberculosis during anti-tuberculosis treatment. Ren Fail. 2022;44(1):625-35. [PubMed] DOI: 1080/0886022X.2022.2058405
- Molla Y, Wubetu M, Dessie B. Anti-Tuberculosis Drug Induced Hepatotoxicity and Associated Factors among Tuberculosis Patients at Selected Hospitals, Ethiopia. Hepat Med. 2021;13:1-8. [PubMed] DOI: 2147/HMER.S290542
- Adefuye MA, Manjunatha N, Ganduri V, Rajasekaran K, Duraiyarasan S, Adefuye BO. Tuberculosis and Cardiovascular Complications: An Overview. Cureus. 2022;14(8):e28268. [PubMed] DOI: 7759/cureus.28268
- Ockenga J, Fuhse K, Chatterjee S, Malykh R, Rippin H, Pirlich M, et al. Tuberculosis and malnutrition: The European perspective. Clin Nutr. 2023;42(4):486-92. [PubMed] DOI: 1016/j.clnu.2023.01.016
- Patti G, Pellegrino C, Ricciardi A, Novara R, Cotugno S, Papagni R, et al. Potential Role of Vitamins A, B, C, D and E in TB Treatment and Prevention: A Narrative Review. Antibiotics. 2021;10(11):1354. [PubMed] DOI: 3390/antibiotics10111354
- Febi AR, Manu MK, Mohapatra AK, Praharaj SK, Guddattu V. Psychological stress and health-related quality of life among tuberculosis patients: a prospective cohort study. ERJ Open Res. 2021;7(3):00251-2021. [PubMed] DOI: 1183/23120541.00251-2021
- Ramos JP, Vieira M, Pimentel C, Argel M, Barbosa P, Duarte R. Building bridges: multidisciplinary teams in tuberculosis prevention and care. Breathe. 2023;19(3):230092. [PubMed] DOI: 1183/20734735.0092-2023
- Peetluk LS, Ridolfi FM, Rebeiro PF, Liu D, Rolla VC, Sterling TR. Systematic review of prediction models for pulmonary tuberculosis treatment outcomes in adults. BMJ Open. 2021;11(3):e044687. [PubMed] DOI: 1136/bmjopen-2020-044687
- Akalu TY, Clements ACA, Gebreyohannes EA, Gilmour B, Alene KA. Prevalence of tuberculosis infection among contacts of drug-resistant tuberculosis patients: A systematic review and meta-analysis. J Infect. 2024;89(2):106198. [PubMed] DOI: 1016/j.jinf.2024.106198
- Alemu A, Bitew ZW, Worku T, Gamtesa DF, Alebel A. Predictors of mortality in patients with drug-resistant tuberculosis: A systematic review and meta-analysis. PLoS One. 2021;16(6):e0253848. [PubMed] DOI: 1371/journal.pone.0253848
- Naidoo K, Perumal R, Ngema SL, Shunmugam L, Somboro AM. Rapid Diagnosis of Drug-Resistant Tuberculosis-Opportunities and Challenges. Pathogens. 2023;13(1):27. [PubMed] DOI: 3390/pathogens13010027
- Nandlal L, Perumal R, Naidoo K. Rapid Molecular Assays for the Diagnosis of Drug-Resistant Tuberculosis. Infect Drug Resist. 2022;15:4971-84. [PubMed] DOI: 2147/IDR.S381643
- Khan U, Huerga H, Khan AJ, Mitnick CD, Hewison C, Varaine F, et al. The endTB observational study protocol: treatment of MDR-TB with bedaquiline or delamanid containing regimens. BMC Infect Dis. 2019;19(1):733. [PubMed] DOI: 1186/s12879-019-4378-4
- Migliori GB, Pontali E, Sotgiu G, Centis R, D'Ambrosio L, Tiberi S, et al. Combined Use of Delamanid and Bedaquiline to Treat Multidrug-Resistant and Extensively Drug-Resistant Tuberculosis: A Systematic Review. Int J Mol Sci. 2017;18(2):341. [PubMed] DOI: 3390/ijms18020341
- Naidoo K, Perumal R, Cox H, Mathema B, Loveday M, Ismail N, et al. The epidemiology, transmission, diagnosis, and management of drug-resistant tuberculosis-lessons from the South African experience. Lancet Infect Dis. 2024;24(9):e559-75. [PubMed] DOI: 1016/S1473-3099(24)00144-0
- Saktiawati AMI, Vasiliu A, Saluzzo F, Akkerman OW. Strategies to Enhance Diagnostic Capabilities for the New Drug-Resistant Tuberculosis (DR-TB) Drugs. Pathogens. 2024;13(12):1045. [PubMed] DOI: 3390/pathogens13121045
- Mohd Hassan NZA, Razali A, Shahari MR, Mohd Nor Sham Kunusagaran MSJ, Halili J, Zaimi NA, et al. Cost-Effectiveness Analysis of High-Risk Groups Tuberculosis Screening in Malaysia. Front Public Health. 2021;9:699735. [PubMed] DOI: 3389/fpubh.2021.699735
- López-López JP, Posada-Martínez EL, Saldarriaga C, Wyss F, Ponte-Negretti CI, Alexander B, et al. Tuberculosis and the Heart. J Am Heart Assoc. 2021;10(7):e019435. [PubMed] DOI: 1161/JAHA.120.019435
- TB/COVID-19 Global Study Group. Tuberculosis and COVID-19 co-infection: description of the global cohort. Eur Respir J. 2022;59(3):2102538. [PubMed] DOI: 1183/13993003.02538-2021
- Hosu MC, Faye LM, Apalata T. Comorbidities and Treatment Outcomes in Patients Diagnosed with Drug-Resistant Tuberculosis in Rural Eastern Cape Province, South Africa. Diseases. 2024;12(11):296. [PubMed] DOI: 3390/diseases12110296
- Shin HJ, Chang JS, Kim MS, Koh BG, Park HY, Kim TO, et al. Hypersensitivity reactions to multiple anti-tuberculosis drugs. PLoS One. 2021;16(2):e0246291. [PubMed] DOI: 1371/journal.pone.0246291
- Kapwata T, Breetzke G, Wright CY, Marcus TS, Eales O. Demographic and socio-economic risk factors associated with self-reported TB. Int J Tuberc Lung Dis. 2022;26(1):33-7. [PubMed] DOI: 5588/ijtld.21.0247
- Yuan QW, Li W, Yang K, Guo J, Zheng Y. Predictive Mortality of the Prognostic Nutritional Index Combined with APACHE II Score for Critically Ill Tuberculosis Patients. Am J Trop Med Hyg. 2024;111(5):1027-33. [PubMed] DOI: 4269/ajtmh.23-0661
- Liu Q, Gao J, Luo B, Liu J, Zhang L, Kang W, et al. Prediction model for death in patients with pulmonary tuberculosis accompanied by respiratory failure in ICU: retrospective study. Ann Palliat Med. 2020;9(5):2731-40. [PubMed] DOI: 21037/apm-20-182
- Alonzi T, Petruccioli E, Aiello A, Repele F, Goletti D. Diagnostic tests for tuberculosis infection and predictive indicators of disease progression: Utilizing host and pathogen biomarkers to enhance the tuberculosis elimination strategies. Int J Infect Dis. 2025;155:107880. [PubMed] DOI: 1016/j.ijid.2025.107880
- Cui K, Feng S, Mao Y, Luo H, Yang J, Xu R, et al. The association between blood urea nitrogen to albumin ratio and the 28 day mortality in tuberculosis patients complicated by sepsis. Sci Rep. 2024;14(1):16430. [PubMed] DOI: 1038/s41598-024-65622-z
- Samuels THA, Wyss R, Ongarello S, Moore DAJ, Schumacher SG, Denkinger CM. Evaluation of the diagnostic performance of laboratory-based c-reactive protein as a triage test for active pulmonary tuberculosis. PLoS One. 2021;16(7):e0254002. [PubMed] DOI: 1371/journal.pone.0254002
- Sileshi T, Telele NF, Burkley V, Makonnen E, Aklillu E. Correlation of N-acetyltransferase 2 genotype and acetylation status with plasma isoniazid concentration and its metabolic ratio in ethiopian tuberculosis patients. Sci Rep. 2023;13(1):11438. [PubMed] DOI: 1038/s41598-023-38716-3
- Jian Y, Bao Y, Yang F, Zhu M. The role of isoniazid dosage and NAT2 gene polymorphism in the treatment of tuberculous meningitis. Front Immunol. 2025;15:1535447. [PubMed] DOI: 3389/fimmu.2024.1535447
- de Rooij ML, Lynen L, Decroo T, Henriquez-Trujillo AR, Boyles T, Jacobs BKM. The therapeutic threshold in clinical decision-making for TB. Int Health. 2023;15(6):615-22. [PubMed] DOI: 1093/inthealth/ihad002
- Jaganath D, Beaudry J, Salazar-Austin N. Tuberculosis in Children. Infect Dis Clin North Am. 2022;36(1):49-71. [PubMed] DOI: 1016/j.idc.2021.11.008
- Moore BK, Graham SM, Nandakumar S, Doyle J, Maloney SA. Pediatric Tuberculosis: A Review of Evidence-Based Best Practices for Clinicians and Health Care Providers. Pathogens. 2024;13(6):467. [PubMed] DOI: 3390/pathogens13060467
- Sharma OP. Tuberculosis in elderly persons. Indian J Tuberc. 2022;69(Suppl 2):S196-7. [PubMed] DOI: 1016/j.ijtb.2022.10.005
- Tabernero E, Rodrigo T, Garros J, Altube L, Garay E, Medina JF, et al. TB in the elderly: clinical features and outcomes. Int J Tuberc Lung Dis. 2022;26(9):842-9. [PubMed] DOI: 5588/ijtld.22.0010