Assem Mohamed Fathy Mohamed 1, Nermin Sadek Nasr 2, Salwa Omar El Khattab Amin 3, Ahmad Moustafa Mohamed Mohamed 4
Authors affiliations:
Background & objective: Coronary artery disease (CAD) significantly impacts patients’ life and the surgical outcomes, necessitating effective preoperative risk assessment. Pulmonary artery systolic pressure (PASP) measured intraoperatively via transesophageal echocardiography (TEE) may serve as a critical indicator of postoperative complications in elective on-pump coronary artery bypass grafting (CABG).
We aimed to evaluate the association between elevated intraoperative PASP and postoperative early complications in patients undergoing elective on-pump CABG.
Methodology: This study involved 70 patients undergoing elective on-pump CABG, divided into two groups, based on intraoperative PASP measured using TEE: Group A: 35 patients with PASP ≥ 35 mmHg. Group B: 35 patients with PASP < 35 mmHg. The study analyzed PASP association with early postoperative complications, including respiratory failure, significant inotrope use, intra-aortic balloon pump (IABP) use, prolonged ICU stay (> 48 hours), and extended hospital stay (> 7 days).
Results: Postoperative respiratory failure (P < 0.009), significant inotrope use (P < 0.001), IABP use (P < 0.023), prolonged ICU stay (> 48 hours) (P < 0.001), and extended hospital stay (> 7 days) (P < 0.001) were significantly higher in Group A. No statistically significant differences between groups regarding postoperative anticoagulation, pacemaker dependence, arrhythmias, or myocardial infarction were noted (P = 0.145, 0.164, 0.55, and 0.743, respectively). When comparing PASP grades: severe PASP was associated with higher rates of postoperative respiratory failure, IABP use, prolonged ICU stays, and total hospital stays (P = 0.03, 0.01, 0.01, and < 0.001, respectively). Moderate pulmonary hypertension was associated with the most frequent use of significant inotropes (P = 0.03). No significant differences in postoperative anticoagulation, arrhythmias, myocardial infarction, or pacemaker dependence (P = 0.21, 0.87, 0.84, and 0.19, respectively) was noted.
Conclusion: Intraoperative PASP measurement using TEE is a valuable predictor of early postoperative complications and events in patients undergoing elective on-pump CABG.
Abbreviations: IAPB: intra-aortic balloon pump, IBP: invasive blood pressure, ICU: Intensive care unit, CABG: coronary artery bypass grafting, CAD: Coronary artery disease, PAH: Pulmonary arterial hypertension, PASP: Pulmonary artery systolic pressure, RAP: right atrial pressure, RHC: right heart catheterization, TEE: transesophageal echocardiography
Keywords: Pulmonary artery systolic pressure (PASP), Transesophageal echocardiography (TEE), On-pump coronary artery bypass graft (CABG).
Citation: Mohamed AMF, Nasr NS, Amin SOE, Mohamed AMM. Association of pulmonary artery systolic pressure measured by transesophageal echocardiography with early postoperative morbidity in patients undergoing elective on-pump coronary artery bypass grafting. Anaesth. pain intensive care 2025;29(5):317-327. DOI: 10.35975/apic.v29i5.2856
Received: March 24, 2025; Revised: April 18, 2025; Accepted: May 21, 2025
Coronary artery disease (CAD) is the leading global cause of death and disability, responsible for ~16 million deaths and 278.2 million Disability Adjusted Life Years (DALYs) in 2019, disproportionately affecting low- and middle-income countries (28 of deaths, higher DALY rates vs. wealthy nations).1 In Egypt, CAD accounts for 68.4 of cardiovascular cases.2 Despite advances in treatments like coronary artery bypass grafting (CABG) and percutaneous coronary interventions (PCI), with 200,000 CABG and 965,000 PCI procedures performed in the U.S. in 2018 mortality remains high, especially in patients with comorbidities. Effective secondary prevention strategies, including optimized medical care, lifestyle changes, and cardiac rehabilitation, are critical to improving outcomes.3
Preoperative risk assessment is critical for optimizing surgical outcomes. The Society of Thoracic Surgeons (STS) score outperforms Euro SCORE II in predicting in-hospital mortality and complications.4 Pulmonary hypertension (PH), defined as systolic pressure ≥35 mmHg, is a key risk factor in cardiac surgery, often caused by left heart dysfunction or valvular disease. Doppler echocardiography offers a noninvasive method to monitor PH and stratify risk.5
After coronary artery bypass grafting (CABG), complications like myocardial edema and ischemia-reperfusion injury can lead to low cardiac output, heart attacks, or irregular heart rhythms. Neurological issues, such as stroke or coma (2-5 of cases) and mild cognitive impairment (up to 50 of patients), further contribute to longer hospital stays, higher costs, and increased mortality.6
The primary objective was to determine the association between elevated pulmonary artery systolic pressure (PASP ≥ 35 mmHg), measured intraoperatively by transesophageal echocardiography (TEE), and the incidence of early postoperative complications in patients undergoing elective on-pump coronary artery bypass grafting (CABG). The secondary objective was to identify and evaluate specific preoperative and intraoperative risk factors that significantly contribute to the elevation of PASP ≥ 35 mmHg, during elective on-pump CABG.
This was a prospective and observational clinical study conducted on seventy patients of both sexes aged from 30 to 80 years, who were scheduled for isolated elective CABG on cardiopulmonary bypass (CPB). Enrolled consented patients were allocated into one of the following groups according to intraoperative PASP measured by TEE: Group A: 35 patients (PASP ≥ 35 mmHg; Pulmonary hypertension group), Additionally, individuals with increased PASP (≥ 35 mmHg) were divided into mild, moderate, and severe groups based on grades measured by TEE. Group B: contained 35 patients with PASP < 35 mmHg; Non pulmonary hypertension group.7
The study was conducted in the cardiac surgery operating rooms and cardiothoracic intensive care units (ICU) of Ain Shams University Hospitals from March 2022 to March 2024 It was approved by the Research Ethical Committee, Faculty of Medicine, Ain Shams University (approval number FWA 000017585, FMASU MD 14/2022). Informed and written consent was obtained from all participants. This study was registered with the Pan African Clinical Trial Registry (PACTR) under the identification number [PACTR202408668477631].
Patients with significant right ventricular outflow tract obstruction, contraindications to TEE, non-analyzable tricuspid regurgitation (TR) jets, scheduled for off-pump, urgent, redo, or combined valve CABG procedures, severe pulmonary diseases causing elevated pulmonary pressures such as chronic obstructive pulmonary disease (COPD), pulmonary fibrosis, obstructive sleep apnea, or those diagnosed with primary pulmonary arterial hypertension (PAH) were excluded from the study.
The sample size was determined using the PASS 11 program, with a confidence level set at 90 and a margin of error of +0.15. Previous research findings, particularly those by Subramanian et al. in 2020, highlighted a notable discrepancy in postoperative complications, specifically respiratory failure, between patients with high and low PASP. Based on these insights, a sample size of at least 35 patients with high PASP (≥ 35 mmHg) and 35 patients with low PASP (< 35 mmHg) was sufficient to achieve the study objectives.8
Simple random sampling was used to select the participants. All patients underwent comprehensive evaluation, including medical history review, clinical examination, functional status assessment (e.g., exercise tolerance, heart failure symptoms), and routine tests such as CBC, FBS, liver/kidney function panels, ECG, PT/aPTT, transthoracic echocardiography, cardiac catheterization, and chest X-ray. Volume overload was managed with diuretics to alleviate pulmonary congestion and reduce right ventricular strain, particularly in patients with right heart failure. Beta-blockers and calcium channel blockers were continued to control heart rate and lower myocardial oxygen demand, though careful monitoring was implemented to avoid adverse effects on right ventricular function.
Standard monitoring devices were attached to the patient. The patient was monitored intraoperatively, postoperatively, and in the ICU using the following: electrocardiogram (ECG), pulse oximetry (SpO2), non-invasive blood pressure (NIBP), invasive blood pressure (IBP) monitoring, intraoperative arterial pressure-based cardiac output measurement via arterial line insertion, central venous pressure (CVP), urine output, arterial blood gas (ABG) analysis, activated clotting time (ACT), nasopharyngeal core temperature measurement, and intraoperative TEE after the induction of anesthesia.
Intravenous access (IV) was obtained. All patients were premedicated with morphine sulphate 0.05 to 0.1 mg/kg IV one hour before surgery. After administration of IV midazolam (0.05mg/kg), both groups underwent standardized general anesthesia starting with pre-oxygenation with 100 oxygen before intubation. Smooth cardiac induction with fentanyl (5-10 µg/kg) slowly administered intravenously to both groups over 1-2 min, propofol (1-2 mg/kg) IV injected slowly over 30 sec guided by monitored invasive blood pressure, and pancuronium (0.07-0.1 mg/kg) was used to intubate the patients. Controlled mechanical ventilation (volume-controlled mode; tidal volume (TV) 6-8 mL/kg, respiratory rate 10-12 breath/min, PEEP 3-5 cmH2O) with 100 oxygen and inhalational agent isoflurane adjusted to (0.6 - 1.6) minimal alveolar concentration (MAC) with boluses of fentanyl (1µg/kg) IV and pancuronium (0.015-0.01 mg/kg) IV every 30-60 min, used for maintenance of anesthesia.
US-guided central line was secured in the right internal jugular vein and baseline CVP measurement was done. Unfractionated heparin (UFH) was used as an IV anticoagulant in a dose of 400 IU/kg, monitored with activated clotting time (ACT) to a target of 450-600 sec. Potassium-rich cardioplegia was used to stop the heart beatings. IV fluids (Ringer acetate), blood, and FFP were administered according to the standard practice. The cardiopulmonary bypass pump was managed by a dedicated certified clinical perfusionist who interacts and coordinates with the surgeon and the anesthetist.
No regional and/or fast-track anesthesia protocols were employed in these patients. All the relevant measurements were done 30 min after anesthesia induction, during critical phases of surgery; e.g., after initiation and during weaning from CPB, and immediately before the conclusion of the procedure. This protocol allowed for continuous or intermittent monitoring of PASP and cardiac function throughout the surgical process.
After insertion of the TEE probe, all measurements were obtained 30 min following induction. Preload and afterload were normalized through volume infusion and noradrenaline administration, starting with an initial dose of 8–12 µg/min IV infusion, subsequently titrated to a maintenance dose of 2–4 µg/min IV infusion to maintain filling pressures and invasive arterial blood pressure within ±20 of the pre-induction baseline levels. Anesthesia management and related procedures were independently carried out by an anesthesiologist not involved in the study.
All TEE assessments were conducted by a single experienced anesthesiologist using standardized equipment (TEE Probe: 6TC-RS KN100106-R, Vivid S6 N, General Electric Healthcare, Horten, Norway). Parameters were measured in triplicate and averaged, employing M-mode, 2D, and color Doppler imaging.
After weaning from CPB, heparin was reversed with protamine sulfate (1-1.5 mg per 100 units heparin; max 50 mg) under ACT monitoring. Post-op care included correcting electrolytes, acid-base balance, hypothermia, and coagulopathy, followed by sternal/skin closure. Patients were transferred to the ICU with portable monitoring, continued cardiac support, sedation, intubation, and manual ventilation via an oxygen-connected Ambu bag.
In the ICU, the patients were monitored with standard monitoring, mechanically ventilated and sedated. Clinical and laboratory monitoring of hemodynamic parameters, bleeding, and other complications was done and managed accordingly. Weaning from cardiac support with the improvement of the hemodynamic parameters, extubation, and weaning from mechanical ventilation was done when the criteria of weaning were met.
The study’s primary outcome focused on intraoperative measurement of PASP, calculated using the trans-tricuspid gradient, right atrial pressure (RAP), and tricuspid regurgitation (TR) jet velocity. These values were assessed in real-time during surgery via TEE and central venous pressure (CVP) monitoring. This measurement, alongside related TEE parameters, was correlated with postoperative outcomes.
PASP was calculated using the formula: PASP = 4 × (TR jet max velocity) ² + RAP.
TR jet velocity (m/s) was measured via continuous wave Doppler in mid-esophageal 4-chamber or bicaval views, ensuring a clear signal (alignment ≤ 20° and full Doppler envelope). Trans-tricuspid gradient: Derived using the Bernoulli equation (4 × [TR velocity] ²). RAP (mmHg): Obtained directly from CVP.
These TEE-derived values were primary predictors and allowed the categorization of patients into pulmonary hypertension (PASP ≥ 35 mmHg) and non-pulmonary hypertension (PASP < 35 mmHg) groups. The relationship between elevated PASP and postoperative outcomes was examined.
The secondary outcomes consisted of the early postoperative complications and morbidity indicators. Each of these complications was recorded and evaluated to assess their incidence and relationship to intraoperative PASP:
The collected data were revised, coded, tabulated, and then analyzed using Statistical Package for Social Science (SPSS) version 27.0. Quantitative data were expressed as mean ± SD or median (IQR), and qualitative data as frequencies and percentages. Statistical significance was assessed with independent-sample t-tests and Chi-square tests, with a 95% confidence interval and a significance (S) level set at P < 0.05 and highly significant (HS) at P < 0.01.
Regarding age, sex, ASA physical status, and BMI (kg/m2), there were no statistically significant differences between the two groups.
Baseline physiological and medical characteristics, including NYHA functional class, showed no statistically significant differences between two groups. However, diabetes mellitus (DM) and peripheral vascular disease (PVD) occurred significantly more frequently in Group A compared to Group B (P < 0.001 and < 0.010), as detailed in Table 1.
Cardiac output (COP) and ejection fraction (EF) were significantly higher in Group B compared to Group A (P < 0.001 and P < 0.0076, respectively). Conversely, Group A exhibited markedly elevated E/e’ ratio, tricuspid regurgitation peak velocity (TR max), RAP, and PASP compared to Group B, all with highly significant differences (P < 0.001), as shown in Table 2.
No statistically significant differences were observed between Group A (PASP ≥ 35 mmHg) and Group B (PASP <35 mmHg) in postoperative anticoagulation (heparin infusion), pacemaker dependence, arrhythmias, or myocardial infarction (P > 0.05). However, Group A had significantly higher rates of postoperative respiratory failure (P < 0.009), inotrope use (P < 0.001), IABP requirement (P < 0.023), prolonged ICU stays (P < 0.009), and longer total hospital stays (P < 0.001) compared to Group B, as detailed in Table 3.
Comparison between groups of the PASP grades measured by TEE as regards postoperative complications: Postoperative anticoagulation, arrhythmias, and myocardial infarction rates were comparable across all PH severity groups (P = 0.21, 0.87, and 0.84, respectively). However, respiratory failure and IABP use were significantly higher in the severe PH group (P = 0.03 and P = 0.01, respectively). Inotrope dependency peaked in the Moderate PH group (P = 0.03), while prolonged ICU stays (> 48 hours) and hospital stays (>7 days) were most frequent in the severe PH group (P = 0.01 and P < 0.001), as mentioned in Figure I.
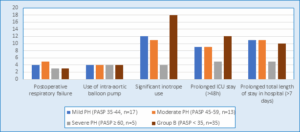
1: Bar graph between groups of the PASP grades as regards postoperative complications with a significant difference.
Pacemaker dependence showed no significant variation (P = 0.19), as detailed in Table 4.
Elevated pulmonary artery systolic pressure (PASP), as previously described, is a known marker of pulmonary hypertension and has been associated with adverse outcomes in cardiac surgery. Our findings reinforce this association, showing that patients with PASP ≥ 35 mmHg experienced higher rates of postoperative complications, highlighting the value of intraoperative PASP monitoring for risk stratification and management.
Our study revealed that postoperative respiratory failure requiring prolonged mechanical ventilation (≥48h) or re-intubation occurred in 12 patients (38.7) in Group A (elevated PASP) compared to 4 patients (10.3) in Group complications, highlighting the value of intraoperative PASP monitoring for risk stratification and management.
Our study revealed that postoperative respiratory failure requiring prolonged mechanical ventilation (≥48h) or re-intubation occurred in 12 patients (38.7) in Group A (elevated PASP) compared to 4 patients (10.3) in Group B (normal PASP), with statistical significance (p = 0.009). This underscores the hemodynamic burden posed by elevated PASP, likely through right ventricular dysfunction and compromised pulmonary perfusion. His inding aligns with Kanwar et al. (2019), who identified pulmonary hypertension as a risk factor for postoperative respiratory complications, attributed to impaired right ventricular function and intraoperative hemodynamic instability.9
Inotropic support for more than 24 hours was required in 16 patients (51.6) in Group A compared to 8 patients (20.5) in Group B (P = 0.001). IABP was needed in 7 patients (22.6) in Group A, versus 2 patients (5.1) in Group B (P = 0.01). These findings indicate that patients with elevated PASP are more likely to experience perioperative hemodynamic instability. This is consistent with Keranovic S et al. (2020), who reported an increased requirement for inotropes and mechanical support in patients with pulmonary hypertension during cardiac surgery.10
Prolonged ICU stay (> 48h) was recorded in 18 (58.1%) patients in Group A compared to 10 (25.6%) patients in Group B (P = 0.009). Similarly, hospital stays longer than 7 days occurred in 21 (67.7%) patients in Group A versus 12 (30.8%) patients in Group B (P < 0.001). These results are consistent with the work of Braksator et al. (2022), who found that pulmonary hypertension contributes to prolonged hospitalizations due to the complexity of postoperative management.11
In our study, patients with non-elevated PASP (Group B) demonstrated a numerically higher requirement for postoperative anticoagulation (P = 0.145). This contrasts with established predictors of thrombotic risk in cardiac surgery, where elevated pulmonary pressures typically correlate with increased anticoagulation demands. Differences in patient cohorts may explain this divergence, as our study focused exclusively on CABG patients. Moreover, this trend may reflect institutional protocols empirically anticoagulating normal PASP patients to optimize graft patency. Collectively, these findings reinforce guideline recommendations that anticoagulation strategies post-CABG should be individualized based on comprehensive patient-specific risks rather than isolated parameters like PASP.12
Postoperative arrhythmias, sustained beyond 5 minutes or requiring intervention, were more common in patients with elevated PASP (29) compared to those with normal PASP (15.4), though this was not statistically significant (p = 0.52). This is in line with previous research by Ruiz-Cano et al. (2020), which highlighted an elevated risk of supraventricular arrhythmias in pulmonary hypertension due to increased right atrial strain and electrical remodeling.13 Additionally, Huang et al. (2020) confirmed similar associations through systematic meta-analysis.14
According to this study, the requirement for postoperative anticoagulation was higher in the mild and moderate pulmonary hypertension (PH) groups compared to the severe PH group, although this difference was not statistically significant (P = 0.21). This finding aligns with current revascularization guidelines emphasizing that anticoagulation management should be tailored to comprehensive patient profiles rather than isolated parameters like PASP.12
The increased anticoagulation needs in mild and moderate PH may be attributed to better hemodynamic stability, as these patients typically exhibit improved right ventricular function and lower pressure overload, allowing for effective anticoagulation without the heightened bleeding risk associated with severe PH.15 In contrast, patients with severe PH experience significant strain on the right ventricle due to elevated pulmonary artery pressures, leading to right ventricular dysfunction and increased bleeding risk, which necessitates more cautious anticoagulation management.16
We found that postoperative arrhythmias were similarly prevalent across all PASP groups, with no statistically significant difference (P = 0.87). This aligns with Braman et al. (2020), who demonstrated that preoperative pulmonary hypertension does not independently predict postoperative atrial fibrillation in cardiac surgery patients after adjusting for surgical confounders (OR 1.1, 95% CI 0.9–1.4). The overall surgical stress and physiological changes during and after cardiac surgery can independently influence the risk of arrhythmias, regardless of baseline PASP levels. Intraoperative factors such as fluid shifts, electrolyte imbalances, and the use of cardiopulmonary bypass can contribute to the development of arrhythmias in all patients, masking potential differences related to PASP.17
Our study did not find significant differences in pacemaker dependence across groups (P = 0.19), which is in line with previous research suggesting that pacemaker needs are not strongly correlated with PASP grades in all groups.
Respiratory failure was significantly more frequent in the severe PH group (P = 0.03), supporting the findings of Kanwar et al. (2017), who reported a higher incidence of postoperative respiratory complications in patients with severe PH compared to those with mild or moderate PH. The increased risk of respiratory failure in these patients is attributed to underlying hemodynamic instability and impaired right ventricular function associated with elevated pulmonary artery pressures. This dysfunction can lead to inadequate pulmonary perfusion and gas exchange, resulting in hypoxemia and respiratory distress during the postoperative period.9
The study demonstrated that the need for significant inotropic support was highest in the moderate PH group (P = 0.03). This finding aligns with Rozado et al. (2019), who identified moderate pulmonary hypertension as the peak-risk subgroup for hemodynamic instability following cardiac surgery, characterized by significant fluctuations in blood pressure and cardiac output. Such instability is most critical in the early postoperative period, where fluid shifts, residual anesthetic effects, and surgical trauma compound cardiovascular strain. Unlike mild cases (where right ventricular adaptation is sufficient) or severe PH (with chronic compensatory remodeling), moderate PH induces a maladaptive RV stress response that significantly increases susceptibility to postoperative instability and inotrope dependence.18
Prolonged ICU stays (> 48 hours) and total hospital stays (>7 days) were highest in the severe PH group, with significant differences (P = 0.01 and P < 0.001, respectively). These results align with the literature, such as Braksator et al. (2022), that patients with severe PH may require more intensive monitoring, advanced pharmacologic support, and careful management of fluid balance and hemodynamics in the ICU, all of which contribute to longer recovery times extended hospital stays.11
The use of an IABP was significantly higher in the severe PH group (P = 0.01). This observation aligns with the pathophysiology of severe pulmonary hypertension, where elevated pulmonary vascular resistance imposes substantial strain on the right ventricle (RV), impairing its ability to effectively pump blood through the pulmonary circulation. This hemodynamic burden increases susceptibility to RV failure perioperatively, particularly under surgical stress. Consistent with this mechanism, Keranovic et al. (2020), reported that mechanical circulatory support (including IABP) is frequently required for critically ill cardiac surgery patients with compromised ventricular function, serving as a bridge to recovery in severe hemodynamic instability.10
Our study highlights the significant impact of different grades of PASP on postoperative outcomes for patients undergoing CABG. By illustrating the relationship between PASP levels and complications, our findings emphasize the importance of classifying patients based on their PASP levels. This strategy can improve clinical decision-making and enhance postoperative care, potentially leading to lower rates of complications and mortality.
Inuzuka et al. (2021) conducted a multicenter study involving 1,056 patients to explore the relationship between PASP, measured by Doppler echocardiography, and morbidity in patients with various heart conditions. They found a significant link between elevated PASP and increased causes of mortality and hospitalizations due to heart failure. This identifies PASP as an independent predictor of these outcomes after adjusting for other clinical factors. In contrast, our study specifically targeted the early perioperative period in patients undergoing CABG, which may result in differences in baseline characteristics and comorbidities when compared to the broader patient population examined by Inuzuka et al.19
Mahmood et al. (2018) demonstrated that using TEE for intraoperative monitoring allows for real-time assessment of pulmonary hemodynamics. This capability facilitates the early detection of hemodynamic instability, enabling timely interventions that can significantly improve patient outcomes. Continuous PASP monitoring provides valuable information that guides clinical decision-making during surgery.20
Price et al. (2021) emphasized the importance of individualized anesthetic protocols in patients with pulmonary hypertension, including early adjustment of positive end-expiratory pressure (PEEP), goal-directed fluid optimization, and tailored inotropic support based on hemodynamic phenotype. While such perioperative strategies are critical for maintaining cardiovascular stability, our study focused specifically on intraoperative monitoring of pulmonary artery systolic pressure (PASP) using transesophageal echocardiography (TEE), rather than implementing a comprehensive anesthetic management protocol.21
Regarding PASP measurement techniques, Tal Abu et al. (2022) demonstrated that right heart catheterization (RHC) remains the gold standard due to its ability to directly measure pulmonary pressures accurately. However, RHC’s invasive nature, need for specialized training, and lack of real-time, continuous monitoring led us to adopt a different approach.22
Cardiac magnetic resonance imaging (CMR) offers detailed anatomical and functional evaluation of the right ventricle and pulmonary circulation providing estimated measures of pulmonary artery pressure with high diagnostic and prognostic accuracy. However, its high cost, limited availability, and incompatibility with intraoperative environments (electromagnetic interference and lack of portability) render CMR unsuitable for continuous intraoperative monitoring. Consequently, our study adopted transesophageal echocardiography (TEE) for real-time pulmonary artery systolic pressure (PASP) measurement instead.23
Our study has several limitations. First, its single-center design and limited sample size reduce statistical power and may restrict the generalizability of findings to broader CABG populations. Second, while TEE-derived PASP measurements were standardized and performed by experienced operators, the technique remains operator-dependent and susceptible to inter-observer variability. TEE is also contraindicated in patients with esophageal pathologies (e.g., varices) or bleeding risks, potentially excluding high-risk subgroups.
Although PASP was measured post-anesthesia induction to establish baseline values, dynamic intraoperative hemodynamic fluctuations (e.g., fluid shifts, vasoactive medications) may have introduced variability. Furthermore, we did not account for confounders such as left ventricular function, pulmonary vascular resistance, or surgical technique Variability. The study’s focus on intraoperative PASP measurements also limited our ability to evaluate long-term clinical endpoints such as mortality or heart failure progression.
To overcome these gaps, future multicenter studies with larger cohorts should integrate multimodal monitoring (e.g., pulmonary artery catheters, advanced echocardiographic parameters) and longer follow-up periods. This would help establish validated PASP thresholds and optimize perioperative management for CABG patients with pulmonary hypertension.
6. Strengths of the Study
One of the key strengths of our study lies in its prospective design and the use of intraoperative TEE for real-time, dynamic assessment of pulmonary artery systolic pressure (PASP), allowing for accurate correlation with early postoperative outcomes. By focusing exclusively on elective on-pump coronary artery bypass grafting (CABG) patients, we minimized heterogeneity and isolated the specific impact of PASP in a uniform surgical population. Additionally, the stratification of patients based on PASP severity (mild, moderate, severe) enabled a nuanced analysis of morbidity risk across clinically relevant thresholds. The study also benefited from standardized anesthetic and surgical protocols, enhancing the internal validity and reliability of the findings. Collectively, these strengths support the robustness of our conclusions and their potential relevance to perioperative risk stratification and management.
Our study demonstrated that elevated intraoperative PASP measurements (≥ 35 mmHg) during elective on-pump coronary artery bypass grafting are associated with increased early postoperative complications, including prolonged mechanical ventilation (> 48h), re-intubation, greater need for inotropic support or intra-aortic balloon pump, and extended ICU (> 48 hr) and hospital stays (> 7 days). Additionally, DM and PVD were identified as independent risk factors for higher PASP. These findings underscore the prognostic value of PASP and highlight the importance of intraoperative hemodynamic monitoring to identify high-risk patients, enabling tailored management and potentially improving perioperative outcomes.
8. Ethical considerations
Ethical and administrative approvals from the Ain Shamas University, Faculty of Medicine, Ethical Committee were obtained, (under the number code FAMSU MD 14/2022 (FWA 000017585) 25/1/2022).
This study was registered with the Pan African Clinical Trial Registry (PACTR) under the identification number [PACTR202408668477631].
-Informed and written consent was obtained from all participants.
9. Availability of data
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
10. Conflict of interest
The authors declare that they have no competing interests.
11. Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
12. Acknowledgment:
The author wishes to thank the staff of the Anesthesia, Intensive Care, and Pain Management department, at Ain Shams University, and all the study participants for their great contribution.
13. Authors' contributions
AMFM: conduction of the study work.
NSN: manuscript editing.
SOAE: literature search.
AMMM: Data analysis
All authors have contributed intellectually to the manuscript and the manuscript has been read and approved by all the authors.
Authors affiliations:
- Assem Mohamed Fathy Mohamed, Assistant lecturer in Anesthesia, Intensive Care & Pain Management. Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: dr.asemelders9@gmail.com; {ORCID:0000-0001-7459-6853}
- Nermin Sadek Nasr, Professor of Anesthesia, Intensive Care & Pain Management, Faculty of Medicine, Ain Shams University Cairo, Egyp Egypt; Email: nerminnasr@yahoo.com
- Salwa Omar El Khattab Amin, Assistant Professor of Anesthesia, Intensive Care & Pain Management, Faculty of Medicine, Ain Shams University. Cairo, Egypt; Email: salwakhattab2011@hotmail.com
- Ahmad Moustafa Mohamed Mohamed, Lecturer of Anesthesia, Intensive Care & Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: drahmed.mostafa.mohamed@med.asu.edu.eg
ABSTRACT
Background & objective: Coronary artery disease (CAD) significantly impacts patients’ life and the surgical outcomes, necessitating effective preoperative risk assessment. Pulmonary artery systolic pressure (PASP) measured intraoperatively via transesophageal echocardiography (TEE) may serve as a critical indicator of postoperative complications in elective on-pump coronary artery bypass grafting (CABG).
We aimed to evaluate the association between elevated intraoperative PASP and postoperative early complications in patients undergoing elective on-pump CABG.
Methodology: This study involved 70 patients undergoing elective on-pump CABG, divided into two groups, based on intraoperative PASP measured using TEE: Group A: 35 patients with PASP ≥ 35 mmHg. Group B: 35 patients with PASP < 35 mmHg. The study analyzed PASP association with early postoperative complications, including respiratory failure, significant inotrope use, intra-aortic balloon pump (IABP) use, prolonged ICU stay (> 48 hours), and extended hospital stay (> 7 days).
Results: Postoperative respiratory failure (P < 0.009), significant inotrope use (P < 0.001), IABP use (P < 0.023), prolonged ICU stay (> 48 hours) (P < 0.001), and extended hospital stay (> 7 days) (P < 0.001) were significantly higher in Group A. No statistically significant differences between groups regarding postoperative anticoagulation, pacemaker dependence, arrhythmias, or myocardial infarction were noted (P = 0.145, 0.164, 0.55, and 0.743, respectively). When comparing PASP grades: severe PASP was associated with higher rates of postoperative respiratory failure, IABP use, prolonged ICU stays, and total hospital stays (P = 0.03, 0.01, 0.01, and < 0.001, respectively). Moderate pulmonary hypertension was associated with the most frequent use of significant inotropes (P = 0.03). No significant differences in postoperative anticoagulation, arrhythmias, myocardial infarction, or pacemaker dependence (P = 0.21, 0.87, 0.84, and 0.19, respectively) was noted.
Conclusion: Intraoperative PASP measurement using TEE is a valuable predictor of early postoperative complications and events in patients undergoing elective on-pump CABG.
Abbreviations: IAPB: intra-aortic balloon pump, IBP: invasive blood pressure, ICU: Intensive care unit, CABG: coronary artery bypass grafting, CAD: Coronary artery disease, PAH: Pulmonary arterial hypertension, PASP: Pulmonary artery systolic pressure, RAP: right atrial pressure, RHC: right heart catheterization, TEE: transesophageal echocardiography
Keywords: Pulmonary artery systolic pressure (PASP), Transesophageal echocardiography (TEE), On-pump coronary artery bypass graft (CABG).
Citation: Mohamed AMF, Nasr NS, Amin SOE, Mohamed AMM. Association of pulmonary artery systolic pressure measured by transesophageal echocardiography with early postoperative morbidity in patients undergoing elective on-pump coronary artery bypass grafting. Anaesth. pain intensive care 2025;29(5):317-327. DOI: 10.35975/apic.v29i5.2856
Received: March 24, 2025; Revised: April 18, 2025; Accepted: May 21, 2025
1. INTRODUCTION
Coronary artery disease (CAD) is the leading global cause of death and disability, responsible for ~16 million deaths and 278.2 million Disability Adjusted Life Years (DALYs) in 2019, disproportionately affecting low- and middle-income countries (28 of deaths, higher DALY rates vs. wealthy nations).1 In Egypt, CAD accounts for 68.4 of cardiovascular cases.2 Despite advances in treatments like coronary artery bypass grafting (CABG) and percutaneous coronary interventions (PCI), with 200,000 CABG and 965,000 PCI procedures performed in the U.S. in 2018 mortality remains high, especially in patients with comorbidities. Effective secondary prevention strategies, including optimized medical care, lifestyle changes, and cardiac rehabilitation, are critical to improving outcomes.3
Preoperative risk assessment is critical for optimizing surgical outcomes. The Society of Thoracic Surgeons (STS) score outperforms Euro SCORE II in predicting in-hospital mortality and complications.4 Pulmonary hypertension (PH), defined as systolic pressure ≥35 mmHg, is a key risk factor in cardiac surgery, often caused by left heart dysfunction or valvular disease. Doppler echocardiography offers a noninvasive method to monitor PH and stratify risk.5
After coronary artery bypass grafting (CABG), complications like myocardial edema and ischemia-reperfusion injury can lead to low cardiac output, heart attacks, or irregular heart rhythms. Neurological issues, such as stroke or coma (2-5 of cases) and mild cognitive impairment (up to 50 of patients), further contribute to longer hospital stays, higher costs, and increased mortality.6
The primary objective was to determine the association between elevated pulmonary artery systolic pressure (PASP ≥ 35 mmHg), measured intraoperatively by transesophageal echocardiography (TEE), and the incidence of early postoperative complications in patients undergoing elective on-pump coronary artery bypass grafting (CABG). The secondary objective was to identify and evaluate specific preoperative and intraoperative risk factors that significantly contribute to the elevation of PASP ≥ 35 mmHg, during elective on-pump CABG.
2. METHODOLOGY
This was a prospective and observational clinical study conducted on seventy patients of both sexes aged from 30 to 80 years, who were scheduled for isolated elective CABG on cardiopulmonary bypass (CPB). Enrolled consented patients were allocated into one of the following groups according to intraoperative PASP measured by TEE: Group A: 35 patients (PASP ≥ 35 mmHg; Pulmonary hypertension group), Additionally, individuals with increased PASP (≥ 35 mmHg) were divided into mild, moderate, and severe groups based on grades measured by TEE. Group B: contained 35 patients with PASP < 35 mmHg; Non pulmonary hypertension group.7
The study was conducted in the cardiac surgery operating rooms and cardiothoracic intensive care units (ICU) of Ain Shams University Hospitals from March 2022 to March 2024 It was approved by the Research Ethical Committee, Faculty of Medicine, Ain Shams University (approval number FWA 000017585, FMASU MD 14/2022). Informed and written consent was obtained from all participants. This study was registered with the Pan African Clinical Trial Registry (PACTR) under the identification number [PACTR202408668477631].
Patients with significant right ventricular outflow tract obstruction, contraindications to TEE, non-analyzable tricuspid regurgitation (TR) jets, scheduled for off-pump, urgent, redo, or combined valve CABG procedures, severe pulmonary diseases causing elevated pulmonary pressures such as chronic obstructive pulmonary disease (COPD), pulmonary fibrosis, obstructive sleep apnea, or those diagnosed with primary pulmonary arterial hypertension (PAH) were excluded from the study.
The sample size was determined using the PASS 11 program, with a confidence level set at 90 and a margin of error of +0.15. Previous research findings, particularly those by Subramanian et al. in 2020, highlighted a notable discrepancy in postoperative complications, specifically respiratory failure, between patients with high and low PASP. Based on these insights, a sample size of at least 35 patients with high PASP (≥ 35 mmHg) and 35 patients with low PASP (< 35 mmHg) was sufficient to achieve the study objectives.8
Simple random sampling was used to select the participants. All patients underwent comprehensive evaluation, including medical history review, clinical examination, functional status assessment (e.g., exercise tolerance, heart failure symptoms), and routine tests such as CBC, FBS, liver/kidney function panels, ECG, PT/aPTT, transthoracic echocardiography, cardiac catheterization, and chest X-ray. Volume overload was managed with diuretics to alleviate pulmonary congestion and reduce right ventricular strain, particularly in patients with right heart failure. Beta-blockers and calcium channel blockers were continued to control heart rate and lower myocardial oxygen demand, though careful monitoring was implemented to avoid adverse effects on right ventricular function.
Standard monitoring devices were attached to the patient. The patient was monitored intraoperatively, postoperatively, and in the ICU using the following: electrocardiogram (ECG), pulse oximetry (SpO2), non-invasive blood pressure (NIBP), invasive blood pressure (IBP) monitoring, intraoperative arterial pressure-based cardiac output measurement via arterial line insertion, central venous pressure (CVP), urine output, arterial blood gas (ABG) analysis, activated clotting time (ACT), nasopharyngeal core temperature measurement, and intraoperative TEE after the induction of anesthesia.
Intravenous access (IV) was obtained. All patients were premedicated with morphine sulphate 0.05 to 0.1 mg/kg IV one hour before surgery. After administration of IV midazolam (0.05mg/kg), both groups underwent standardized general anesthesia starting with pre-oxygenation with 100 oxygen before intubation. Smooth cardiac induction with fentanyl (5-10 µg/kg) slowly administered intravenously to both groups over 1-2 min, propofol (1-2 mg/kg) IV injected slowly over 30 sec guided by monitored invasive blood pressure, and pancuronium (0.07-0.1 mg/kg) was used to intubate the patients. Controlled mechanical ventilation (volume-controlled mode; tidal volume (TV) 6-8 mL/kg, respiratory rate 10-12 breath/min, PEEP 3-5 cmH2O) with 100 oxygen and inhalational agent isoflurane adjusted to (0.6 - 1.6) minimal alveolar concentration (MAC) with boluses of fentanyl (1µg/kg) IV and pancuronium (0.015-0.01 mg/kg) IV every 30-60 min, used for maintenance of anesthesia.
US-guided central line was secured in the right internal jugular vein and baseline CVP measurement was done. Unfractionated heparin (UFH) was used as an IV anticoagulant in a dose of 400 IU/kg, monitored with activated clotting time (ACT) to a target of 450-600 sec. Potassium-rich cardioplegia was used to stop the heart beatings. IV fluids (Ringer acetate), blood, and FFP were administered according to the standard practice. The cardiopulmonary bypass pump was managed by a dedicated certified clinical perfusionist who interacts and coordinates with the surgeon and the anesthetist.
No regional and/or fast-track anesthesia protocols were employed in these patients. All the relevant measurements were done 30 min after anesthesia induction, during critical phases of surgery; e.g., after initiation and during weaning from CPB, and immediately before the conclusion of the procedure. This protocol allowed for continuous or intermittent monitoring of PASP and cardiac function throughout the surgical process.
After insertion of the TEE probe, all measurements were obtained 30 min following induction. Preload and afterload were normalized through volume infusion and noradrenaline administration, starting with an initial dose of 8–12 µg/min IV infusion, subsequently titrated to a maintenance dose of 2–4 µg/min IV infusion to maintain filling pressures and invasive arterial blood pressure within ±20 of the pre-induction baseline levels. Anesthesia management and related procedures were independently carried out by an anesthesiologist not involved in the study.
All TEE assessments were conducted by a single experienced anesthesiologist using standardized equipment (TEE Probe: 6TC-RS KN100106-R, Vivid S6 N, General Electric Healthcare, Horten, Norway). Parameters were measured in triplicate and averaged, employing M-mode, 2D, and color Doppler imaging.
After weaning from CPB, heparin was reversed with protamine sulfate (1-1.5 mg per 100 units heparin; max 50 mg) under ACT monitoring. Post-op care included correcting electrolytes, acid-base balance, hypothermia, and coagulopathy, followed by sternal/skin closure. Patients were transferred to the ICU with portable monitoring, continued cardiac support, sedation, intubation, and manual ventilation via an oxygen-connected Ambu bag.
In the ICU, the patients were monitored with standard monitoring, mechanically ventilated and sedated. Clinical and laboratory monitoring of hemodynamic parameters, bleeding, and other complications was done and managed accordingly. Weaning from cardiac support with the improvement of the hemodynamic parameters, extubation, and weaning from mechanical ventilation was done when the criteria of weaning were met.
The study’s primary outcome focused on intraoperative measurement of PASP, calculated using the trans-tricuspid gradient, right atrial pressure (RAP), and tricuspid regurgitation (TR) jet velocity. These values were assessed in real-time during surgery via TEE and central venous pressure (CVP) monitoring. This measurement, alongside related TEE parameters, was correlated with postoperative outcomes.
PASP was calculated using the formula: PASP = 4 × (TR jet max velocity) ² + RAP.
TR jet velocity (m/s) was measured via continuous wave Doppler in mid-esophageal 4-chamber or bicaval views, ensuring a clear signal (alignment ≤ 20° and full Doppler envelope). Trans-tricuspid gradient: Derived using the Bernoulli equation (4 × [TR velocity] ²). RAP (mmHg): Obtained directly from CVP.
These TEE-derived values were primary predictors and allowed the categorization of patients into pulmonary hypertension (PASP ≥ 35 mmHg) and non-pulmonary hypertension (PASP < 35 mmHg) groups. The relationship between elevated PASP and postoperative outcomes was examined.
The secondary outcomes consisted of the early postoperative complications and morbidity indicators. Each of these complications was recorded and evaluated to assess their incidence and relationship to intraoperative PASP:
- Postoperative heparin anticoagulation was used as an indicator of suboptimal coronary revascularization (e.g., inadequate graft function or endarterectomy) and as a bridge to initiating antiplatelet therapy (typically started on the first postoperative morning).
- Postoperative arrhythmias: were recorded if sustained beyond 5 min in the postoperative period or required pharmacological or electrical therapy.
- Postoperative myocardial infarction (MI): Defined as the appearance in the ECG of a significant new Q wave (≥ 30 ms and ≥ 0.1 mV in two or more contiguous leads) and an echocardiographic image of disturbances in the segmental contractility in an area consistent with the ECG disturbances.
- Postoperative respiratory failure: Defined as the duration of mechanical ventilation more than 48 hours or re-intubation following cardiac surgery after successful extubation.
- The use of an intra-aortic balloon pump (IABP): A temporary mechanical support device for cardiac dysfunction, inserted via the femoral artery into the descending aorta. The balloon inflates during diastole (improving coronary blood flow) and deflates during systole (reducing cardiac workload), synchronized with the cardiac cycle to enhance heart function.
- The patient`s pacemaker dependency is confirmed when a patient’s intrinsic (natural) heart rate falls below a safe threshold or becomes ineffective at sustaining adequate cardiac output, necessitating continuous pacemaker support to maintain hemodynamic stability.
- Significant inotropes use: Defined as the requirement of inotropes beyond 24 hours of completion of surgery to maintain adequate cardiac output and BP.
- Prolonged ICU stay: defined to be > 48 hours postoperative.
- Prolonged total length of stay in hospital: defined to be more than seven days postoperative.
The collected data were revised, coded, tabulated, and then analyzed using Statistical Package for Social Science (SPSS) version 27.0. Quantitative data were expressed as mean ± SD or median (IQR), and qualitative data as frequencies and percentages. Statistical significance was assessed with independent-sample t-tests and Chi-square tests, with a 95% confidence interval and a significance (S) level set at P < 0.05 and highly significant (HS) at P < 0.01.
3. RESULTS
Regarding age, sex, ASA physical status, and BMI (kg/m2), there were no statistically significant differences between the two groups.
Baseline physiological and medical characteristics, including NYHA functional class, showed no statistically significant differences between two groups. However, diabetes mellitus (DM) and peripheral vascular disease (PVD) occurred significantly more frequently in Group A compared to Group B (P < 0.001 and < 0.010), as detailed in Table 1.
| Table 1: Comparative risk factors between groups | |||
| Risk factors for coronary artery disease (CAD) | Group A (PASP ≥ 35)
(n = 35) |
Group B (PASP < 35)
(n = 35) |
P-value |
| Family history of CAD | 6 (17) | 5 (14.3) | 0.743 |
| Smoking | 20 (57) | 13 (37) | 0.094 |
| DM | 29 (83) | 16 (45.7) | 0.001 |
| Opioid addiction | 0 (0) | 0 (0) | - |
| Hyperlipidemia | 6 (17) | 3 (8.5) | 0.284 |
| Hypertension | 18 (51.4) | 15 (43) | 0.473 |
| PVD | 6 (17) | 0 (0) | 0.010 |
| CVD | 3 (8.5) | 2 (5.7) | 0.643 |
| Recent MI | 26 (74) | 19 (54) | 0.081 |
| New York Heart Association (NYHA) Functional Class | |||
| 1 | 0 (0) | 0 (0) | - |
| 2 | 15 (42.8) | 22 (62.8) | 0.094 |
| 3 | 20 (57) | 13 (37) | 0.094 |
| 4 | 0 (0) | 1 (2.8) | 0.314 |
| Number of Coronary involvements | |||
| 1 | 0 (0) | 0 (0) | - |
| 2 | 6 (17) | 2 (5.7) | 0.133 |
| 3 | 29 (82.8) | 33 (94) | 0.133 |
| Data presented as n (%); P < 0.05 considered as significant | |||
Cardiac output (COP) and ejection fraction (EF) were significantly higher in Group B compared to Group A (P < 0.001 and P < 0.0076, respectively). Conversely, Group A exhibited markedly elevated E/e’ ratio, tricuspid regurgitation peak velocity (TR max), RAP, and PASP compared to Group B, all with highly significant differences (P < 0.001), as shown in Table 2.
| Table 2: Comparative intraoperative echo findings between groups | |||
| Intraoperative echo finding | Group A (PASP ≥ 35) (n = 35) | Group B (PASP < 35) (n = 35) | P-value t |
| COP (L/min) | 3.65 ± 0.65 | 4.79 ± 0.64 | < 0.001 |
| E/e’ ratio | 12.45 ± 1.29 | 9.00 ± 1.34 | < 0.001 |
| TR max (m/s) | 4.30 ± 0.56 | 1.96 ± 0.52 | < 0.001 |
| RAP (mmHg) | 18.37 ± 3.41 | 8.09 ± 2.02 | < 0.001 |
| PASP (mmHg) | 93.46 ± 19.05 | 24.56 ± 7.57 | < 0.001 |
| EF | 49.21 ± 5.47 | 53.41 ± 7.20 | 0.0076 |
| Data expressed as mean ± SD; t = student’s t-test; P < 0.05 considered as significant
COP: cardiac output, TR max: tricuspid regurgitation peak velocity, PASP: Pulmonary artery systolic pressure, RAP: right atrial pressure EF: ejection fraction, |
|||
No statistically significant differences were observed between Group A (PASP ≥ 35 mmHg) and Group B (PASP <35 mmHg) in postoperative anticoagulation (heparin infusion), pacemaker dependence, arrhythmias, or myocardial infarction (P > 0.05). However, Group A had significantly higher rates of postoperative respiratory failure (P < 0.009), inotrope use (P < 0.001), IABP requirement (P < 0.023), prolonged ICU stays (P < 0.009), and longer total hospital stays (P < 0.001) compared to Group B, as detailed in Table 3.
| Table 3: Comparison between groups as regards postoperative complications | |||
| Postoperative complications | Group A (PASP ≥ 35)
(n = 35) |
Group B (PASP < 35)
(n = 35) |
P-value ꭓ2 |
| The requirement of postoperative anticoagulation | 5 (13.4) | 10 (28.6) | 0.145 |
| Postoperative arrhythmias | 6 (17.1) | 8 (22.9) | 0.55 |
| Postoperative myocardial infarction | 6 (17.1) | 5 (13.4) | 0.743 |
| Postoperative respiratory failure | 12 (34.3) | 3 (8.6) | 0.009 |
| The use of intra-aortic balloon pump | 12 (34.3) | 4 (11.4) | 0.023 |
| Pacemaker dependence | 1 (2.9) | 4 (11.4) | 0.164 |
| Significant inotrope use | 31 (88.6) | 18 (51.4) | 0.001 |
| Prolonged ICU stay > 48 h | 23 (65.7) | 12 (34.3) | 0.009 |
| Total stay in hospital > 7 days | 29 (82.9) | 10 (28.6) | < 0.001 |
| Data presented as n (%); P < 0.05 considered as significant; X2 = chi-square. | |||
Comparison between groups of the PASP grades measured by TEE as regards postoperative complications: Postoperative anticoagulation, arrhythmias, and myocardial infarction rates were comparable across all PH severity groups (P = 0.21, 0.87, and 0.84, respectively). However, respiratory failure and IABP use were significantly higher in the severe PH group (P = 0.03 and P = 0.01, respectively). Inotrope dependency peaked in the Moderate PH group (P = 0.03), while prolonged ICU stays (> 48 hours) and hospital stays (>7 days) were most frequent in the severe PH group (P = 0.01 and P < 0.001), as mentioned in Figure I.

1: Bar graph between groups of the PASP grades as regards postoperative complications with a significant difference.
Pacemaker dependence showed no significant variation (P = 0.19), as detailed in Table 4.
| Table 4: Comparative PASP grades measured by TEE in groups as regards postoperative complications | |||||
| Postoperative Complications | Mild PH
(PASP 35-44) (n = 17) |
Moderate PH (PASP 45-59)
(n = 13) |
Severe PH (PASP ≥ 60)
(n = 5) |
Group B (PASP < 35)
(n = 35) |
P-value |
| Requirement of postoperative anticoagulation | 2 (11.8) | 3 (23.1) | 1 (20) | 10 (28.6) | 0.21 |
| Postoperative arrhythmias | 3 (17.6) | 3 (23.1) | 1 (20) | 8 (22.9) | 0.87 |
| Postoperative myocardial infarction | 2 (11.8) | 3 (23.1) | 1 (20) | 5 (13.4) | 0.84 |
| Postoperative respiratory failure | 4 (23.5) | 5 (38.5) | 3 (60) | 3 (8.6) | 0.03 |
| Use of intra-aortic balloon pump | 4 (23.5) | 4 (30.8) | 4 (80) | 4 (11.4) | 0.01 |
| Pacemaker dependence | 0 (0) | 1 (7.7) | 0 (0) | 4 (11.4) | 0.19 |
| Significant inotrope use | 12 (70.6) | 11 (84.6) | 4 (80) | 18 (51.4) | 0.03 |
| Prolonged ICU stay (> 48 h) | 9 (52.9) | 9 (69.2) | 5 (100) | 12 (34.3) | 0.01 |
| Prolonged total length of stay in hospital (> 7 days) | 11 (64.7) | 11 (84.6) | 5 (100) | 10 (28.6) | < 0.001 |
| Data presented as n (%); P < 0.05 considered as significant | |||||
4. DISCUSSION
Elevated pulmonary artery systolic pressure (PASP), as previously described, is a known marker of pulmonary hypertension and has been associated with adverse outcomes in cardiac surgery. Our findings reinforce this association, showing that patients with PASP ≥ 35 mmHg experienced higher rates of postoperative complications, highlighting the value of intraoperative PASP monitoring for risk stratification and management.
Our study revealed that postoperative respiratory failure requiring prolonged mechanical ventilation (≥48h) or re-intubation occurred in 12 patients (38.7) in Group A (elevated PASP) compared to 4 patients (10.3) in Group complications, highlighting the value of intraoperative PASP monitoring for risk stratification and management.
Our study revealed that postoperative respiratory failure requiring prolonged mechanical ventilation (≥48h) or re-intubation occurred in 12 patients (38.7) in Group A (elevated PASP) compared to 4 patients (10.3) in Group B (normal PASP), with statistical significance (p = 0.009). This underscores the hemodynamic burden posed by elevated PASP, likely through right ventricular dysfunction and compromised pulmonary perfusion. His inding aligns with Kanwar et al. (2019), who identified pulmonary hypertension as a risk factor for postoperative respiratory complications, attributed to impaired right ventricular function and intraoperative hemodynamic instability.9
Inotropic support for more than 24 hours was required in 16 patients (51.6) in Group A compared to 8 patients (20.5) in Group B (P = 0.001). IABP was needed in 7 patients (22.6) in Group A, versus 2 patients (5.1) in Group B (P = 0.01). These findings indicate that patients with elevated PASP are more likely to experience perioperative hemodynamic instability. This is consistent with Keranovic S et al. (2020), who reported an increased requirement for inotropes and mechanical support in patients with pulmonary hypertension during cardiac surgery.10
Prolonged ICU stay (> 48h) was recorded in 18 (58.1%) patients in Group A compared to 10 (25.6%) patients in Group B (P = 0.009). Similarly, hospital stays longer than 7 days occurred in 21 (67.7%) patients in Group A versus 12 (30.8%) patients in Group B (P < 0.001). These results are consistent with the work of Braksator et al. (2022), who found that pulmonary hypertension contributes to prolonged hospitalizations due to the complexity of postoperative management.11
In our study, patients with non-elevated PASP (Group B) demonstrated a numerically higher requirement for postoperative anticoagulation (P = 0.145). This contrasts with established predictors of thrombotic risk in cardiac surgery, where elevated pulmonary pressures typically correlate with increased anticoagulation demands. Differences in patient cohorts may explain this divergence, as our study focused exclusively on CABG patients. Moreover, this trend may reflect institutional protocols empirically anticoagulating normal PASP patients to optimize graft patency. Collectively, these findings reinforce guideline recommendations that anticoagulation strategies post-CABG should be individualized based on comprehensive patient-specific risks rather than isolated parameters like PASP.12
Postoperative arrhythmias, sustained beyond 5 minutes or requiring intervention, were more common in patients with elevated PASP (29) compared to those with normal PASP (15.4), though this was not statistically significant (p = 0.52). This is in line with previous research by Ruiz-Cano et al. (2020), which highlighted an elevated risk of supraventricular arrhythmias in pulmonary hypertension due to increased right atrial strain and electrical remodeling.13 Additionally, Huang et al. (2020) confirmed similar associations through systematic meta-analysis.14
According to this study, the requirement for postoperative anticoagulation was higher in the mild and moderate pulmonary hypertension (PH) groups compared to the severe PH group, although this difference was not statistically significant (P = 0.21). This finding aligns with current revascularization guidelines emphasizing that anticoagulation management should be tailored to comprehensive patient profiles rather than isolated parameters like PASP.12
The increased anticoagulation needs in mild and moderate PH may be attributed to better hemodynamic stability, as these patients typically exhibit improved right ventricular function and lower pressure overload, allowing for effective anticoagulation without the heightened bleeding risk associated with severe PH.15 In contrast, patients with severe PH experience significant strain on the right ventricle due to elevated pulmonary artery pressures, leading to right ventricular dysfunction and increased bleeding risk, which necessitates more cautious anticoagulation management.16
We found that postoperative arrhythmias were similarly prevalent across all PASP groups, with no statistically significant difference (P = 0.87). This aligns with Braman et al. (2020), who demonstrated that preoperative pulmonary hypertension does not independently predict postoperative atrial fibrillation in cardiac surgery patients after adjusting for surgical confounders (OR 1.1, 95% CI 0.9–1.4). The overall surgical stress and physiological changes during and after cardiac surgery can independently influence the risk of arrhythmias, regardless of baseline PASP levels. Intraoperative factors such as fluid shifts, electrolyte imbalances, and the use of cardiopulmonary bypass can contribute to the development of arrhythmias in all patients, masking potential differences related to PASP.17
Our study did not find significant differences in pacemaker dependence across groups (P = 0.19), which is in line with previous research suggesting that pacemaker needs are not strongly correlated with PASP grades in all groups.
Respiratory failure was significantly more frequent in the severe PH group (P = 0.03), supporting the findings of Kanwar et al. (2017), who reported a higher incidence of postoperative respiratory complications in patients with severe PH compared to those with mild or moderate PH. The increased risk of respiratory failure in these patients is attributed to underlying hemodynamic instability and impaired right ventricular function associated with elevated pulmonary artery pressures. This dysfunction can lead to inadequate pulmonary perfusion and gas exchange, resulting in hypoxemia and respiratory distress during the postoperative period.9
The study demonstrated that the need for significant inotropic support was highest in the moderate PH group (P = 0.03). This finding aligns with Rozado et al. (2019), who identified moderate pulmonary hypertension as the peak-risk subgroup for hemodynamic instability following cardiac surgery, characterized by significant fluctuations in blood pressure and cardiac output. Such instability is most critical in the early postoperative period, where fluid shifts, residual anesthetic effects, and surgical trauma compound cardiovascular strain. Unlike mild cases (where right ventricular adaptation is sufficient) or severe PH (with chronic compensatory remodeling), moderate PH induces a maladaptive RV stress response that significantly increases susceptibility to postoperative instability and inotrope dependence.18
Prolonged ICU stays (> 48 hours) and total hospital stays (>7 days) were highest in the severe PH group, with significant differences (P = 0.01 and P < 0.001, respectively). These results align with the literature, such as Braksator et al. (2022), that patients with severe PH may require more intensive monitoring, advanced pharmacologic support, and careful management of fluid balance and hemodynamics in the ICU, all of which contribute to longer recovery times extended hospital stays.11
The use of an IABP was significantly higher in the severe PH group (P = 0.01). This observation aligns with the pathophysiology of severe pulmonary hypertension, where elevated pulmonary vascular resistance imposes substantial strain on the right ventricle (RV), impairing its ability to effectively pump blood through the pulmonary circulation. This hemodynamic burden increases susceptibility to RV failure perioperatively, particularly under surgical stress. Consistent with this mechanism, Keranovic et al. (2020), reported that mechanical circulatory support (including IABP) is frequently required for critically ill cardiac surgery patients with compromised ventricular function, serving as a bridge to recovery in severe hemodynamic instability.10
Our study highlights the significant impact of different grades of PASP on postoperative outcomes for patients undergoing CABG. By illustrating the relationship between PASP levels and complications, our findings emphasize the importance of classifying patients based on their PASP levels. This strategy can improve clinical decision-making and enhance postoperative care, potentially leading to lower rates of complications and mortality.
Inuzuka et al. (2021) conducted a multicenter study involving 1,056 patients to explore the relationship between PASP, measured by Doppler echocardiography, and morbidity in patients with various heart conditions. They found a significant link between elevated PASP and increased causes of mortality and hospitalizations due to heart failure. This identifies PASP as an independent predictor of these outcomes after adjusting for other clinical factors. In contrast, our study specifically targeted the early perioperative period in patients undergoing CABG, which may result in differences in baseline characteristics and comorbidities when compared to the broader patient population examined by Inuzuka et al.19
Mahmood et al. (2018) demonstrated that using TEE for intraoperative monitoring allows for real-time assessment of pulmonary hemodynamics. This capability facilitates the early detection of hemodynamic instability, enabling timely interventions that can significantly improve patient outcomes. Continuous PASP monitoring provides valuable information that guides clinical decision-making during surgery.20
Price et al. (2021) emphasized the importance of individualized anesthetic protocols in patients with pulmonary hypertension, including early adjustment of positive end-expiratory pressure (PEEP), goal-directed fluid optimization, and tailored inotropic support based on hemodynamic phenotype. While such perioperative strategies are critical for maintaining cardiovascular stability, our study focused specifically on intraoperative monitoring of pulmonary artery systolic pressure (PASP) using transesophageal echocardiography (TEE), rather than implementing a comprehensive anesthetic management protocol.21
Regarding PASP measurement techniques, Tal Abu et al. (2022) demonstrated that right heart catheterization (RHC) remains the gold standard due to its ability to directly measure pulmonary pressures accurately. However, RHC’s invasive nature, need for specialized training, and lack of real-time, continuous monitoring led us to adopt a different approach.22
Cardiac magnetic resonance imaging (CMR) offers detailed anatomical and functional evaluation of the right ventricle and pulmonary circulation providing estimated measures of pulmonary artery pressure with high diagnostic and prognostic accuracy. However, its high cost, limited availability, and incompatibility with intraoperative environments (electromagnetic interference and lack of portability) render CMR unsuitable for continuous intraoperative monitoring. Consequently, our study adopted transesophageal echocardiography (TEE) for real-time pulmonary artery systolic pressure (PASP) measurement instead.23
5. LIMITATIONS
Our study has several limitations. First, its single-center design and limited sample size reduce statistical power and may restrict the generalizability of findings to broader CABG populations. Second, while TEE-derived PASP measurements were standardized and performed by experienced operators, the technique remains operator-dependent and susceptible to inter-observer variability. TEE is also contraindicated in patients with esophageal pathologies (e.g., varices) or bleeding risks, potentially excluding high-risk subgroups.
Although PASP was measured post-anesthesia induction to establish baseline values, dynamic intraoperative hemodynamic fluctuations (e.g., fluid shifts, vasoactive medications) may have introduced variability. Furthermore, we did not account for confounders such as left ventricular function, pulmonary vascular resistance, or surgical technique Variability. The study’s focus on intraoperative PASP measurements also limited our ability to evaluate long-term clinical endpoints such as mortality or heart failure progression.
To overcome these gaps, future multicenter studies with larger cohorts should integrate multimodal monitoring (e.g., pulmonary artery catheters, advanced echocardiographic parameters) and longer follow-up periods. This would help establish validated PASP thresholds and optimize perioperative management for CABG patients with pulmonary hypertension.
6. Strengths of the Study
One of the key strengths of our study lies in its prospective design and the use of intraoperative TEE for real-time, dynamic assessment of pulmonary artery systolic pressure (PASP), allowing for accurate correlation with early postoperative outcomes. By focusing exclusively on elective on-pump coronary artery bypass grafting (CABG) patients, we minimized heterogeneity and isolated the specific impact of PASP in a uniform surgical population. Additionally, the stratification of patients based on PASP severity (mild, moderate, severe) enabled a nuanced analysis of morbidity risk across clinically relevant thresholds. The study also benefited from standardized anesthetic and surgical protocols, enhancing the internal validity and reliability of the findings. Collectively, these strengths support the robustness of our conclusions and their potential relevance to perioperative risk stratification and management.
7. CONCLUSION
Our study demonstrated that elevated intraoperative PASP measurements (≥ 35 mmHg) during elective on-pump coronary artery bypass grafting are associated with increased early postoperative complications, including prolonged mechanical ventilation (> 48h), re-intubation, greater need for inotropic support or intra-aortic balloon pump, and extended ICU (> 48 hr) and hospital stays (> 7 days). Additionally, DM and PVD were identified as independent risk factors for higher PASP. These findings underscore the prognostic value of PASP and highlight the importance of intraoperative hemodynamic monitoring to identify high-risk patients, enabling tailored management and potentially improving perioperative outcomes.
8. Ethical considerations
Ethical and administrative approvals from the Ain Shamas University, Faculty of Medicine, Ethical Committee were obtained, (under the number code FAMSU MD 14/2022 (FWA 000017585) 25/1/2022).
This study was registered with the Pan African Clinical Trial Registry (PACTR) under the identification number [PACTR202408668477631].
-Informed and written consent was obtained from all participants.
9. Availability of data
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
10. Conflict of interest
The authors declare that they have no competing interests.
11. Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
12. Acknowledgment:
The author wishes to thank the staff of the Anesthesia, Intensive Care, and Pain Management department, at Ain Shams University, and all the study participants for their great contribution.
13. Authors' contributions
AMFM: conduction of the study work.
NSN: manuscript editing.
SOAE: literature search.
AMMM: Data analysis
All authors have contributed intellectually to the manuscript and the manuscript has been read and approved by all the authors.
14. REFERENCES
- GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020 Oct 17;396(10258):1204-1222. PMID: 33069326 PMCID: PMC7567026 DOI: 1016/S0140-6736(20)30925-9
- Awadallah S, Fekry A, El Deeb A, Elghazaly H, Elgazzar H, Basyuni M, et al. Atherosclerotic cardiovascular diseases in Middle Delta of Egypt: a systematic analysis of risk factors associated with the rising burden of the disease. Glob Heart. 2025 Feb 3;20(1):11. PMID: 39925839 PMCID: PMC11804180 DOI: 5334/gh.1395
- Virani SS, Alonso A, Aparicio HJ, Benjamin EJ, Bittencourt MS, Callaway CW, et al. Heart disease and stroke statistics—2021 update: A report from the American Heart Association. Circulation. 2021;143(8):e254–e743. DOI:1161/CIR.0000000000000950. PMID:33501848.
- Ad N, Holmes SD, Patel J, Pritchard G, Shuman DJ, Halpin L, et al. Comparison of EuroSCORE II, original EuroSCORE, and the Society of Thoracic Surgeons risk score in cardiac surgery patients. Ann Thorac Surg. 2016;102(2):573–9. DOI: 1016/j.athoracsur.2016.01.105 . PMID: 27112651
- Gozdzik A, Czechowski P, Zielinski K, Zembala M, Knapczyk M, Wiechetek A, et al. Echocardiographic probability of pulmonary hypertension in cardiac surgery patients—Occurrence and association with respiratory adverse events—An observational prospective single-center study. J Clin Med. 2022 Sep 28;11(19):5749. PMID: 36233617 PMCID: PMC9573503 DOI: 3390/jcm11195749
- Thielmann M, Sharma V, Al-Attar N, Bulluck H, Bisleri G, Bunge JJH, et al. ESC Joint Working Groups on Cardiovascular Surgery and the Cellular Biology of the Heart Position Paper: Peri-operative myocardial injury and infarction in patients undergoing coronary artery bypass graft surgery. Eur Heart J. 2017 Jul 25;38(30):2392-2407. PMID: 28821170 PMCID: PMC5808635 DOI: 1093/eurheartj/ehx383
- Galiè N, Humbert M, Vachiery JL, Gibbs S, Lang I, Torbicki A, et al. The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Heart J. 2016 Jan 1;37(1):67-119.. PMID: 26320113 DOI: 1093/eurheartj/ehv317
- Subramanian H, Parida S, Thangaswamy CR, Subramaniam K, Kumar A, Kapoor PM. Relationship between transoesophageal echocardiography-derived pulmonary artery systolic pressure measurements and early morbidity in patients undergoing coronary artery bypass grafting. Ann Card Anaesth. 2020;23(4):453-9. PMID: 33109803 PMCID: PMC7879900 DOI: 4103/aca.ACA_161_19
- Kanwar M, Tedford RJ, Brensinger CM, Halushka MK, Shah SJ, Hassoun PM, et al. Postoperative pulmonary complications in contemporary cohort of patients with pulmonary hypertension. Bosn J Basic Med Sci. 2019;19(2):231-237. PMID: 31301223 PMCID: PMC6868488 DOI: 17305/bjbms.2019.4332
- Keranovic S, Salihovic E, Zrnanovic D, Prelic M, Piric A, Kikanovic T. Inotropic and Mechanical Support of Critically Ill Patient after Cardiac Surgery. Med Arch. 2020 Jun;74(3):236–239. PMID: 32801443 PMCID: PMC7406010 DOI: 5455/medarh.2020.74.236-239
- Braksator M, Jachymek M, Witkiewicz K, Piątek P, Witkiewicz W, Peregud-Pogorzelska M, et al. Echocardiographic Probability of Pulmonary Hypertension in Cardiac Surgery Patients—Occurrence and Association with Respiratory Adverse Events—An Observational Prospective Single‑Center Study. J Clin Med. 2022;11(19):5749. PMID: 36233617 PMCID: PMC9573503 DOI: 3390/jcm11195749
- Lawton JS, Tamis-Holland JE, Bangalore S, et al. 2021 ACC/AHA/SCAI Guideline for Coronary Artery Revascularization: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145(3):e18-e114. DOI:1161/CIR.0000000000001038. PMID: 34882435.
- Ruiz-Cano MJ, Gonzalez-Mansilla A, Escribano P, Delgado J, Arribas F, Torres J, et al. Clinical implications of supraventricular arrhythmias in patients with severe pulmonary arterial hypertension. Int J Cardiol. 2021 Jan 1;322:190-198. DOI:1016/j.ijcard.2020.08.058. PMID: 32827597.
- Huang W, Chai Y, Zhuang Y. Postoperative outcomes and arrhythmias in patients with pulmonary hypertension: a systematic review and meta-analysis. Heart Lung. 2020;49(5):700-707. DOI: 1016/j.hrtlng.2020.04.018. PMID: 32417076.
- McLaughlin VV, Archer SL, Badesch DB, Barst RJ, Farber HW, Lindner JR, et al. ACCF/AHA 2009 expert consensus document on pulmonary hypertension: a report of the American College of Cardiology Foundation Task Force on Expert Consensus Documents and the American Heart Association developed in collaboration with the American College of Chest Physicians; American Thoracic Society, Inc.; and the Pulmonary Hypertension Association. J Am Coll Cardiol. 2009;53(17):1573-619. DOI: 11016/j.jacc.2009.01.004. PMID: 19389575.
- Klinger JR, Badesch DB, Frantz RP. Diagnosis and management of pulmonary hypertension: a review. J Am Coll Cardiol. 2018;71(25):2980-2996. DOI: 1016/j.jacc.2018.04.006. PMID: 29929630.
- Braman JG, Wilkins KT, Halperin JL. Atrial fibrillation following cardiac surgery: an update. Curr Cardiol Rep. 2020;22(4):37. DOI:1007/s11886-020-01284-3. PMID: 32185565.
- Ogawa S, Maeda M, Hosoba S, Goto Y, Okawa Y. Management of Bilateral Partial Anomalous Pulmonary Venous Connection. Ann Thorac Surg. 2019 May;107(5):e309-e310.. DOI: 1016/j.athoracsur.2018.09.047 PMID: 30403977.
- Russcher A, Verdonschot J, Molenaar-de Backer MWA, Heymans SRB, Kroes ACM, Zaaijer HL. Parvovirus B19 DNA detectable in hearts of patients with dilated cardiomyopathy, but absent or inactive in blood. ESC Heart Fail. 2021 Aug;8(4):2723-2730.. PMID: 33931945 PMCID: PMC8318422 DOI: 1002/ehf2.13341
- Augustine DX, Coates-Bradshaw LD, Willis J, Harkness A, Ring L, Grapsa J, et al. Echocardiographic assessment of pulmonary hypertension: a guideline protocol from the British Society of Echocardiography. Echo Res Pract. 2018 Sep;5(3):G11-G24. PMCID: PMC6055509 PMID: 30012832 DOI: 1530/ERP-17-0071
- Price LC, Martinez G, Brame A, Pickworth T, Samaranayake C, Alexander D, et al. Perioperative management of patients with pulmonary hypertension undergoing non-cardiothoracic, non-obstetric surgery: a systematic review and expert consensus statement. Br J Anaesth. 2021 Apr;126(4):774–790. DOI: 1016/j.bja.2021.01.005 . PMID: 33612249
- Abu T, Levi A, Hasdai D, Kramer MR, Bental T, Bdolah-Abram T, et al. Preoperative evaluation of pulmonary hypertension in lung transplant candidates: echocardiography versus right heart catheterization. BMC Cardiovasc Disord. 2022;22:53. PMID: 35172724 PMCID: PMC8851783 DOI: 1186/s12872-022-02495-y
- Banjade A, Subedi A, Acharya S, Itani A, Sharma M, Kassam N, et al. The role of cardiac MRI in pulmonary hypertension – is it still an underutilized tool? Open Respir Med J. 2024;18:e18743064288565. PMID: 39136034 PMCID: PMC11318157 DOI: 2174/0118743064288565240515115239