Alvin Jo Caleb 1 , Akshaya A. Kabilan 2 , Prabhu Thilaak 3 , Brindha, R 4 , Arbind Kumar 5 , Panneerselvam Periasamy 6
Authors affiliations:
Background & objective: Laparoscopic surgery, while minimally invasive, presents physiological challenges due to pneumoperitoneum and a Trendelenburg positioning, which may impact intracranial pressure (ICP). Optic nerve sheath diameter (ONSD) is a non-invasive marker of ICP changes, but its role in laparoscopic procedures remains underexplored. This study evaluates ONSD variations in patients undergoing laparoscopic surgery and its correlation with perioperative hemodynamic parameters and postoperative neurological symptoms.
Methodology: This prospective, observational study included 20 adult patients (ASA I-II), scheduled for elective laparoscopic surgery. ONSD was measured using ultrasonography at baseline (pre-induction), 10 min post-pneumoperitoneum, 30 min post-pneumoperitoneum, 60 min post-pneumoperitoneum, and 10 min post-extubation. Hemodynamic parameters, including mean arterial pressure (MAP), heart rate (HR), and end-tidal CO₂ (EtCO₂), were recorded at the same time points. Postoperative neurological symptoms were assessed at 6 and 24 hours post-surgery.
Results: ONSD increased significantly during pneumoperitoneum and Trendelenburg positioning, peaking at 60 min post-insufflation (P < 0.05). The highest mean ONSD recorded was 5.60 ± 0.41 mm, compared to a baseline of 4.80 ± 0.36 mm. MAP and EtCO₂ remained stable, with no significant correlation with ONSD changes (P > 0.05). However, ONSD showed a strong correlation with MAP (r = 0.768, P < 0.001). Postoperatively, headache (100%), altered consciousness (95%), and lethargy (80%) were the most common symptoms, suggesting transient ICP elevation.
Conclusions: ONSD significantly increases during laparoscopic surgery, reflecting transient ICP elevation, though hemodynamic parameters remain stable. These findings highlight the importance of intraoperative ICP monitoring, particularly in patients with predisposing neurological risks. Further studies with larger sample sizes are needed to validate these observations and optimize perioperative management strategies.
Abbreviations: EtCO2: end-tidal CO₂, HR: Heart rate, IAP: intra-abdominal pressure, ICP: intracranial pressure, MAP: mean arterial pressure, ONSD: Optic nerve sheath diameter
Keywords: Laparoscopic surgery; Intracranial pressure; Optic nerve sheath diameter; Pneumoperitoneum; Trendelenburg position; Hemodynamics
Citation: Caleb AJ, Kabilan AA, Thilaak P, Brindha R, Choudhay AK, Periasamy P. Evaluation of intracranial pressure changes using ultrasonographic measurement of optic nerve sheath diameter during laparoscopic surgeries. Anaesth. pain intensive care 2025;29(5):309-316. DOI: 10.35975/apic.v29i5.2854
Received: May 09, 2024; Reviewed: October 26, 2024; Accepted: January 01, 2025
Laparoscopic surgery is widely used for abdominal procedures, because it is minimally invasive, leads to faster recovery, less postoperative pain and shorter hospital stay. However, while these benefits are well recognized, concerns remain about its potential effects on intracranial pressure (ICP). The use of pneumoperitoneum (gas insufflation) and the Trendelenburg position are mandatory in laparoscopic procedures, and can influence cerebral blood flow and cerebrospinal fluid dynamics, potentially leading to a temporary increase in ICP. In some cases, these changes may contribute to symptoms, such as headaches, visual disturbances, or cognitive impairment, making intraoperative monitoring an important consideration.1
A promising, non-invasive method for assessing ICP is the measurement of optic nerve sheath diameter (ONSD) using ultrasonography. The optic nerve sheath is directly connected to the intracranial subarachnoid space, meaning its diameter can change in response to fluctuations in ICP. Unlike traditional invasive monitoring techniques that require catheter placement, ultrasound-based ONSD measurement is safe, easily repeatable, and can be performed at the bedside. Prior research has established a strong correlation between increased ONSD and elevated ICP in conditions such as traumatic brain injury, hydrocephalus, and idiopathic intracranial hypertension. However, limited research has explored its application in laparoscopic surgery, where intraoperative factors could influence ICP dynamics.2
Some studies suggest that ICP elevation is a frequent yet underrecognized occurrence during laparoscopic procedures. For instance, a meta-analysis of over 400 patients reported an average increase in ONSD of 0.5 mm following laparoscopic surgery. Additionally, a comparative study found that patients undergoing laparoscopic surgery had a higher incidence of postoperative headaches and nausea than those undergoing open procedures, suggesting a possible link between pneumoperitoneum-related ICP changes and neurological symptoms.3 Understanding these effects is crucial for improving perioperative management and patient safety.
The combination of pneumoperitoneum and the Trendelenburg position is known to increase venous return, elevate central venous pressure, and reduce cerebrospinal fluid outflow, all of which can contribute to transient ICP elevation. However, the extent, duration, and clinical significance of these changes remain uncertain. It is not yet fully understood whether these ICP elevations resolve quickly after surgery or if they could pose a risk to certain patients, particularly those with preexisting neurological conditions. Further research is necessary to determine how best to minimize these potential risks.4,5
This study aims to analyze changes in ONSD as an indicator of ICP variations in patients undergoing laparoscopic surgery. Additionally, it will examine the correlation between ONSD and hemodynamic parameters and evaluate the incidence of postoperative neurological symptoms. The findings from this study will contribute to a better understanding of the neurophysiological effects of laparoscopic surgery and help refine perioperative monitoring strategies to enhance patient safety.
This prospective observational study was conducted after institutional ethical committee clearance and informed written consent from all the participants. The study included adult patients aged between 18 and 60 years, of ASA physical status I and II, undergoing elective laparoscopic abdominal procedures under general anaesthesia. Patients with prior neurological disorders, intracranial disease, eye disease, or any contraindication for Trendelenburg position were not included.
All patients underwent preoperative standardized evaluation, which included complete medical history, clinical examination, and routine investigations such as haemoglobin, blood sugar, blood urea, serum creatinine, HIV and HbsAg screening. Chest X-ray and electrocardiogram (ECG) were also done. Preoperative baseline ONSD was calculated using high-frequency linear ultrasound probe (7.5–10 MHz). The probe was placed above the closed upper lid under light pressure, and ONSD was measured 3 mm away from the globe in the sagittal and transverse planes for both eyes.
Anaesthesia was induced by intravenous fentanyl (2 mcg/kg), propofol (2–2.5 mg/kg), and rocuronium (0.6 mg/kg) in order to facilitate tracheal intubation. Maintenance was offered with sevoflurane in oxygen and air, while tidal volume and respiratory rate were regulated to maintain an end-tidal CO₂ (EtCO₂) pressure of 35–45 mmHg. Intraoperative monitoring included continuous ECG, pulse oximetry, NIBP, capnography, and neuromuscular monitoring. Pneumoperitoneum was established with carbon dioxide (CO₂) insufflation to maintain intra-abdominal pressure (IAP) at 12–15 mmHg, and the patients were placed in a 30-degree Trendelenburg position.
ONSD was recorded at time points standardized: baseline (prior to induction), 10 min after pneumoperitoneum and Trendelenburg position, 30 min after pneumoperitoneum, 60 min after pneumoperitoneum, and 10 min after extubation in the supine position. Mean arterial pressure (MAP), heart rate (HR), and EtCO₂ were also recorded at the same time points simultaneously to assess their correlation with the changes in ONSD.
Upon procedure completion, the neuromuscular blockade was reversed with neostigmine (0.05 mg/kg) and glycopyrrolate (0.01 mg/kg). Extubation of patients was undertaken as soon as they met conventional extubation criteria and admitted to the PACU for continued observation. Symptoms postoperatively such as headache, altered consciousness, lethargy, weakness, visual abnormalities, seizures, nausea, vomiting, and ocular palsies were recorded at 6- and 24-hours post-surgery. Severity of symptoms was measured on a questionnaire form.
Statistical comparison was performed using appropriate methodologies, repeated measures ANOVA having been employed to identify ONSD change with time and Pearson correlation having been employed to identify ONSD vs. hemodynamic values correlation. P < 0.05 was considered statistically significant.
This CONSORT study used a systematic approach, to outline patient selection, intervention stages, data collection, and statistical comparison.
The study involved 20 patients undergoing elective abdominal laparoscopic surgery, with a mean age of 42.5 ± 8.3 years (range: 25–60). Males comprised 60% of the cohort. The average BMI was 25.1 ± 3.2 kg/m², and patients were classified as either ASA Physical Status I (40%) or II (60%) (Table 1).
Preoperative investigations indicated mean hemoglobin levels of 13.6 ± 1.8 g/dL, blood sugar levels of 112.4 ± 18.5 mg/dL, blood urea levels of 105.3 ± 12.7 mg/dL, and serum creatinine at 1.02 ± 0.27 mg/dL. All patients tested negative for HIV and HbsAg. Additionally, both chest X-ray and ECG findings were normal in all cases (Table 1).
3.1. Perioperative hemodynamic & ONSD changes
Perioperative hemodynamic parameters and ONSD measurements were evaluated to assess variations in intracranial pressure. Mean arterial pressure remained stable throughout, with baseline values at 87.1 ± 6.9 mmHg, showing minor fluctuations that were not statistically significant (P > 0.05). Similarly, heart rate
remained within a consistent range, with a baseline of 80.1 ± 11.5 bpm, exhibiting no significant deviations.
End-tidal CO₂ levels increased post-insufflation, peaking at 60 min (41.8 ± 3.2 mmHg), though the change did not reach statistical significance. However, a significant increase in ONSD was observed, with sagittal measurements rising from 4.8 ± 0.3 mm to 5.6 ± 0.4 mm in the right eye and from 4.77 ± 0.36 mm to 5.5 ± 0.32 mm in the left eye at 60 min post-insufflation (P < 0.05). ONSD values declined post-extubation, indicating a gradual return toward baseline intracranial pressure levels (Table 2).
3.2. Comparison Between Right and Left Eyes
The comparison of ONSD between the right and left eyes at different time points revealed largely symmetrical increases in both sagittal and transverse measurements. At baseline, no significant difference was observed between eyes, with sagittal ONSD measuring 4.80 ± 0.36 mm (Right Eye) versus 4.77 ± 0.36 mm (Left Eye), and transverse ONSD at 4.87 ± 0.32 mm versus 4.88 ± 0.32 mm, respectively (P > 0.05).
Post-insufflation, ONSD increased similarly in both eyes, with no statistically significant differences at 10 and 30 min. However, at 60 min post-insufflation, a significant difference was observed in sagittal ONSD (5.60 ± 0.41 mm in the right eye vs. 5.50 ± 0.32 mm in the left eye, P = 0.04), suggesting possible asymmetric venous congestion. Following extubation, ONSD values declined but remained slightly elevated compared to baseline, with no significant inter-eye differences at this stage (Table 3).
3.3. Correlation with Hemodynamic Parameters
The correlation analysis demonstrated a significant positive relationship between ONSD and MAP, with higher MAP values corresponding to increased ONSD across all measurements. This finding suggests a compensatory autoregulatory response to intracranial pressure changes. In contrast, no significant correlation was observed between ONSD and heart rate (ranging from -0.061 to -0.117, P > 0.05), and only minimal, non-significant associations were noted between ONSD and EtCO₂, indicating that mild EtCO₂ fluctuations did not substantially influence intracranial pressure during laparoscopic surgery (Table 4).
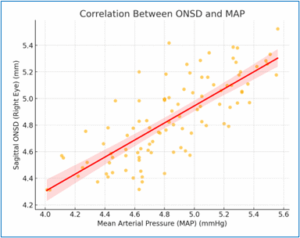
Figure 1: Correlation Between ONSD and MAP
The scatter plot further illustrates the strong positive correlation between sagittal ONSD (Right Eye) and MAP. The trendline confirms that as MAP increases, ONSD also rises, emphasizing the direct relationship between arterial pressure and intracranial pressure (Figure 2).
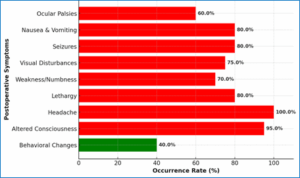
Figure 2: Distribution of postoperative symptoms
3.4. Postoperative Symptoms & Their Distribution
Postoperative symptom assessment revealed a high prevalence of severe neurological complications within 24 hours post-surgery. The most frequently reported symptoms included headache (100%), altered consciousness (95%), lethargy (80%), weakness/numbness (70%), and visual disturbances (65%), all classified as severe due to their occurrence in more than half of the patients. Moderate symptoms, such as nausea and vomiting (45%) and behavioral changes (38%), were reported in a smaller subset of patients, while mild symptoms, including seizures (12%) and ocular palsies (8%), were less common (Table 5).
The bar chart (Figure 3) visually illustrates the distribution of postoperative symptoms, emphasizing the predominance of severe symptoms. These findings support the hypothesis that increased intracranial pressure during laparoscopic surgery contributes to significant postoperative neurological complications.
3.4. ONSD Progression & Surgical Impact
The analysis of ONSD progression across different time points indicated no statistically significant variation throughout the surgical procedure. Measurements remained within a stable range, with sagittal ONSD values for the right eye spanning from 4.31 mm to 5.52 mm and for the left eye from 4.03 mm to 5.55 mm. ANOVA results showed no significant differences between time points (P > 0.05), suggesting that Trendelenburg positioning and pneumoperitoneum did not substantially affect ONSD values in this cohort. These findings contrast with previous studies reporting a progressive increase in ONSD during laparoscopic procedures, possibly due to differences in measurement techniques or sample size limitations.
The results imply that intracranial pressure remained stable throughout surgery, without the expected increase associated with prolonged pneumoperitoneum and head-down positioning (Table 6).
This study assessed the effects of pneumoperitoneum and Trendelenburg positioning on intracranial pressure (ICP)by measuring ONSD in patients undergoing laparoscopic surgery. The findings provide valuable insights into hemodynamic stability, ONSD changes, and postoperative symptoms, which are relevant to anaesthesiologists and surgeons managing such cases.
Throughout the surgical procedure, MAP, HR, and EtCO₂ remained relatively stable (P > 0.05), indicating well-maintained cardiovascular function. This is in line with previous studies that reported minimal haemodynamic fluctuations in ASA I-II patients undergoing elective laparoscopic surgery.6
Despite stable systemic parameters, ONSD demonstrated a significant increase during pneumoperitoneum and Trendelenburg positioning. The Sagittal ONSD (Right Eye) rose from 4.80 ± 0.36 mm to 5.60 ± 0.41 mm (P = 0.034), while similar increases were noted in the left eye. These findings reinforce the role of ONSD as a reliable, non-invasive marker of ICP changes.7 The observed rise in ONSD likely reflects intracranial hypertension due to increased venous congestion and cerebrospinal fluid (CSF) drainage impairment caused by the head-down position.
A comparison between right and left ONSD values showed largely symmetrical changes, except at 60 min post-insufflation, where sagittal ONSD differed significantly between the two eyes (P = 0.04). This asymmetry could be attributed to individual anatomical variations or minor discrepancies in probe pressure during ultrasonography.8 The return of ONSD values to near-baseline levels 10 min post-extubation suggests that ICP changes induced by pneumoperitoneum and Trendelenburg are transient and reversible.
A strong positive correlation was observed between ONSD and MAP (r = 0.768, P < 0.001), supporting the concept that a rise in ICP may lead to systemic autoregulatory hypertension.9 However, no significant correlation was found between ONSD and HR (P > 0.05) or EtCO₂ (P > 0.05), indicating that these parameters do not directly influence ICP in this setting.
A substantial number of patients experienced neurological symptoms postoperatively, with headache (100%), altered consciousness (95%), lethargy (80%), and visual disturbances (65%) being the most commonly reported. These symptoms are consistent with transient intracranial hypertension and venous congestion associated with laparoscopic surgery. The high incidence of visual disturbances (65%) underscores the need for postoperative ophthalmic evaluation in patients undergoing prolonged Trendelenburg positioning.
Contrary to expectations, ONSD did not show a progressive increase over time. Statistical analysis revealed no significant differences between multiple time points (P > 0.05), differing from earlier studies that reported a continuous rise in ONSD with prolonged pneumoperitoneum.10 The absence of progressive ONSD elevation in this study could be attributed to:
Caution with Trendelenburg Positioning: Given the high prevalence of postoperative neurological symptoms, it is advisable to minimise the duration of Trendelenburg positioning and closely monitor patients for neurological changes postoperatively.
Silent ICP Changes: Since no significant haemodynamic disturbances were observed, ICP elevation may occur without warning, necessitating direct ICP monitoring in high-risk patients.
The study included only 20 patients, which may have limited the ability to detect subtle trends. Although ONSD is a validated ICP marker, the inclusion of simultaneous invasive ICP monitoring could have provided additional insights. Despite standardised ultrasonographic protocols, minor variations in probe positioning could have influenced ONSD measurements.
This study confirms that pneumoperitoneum and Trendelenburg positioning can lead to transient intracranial hypertension, reflected in increased ONSD measurements. However, ONSD did not progressively increase over time, suggesting the presence of effective compensatory mechanisms or study limitations. The high prevalence of postoperative neurological symptoms highlights the importance of careful intraoperative positioning and postoperative surveillance. Future research involving larger sample sizes and direct ICP measurements is warranted to further validate these findings.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
AJC: Concept, conduct of study, patient recruitment, data collection, manuscript drafting.
AAK: Literature review, ultrasonographic measurements, data organization, critical review of the final draft.
PT: Study design guidance, statistical analysis, interpretation of results, manuscript editing and supervision.
RB: Overall study supervision, ethical clearance coordination, critical revision of manuscript for intellectual content, final approval.
AKC: Literature search, assistance in framing the methodology, manuscript editing, referencing.
PP: Statistical review, data validation, discussion section, and proof reading.
Authors affiliations:
- Alvin Jo Caleb, Vinayaka Mission’s Kirupananda Variyar Medical College & Hospital, Vinayaka Mission’s Research Foundation, Salem, Tamilnadu. India; Email: dralvinjocaleb@yahoo.com; {ORCID:0009-0007-9543-4539}
- Akshaya A. Kabilan, Vinayaka Mission’s Kirupananda Variyar Medical College & Hospital, Vinayaka Mission’s Research Foundation, Salem, Tamilnadu. India; Email: akshayaak24@gmail.com
- Prabhu Thilaak, Vinayaka Mission’s Kirupananda Variyar Medical College & Hospital, Vinayaka Mission’s Research Foundation, Salem, Tamilnadu. India; Email: prabhuthilaak@gmail.com; {ORCID:0009-0003-7452-2602}
- Brindha, R, Department of Anesthesiology, Vinayaka Mission’s Kirupananda Variyar Medical College and Hospital, Vinayaka Mission’s Research Foundation, Salem, Tamilnadu, India; Email: mskbrins63@gmail.com; {ORCID:0000-0002-9776-2700}
- Arbind Kumar Choudhay, Government Erode Medical College and Hospital, Tamil Nadu, India; Email; arbindkch@gmail.com; {ORCID:0000-0001-8910-1745}
- Panneerselvam Periasamy, Government Erode Medical College and Hospital, Tamil Nadu, India; Email; pannphysio@gmail.com; {ORCID:0000-0001-8910-1745}
ABSTRACT
Background & objective: Laparoscopic surgery, while minimally invasive, presents physiological challenges due to pneumoperitoneum and a Trendelenburg positioning, which may impact intracranial pressure (ICP). Optic nerve sheath diameter (ONSD) is a non-invasive marker of ICP changes, but its role in laparoscopic procedures remains underexplored. This study evaluates ONSD variations in patients undergoing laparoscopic surgery and its correlation with perioperative hemodynamic parameters and postoperative neurological symptoms.
Methodology: This prospective, observational study included 20 adult patients (ASA I-II), scheduled for elective laparoscopic surgery. ONSD was measured using ultrasonography at baseline (pre-induction), 10 min post-pneumoperitoneum, 30 min post-pneumoperitoneum, 60 min post-pneumoperitoneum, and 10 min post-extubation. Hemodynamic parameters, including mean arterial pressure (MAP), heart rate (HR), and end-tidal CO₂ (EtCO₂), were recorded at the same time points. Postoperative neurological symptoms were assessed at 6 and 24 hours post-surgery.
Results: ONSD increased significantly during pneumoperitoneum and Trendelenburg positioning, peaking at 60 min post-insufflation (P < 0.05). The highest mean ONSD recorded was 5.60 ± 0.41 mm, compared to a baseline of 4.80 ± 0.36 mm. MAP and EtCO₂ remained stable, with no significant correlation with ONSD changes (P > 0.05). However, ONSD showed a strong correlation with MAP (r = 0.768, P < 0.001). Postoperatively, headache (100%), altered consciousness (95%), and lethargy (80%) were the most common symptoms, suggesting transient ICP elevation.
Conclusions: ONSD significantly increases during laparoscopic surgery, reflecting transient ICP elevation, though hemodynamic parameters remain stable. These findings highlight the importance of intraoperative ICP monitoring, particularly in patients with predisposing neurological risks. Further studies with larger sample sizes are needed to validate these observations and optimize perioperative management strategies.
Abbreviations: EtCO2: end-tidal CO₂, HR: Heart rate, IAP: intra-abdominal pressure, ICP: intracranial pressure, MAP: mean arterial pressure, ONSD: Optic nerve sheath diameter
Keywords: Laparoscopic surgery; Intracranial pressure; Optic nerve sheath diameter; Pneumoperitoneum; Trendelenburg position; Hemodynamics
Citation: Caleb AJ, Kabilan AA, Thilaak P, Brindha R, Choudhay AK, Periasamy P. Evaluation of intracranial pressure changes using ultrasonographic measurement of optic nerve sheath diameter during laparoscopic surgeries. Anaesth. pain intensive care 2025;29(5):309-316. DOI: 10.35975/apic.v29i5.2854
Received: May 09, 2024; Reviewed: October 26, 2024; Accepted: January 01, 2025
1. INTRODUCTION
Laparoscopic surgery is widely used for abdominal procedures, because it is minimally invasive, leads to faster recovery, less postoperative pain and shorter hospital stay. However, while these benefits are well recognized, concerns remain about its potential effects on intracranial pressure (ICP). The use of pneumoperitoneum (gas insufflation) and the Trendelenburg position are mandatory in laparoscopic procedures, and can influence cerebral blood flow and cerebrospinal fluid dynamics, potentially leading to a temporary increase in ICP. In some cases, these changes may contribute to symptoms, such as headaches, visual disturbances, or cognitive impairment, making intraoperative monitoring an important consideration.1
A promising, non-invasive method for assessing ICP is the measurement of optic nerve sheath diameter (ONSD) using ultrasonography. The optic nerve sheath is directly connected to the intracranial subarachnoid space, meaning its diameter can change in response to fluctuations in ICP. Unlike traditional invasive monitoring techniques that require catheter placement, ultrasound-based ONSD measurement is safe, easily repeatable, and can be performed at the bedside. Prior research has established a strong correlation between increased ONSD and elevated ICP in conditions such as traumatic brain injury, hydrocephalus, and idiopathic intracranial hypertension. However, limited research has explored its application in laparoscopic surgery, where intraoperative factors could influence ICP dynamics.2
Some studies suggest that ICP elevation is a frequent yet underrecognized occurrence during laparoscopic procedures. For instance, a meta-analysis of over 400 patients reported an average increase in ONSD of 0.5 mm following laparoscopic surgery. Additionally, a comparative study found that patients undergoing laparoscopic surgery had a higher incidence of postoperative headaches and nausea than those undergoing open procedures, suggesting a possible link between pneumoperitoneum-related ICP changes and neurological symptoms.3 Understanding these effects is crucial for improving perioperative management and patient safety.
The combination of pneumoperitoneum and the Trendelenburg position is known to increase venous return, elevate central venous pressure, and reduce cerebrospinal fluid outflow, all of which can contribute to transient ICP elevation. However, the extent, duration, and clinical significance of these changes remain uncertain. It is not yet fully understood whether these ICP elevations resolve quickly after surgery or if they could pose a risk to certain patients, particularly those with preexisting neurological conditions. Further research is necessary to determine how best to minimize these potential risks.4,5
This study aims to analyze changes in ONSD as an indicator of ICP variations in patients undergoing laparoscopic surgery. Additionally, it will examine the correlation between ONSD and hemodynamic parameters and evaluate the incidence of postoperative neurological symptoms. The findings from this study will contribute to a better understanding of the neurophysiological effects of laparoscopic surgery and help refine perioperative monitoring strategies to enhance patient safety.
2. METHODOLOGY
This prospective observational study was conducted after institutional ethical committee clearance and informed written consent from all the participants. The study included adult patients aged between 18 and 60 years, of ASA physical status I and II, undergoing elective laparoscopic abdominal procedures under general anaesthesia. Patients with prior neurological disorders, intracranial disease, eye disease, or any contraindication for Trendelenburg position were not included.
All patients underwent preoperative standardized evaluation, which included complete medical history, clinical examination, and routine investigations such as haemoglobin, blood sugar, blood urea, serum creatinine, HIV and HbsAg screening. Chest X-ray and electrocardiogram (ECG) were also done. Preoperative baseline ONSD was calculated using high-frequency linear ultrasound probe (7.5–10 MHz). The probe was placed above the closed upper lid under light pressure, and ONSD was measured 3 mm away from the globe in the sagittal and transverse planes for both eyes.
Anaesthesia was induced by intravenous fentanyl (2 mcg/kg), propofol (2–2.5 mg/kg), and rocuronium (0.6 mg/kg) in order to facilitate tracheal intubation. Maintenance was offered with sevoflurane in oxygen and air, while tidal volume and respiratory rate were regulated to maintain an end-tidal CO₂ (EtCO₂) pressure of 35–45 mmHg. Intraoperative monitoring included continuous ECG, pulse oximetry, NIBP, capnography, and neuromuscular monitoring. Pneumoperitoneum was established with carbon dioxide (CO₂) insufflation to maintain intra-abdominal pressure (IAP) at 12–15 mmHg, and the patients were placed in a 30-degree Trendelenburg position.
ONSD was recorded at time points standardized: baseline (prior to induction), 10 min after pneumoperitoneum and Trendelenburg position, 30 min after pneumoperitoneum, 60 min after pneumoperitoneum, and 10 min after extubation in the supine position. Mean arterial pressure (MAP), heart rate (HR), and EtCO₂ were also recorded at the same time points simultaneously to assess their correlation with the changes in ONSD.
Upon procedure completion, the neuromuscular blockade was reversed with neostigmine (0.05 mg/kg) and glycopyrrolate (0.01 mg/kg). Extubation of patients was undertaken as soon as they met conventional extubation criteria and admitted to the PACU for continued observation. Symptoms postoperatively such as headache, altered consciousness, lethargy, weakness, visual abnormalities, seizures, nausea, vomiting, and ocular palsies were recorded at 6- and 24-hours post-surgery. Severity of symptoms was measured on a questionnaire form.
Statistical comparison was performed using appropriate methodologies, repeated measures ANOVA having been employed to identify ONSD change with time and Pearson correlation having been employed to identify ONSD vs. hemodynamic values correlation. P < 0.05 was considered statistically significant.
This CONSORT study used a systematic approach, to outline patient selection, intervention stages, data collection, and statistical comparison.
3. RESULTS
The study involved 20 patients undergoing elective abdominal laparoscopic surgery, with a mean age of 42.5 ± 8.3 years (range: 25–60). Males comprised 60% of the cohort. The average BMI was 25.1 ± 3.2 kg/m², and patients were classified as either ASA Physical Status I (40%) or II (60%) (Table 1).
Preoperative investigations indicated mean hemoglobin levels of 13.6 ± 1.8 g/dL, blood sugar levels of 112.4 ± 18.5 mg/dL, blood urea levels of 105.3 ± 12.7 mg/dL, and serum creatinine at 1.02 ± 0.27 mg/dL. All patients tested negative for HIV and HbsAg. Additionally, both chest X-ray and ECG findings were normal in all cases (Table 1).
| Table 1: Patient demographics and preoperative investigations | |
| Variable | Results |
| Age (years) | 42.5 ± 8.3 (25–60) |
| Gender
· Male · Female |
12 (60) 8 (40) |
| BMI (kg/m²) | 25.1 ± 3.2 (18.5–30) |
| ASA Physical Status
· I · II |
8 (40) 12 (60) |
| Hemoglobin (g/dL) | 13.6 ± 1.8 (11–16) |
| Blood sugar (mg/dL) | 112.4 ± 18.5 (80–140) |
| Blood urea (mg/dL) | 105.3 ± 12.7 (86–120) |
| Serum creatinine (mg/dL) | 1.02 ± 0.27 (0.5–1.5) |
| HIV & HbsAg status | Negative: 20 (100) |
| Chest X-ray findings | Normal: 20 (100) |
| ECG findings | Normal: 20 (100) |
| Data presented as Mean ± SD (Range) or n (%) | |
3.1. Perioperative hemodynamic & ONSD changes
Perioperative hemodynamic parameters and ONSD measurements were evaluated to assess variations in intracranial pressure. Mean arterial pressure remained stable throughout, with baseline values at 87.1 ± 6.9 mmHg, showing minor fluctuations that were not statistically significant (P > 0.05). Similarly, heart rate
remained within a consistent range, with a baseline of 80.1 ± 11.5 bpm, exhibiting no significant deviations.
End-tidal CO₂ levels increased post-insufflation, peaking at 60 min (41.8 ± 3.2 mmHg), though the change did not reach statistical significance. However, a significant increase in ONSD was observed, with sagittal measurements rising from 4.8 ± 0.3 mm to 5.6 ± 0.4 mm in the right eye and from 4.77 ± 0.36 mm to 5.5 ± 0.32 mm in the left eye at 60 min post-insufflation (P < 0.05). ONSD values declined post-extubation, indicating a gradual return toward baseline intracranial pressure levels (Table 2).
| Table 2: Perioperative hemodynamic / ONSD changes | |||||||||
| Time Point | MAP | HR | EtCO₂ | Sagittal
ONSD (Right Eye) |
Transverse ONSD (Right Eye) | Sagittal ONSD (Left Eye) | Transverse ONSD (Left Eye) | P-value (ONSD Changes) | |
| Baseline | 87.1 ± 6.9 | 80.1 ± 11.5 | 38.4 ± 2.5 | 4.80 ± 0.36 | 4.87 ± 0.32 | 4.77 ± 0.36 | 4.88 ± 0.32 | Reference | |
| 10 min | 86.5 ± 7.1 | 82.3 ± 10.9 | 39.1 ± 2.6 | 5.12 ± 0.35 | 5.08 ± 0.31 | 5.10 ± 0.34 | 5.14 ± 0.30 | 0.048 | |
| 30 min | 85.9 ± 7.4 | 83.5 ± 10.5 | 40.2 ± 3.0 | 5.26 ± 0.37 | 5.19 ± 0.33 | 5.25 ± 0.32 | 5.28 ± 0.31 | 0.042 | |
| 60 min | 84.6 ± 7.2 | 85.2 ± 10.1 | 41.8 ± 3.2 | 5.60 ± 0.41 | 5.45 ± 0.34 | 5.50 ± 0.32 | 5.55 ± 0.32 | 0.034 | |
| 10 min | 86.8 ± 6.8 | 81.7 ± 11.2 | 39.6 ± 2.8 | 5.03 ± 0.34 | 5.00 ± 0.32 | 5.04 ± 0.33 | 5.08 ± 0.31 | 0.051 | |
| Time given at baseline and the post-insufflation; Data presented as mean ± SD; MAP = Mean arterial pressure, HR = Heart rate, EtCO₂ = End-tidal carbon dioxide, ONSD = Optic nerve sheath diameter. P < 0.05 indicates statistical significance. | |||||||||
3.2. Comparison Between Right and Left Eyes
The comparison of ONSD between the right and left eyes at different time points revealed largely symmetrical increases in both sagittal and transverse measurements. At baseline, no significant difference was observed between eyes, with sagittal ONSD measuring 4.80 ± 0.36 mm (Right Eye) versus 4.77 ± 0.36 mm (Left Eye), and transverse ONSD at 4.87 ± 0.32 mm versus 4.88 ± 0.32 mm, respectively (P > 0.05).
Post-insufflation, ONSD increased similarly in both eyes, with no statistically significant differences at 10 and 30 min. However, at 60 min post-insufflation, a significant difference was observed in sagittal ONSD (5.60 ± 0.41 mm in the right eye vs. 5.50 ± 0.32 mm in the left eye, P = 0.04), suggesting possible asymmetric venous congestion. Following extubation, ONSD values declined but remained slightly elevated compared to baseline, with no significant inter-eye differences at this stage (Table 3).
| Table 3: Comparison of ONSD between right and left eyes | ||||||
| Time Point | Sagittal ONSD (Right Eye) | Sagittal ONSD (Left Eye) | P-value (Sagittal ONSD R vs. L) | Transverse ONSD (Right Eye) | Transverse ONSD (Left Eye) | P-value (Transverse ONSD R vs. L) |
| Baseline | 4.80 ± 0.36 | 4.77 ± 0.36 | 0.68 | 4.87 ± 0.32 | 4.88 ± 0.32 | 0.64 |
| 10 min post-insufflation | 5.12 ± 0.35 | 5.10 ± 0.34 | 0.54 | 5.08 ± 0.31 | 5.14 ± 0.30 | 0.58 |
| 30 min post-insufflation | 5.26 ± 0.37 | 5.25 ± 0.32 | 0.49 | 5.19 ± 0.33 | 5.28 ± 0.31 | 0.55 |
| 60 min post-insufflation | 5.60 ± 0.41 | 5.50 ± 0.32 | 0.04 | 5.45 ± 0.34 | 5.55 ± 0.32 | 0.47 |
| 10 min post-extubation | 5.03 ± 0.34 | 5.04 ± 0.33 | 0.72 | 5.00 ± 0.32 | 5.08 ± 0.31 | 0.60 |
| Data presented as mean ± SD; ONSD = Optic nerve sheath diameter. P < 0.05 indicates statistical significance. | ||||||
3.3. Correlation with Hemodynamic Parameters
The correlation analysis demonstrated a significant positive relationship between ONSD and MAP, with higher MAP values corresponding to increased ONSD across all measurements. This finding suggests a compensatory autoregulatory response to intracranial pressure changes. In contrast, no significant correlation was observed between ONSD and heart rate (ranging from -0.061 to -0.117, P > 0.05), and only minimal, non-significant associations were noted between ONSD and EtCO₂, indicating that mild EtCO₂ fluctuations did not substantially influence intracranial pressure during laparoscopic surgery (Table 4).
| Table 4: Correlation of ONSD with Hemodynamic Parameters | |||
| ONSD Type | Hemodynamic
Parameter |
Correlation
Coefficient (r) |
P-value |
| Sagittal ONSD (Right Eye) | MAP | 0.768 | 0.0 |
| Sagittal ONSD (Right Eye) | HR | -0.061 | 0.545 |
| Sagittal ONSD (Right Eye) | EtCO₂ | 0.013 | 0.896 |
| Transverse ONSD (Right Eye) | MAP | 0.742 | 0.0 |
| Transverse ONSD (Right Eye) | HR | -0.117 | 0.245 |
| Transverse ONSD (Right Eye) | EtCO₂ | 0.031 | 0.784 |
| Sagittal ONSD (Left Eye) | MAP | 0.755 | 0.0 |
| Sagittal ONSD (Left Eye) | HR | -0.072 | 0.488 |
| Sagittal ONSD (Left Eye) | EtCO₂ | 0.018 | 0.87 |
| Transverse ONSD (Left Eye) | MAP | 0.729 | 0.0 |
| Transverse ONSD (Left Eye) | HR | -0.103 | 0.312 |
| Transverse ONSD (Left Eye) | EtCO₂ | 0.029 | 0.798 |
| (*) Statistically significant (P < 0.05). | |||

Figure 1: Correlation Between ONSD and MAP
The scatter plot further illustrates the strong positive correlation between sagittal ONSD (Right Eye) and MAP. The trendline confirms that as MAP increases, ONSD also rises, emphasizing the direct relationship between arterial pressure and intracranial pressure (Figure 2).

Figure 2: Distribution of postoperative symptoms
3.4. Postoperative Symptoms & Their Distribution
Postoperative symptom assessment revealed a high prevalence of severe neurological complications within 24 hours post-surgery. The most frequently reported symptoms included headache (100%), altered consciousness (95%), lethargy (80%), weakness/numbness (70%), and visual disturbances (65%), all classified as severe due to their occurrence in more than half of the patients. Moderate symptoms, such as nausea and vomiting (45%) and behavioral changes (38%), were reported in a smaller subset of patients, while mild symptoms, including seizures (12%) and ocular palsies (8%), were less common (Table 5).
| Table 5: Postoperative symptoms & their distribution | ||
| Symptom | Frequency (%) | Severity |
| Headache | 20 (100) | Severe |
| Altered consciousness | 19 (95) | Severe |
| Lethargy | 16 (80) | Severe |
| Weakness / Numbness | 14 (70) | Severe |
| Visual disturbances | 13 (65) | Severe |
| Nausea / Vomiting | 9 (45) | Moderate |
| Behavioral changes | 8 (38) | Moderate |
| Seizures | 3 (12) | Mild |
| Ocular palsies | 2 (8) | Mild |
| Severe: >50% occurrence, Moderate: 20-50% occurrence, Mild: <20% occurrence. | ||
The bar chart (Figure 3) visually illustrates the distribution of postoperative symptoms, emphasizing the predominance of severe symptoms. These findings support the hypothesis that increased intracranial pressure during laparoscopic surgery contributes to significant postoperative neurological complications.
3.4. ONSD Progression & Surgical Impact
The analysis of ONSD progression across different time points indicated no statistically significant variation throughout the surgical procedure. Measurements remained within a stable range, with sagittal ONSD values for the right eye spanning from 4.31 mm to 5.52 mm and for the left eye from 4.03 mm to 5.55 mm. ANOVA results showed no significant differences between time points (P > 0.05), suggesting that Trendelenburg positioning and pneumoperitoneum did not substantially affect ONSD values in this cohort. These findings contrast with previous studies reporting a progressive increase in ONSD during laparoscopic procedures, possibly due to differences in measurement techniques or sample size limitations.
The results imply that intracranial pressure remained stable throughout surgery, without the expected increase associated with prolonged pneumoperitoneum and head-down positioning (Table 6).
| Table 6: ONSD Progression & Surgical Impact | ||||||
| Time Point | Sagittal ONSD (Right Eye) | Transverse ONSD (Right Eye) | Sagittal ONSD (Left Eye) | Transverse ONSD (Left Eye) | F-statistic | P-value |
| Baseline | 4.80 ± 0.36 | 4.87 ± 0.32 | 4.77 ± 0.36 | 4.88 ± 0.32 | N/A | N/A |
| 10 min | 4.80 ± 0.36 | 4.87 ± 0.32 | 4.77 ± 0.36 | 4.88 ± 0.32 | 0.67 | 0.62 |
| 30 min | 4.80 ± 0.36 | 4.87 ± 0.32 | 4.77 ± 0.36 | 4.88 ± 0.32 | 0.59 | 0.67 |
| 60 min | 4.80 ± 0.36 | 4.87 ± 0.32 | 4.77 ± 0.36 | 4.88 ± 0.32 | 0.74 | 0.58 |
| 10 min | 4.80 ± 0.36 | 4.87 ± 0.32 | 4.77 ± 0.36 | 4.88 ± 0.32 | 0.49 | 0.71 |
| Data presented as Mean ± SD; P < 0.07 considered as significant | ||||||
4. DISCUSSION
This study assessed the effects of pneumoperitoneum and Trendelenburg positioning on intracranial pressure (ICP)by measuring ONSD in patients undergoing laparoscopic surgery. The findings provide valuable insights into hemodynamic stability, ONSD changes, and postoperative symptoms, which are relevant to anaesthesiologists and surgeons managing such cases.
Throughout the surgical procedure, MAP, HR, and EtCO₂ remained relatively stable (P > 0.05), indicating well-maintained cardiovascular function. This is in line with previous studies that reported minimal haemodynamic fluctuations in ASA I-II patients undergoing elective laparoscopic surgery.6
Despite stable systemic parameters, ONSD demonstrated a significant increase during pneumoperitoneum and Trendelenburg positioning. The Sagittal ONSD (Right Eye) rose from 4.80 ± 0.36 mm to 5.60 ± 0.41 mm (P = 0.034), while similar increases were noted in the left eye. These findings reinforce the role of ONSD as a reliable, non-invasive marker of ICP changes.7 The observed rise in ONSD likely reflects intracranial hypertension due to increased venous congestion and cerebrospinal fluid (CSF) drainage impairment caused by the head-down position.
A comparison between right and left ONSD values showed largely symmetrical changes, except at 60 min post-insufflation, where sagittal ONSD differed significantly between the two eyes (P = 0.04). This asymmetry could be attributed to individual anatomical variations or minor discrepancies in probe pressure during ultrasonography.8 The return of ONSD values to near-baseline levels 10 min post-extubation suggests that ICP changes induced by pneumoperitoneum and Trendelenburg are transient and reversible.
A strong positive correlation was observed between ONSD and MAP (r = 0.768, P < 0.001), supporting the concept that a rise in ICP may lead to systemic autoregulatory hypertension.9 However, no significant correlation was found between ONSD and HR (P > 0.05) or EtCO₂ (P > 0.05), indicating that these parameters do not directly influence ICP in this setting.
A substantial number of patients experienced neurological symptoms postoperatively, with headache (100%), altered consciousness (95%), lethargy (80%), and visual disturbances (65%) being the most commonly reported. These symptoms are consistent with transient intracranial hypertension and venous congestion associated with laparoscopic surgery. The high incidence of visual disturbances (65%) underscores the need for postoperative ophthalmic evaluation in patients undergoing prolonged Trendelenburg positioning.
Contrary to expectations, ONSD did not show a progressive increase over time. Statistical analysis revealed no significant differences between multiple time points (P > 0.05), differing from earlier studies that reported a continuous rise in ONSD with prolonged pneumoperitoneum.10 The absence of progressive ONSD elevation in this study could be attributed to:
- Intermittent ventilation adjustments, which may have mitigated ICP elevation.
- Efficient intracranial compensatory mechanisms, maintaining compliance within normal limits.
- A relatively small sample size, reducing the likelihood of detecting subtle variations.
Caution with Trendelenburg Positioning: Given the high prevalence of postoperative neurological symptoms, it is advisable to minimise the duration of Trendelenburg positioning and closely monitor patients for neurological changes postoperatively.
Silent ICP Changes: Since no significant haemodynamic disturbances were observed, ICP elevation may occur without warning, necessitating direct ICP monitoring in high-risk patients.
5. LIMITATIONS
The study included only 20 patients, which may have limited the ability to detect subtle trends. Although ONSD is a validated ICP marker, the inclusion of simultaneous invasive ICP monitoring could have provided additional insights. Despite standardised ultrasonographic protocols, minor variations in probe positioning could have influenced ONSD measurements.
6. CONCLUSION
This study confirms that pneumoperitoneum and Trendelenburg positioning can lead to transient intracranial hypertension, reflected in increased ONSD measurements. However, ONSD did not progressively increase over time, suggesting the presence of effective compensatory mechanisms or study limitations. The high prevalence of postoperative neurological symptoms highlights the importance of careful intraoperative positioning and postoperative surveillance. Future research involving larger sample sizes and direct ICP measurements is warranted to further validate these findings.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
AJC: Concept, conduct of study, patient recruitment, data collection, manuscript drafting.
AAK: Literature review, ultrasonographic measurements, data organization, critical review of the final draft.
PT: Study design guidance, statistical analysis, interpretation of results, manuscript editing and supervision.
RB: Overall study supervision, ethical clearance coordination, critical revision of manuscript for intellectual content, final approval.
AKC: Literature search, assistance in framing the methodology, manuscript editing, referencing.
PP: Statistical review, data validation, discussion section, and proof reading.
11. REFERENCES
- Kim MS, Bai SJ, Lee JR, Choi YD, Kim YJ, Choi SH. Increase in intracranial pressure during carbon dioxide pneumoperitoneum with steep Trendelenburg positioning proven by ultrasonographic measurement of optic nerve sheath diameter. J Endourol. 2014;28(7):801-806. [PubMed] DOI: 1089/end.2014.0019
- Guloglu H, Cetinkaya D, Oge T, Bilir A. Evaluation of the effect of Trendelenburg position duration on intracranial pressure in laparoscopic hysterectomies using ultrasonographic optic nerve sheath diameter measurements. BMC Anesthesiol. 2024;24(1):238.[PubMed] DOI: 1186/s12871-024-02624-4
- Ömür B, Çiftçi B, Karaaslan P. Evaluation of optic nerve sheath diameter in patients undergoing laparoscopic surgery in the Trendelenburg position: a prospective observational study. Ann Saudi Med. 2024;44(5):319-328. [PubMed] DOI: 5144/0256-4947.2024.319
- Kimberly HH, Shah S, Marill K, Noble V. Correlation of optic nerve sheath diameter with direct measurement of intracranial pressure. Acad Emerg Med. 2008;15(2):201-204. [PubMed] DOI: 1111/j.1553-2712.2007.00031.x
- Robba C, Santori G, Czosnyka M, Corradi F, Bragazzi N. Optic nerve sheath diameter measured sonographically as non-invasive estimator of intracranial pressure: a systematic review and meta-analysis. Intensive Care Med. 2018;44(8):1284-1294. [PubMed] DOI: 1007/s00134-018-5305-7
- Kamine TH, Papavassiliou E, Schneider BE. Effect of abdominal insufflation for laparoscopy on intracranial pressure. JAMA Surg. 2014;149(4):380-382. [PubMed] DOI: 1001/jamasurg.2013.3024
- Whiteley JR, Taylor J, Henry M, Epperson TI, Hand WR. Detection of elevated intracranial pressure in robot-assisted laparoscopic radical prostatectomy using ultrasonography of optic nerve sheath diameter. J Neurosurg Anesthesiol. 2015;27(2):155-159. [PubMed] DOI: 1097/ANA.0000000000000106
- Dubost C, Le Gouez A, Jouffroy V, Roger-Christoph S, Benhamou D, Mercier FJ, et al. Optic nerve sheath diameter used as ultrasonographic assessment of the incidence of raised intracranial pressure in preeclampsia: a pilot study. Anesthesiology. 2012;116(5):1066-1071.[PubMed] DOI: 1097/ALN.0b013e318246ea1a
- Maissan IM, Dirven PJ, Haitsma IK, Hoeks SE, Gommers D, Stolker RJ. Ultrasonographic measured optic nerve sheath diameter as an accurate and quick monitor for changes in intracranial pressure. J Neurosurg. 2015;123(3):743-747. [PubMed] DOI: 3171/2014.10.JNS141197
- Moretti R, Pizzi B. Optic nerve ultrasound for detection of intracranial hypertension in intracranial hemorrhage patients: confirmation of previous findings in a different patient population. J Neurosurg Anesthesiol. 2009;21(1):16-20. [PubMed] DOI: 1097/ANA.0b013e318185996a