Sana Shah, BS 1, Taizoon Q. Dhoon, MD 2, Yeyoon Choi, MD 3, George Wang, MD 4, Govind Rajan MD 5
Author affiliations:
Malignant hyperthermia (MH) is a rare, life-threatening pharmacogenetic disorder triggered by exposure to volatile anesthetics and succinylcholine. It results from mutations in the RYR1 gene, leading to excessive intracellular calcium release, hypermetabolism, and potential multiorgan dysfunction. Prompt recognition and treatment are essential to reduce morbidity and mortality.
We report the case of an 11-year-old female undergoing pancreaticoduodenectomy who developed intraoperative MH following sevoflurane exposure. Early signs included rising end-tidal CO₂ (EtCO₂) despite increased ventilation, followed by tachycardia and metabolic acidosis, with a peak temperature of 37.1°C. MH was suspected approximately two hours after induction, leading to immediate discontinuation of sevoflurane, transition to total intravenous anesthesia, and administration dantrolene. The patient's vital signs stabilized, and the surgery was completed without further complications.
This case underscores the importance of early MH recognition, especially in pediatric patients, who may present with subtle or atypical symptoms. The MH Clinical Grading Score, designed for adults, may not reliably predict MH likelihood in children, necessitating a high index of suspicion. Given the variable presentation of MH in pediatric patients, anesthesiologists must be vigilant in monitoring intraoperative changes. Timely intervention and adherence to MH crisis protocols significantly improve outcomes. Further research is needed to optimize pediatric-specific MH diagnostic criteria and management strategies.
Abbreviations: MH: Malignant Hyperthermia, EtCO2: End Tidal Carbon Dioxide, RYR1: Ryanodine Receptor Gene
Keywords: Malignant Hyperthermia; Anesthesia; Pediatric Anesthesia; General Anesthesia; Dantrolene; RYR1 gene
Citation: Shah S, Dhoon T, Choi Y, Wang G, Rajan G. Pediatric malignant hyperthermia: recognizing early warning signs – a case report. Anaesth. pain intensive care 2025;29(4):446-452. DOI: 10.35975/apic.v29i4.2835
Received: May 09, 2024; Revised: October 26, 2024; Accepted: January 01, 2025
Malignant hyperthermia (MH) is a rare pharmacogenetic disorder of skeletal muscle characterized by hypermetabolism secondary to exposure to volatile anesthetic agents and succinylcholine.1 Most affected individuals harbor mutations in the ryanodine receptor RYR1, leading to unregulated accumulation of intracellular calcium and subsequent abnormal skeletal muscle metabolism and contraction.1 The incidence of MH during anesthesia is between 1:100,000 in adults and 1:30,000 in children, and is more likely to present in males than females (2:1).2 The mean age of affected patients is 18.3 years, with 52.1% of all reactions noted to be in children under the age of 15.1
The mortality rate of MH is reported at 80% without treatment and up to 15% even with proper treatment.2,3 Although confirmation can be obtained after an MH event by the halothane-caffeine contracture test or genetic studies, intraoperatively, MH is a purely clinical diagnosis necessitating the need for a high degree of clinical suspicion.4
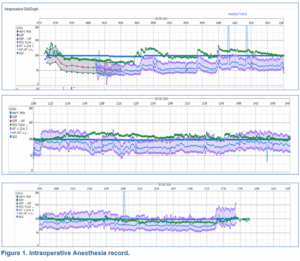
Figure 1: Intraoperative Anesthesia record.
An 11-year-old previously healthy female was incidentally diagnosed with a primary solid pseudopapillary epithelial neoplasm of the pancreas and presented for a pancreaticoduodenectomy. Preoperatively, a thoracic epidural was administered at the T8-9 level. Induction of anesthesia was achieved with 30 mg of lidocaine, 80 mg of propofol, 40 mg of rocuronium, and 25 mg of fentanyl, followed by an uneventful intubation. Sevoflurane was subsequently initiated for maintenance of anesthesia. Post-induction, a left radial arterial line and a right internal jugular double-lumen central line were successfully placed without complication.
One hour after sevoflurane was started, there was a rise in end-tidal carbon dioxide (EtCO2) that reached 45 mmHg despite increased ventilatory measures. The changes in EtCO2 preceded a rise in temperature by 15 minutes, which peaked at 37.1° C despite discontinuation of heating elements. Heart rate and blood pressure started increasing approximately 1 hour after sevoflurane initiation, which coincided with the rise in EtCO2; 1.5 hours after induction, initial arterial blood gas showed evidence of acidemia (pH 7.317, PCO2 37.7, PO2 186, HCO3 19.3, BE -7).
Approximately two hours after induction, MH was suspected. In response, sevoflurane was discontinued, and anesthesia was transitioned to total intravenous anesthesia. The anesthesia breathing circuit was replaced and flushed with high fresh gas flow, and charcoal filters (Vapor-Clean; Dynasthetics, Salt Lake City, UT) were applied. The surgical team was promptly notified, and the MH cart was brought to the operating room. The patient received four rounds of dantrolene and sodium bicarbonate (Figure 2), and the MH hotline was contacted for further guidance.
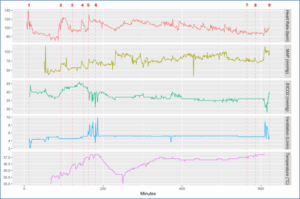
Figure 2: Minute by minute analysis of Heart Rate, MAP ART A-line, EtCO2 (mmHg), Minute Ventilation (liters/minute), and Temperature (°C) during surgery. For details regarding time points 1-9, refer to Table 1.
Within 15 minutes of discontinuing sevoflurane, the patient's EtCO2, heart rate, and blood pressure returned to normal ranges, while the decrease in temperature followed approximately 45 minutes later. Subsequent arterial blood gas analyses indicated improvement in acidemia. The surgery was paused for 40 minutes to allow for stabilization and then resumed without further complications. Postoperatively, the patient was admitted to the surgical intensive care unit (SICU), where elevated liver function tests were noted, likely as a side effect of dantrolene administration. Her recovery was otherwise uneventful, and she was discharged home on postoperative day 11.
Summarizing Patient Data and Vitals
The patient's vitals were first recorded at 7:17 AM, with sevoflurane induction starting at 7:28 AM. Heart rate spiked from 70 to 123 bpm between 8:34 and 8:53 AM, followed by a temperature increase from 35.7°C to 36.4°C between 9:09 and 9:18 AM. At 9:46 AM, a malignant hyperthermia (MH) alert was triggered due to rising temperature and unresponsive elevated EtCO2. Sevoflurane was discontinued, TIVA initiated, charcoal filters placed, and 80 mg IV Dantrolene administered at 9:59 AM, with a second dose at 10:17 AM. At 10:35 AM, the MH hotline was contacted to report the case. Between 11:06 and 11:12 AM, MAP-ART A-line pressure increased from 68 to 93, peaking at 104. Over the remainder of the operation, the patient’s vitals gradually stabilized. Additional doses of 15 mg IV Dantrolene were given at 4:40 PM and 5:02 PM. The patient was safely extubated at 5:37 PM.
MH is a rare pharmacogenetic disorder that manifests as a hypermetabolic response during or after exposure to volatile halogenated anesthetic agents and succinylcholine.1 Most affected individuals harbor a mutation in the ryanodine receptor gene RYR1, which is inherited in an autosomal dominant manner.5 Determining the prevalence of MH susceptibility in the general population is challenging due to its infrequent occurrence, unknown family history, and variable expressivity in response to triggering agents.4 Many individuals with this genetic predisposition may be unaware of their susceptibility until an MH episode occurs.
The mutation in MH primarily affects skeletal muscle, leading to an excessive release of intracellular calcium ions that triggers unregulated contraction of sarcomeres. This leads to a hypermetabolic state, resulting in increased carbon dioxide production and oxygen consumption, increased respiratory rate, acidosis, hyperkalemia, rhabdomyolysis, and muscle rigidity.6,7 MH is a condition that can lead to severe organ dysfunction and death, in extreme cases. Even with treatment, the mortality rate associated with MH can be as high as 15%.8,9 This rate may be influenced by various factors, including patient age and muscle mass, but the timeliness of diagnosis and treatment are likely key factors. As such, the ability to diagnose and treat MH swiftly and effectively is paramount in improving patient outcomes.
When MH is suspected, all triggering agents must be discontinued, the patient should be hyperventilated, and dantrolene should be given at an initial dose of 2.5 mg/kg and repeated every 10-15 minutes thereafter until acidosis, hyperthermia, electrolyte abnormalities, and muscle rigidity are resolved.10 Core temperature monitoring and cooling measures should be implemented, as well as laboratory tests, including arterial blood gases and electrolytes, should be closely monitored.11 The MH hotline is a vital resource during an acute crisis. Post-crisis, patients should be observed in the ICU for any recurrence or complications. It also is essential to inform patients and their families about this complication. Additionally, outpatient testing should be arranged to ensure comprehensive evaluation and management.
3.1. Dantrolene Sodium
Dantrolene sodium is a postsynaptic muscle relaxant that inhibits the release of intracellular calcium from the sarcoplasmic reticulum of skeletal muscle cells mediated by the RyR1 receptor, counteracting the effects of MH.8 Since its introduction in the 1970s, dantrolene has revolutionized the treatment of MH, significantly reducing mortality rates from 70–80% to approximately 10–15%.12 Dantrolene is typically supplied as a lyophilized powder with 20 mg of dantrolene sodium and 3000 mg mannitol, with each vial requiring reconstitution with 60 mL of sterile water to reach a pH of approximately 9.5. Dantrolene’s reconstitution can be time-consuming and risks fluid overload, particularly in pediatric patients. The drug has a half-life of 6–8 hours, necessitating repeated dosing to ensure sustained treatment of MH.13,14
Common side effects include muscle weakness, nausea, vomiting, and diarrhea. Rarely, dantrolene can cause liver toxicity, dizziness, fatigue, and injection site reactions, and is contraindicated in patients with hepatic disease. There is no antidote for dantrolene toxicity, so treatment focuses on symptom management.14,15 Despite its relatively cumbersome reconstitution process, dantrolene remains a safe and reliable formulation with minimal risk to the general population.
3.2. Ryanodex
Ryanodex, a newer Dantrolene formulation, offers significant advantages over previous versions, including room temperature storage, reduced dosage requirements, faster reconstitution, and improved solubility. It is available in 20 mL vials which contain 250 mg of dantrolene sodium, requiring just 5 mL of sterile water for reconstitution.15 Its lower volume and rapid administration make it crucial for emergencies like MH. While associated with fewer gastrointestinal issues and lower liver toxicity, it still poses risks like muscle weakness, fatigue, dizziness, flushing, somnolence, dysphonia, and potential hepatotoxicity with prolonged use.14,15 This newer product is also more expensive and has a shorter shelf life than traditional Dantrolene.16
3.3. Pediatric Malignant Hyperthermia
MH is estimated to occur in 1 in 15,000 pediatric anesthetic procedures and 52% of all reactions are noted be in children under the age of 15.1,17 The variability in clinical presentation of MH, especially among age cohorts during childhood, highlights the importance of having a high index of suspicion.
A retrospective analysis of data from the North American Malignant Hyperthermia Registry on patients under 18 years of age demonstrated age-related differences in presentation of MH.17
In infants and young children, initial signs of MH are often subtle, with tachycardia and increased EtCO2 being early indicators.18 The youngest age group (0 to 24 months) displayed less frequent muscle rigidity, but more often showed skin mottling, higher peak lactic acid levels, and lower peak creatine kinase (CK) levels compared to older children.18 Children aged 25 months to 12 years exhibited lower maximum EtCO2 and carbon dioxide tension (PaCO2) compared to the youngest and oldest age groups but were more than three times as likely to experience masseter spasm.18 Adolescents aged 12 to 18 years had higher peak potassium levels, higher maximum temperatures, were more prone to sweating, and took longer to reach their maximum EtCO2 levels.18 As children grow older, classical signs such as masseter muscle rigidity and hyperthermia become more pronounced. Across all age groups, the most observed findings were sinus tachycardia (73.1%), hypercarbia (68.6%), and rapid temperature increase (48.5%).18 These age-related differences in MH presentation may be attributable to differences in body composition, muscle mass, and inherent thermoregulatory changes throughout development.18 Alternate studies also suggest these differences may be attributed to age-related differences in hemodynamics, anatomy, and physiology, such as smaller functional residual capacity and higher oxygen consumption in pediatric patients.19
3.4. Malignant Hyperthermia (MH) Clinical Grading Score
Diagnosis and assessment of MH risk, including in pediatric patients, are often guided by clinical signs and symptoms observed during anesthesia, laboratory tests, and MH Clinical Grading Score. The clinical grading scale for MH, developed by Larach et al., is used to determine the likelihood of an MH event based on clinical presentation in adults. Currently, there is no pediatric specific MH clinical grading score in clinical practice. This scale considers various factors such as muscle rigidity, elevated temperature, acidosis, and hypermetabolism.20
Although this scale is not specifically designed for pediatric use, it can be applied to patients of all ages to assess the probability of MH, with the understanding that pediatric and adult patients may exhibit different symptoms. For children, particular attention is paid to age-specific presentations, such as those mentioned previously: subtle initial signs in infants, differences in muscle rigidity, and variations in biochemical markers like creatine kinase and lactic acid levels.21
MH often occurs in patients without a known personal or family history, highlighting the critical importance of early recognition and intervention when noting signs of hemodynamic instability and increases in minute ventilation and EtCO2. The currently available MH grading score used clinically to diagnose MH is only fashioned for adults, which can lead to delayed management in pediatric patients. Awareness of age-related differences between children and adults is crucial for anesthesia providers to offer prompt and appropriate treatment via Dantrolene or Ryanodex, greatly improving patient outcomes.
5. Conflict of interest:
Authors declare no conflict of interest. No external or industry funding was involved.
6. Ethical considerations
Written informed consent was obtained from the father of the child to use this case report for academic interest.
7. Authors contribution
All authors were involved in the management of this patient. Expert advice was sought from the seniors in the preparation of this manuscript.
Author affiliations:
- Sana Shah, 101 The City Dr S, Orange, CA 92868, USA; Email: sana.sanjit.shah@gmail.com; {ORCID:0000-0003-2904-4827}
- Taizoon Dhoon, 101 The City Dr S, Orange, CA 92868, USA; Email: tdhoon@hs.uci.edu; {ORCID:0009-0003-8925-3652}
- Yeyoon Choi, 101 The City Dr S, Orange, CA 92868, USA; Email: yeyoonc1@hs.uci.edu; {ORCID:0000-0002-3685-7933}
- George Wang, 101 The City Dr S, Orange, CA 92868, USA; Email: wanggs@hs.uci.edu; {ORCID:0000-0002-1521-9799}
- Govind Rajan, 101 The City Dr S, Orange, CA 92868, USA; Email: grajan@hs.uci.edu; {ORCID:0000-0002-9498-3241}
ABSTRACT
Malignant hyperthermia (MH) is a rare, life-threatening pharmacogenetic disorder triggered by exposure to volatile anesthetics and succinylcholine. It results from mutations in the RYR1 gene, leading to excessive intracellular calcium release, hypermetabolism, and potential multiorgan dysfunction. Prompt recognition and treatment are essential to reduce morbidity and mortality.
We report the case of an 11-year-old female undergoing pancreaticoduodenectomy who developed intraoperative MH following sevoflurane exposure. Early signs included rising end-tidal CO₂ (EtCO₂) despite increased ventilation, followed by tachycardia and metabolic acidosis, with a peak temperature of 37.1°C. MH was suspected approximately two hours after induction, leading to immediate discontinuation of sevoflurane, transition to total intravenous anesthesia, and administration dantrolene. The patient's vital signs stabilized, and the surgery was completed without further complications.
This case underscores the importance of early MH recognition, especially in pediatric patients, who may present with subtle or atypical symptoms. The MH Clinical Grading Score, designed for adults, may not reliably predict MH likelihood in children, necessitating a high index of suspicion. Given the variable presentation of MH in pediatric patients, anesthesiologists must be vigilant in monitoring intraoperative changes. Timely intervention and adherence to MH crisis protocols significantly improve outcomes. Further research is needed to optimize pediatric-specific MH diagnostic criteria and management strategies.
Abbreviations: MH: Malignant Hyperthermia, EtCO2: End Tidal Carbon Dioxide, RYR1: Ryanodine Receptor Gene
Keywords: Malignant Hyperthermia; Anesthesia; Pediatric Anesthesia; General Anesthesia; Dantrolene; RYR1 gene
Citation: Shah S, Dhoon T, Choi Y, Wang G, Rajan G. Pediatric malignant hyperthermia: recognizing early warning signs – a case report. Anaesth. pain intensive care 2025;29(4):446-452. DOI: 10.35975/apic.v29i4.2835
Received: May 09, 2024; Revised: October 26, 2024; Accepted: January 01, 2025
1. INTRODUCTION
Malignant hyperthermia (MH) is a rare pharmacogenetic disorder of skeletal muscle characterized by hypermetabolism secondary to exposure to volatile anesthetic agents and succinylcholine.1 Most affected individuals harbor mutations in the ryanodine receptor RYR1, leading to unregulated accumulation of intracellular calcium and subsequent abnormal skeletal muscle metabolism and contraction.1 The incidence of MH during anesthesia is between 1:100,000 in adults and 1:30,000 in children, and is more likely to present in males than females (2:1).2 The mean age of affected patients is 18.3 years, with 52.1% of all reactions noted to be in children under the age of 15.1
The mortality rate of MH is reported at 80% without treatment and up to 15% even with proper treatment.2,3 Although confirmation can be obtained after an MH event by the halothane-caffeine contracture test or genetic studies, intraoperatively, MH is a purely clinical diagnosis necessitating the need for a high degree of clinical suspicion.4

Figure 1: Intraoperative Anesthesia record.
2. CASE REPORT
An 11-year-old previously healthy female was incidentally diagnosed with a primary solid pseudopapillary epithelial neoplasm of the pancreas and presented for a pancreaticoduodenectomy. Preoperatively, a thoracic epidural was administered at the T8-9 level. Induction of anesthesia was achieved with 30 mg of lidocaine, 80 mg of propofol, 40 mg of rocuronium, and 25 mg of fentanyl, followed by an uneventful intubation. Sevoflurane was subsequently initiated for maintenance of anesthesia. Post-induction, a left radial arterial line and a right internal jugular double-lumen central line were successfully placed without complication.
One hour after sevoflurane was started, there was a rise in end-tidal carbon dioxide (EtCO2) that reached 45 mmHg despite increased ventilatory measures. The changes in EtCO2 preceded a rise in temperature by 15 minutes, which peaked at 37.1° C despite discontinuation of heating elements. Heart rate and blood pressure started increasing approximately 1 hour after sevoflurane initiation, which coincided with the rise in EtCO2; 1.5 hours after induction, initial arterial blood gas showed evidence of acidemia (pH 7.317, PCO2 37.7, PO2 186, HCO3 19.3, BE -7).
Approximately two hours after induction, MH was suspected. In response, sevoflurane was discontinued, and anesthesia was transitioned to total intravenous anesthesia. The anesthesia breathing circuit was replaced and flushed with high fresh gas flow, and charcoal filters (Vapor-Clean; Dynasthetics, Salt Lake City, UT) were applied. The surgical team was promptly notified, and the MH cart was brought to the operating room. The patient received four rounds of dantrolene and sodium bicarbonate (Figure 2), and the MH hotline was contacted for further guidance.

Figure 2: Minute by minute analysis of Heart Rate, MAP ART A-line, EtCO2 (mmHg), Minute Ventilation (liters/minute), and Temperature (°C) during surgery. For details regarding time points 1-9, refer to Table 1.
| Table 1: Timestamps of vitals throughout points of interest during surgery and corresponding clinical indicator score for MH. | ||||||||
| Time Points | HR | MAP-ART | EtCO2 (mmHg) | Minute ventilation (L/min) | Temp (°C) | MH Points Attributed | Total MH Score | Likelihood of MH |
| #1: Induction | 106 | N/A | N/A | N/A | N/A | 0 | 0 | Almost Never |
| #2: HR Elevation | 118 | 63 | 40 | 4.6 | 35.9 | 3 | 3 | Unlikely |
| #3: Temperature Elevation | 90 | 55 | 43 | 4.6 | 36.4 | 15 | 18 | Somewhat less than likely |
| #4: MH Suspected | 109 | 63 | 44 | 5.1 | 36.6 | 30 | 48 | Very Likely |
| #5: Dantrolene Administration | 113 | 74 | 40 | 5.5 | 37 | 5 | 53 | Almost Certain |
| #6: Dantrolene Administration | 119 | 77 | 39 | 8.5 | 37.2 | |||
| #7: Dantrolene Administration | 88 | 77 | 36 | 5 | 37.1 | |||
| #8: Dantrolene Administration | 88 | 77 | 36 | 5 | 37.2 | |||
| #9: Extubation | 102 | 105 | 40 | 5.4 | N/A | |||
Within 15 minutes of discontinuing sevoflurane, the patient's EtCO2, heart rate, and blood pressure returned to normal ranges, while the decrease in temperature followed approximately 45 minutes later. Subsequent arterial blood gas analyses indicated improvement in acidemia. The surgery was paused for 40 minutes to allow for stabilization and then resumed without further complications. Postoperatively, the patient was admitted to the surgical intensive care unit (SICU), where elevated liver function tests were noted, likely as a side effect of dantrolene administration. Her recovery was otherwise uneventful, and she was discharged home on postoperative day 11.
Summarizing Patient Data and Vitals
The patient's vitals were first recorded at 7:17 AM, with sevoflurane induction starting at 7:28 AM. Heart rate spiked from 70 to 123 bpm between 8:34 and 8:53 AM, followed by a temperature increase from 35.7°C to 36.4°C between 9:09 and 9:18 AM. At 9:46 AM, a malignant hyperthermia (MH) alert was triggered due to rising temperature and unresponsive elevated EtCO2. Sevoflurane was discontinued, TIVA initiated, charcoal filters placed, and 80 mg IV Dantrolene administered at 9:59 AM, with a second dose at 10:17 AM. At 10:35 AM, the MH hotline was contacted to report the case. Between 11:06 and 11:12 AM, MAP-ART A-line pressure increased from 68 to 93, peaking at 104. Over the remainder of the operation, the patient’s vitals gradually stabilized. Additional doses of 15 mg IV Dantrolene were given at 4:40 PM and 5:02 PM. The patient was safely extubated at 5:37 PM.
3. DISCUSSION
MH is a rare pharmacogenetic disorder that manifests as a hypermetabolic response during or after exposure to volatile halogenated anesthetic agents and succinylcholine.1 Most affected individuals harbor a mutation in the ryanodine receptor gene RYR1, which is inherited in an autosomal dominant manner.5 Determining the prevalence of MH susceptibility in the general population is challenging due to its infrequent occurrence, unknown family history, and variable expressivity in response to triggering agents.4 Many individuals with this genetic predisposition may be unaware of their susceptibility until an MH episode occurs.
The mutation in MH primarily affects skeletal muscle, leading to an excessive release of intracellular calcium ions that triggers unregulated contraction of sarcomeres. This leads to a hypermetabolic state, resulting in increased carbon dioxide production and oxygen consumption, increased respiratory rate, acidosis, hyperkalemia, rhabdomyolysis, and muscle rigidity.6,7 MH is a condition that can lead to severe organ dysfunction and death, in extreme cases. Even with treatment, the mortality rate associated with MH can be as high as 15%.8,9 This rate may be influenced by various factors, including patient age and muscle mass, but the timeliness of diagnosis and treatment are likely key factors. As such, the ability to diagnose and treat MH swiftly and effectively is paramount in improving patient outcomes.
When MH is suspected, all triggering agents must be discontinued, the patient should be hyperventilated, and dantrolene should be given at an initial dose of 2.5 mg/kg and repeated every 10-15 minutes thereafter until acidosis, hyperthermia, electrolyte abnormalities, and muscle rigidity are resolved.10 Core temperature monitoring and cooling measures should be implemented, as well as laboratory tests, including arterial blood gases and electrolytes, should be closely monitored.11 The MH hotline is a vital resource during an acute crisis. Post-crisis, patients should be observed in the ICU for any recurrence or complications. It also is essential to inform patients and their families about this complication. Additionally, outpatient testing should be arranged to ensure comprehensive evaluation and management.
3.1. Dantrolene Sodium
Dantrolene sodium is a postsynaptic muscle relaxant that inhibits the release of intracellular calcium from the sarcoplasmic reticulum of skeletal muscle cells mediated by the RyR1 receptor, counteracting the effects of MH.8 Since its introduction in the 1970s, dantrolene has revolutionized the treatment of MH, significantly reducing mortality rates from 70–80% to approximately 10–15%.12 Dantrolene is typically supplied as a lyophilized powder with 20 mg of dantrolene sodium and 3000 mg mannitol, with each vial requiring reconstitution with 60 mL of sterile water to reach a pH of approximately 9.5. Dantrolene’s reconstitution can be time-consuming and risks fluid overload, particularly in pediatric patients. The drug has a half-life of 6–8 hours, necessitating repeated dosing to ensure sustained treatment of MH.13,14
Common side effects include muscle weakness, nausea, vomiting, and diarrhea. Rarely, dantrolene can cause liver toxicity, dizziness, fatigue, and injection site reactions, and is contraindicated in patients with hepatic disease. There is no antidote for dantrolene toxicity, so treatment focuses on symptom management.14,15 Despite its relatively cumbersome reconstitution process, dantrolene remains a safe and reliable formulation with minimal risk to the general population.
3.2. Ryanodex
Ryanodex, a newer Dantrolene formulation, offers significant advantages over previous versions, including room temperature storage, reduced dosage requirements, faster reconstitution, and improved solubility. It is available in 20 mL vials which contain 250 mg of dantrolene sodium, requiring just 5 mL of sterile water for reconstitution.15 Its lower volume and rapid administration make it crucial for emergencies like MH. While associated with fewer gastrointestinal issues and lower liver toxicity, it still poses risks like muscle weakness, fatigue, dizziness, flushing, somnolence, dysphonia, and potential hepatotoxicity with prolonged use.14,15 This newer product is also more expensive and has a shorter shelf life than traditional Dantrolene.16
3.3. Pediatric Malignant Hyperthermia
MH is estimated to occur in 1 in 15,000 pediatric anesthetic procedures and 52% of all reactions are noted be in children under the age of 15.1,17 The variability in clinical presentation of MH, especially among age cohorts during childhood, highlights the importance of having a high index of suspicion.
A retrospective analysis of data from the North American Malignant Hyperthermia Registry on patients under 18 years of age demonstrated age-related differences in presentation of MH.17
In infants and young children, initial signs of MH are often subtle, with tachycardia and increased EtCO2 being early indicators.18 The youngest age group (0 to 24 months) displayed less frequent muscle rigidity, but more often showed skin mottling, higher peak lactic acid levels, and lower peak creatine kinase (CK) levels compared to older children.18 Children aged 25 months to 12 years exhibited lower maximum EtCO2 and carbon dioxide tension (PaCO2) compared to the youngest and oldest age groups but were more than three times as likely to experience masseter spasm.18 Adolescents aged 12 to 18 years had higher peak potassium levels, higher maximum temperatures, were more prone to sweating, and took longer to reach their maximum EtCO2 levels.18 As children grow older, classical signs such as masseter muscle rigidity and hyperthermia become more pronounced. Across all age groups, the most observed findings were sinus tachycardia (73.1%), hypercarbia (68.6%), and rapid temperature increase (48.5%).18 These age-related differences in MH presentation may be attributable to differences in body composition, muscle mass, and inherent thermoregulatory changes throughout development.18 Alternate studies also suggest these differences may be attributed to age-related differences in hemodynamics, anatomy, and physiology, such as smaller functional residual capacity and higher oxygen consumption in pediatric patients.19
| Appendix I: Calculator: Clinical Indicators for Malignant Hyperthermia in Adults.17 | |||
| Symptoms | Points Attributed | Pediatric Patient | |
| Rigidity | Generalized muscle rigidity without shivering during or immediately following emergence from inhalational anesthesia | 15 | 0 |
| Masseter muscle spasm shortly following succinylcholine administration | 15 | 0 | |
| Rhabdomyolysis | CK > 20,000 units after anesthesia with succinylcholine | 15 | 0 |
| CK > 10,000 units after anesthesia without succinylcholine | 15 | 0 | |
| Cola-colored urine in perioperative period | 10 | 0 | |
| Urine myoglobin >60 mcg/L | 5 | 0 | |
| Serum myoglobin >170 mcg/L | 5 | 0 | |
| Serum K >6 mEq/L | 3 | 0 | |
| Respiratory acidosis | Controlled ventilation ETCO2 >55 mmHg | 15 | 0 |
| Controlled ventilation PaCO2 >60 mmHg | 15 | 0 | |
| Spontaneous ventilation ETCO2 >60 mmH | 15 | 0 | |
| Spontaneous ventilation PaCO2 >65 mmH | 15 | 0 | |
| Inappropriate hypercarbia (judgement) | 15 | 15 | |
| Inappropriate tachypnea (judgement) | 10 | 15 | |
| Fever | Inappropriate rapid increase in temperature (judgment) | 15 | 15 |
| Inappropriate perioperative temperature >38.8 degrees Celsius (>101.8 F) | 10 | 0 | |
| Tachycardia | Inappropriate sinus tachycardia | 3 | 3 |
| Ventricular tachycardia or ventricular fibrillation | 3 | 0 | |
| Other | Base excess below ~8 mEq/L | 10 | 0 |
| Arterial pH <7.25 | 10 | 0 | |
| Rapid reversal of MH signs with dantrolene | 5 | 5 | |
| Positive family history together with positive personal anesthetic history, not including elevated resting CK | 10 | 0 | |
| Resting elevated serum CK (in patient with positive family history) | 10 | 0 | |
| Total Score | 53 | ||
3.4. Malignant Hyperthermia (MH) Clinical Grading Score
Diagnosis and assessment of MH risk, including in pediatric patients, are often guided by clinical signs and symptoms observed during anesthesia, laboratory tests, and MH Clinical Grading Score. The clinical grading scale for MH, developed by Larach et al., is used to determine the likelihood of an MH event based on clinical presentation in adults. Currently, there is no pediatric specific MH clinical grading score in clinical practice. This scale considers various factors such as muscle rigidity, elevated temperature, acidosis, and hypermetabolism.20
| Appendix II: MH Likelihood Based on Clinical Indicator Score17. The pediatric patient in this case reached a score of 53. | ||
| Points | MH Rank | Likelihood |
| 0 | 1 | Almost never |
| 3-9 | 2 | Unlikely |
| 10-19 | 3 | Somewhat less than likely |
| 20-34 | 4 | Somewhat greater than likely |
| 35-49 | 5 | Very likely |
| 50-108 | 6 | Almost certain |
Although this scale is not specifically designed for pediatric use, it can be applied to patients of all ages to assess the probability of MH, with the understanding that pediatric and adult patients may exhibit different symptoms. For children, particular attention is paid to age-specific presentations, such as those mentioned previously: subtle initial signs in infants, differences in muscle rigidity, and variations in biochemical markers like creatine kinase and lactic acid levels.21
4. CONCLUSION
MH often occurs in patients without a known personal or family history, highlighting the critical importance of early recognition and intervention when noting signs of hemodynamic instability and increases in minute ventilation and EtCO2. The currently available MH grading score used clinically to diagnose MH is only fashioned for adults, which can lead to delayed management in pediatric patients. Awareness of age-related differences between children and adults is crucial for anesthesia providers to offer prompt and appropriate treatment via Dantrolene or Ryanodex, greatly improving patient outcomes.
5. Conflict of interest:
Authors declare no conflict of interest. No external or industry funding was involved.
6. Ethical considerations
Written informed consent was obtained from the father of the child to use this case report for academic interest.
7. Authors contribution
All authors were involved in the management of this patient. Expert advice was sought from the seniors in the preparation of this manuscript.
8. REFERENCES
- Rosenberg H, Pollock N, Schiemann A, Bulger T, Stowell K. Malignant hyperthermia: a review. Orphanet J Rare Dis. 2015;10:93. [PubMed] DOI: 1186/s13023-015-0310-1
- Watt S, McAllister RK. Malignant hyperthermia. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430828/
- Kaur H, Katyal N, Yelam A, Kumar K, Srivastava H, Govindarajan R. Malignant hyperthermia. Mo Med. 2019;116(2):154–9. [PubMed]
- Schneiderbanger D, Johannsen S, Roewer N, Schuster F. Management of malignant hyperthermia: diagnosis and treatment. Ther Clin Risk Manag. 2014;10:355–62. [PubMed] DOI: 2147/TCRM.S47632
- Gonsalves SG, Ng D, Johnston JJ, Teer JK, Stenson PD, Cooper DN, et al. Using exome data to identify malignant hyperthermia susceptibility mutations. Anesthesiology. 2013;119(5):1043–53. [PubMed] DOI: 1097/ALN.0b013e3182a8a8e7
- Larach MG. A primer for diagnosing and managing malignant hyperthermia susceptibility. Anesthesiology. 2018;128:8–10. [PubMed] DOI: 1097/ALN.0000000000001879
- Hopkins PM, Girard T, Dalay S, et al. Malignant hyperthermia 2020: guideline from the Association of Anaesthetists. Anaesthesia. 2021;76(5):655–64. [PubMed] DOI: 1111/anae.15317
- Frassanito L, Sbaraglia F, Piersanti A, Vassalli F, Lucente M, Filetici N, et al. Real evidence and misconceptions about malignant hyperthermia in children: a narrative review. J Clin Med. 2023;12(12):3869. [PubMed] DOI: 3390/jcm12123869
- Malignant Hyperthermia Association of the United States (MHAUS). Managing a crisis. Accessed 2025. Full Text
- Riazi S, Kraeva N, Hopkins PM. Updated guide for the management of malignant hyperthermia. Can J Anaesth. 2018 Jun;65(6):709-721. [PubMed] DOI: 1007/s12630-018-1108-0
- Larach MG, Brandom BW, Allen GC, et al. Clinical presentation, treatment, and complications of malignant hyperthermia in North America from 1987 to 2006. Anesth Analg. 2010;110(2):498–507. [PubMed] DOI: 1213/ANE.0b013e3181c6b9b2
- Hirshey Dirksen SJ, Van Wicklin SA, Mashman DL, Neiderer P, Merritt DR. Developing effective malignant hyperthermia protocols: an evidence-based approach. AORN J. 2013;97(3):329–53. [PubMed] DOI: 1016/j.aorn.2012.12.009
- Malignant Hyperthermia Association of the United States (MHAUS). What should be on an MH cart? Accessed 2025. Full Text
- Ryanodex - dantrolene sodium injection. Accessed 2025. Available from: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8f7b3ac0-604d-4c78-b545-5e0f8ea3d698
- Revonto - dantrolene sodium injection. Accessed 2025. Available from: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=f1650487-32bd-4017-b9c2-b38829a531af
- Khan TH. Malignant hyperthermia, dantrolene and apathy. Anaesth Pain Intensive Care. 2016;20(2):129–30. Full Text
- Brady JE, Sun LS, Rosenberg H, Li G. Prevalence of malignant hyperthermia due to anesthesia in New York State, 2001–2005. Anesth Analg. 2009;109(4):1162–6. [PubMed] DOI: 1213/ane.0b013e3181ac1548
- Nelson P, Litman RS. Malignant hyperthermia in children: an analysis of the North American Malignant Hyperthermia Registry. Anesth Analg. 2014;118(2):369–74. [PubMed] DOI: 1213/ANE.0b013e3182a8fad0
- Otsuki S, Miyoshi H, Mukaida K, Yasuda T, Nakamura R, Tsutsumi YM. Age-specific clinical features of pediatric malignant hyperthermia: a review of 187 cases over 60 years in Japan. Anesth Analg. 2022;135(1):128–35. [PubMed] DOI: 1213/ANE.0000000000005837
- Larach MG, Localio AR, Allen GC, Denborough MA, Ellis FR, Gronert GA, et al. A clinical grading scale to predict malignant hyperthermia susceptibility. Anesthesiology. 1994;80:771–9. [PubMed] DOI: 1097/00000542-199404000-00008
- Lodhi HS, Rafique Z. An unusual presentation of postoperative malignant hyperthermia. Anaesth Pain Intensive Care. 2016;20(2):230–2. Full Text