Ghufran Abd Omran 1, Maha Abdul Saheb Ridha 2, Zainab Hussein Alhillawi 3, Furqan Moein Auda 4, Basim M. Ali 5, Nuha Abd Sahab 6
Author affiliations:
Multiple sclerosis (MS) is a progressive demyelinating disease of the central nervous system with variable geographic prevalence. Symptoms vary widely between patients and over time, often causing neurological impairments due to damage anywhere in the CNS. MS diagnosis can be delayed after initial symptoms. The disease involves axonal damage, oligodendrocyte loss, and demyelination caused by adaptive immunity, primarily antigen-specific B and T cells targeting neural tissue. Activated lymphocytes in secondary lymphoid organs trigger disease onset. Although MS etiology remains unclear, morphological, genetic, and immunological evidence supports an autoimmune basis. Familial MS (FMS) suggests genetic susceptibility and may indicate a more severe course compared to sporadic cases, a distinction important for diagnosis and treatment, especially in Iraqi patients. Biomarkers that monitor disease activity, predict disability progression, and assess treatment response are vital for managing MS. This review summarizes MS prevalence, risk factors, and clinically relevant biomarkers among the Iraqi population.
Abbreviations: MS - Multiple sclerosis; FMS- familial Multiple sclerosis
Keywords: Multiple Sclerosis; Demyelination; Epidemiology; Pathophysiology; Diagnosis; Treatment; Occupational therapy; Physical therapy
Citation: Omran GA, Ridha MAS, Alhillawi ZH, Auda FM, Ali BM, Sahab NA. A clinical perspective on multiple sclerosis for enhancing diagnosis and treatment protocols. Anaesth. pain intensive care 2025;29(4):418-424. DOI: 10.35975/apic.v29i4.2830
Received: March 20, 2025; Revised: May 12, 2024; Accepted: May 23, 2025
Multiple sclerosis (MS) is a chronic autoimmune disease of the central nervous system (CNS) characterized by demyelination, neurodegeneration, and inflammation.1,2 Pathological features include reactive gliosis, demyelination, and loss of myelin-producing oligodendrocytes, resulting in neuroaxonal degeneration.1 The disease typically presents between ages 20 and 40, affecting women about twice as often as men, and is marked by isolated lesions in the brain and spinal cord.2,3 A hallmark of MS is the breakdown of the myelin sheath surrounding axons, leading to synaptic loss and impaired neural conduction.1,4
MS is thought to be triggered by an autoimmune response, often involving CD4⁺ T helper cells reacting to CNS antigens, possibly after viral infections.2,5 Disruption of the blood-brain barrier (BBB), caused by genetic and environmental factors such as vitamin D deficiency, microbiota imbalance, or diet, allows infiltration of immune cells and contributes to lesion formation.4,6 Therapeutic strategies targeting fibrinogen or adhesion molecules aim to restore BBB integrity.6
Symptoms vary widely among individuals and include fatigue, cognitive impairment, tremor, visual disturbances, and bowel or bladder dysfunction, often fluctuating over time.7 As the disease progresses, patients may experience accumulating disability.8
The etiology of MS is multifactorial, involving genetic susceptibility, environmental exposures, and immune dysregulation.2,5,9 Key immune players include autoreactive T and B lymphocytes, as well as microglia, which promote inflammation and lesion formation.2,5 B cells also contribute through antibody production and cytokine release.5,10 Migration of lymphocytes across a compromised BBB and reduced function of regulatory T cells play pivotal roles in the autoimmune cascade.5
According to the World Health Organization, about 2.3 million people worldwide had multiple sclerosis (MS) in 2013. In Arab countries, MS prevalence ranges from 4 to 8 cases per 100,000. The epidemiology of MS has shifted in recent decades in Iran and other Middle Eastern countries, including western Asia and Egypt (excluding Caucasia). By 2020, the global MS population rose to 2.8 million, a 30% increase since 2013, with a prevalence of 35.9 per 100,000.1 Nearly one million people in the US live with MS, which is more common in northern regions. It is estimated that neurodegenerative diseases affect 50 million globally and they are expected to rise to 115 million by 2050.11
In Iraq, comprehensive epidemiological studies began after establishing the MS Clinic in Baghdad. The rising number of cases may reflect improved disease awareness, diagnostic capacity, and more specialists. Over the past 30 years, MS prevalence in Iraq has steadily increased, though many cases are recent. The higher female-to-male ratio in the West may partly explain this trend, alongside changing environmental factors like westernized lifestyles, as seen in Japan.12
Iraq has undergone significant socio-economic changes since the 1970s, potentially influencing MS prevalence.13 Although overall prevalence remains lower than neighboring countries, incidence rates are higher. Visual symptoms commonly present initially, and relapsing-remitting MS (RRMS) is the most frequent type. A nationwide study reported a prevalence of 11.73 per 100,000, with 0.26 per 100,000 in children under 12.14 In Ninevah Governorate, prevalence is 10.3 per 100,000, with an incidence ranging from 4.6 to 11.57 per million annually. The average age of symptom onset is 27.7 years, with women diagnosed twice as often as men.14 MS is most prevalent among Caucasians of northern European descent, followed by African-Americans, Asians, and Latinos.15 It is rare among Norwegian Lapps, Inuit, Australian Aborigines, and New Zealanders. Regions farther from the equator, such as northern US, Europe, southern Canada, New Zealand, and Australia, report higher prevalence. Equatorial regions like Southeast Asia and sub-Saharan Africa show lower rates, though exceptions exist, such as the Inuit in Canada and Sardinians in Italy.15
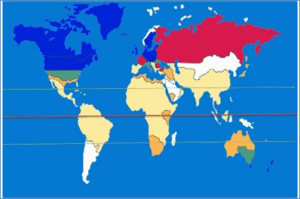
Figure: 1: The prevalence of MS in different regions of the world.13
MS symptoms can vary significantly from person to person, throughout the course of the illness, and depending on the location and extent of the damaged nerve fibers. The sensory and cognitive symptoms of MS demonstrate its profound impact. While some individuals with severe MS may go for extended periods of remission without developing any new symptoms, others may lose the ability to walk on their own or at all. In multiple sclerosis, the transmission of nerve impulses between the brain, spinal cord, and the rest of the body is disrupted by damage to the myelin sheath surrounding the nerve fibers in the central nervous system (CNS) as well as to the nerve fibers themselves. The symptoms of multiple sclerosis (MS) are caused by disrupted nerve signals and vary from person to person and throughout time for each individual based on the location and timing of the damage. Although virtually every neurological symptom or sign might be present in an MS patient, autonomic, ocular, motor, and sensory issues are the most prevalent. Some or several of the following symptoms could be present: fatigue and weakness, pain and spasms in the body, vision problems, speech problems, sensory problems, motor problems, cognitive problems, psychological problems, postural problems, metabolic problems, and convulsions.16
The exact cause of MS remains unclear, but current research suggests it arises from a combination of genetic susceptibility and environmental factors from childhood to early adulthood. Key environmental contributors include tobacco use; Epstein-Barr virus (EBV) infection, vitamin D deficiency, and birth season.17 Individuals seronegative for EBV have a very low MS risk, whereas late-life EBV infection is linked to higher prevalence. Genome-wide studies have identified 52 risk alleles for MS, with the strongest association to HLA-DRB1 status.18
MS risk factors are complex and multifactorial. No single factor alone causes MS; both genetic predisposition and environmental exposures contribute. Smoking and obesity are associated with increased MS risk but are not sole causes, as many MS patients are neither obese nor smokers. Migration studies suggest environmental factors may outweigh genetics in overall MS risk (Fig 2).19 However, familial clustering confirms genetics plays a significant role.
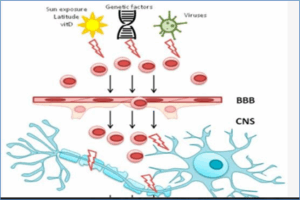
Figure 2: Various MS risk factors.17
MHC class II molecules present unknown antigens to T-cells, triggering autoreactive inflammation. MS involves inflammation, demyelination, axonal damage, and plaques mainly in gray matter, spinal cord, brainstem, corpus callosum, cerebellar peduncles, white matter near ventricles, and optic nerves. These plaques vary between progressive and relapsing-remitting MS, reflecting differences in demyelination and oligodendrocyte damage. MS is an autoimmune disease where autoreactive immune cells cross the blood-brain barrier (BBB). Normally, central and peripheral tolerance eliminate or suppress these cells, but failure occurs due to resistant autoreactive T cells and dysfunctional regulatory T cells. Genetic and environmental factors influence this activation.20 Key T cell subsets in MS are CD8+, CD4+ Th1, and Th17 cells, producing cytokines like interferon-gamma, IL-17, and GM-CSF.21 B cells contribute to MS, indicated by increased immunoglobulins and oligoclonal bands (OCBs) in cerebrospinal fluid (CSF). Memory B cells predominate in brain and CSF, showing clonal expansion and antibody production, underlying intrathecal immunoglobulin synthesis.22,23 Inflammation is central to MS pathophysiology, driven by peripheral autoreactive T cells, possibly activated by bacterial/viral antigens or molecular mimicry. These T cells enter the CNS by binding to endothelial VCAM-1 and integrins. They interact with MHC II on antigen-presenting cells, and promote myelin breakdown.17 This recruits other immune cells and myelin-targeting autoantibodies, amplifying damage.24 The resulting cytokines and antibodies disrupt the BBB, allowing more autoreactive cells to infiltrate and induce neuronal dysfunction.24 These processes cause axonal injury and progressive myelin loss (Figure 3).
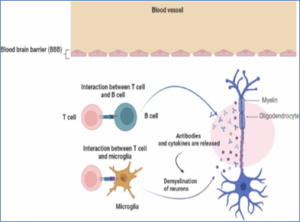
Figure 3: Pathophysiology of MS.21
MS is classified by disease progression into several types: relapsing-remitting MS (RRMS), primary progressive MS (PPMS), secondary progressive MS (SPMS), clinically isolated syndrome (CIS), progressive relapsing MS (now considered PPMS with exacerbation), benign, and fulminant. Initial presentations vary: 5% are progressive-relapsing, 15-20% primary progressive, and 70-80% relapsing-remitting.25 The four main clinical subtypes are:
Diagnosis of multiple sclerosis (MS) relies on clinical evaluation, MRI, and cerebrospinal fluid (CSF) analysis to detect characteristic lesions and immunological markers.28 Routine blood tests exclude other conditions. Diagnosis can be delayed after symptom onset. MRI is crucial due to its high sensitivity and specificity for MS detection and monitoring.MS presents with diverse symptoms—visual, motor, sensory, cognitive, and urinary—due to multifocal CNS lesions, often following a relapsing-remitting course. Disability is measured by the Expanded Disability Status Scale (EDSS) in both familial and sporadic cases.29 Delayed diagnosis reduces early access to disease-modifying therapies. Lumbar puncture and MRI remain primary diagnostic tools, with new criteria facilitating earlier detection. Emerging biomarkers improve diagnosis and prognosis: CHI3L1, linked to inflammation and tissue remodeling, is detected by ELISA and aids in monitoring nerve damage.30 Elevated serum GDF-15 correlates with disease stability. Other candidates include nitric oxide, heat shock proteins, tau, uric acid, neurofilament light chain, and interleukins.31 Although some biomarkers show promise, larger studies are needed to validate their clinical utility. Biomarkers are key to advancing MS care, aiming for accurate diagnosis, prognosis, therapy response prediction, and non-invasive, cost-effective screening of at-risk populations.32
8.1. Medications
There is currently no cure for MS despite extensive research. Treatment includes injectable drugs like Glatiramer Acetate and Interferon Beta, oral medications such as Dimethyl Fumarate and Fingolimod, and monoclonal antibodies including Natalizumab, Ocrelizumab, and Alemtuzumab.33 Oral DMT Siponimod has shown promise in slowing disability progression in secondary progressive MS. New therapies continue to emerge driven by biomarker research.
8.2 Physical and Occupational Therapy
Occupational therapy and rehabilitation are vital for MS management, with advances like telerehabilitation, robot-assisted gait, virtual reality, and brain stimulation techniques enhancing patient care.34,35 Rehabilitation improves neurological function, reduces disability, and supports social integration from early to advanced disease stages. Neurofilament light protein, measured in CSF, is a promising biomarker for predicting disease progression.
Symptomatic treatment, disease-modifying therapies, and relapse management are core strategies. Cannabinoids reduce pain, spasms, and tremors while offering neuroprotection by modulating the immune system.36 Though no drug halts MS completely, immunosuppressants slow progression. Rehabilitation remains essential to improve function and quality of life, with tailored plans involving occupational, speech, respiratory, and physical therapies such as balance and gait training.37,38
Chronic demyelinating MS remains common, complicating understanding, diagnosis, and treatment. A novel nano sandwich ELISA using photo-irradiated gold nanoparticles measures CHI3L1 in serum and cerebrospinal fluid, enhancing sensitivity and shortening detection time compared to traditional methods. CHI3L1 is a promising biomarker for MS diagnosis, progression, and treatment response. Machine learning models analyzing clinical, demographic, and sensor data help predict MS progression and differentiate benign from progressive forms, improving automated diagnosis and prognostication.39,40 New MRI markers, like the central vein sign and paramagnetic rim lesions, could increase diagnostic specificity, though further validation is needed before clinical use. Artificial intelligence (AI) may augment MRI interpretation by detecting subtle features beyond human perception, enhancing diagnosis and patient classification.41 MRI diagnosis is time-consuming and error-prone. AI-driven computer-aided diagnosis systems using traditional machine learning and deep learning (DL) show promise; DL automatically learns features for better classification.42 Future research should focus on expanding datasets, developing novel DL models, and advancing rehabilitation programs for MS patients. MRI detects myelin damage—an MS hallmark—by imaging water accumulation where the hydrophobic myelin layer is lost. This technique enables visualization of brain injury extent, correlating with patient symptoms.43
In conclusion, recent research indicates that the frequency of MS in Iraq is moderate to high, and it has recently sharply increased. This review concludes that both hereditary and environmental variables have a role in the development of multiple sclerosis. Finding the best MS biomarker will help doctors choose early treatment options for newly diagnosed patients, create individualized treatment plans for patients, and significantly reduce clinical trial costs, but the disease's heterogeneity in MS makes finding biomarkers identification more difficult. As knowledge of the pathophysiology and progression of MS has increased, remarkable strides have been made in its treatment. Relapsing illness and focal brain inflammation are now almost entirely under control thanks to the introduction of very efficient treatments. Nevertheless, because existing treatments only partially guard against the neurodegenerative aspect of MS, there is still a need for effective therapy of progression.
11. Acknowledgement
We thank all the participants who were enrolled in this study.
12. Conflict of interest
The authors indicate no conflicts of interest.
13. Authors’ contribution
All authors took equal part in the conduct of the study and in the preparation of this manuscript.
Author affiliations:
- Ghufran Abd Omran, Department of Clinical Chemistry, Hammurabi College of Medicine, University of Babylon, Babylon, Iraq; Email:
ham2977.qufran.abad@uobabylon.com - Maha Abdul Saheb Ridha, Dept of Chemistry, Faculty of Science, University of Kufa, Najaf, Iraq; Email: mahaa.muhamad@uokufa.edu.iq
- Zainab Hussein Alhillawi, Dept of Chemistry, Faculty of Science, University of Kufa, Najaf, Iraq; Email: zainab.alhillawii@uokufa.edu.iq
- Furqan Moein Auda, Dept of Chemistry, Faculty of Science, University of Kufa, Najaf, Iraq; Email: furqan.chmalia@uokufa.edu.iq
- Basim M. Ali, Dept of Chemistry, Faculty of Science, University of Kufa, Najaf, Iraq; Email: basimm.radhi11@uokufa.edu.iq
- Nuha Abd Sahab, Department of Chemistry, Faculty of Science, University of Kufa, Najaf, Iraq; Email: nuha.alandaleb@uokufa.edu.iq
ABSTRACT
Multiple sclerosis (MS) is a progressive demyelinating disease of the central nervous system with variable geographic prevalence. Symptoms vary widely between patients and over time, often causing neurological impairments due to damage anywhere in the CNS. MS diagnosis can be delayed after initial symptoms. The disease involves axonal damage, oligodendrocyte loss, and demyelination caused by adaptive immunity, primarily antigen-specific B and T cells targeting neural tissue. Activated lymphocytes in secondary lymphoid organs trigger disease onset. Although MS etiology remains unclear, morphological, genetic, and immunological evidence supports an autoimmune basis. Familial MS (FMS) suggests genetic susceptibility and may indicate a more severe course compared to sporadic cases, a distinction important for diagnosis and treatment, especially in Iraqi patients. Biomarkers that monitor disease activity, predict disability progression, and assess treatment response are vital for managing MS. This review summarizes MS prevalence, risk factors, and clinically relevant biomarkers among the Iraqi population.
Abbreviations: MS - Multiple sclerosis; FMS- familial Multiple sclerosis
Keywords: Multiple Sclerosis; Demyelination; Epidemiology; Pathophysiology; Diagnosis; Treatment; Occupational therapy; Physical therapy
Citation: Omran GA, Ridha MAS, Alhillawi ZH, Auda FM, Ali BM, Sahab NA. A clinical perspective on multiple sclerosis for enhancing diagnosis and treatment protocols. Anaesth. pain intensive care 2025;29(4):418-424. DOI: 10.35975/apic.v29i4.2830
Received: March 20, 2025; Revised: May 12, 2024; Accepted: May 23, 2025
1. INTRODUCTION
Multiple sclerosis (MS) is a chronic autoimmune disease of the central nervous system (CNS) characterized by demyelination, neurodegeneration, and inflammation.1,2 Pathological features include reactive gliosis, demyelination, and loss of myelin-producing oligodendrocytes, resulting in neuroaxonal degeneration.1 The disease typically presents between ages 20 and 40, affecting women about twice as often as men, and is marked by isolated lesions in the brain and spinal cord.2,3 A hallmark of MS is the breakdown of the myelin sheath surrounding axons, leading to synaptic loss and impaired neural conduction.1,4
MS is thought to be triggered by an autoimmune response, often involving CD4⁺ T helper cells reacting to CNS antigens, possibly after viral infections.2,5 Disruption of the blood-brain barrier (BBB), caused by genetic and environmental factors such as vitamin D deficiency, microbiota imbalance, or diet, allows infiltration of immune cells and contributes to lesion formation.4,6 Therapeutic strategies targeting fibrinogen or adhesion molecules aim to restore BBB integrity.6
Symptoms vary widely among individuals and include fatigue, cognitive impairment, tremor, visual disturbances, and bowel or bladder dysfunction, often fluctuating over time.7 As the disease progresses, patients may experience accumulating disability.8
The etiology of MS is multifactorial, involving genetic susceptibility, environmental exposures, and immune dysregulation.2,5,9 Key immune players include autoreactive T and B lymphocytes, as well as microglia, which promote inflammation and lesion formation.2,5 B cells also contribute through antibody production and cytokine release.5,10 Migration of lymphocytes across a compromised BBB and reduced function of regulatory T cells play pivotal roles in the autoimmune cascade.5
2. EPIDEMIOLOGY
According to the World Health Organization, about 2.3 million people worldwide had multiple sclerosis (MS) in 2013. In Arab countries, MS prevalence ranges from 4 to 8 cases per 100,000. The epidemiology of MS has shifted in recent decades in Iran and other Middle Eastern countries, including western Asia and Egypt (excluding Caucasia). By 2020, the global MS population rose to 2.8 million, a 30% increase since 2013, with a prevalence of 35.9 per 100,000.1 Nearly one million people in the US live with MS, which is more common in northern regions. It is estimated that neurodegenerative diseases affect 50 million globally and they are expected to rise to 115 million by 2050.11
In Iraq, comprehensive epidemiological studies began after establishing the MS Clinic in Baghdad. The rising number of cases may reflect improved disease awareness, diagnostic capacity, and more specialists. Over the past 30 years, MS prevalence in Iraq has steadily increased, though many cases are recent. The higher female-to-male ratio in the West may partly explain this trend, alongside changing environmental factors like westernized lifestyles, as seen in Japan.12
Iraq has undergone significant socio-economic changes since the 1970s, potentially influencing MS prevalence.13 Although overall prevalence remains lower than neighboring countries, incidence rates are higher. Visual symptoms commonly present initially, and relapsing-remitting MS (RRMS) is the most frequent type. A nationwide study reported a prevalence of 11.73 per 100,000, with 0.26 per 100,000 in children under 12.14 In Ninevah Governorate, prevalence is 10.3 per 100,000, with an incidence ranging from 4.6 to 11.57 per million annually. The average age of symptom onset is 27.7 years, with women diagnosed twice as often as men.14 MS is most prevalent among Caucasians of northern European descent, followed by African-Americans, Asians, and Latinos.15 It is rare among Norwegian Lapps, Inuit, Australian Aborigines, and New Zealanders. Regions farther from the equator, such as northern US, Europe, southern Canada, New Zealand, and Australia, report higher prevalence. Equatorial regions like Southeast Asia and sub-Saharan Africa show lower rates, though exceptions exist, such as the Inuit in Canada and Sardinians in Italy.15

Figure: 1: The prevalence of MS in different regions of the world.13
3. CLINICAL FEATURES
MS symptoms can vary significantly from person to person, throughout the course of the illness, and depending on the location and extent of the damaged nerve fibers. The sensory and cognitive symptoms of MS demonstrate its profound impact. While some individuals with severe MS may go for extended periods of remission without developing any new symptoms, others may lose the ability to walk on their own or at all. In multiple sclerosis, the transmission of nerve impulses between the brain, spinal cord, and the rest of the body is disrupted by damage to the myelin sheath surrounding the nerve fibers in the central nervous system (CNS) as well as to the nerve fibers themselves. The symptoms of multiple sclerosis (MS) are caused by disrupted nerve signals and vary from person to person and throughout time for each individual based on the location and timing of the damage. Although virtually every neurological symptom or sign might be present in an MS patient, autonomic, ocular, motor, and sensory issues are the most prevalent. Some or several of the following symptoms could be present: fatigue and weakness, pain and spasms in the body, vision problems, speech problems, sensory problems, motor problems, cognitive problems, psychological problems, postural problems, metabolic problems, and convulsions.16
4. ETIOLOGY & RISK FACTORS
The exact cause of MS remains unclear, but current research suggests it arises from a combination of genetic susceptibility and environmental factors from childhood to early adulthood. Key environmental contributors include tobacco use; Epstein-Barr virus (EBV) infection, vitamin D deficiency, and birth season.17 Individuals seronegative for EBV have a very low MS risk, whereas late-life EBV infection is linked to higher prevalence. Genome-wide studies have identified 52 risk alleles for MS, with the strongest association to HLA-DRB1 status.18
MS risk factors are complex and multifactorial. No single factor alone causes MS; both genetic predisposition and environmental exposures contribute. Smoking and obesity are associated with increased MS risk but are not sole causes, as many MS patients are neither obese nor smokers. Migration studies suggest environmental factors may outweigh genetics in overall MS risk (Fig 2).19 However, familial clustering confirms genetics plays a significant role.

Figure 2: Various MS risk factors.17
5. PATHOPHYSIOLOGY
MHC class II molecules present unknown antigens to T-cells, triggering autoreactive inflammation. MS involves inflammation, demyelination, axonal damage, and plaques mainly in gray matter, spinal cord, brainstem, corpus callosum, cerebellar peduncles, white matter near ventricles, and optic nerves. These plaques vary between progressive and relapsing-remitting MS, reflecting differences in demyelination and oligodendrocyte damage. MS is an autoimmune disease where autoreactive immune cells cross the blood-brain barrier (BBB). Normally, central and peripheral tolerance eliminate or suppress these cells, but failure occurs due to resistant autoreactive T cells and dysfunctional regulatory T cells. Genetic and environmental factors influence this activation.20 Key T cell subsets in MS are CD8+, CD4+ Th1, and Th17 cells, producing cytokines like interferon-gamma, IL-17, and GM-CSF.21 B cells contribute to MS, indicated by increased immunoglobulins and oligoclonal bands (OCBs) in cerebrospinal fluid (CSF). Memory B cells predominate in brain and CSF, showing clonal expansion and antibody production, underlying intrathecal immunoglobulin synthesis.22,23 Inflammation is central to MS pathophysiology, driven by peripheral autoreactive T cells, possibly activated by bacterial/viral antigens or molecular mimicry. These T cells enter the CNS by binding to endothelial VCAM-1 and integrins. They interact with MHC II on antigen-presenting cells, and promote myelin breakdown.17 This recruits other immune cells and myelin-targeting autoantibodies, amplifying damage.24 The resulting cytokines and antibodies disrupt the BBB, allowing more autoreactive cells to infiltrate and induce neuronal dysfunction.24 These processes cause axonal injury and progressive myelin loss (Figure 3).

Figure 3: Pathophysiology of MS.21
6. COMMON CLINICAL PATTERNS
MS is classified by disease progression into several types: relapsing-remitting MS (RRMS), primary progressive MS (PPMS), secondary progressive MS (SPMS), clinically isolated syndrome (CIS), progressive relapsing MS (now considered PPMS with exacerbation), benign, and fulminant. Initial presentations vary: 5% are progressive-relapsing, 15-20% primary progressive, and 70-80% relapsing-remitting.25 The four main clinical subtypes are:
- Clinically Isolated Syndrome (CIS): The first MS episode with no prior symptoms or infection, characterized by sudden symptoms lasting days to weeks and persisting for at least 24 hours.26
- Relapsing-Remitting MS (RRMS): The most common form (85-90%), marked by episodes of symptom worsening followed by remission, with symptoms typically improving within weeks or months.27
- Primary Progressive MS (PPMS): Characterized by steady neurological decline without relapses.26
- Secondary Progressive MS (SPMS): Develops after RRMS, with gradual worsening neurological function, possibly with or without relapses or MRI changes; active SPMS involves relapses/MRI abnormalities, inactive does not.26
7. DIAGNOSIS
Diagnosis of multiple sclerosis (MS) relies on clinical evaluation, MRI, and cerebrospinal fluid (CSF) analysis to detect characteristic lesions and immunological markers.28 Routine blood tests exclude other conditions. Diagnosis can be delayed after symptom onset. MRI is crucial due to its high sensitivity and specificity for MS detection and monitoring.MS presents with diverse symptoms—visual, motor, sensory, cognitive, and urinary—due to multifocal CNS lesions, often following a relapsing-remitting course. Disability is measured by the Expanded Disability Status Scale (EDSS) in both familial and sporadic cases.29 Delayed diagnosis reduces early access to disease-modifying therapies. Lumbar puncture and MRI remain primary diagnostic tools, with new criteria facilitating earlier detection. Emerging biomarkers improve diagnosis and prognosis: CHI3L1, linked to inflammation and tissue remodeling, is detected by ELISA and aids in monitoring nerve damage.30 Elevated serum GDF-15 correlates with disease stability. Other candidates include nitric oxide, heat shock proteins, tau, uric acid, neurofilament light chain, and interleukins.31 Although some biomarkers show promise, larger studies are needed to validate their clinical utility. Biomarkers are key to advancing MS care, aiming for accurate diagnosis, prognosis, therapy response prediction, and non-invasive, cost-effective screening of at-risk populations.32
8. CURRENT TREATMENT OPTIONS
8.1. Medications
There is currently no cure for MS despite extensive research. Treatment includes injectable drugs like Glatiramer Acetate and Interferon Beta, oral medications such as Dimethyl Fumarate and Fingolimod, and monoclonal antibodies including Natalizumab, Ocrelizumab, and Alemtuzumab.33 Oral DMT Siponimod has shown promise in slowing disability progression in secondary progressive MS. New therapies continue to emerge driven by biomarker research.
8.2 Physical and Occupational Therapy
Occupational therapy and rehabilitation are vital for MS management, with advances like telerehabilitation, robot-assisted gait, virtual reality, and brain stimulation techniques enhancing patient care.34,35 Rehabilitation improves neurological function, reduces disability, and supports social integration from early to advanced disease stages. Neurofilament light protein, measured in CSF, is a promising biomarker for predicting disease progression.
Symptomatic treatment, disease-modifying therapies, and relapse management are core strategies. Cannabinoids reduce pain, spasms, and tremors while offering neuroprotection by modulating the immune system.36 Though no drug halts MS completely, immunosuppressants slow progression. Rehabilitation remains essential to improve function and quality of life, with tailored plans involving occupational, speech, respiratory, and physical therapies such as balance and gait training.37,38
9. FUTURE STUDIES AND DEVELOPMENTS
Chronic demyelinating MS remains common, complicating understanding, diagnosis, and treatment. A novel nano sandwich ELISA using photo-irradiated gold nanoparticles measures CHI3L1 in serum and cerebrospinal fluid, enhancing sensitivity and shortening detection time compared to traditional methods. CHI3L1 is a promising biomarker for MS diagnosis, progression, and treatment response. Machine learning models analyzing clinical, demographic, and sensor data help predict MS progression and differentiate benign from progressive forms, improving automated diagnosis and prognostication.39,40 New MRI markers, like the central vein sign and paramagnetic rim lesions, could increase diagnostic specificity, though further validation is needed before clinical use. Artificial intelligence (AI) may augment MRI interpretation by detecting subtle features beyond human perception, enhancing diagnosis and patient classification.41 MRI diagnosis is time-consuming and error-prone. AI-driven computer-aided diagnosis systems using traditional machine learning and deep learning (DL) show promise; DL automatically learns features for better classification.42 Future research should focus on expanding datasets, developing novel DL models, and advancing rehabilitation programs for MS patients. MRI detects myelin damage—an MS hallmark—by imaging water accumulation where the hydrophobic myelin layer is lost. This technique enables visualization of brain injury extent, correlating with patient symptoms.43
10. CONCLUSIONS
In conclusion, recent research indicates that the frequency of MS in Iraq is moderate to high, and it has recently sharply increased. This review concludes that both hereditary and environmental variables have a role in the development of multiple sclerosis. Finding the best MS biomarker will help doctors choose early treatment options for newly diagnosed patients, create individualized treatment plans for patients, and significantly reduce clinical trial costs, but the disease's heterogeneity in MS makes finding biomarkers identification more difficult. As knowledge of the pathophysiology and progression of MS has increased, remarkable strides have been made in its treatment. Relapsing illness and focal brain inflammation are now almost entirely under control thanks to the introduction of very efficient treatments. Nevertheless, because existing treatments only partially guard against the neurodegenerative aspect of MS, there is still a need for effective therapy of progression.
11. Acknowledgement
We thank all the participants who were enrolled in this study.
12. Conflict of interest
The authors indicate no conflicts of interest.
13. Authors’ contribution
All authors took equal part in the conduct of the study and in the preparation of this manuscript.
14. REFERENCES
- Bjørklund G, Wallace DR, Hangan T, Butnariu M, Gurgas L, Peana MJ. Cerebral iron accumulation in multiple sclerosis: Pathophysiology and therapeutic implications. Autoimmun Rev. 2025;24:103741. PubMed DOI: 1016/j.autrev.2025.103741
- Algburi SM, Ebdan WR, Ali RM. Prevalence of primary headache in Iraqi patients with relapsing remitting multiple sclerosis. Mult Scler Relat Disord. 2024;92:106036.
- Salim AA, Ali SH, Hussain AM, Ibrahim WN. Electroencephalographic evidence of gray matter lesions among multiple sclerosis patients: A case-control study. Med (Baltimore). 2021;100(33):e27001. PubMed DOI: 1097/MD.0000000000027001
- Catalá-López F, Tejedor-Romero L, Driver JA, Hutton B, Sánchez-Ortí JV, Ridao M, et al. Risk of cancer development associated with disease-modifying therapies for multiple sclerosis: Study protocol for a systematic review and meta-analysis. Syst Rev. 2024;13(1):263. PubMed DOI: 1186/s13643-024-02677-z
- Di Filippo M, Gaetani L, Centonze D, Hegen H, Kuhle J, Teunissen CE, et al. Fluid biomarkers in multiple sclerosis: From current to future applications. Lancet Reg Health Eur. 2024;44:101009. PubMed DOI: 1016/j.lanepe.2024.101009
- Simonsen CS, Flemmen HØ, Lauritzen T, Berg-Hansen P, Moen SM, Celius EG, et al. The diagnostic value of IgG index versus oligoclonal bands in cerebrospinal fluid of patients with multiple sclerosis. Mult Scler J Exp Transl Clin. 2020;6(1):2055217319901291. PubMed DOI: 1177/2055217319901291
- Bisht P, Rathore C, Rathee A, Kabra A. Astrocyte activation and drug target in pathophysiology of multiple sclerosis. In: Dhawan A, editor. Neuroprotection: Methods and Protocols. New York: Springer; 2024. p. 431-55.
- Ramaglia V, Rojas O, Naouar I, Gommerman JL. The ins and outs of central nervous system inflammation—Lessons learned from multiple sclerosis. Annu Rev Immunol. 2021;39:199-226. PubMed DOI: 1146/annurev-immunol-093019-124155
- Haase S, Linker RA. Inflammation in multiple sclerosis. Ther Adv Neurol Disord. 2021;14:17562864211007687. PubMed DOI: 1177/17562864211007687
- Haki M, Al-Biati HA, Al-Tameemi ZS, Ali IS, Al-Hussaniy HA. Review of multiple sclerosis: Epidemiology, etiology, pathophysiology, and treatment. Med (Baltimore). 2024;103(8):e37297. PubMed DOI: 1097/MD.0000000000037297
- Cengiz EK, Fırat YE, Onur E, Denkçeken T. The role of E2F transcription factor 1 protein in multiple sclerosis (MS): A comparative study of serum levels in MS patients and healthy controls. Cureus. 2025;17(1):e76889. PubMed DOI: 7759/cureus.76889
- Sahebi R, Amiri M, Jami MS. Multiple sclerosis in Iran. Epidemiol Health Sys J. 2018;5(1):30-3. DOI: 15171/ijer.2018.07
- Walton C, King R, Rechtman L, Kaye W, Leray E, Marrie RA, et al. Rising prevalence of multiple sclerosis worldwide: Insights from the Atlas of MS. Mult Scler. 2020;26(14):1816-21. PubMed DOI: 1177/1352458520970841
- Wallin MT, Culpepper WJ, Campbell JD, Nelson LM, Langer-Gould A, Marrie RA, et al. The prevalence of MS in the United States: A population-based estimate using health claims data. Neurology. 2019;92(10):e1029-40. PubMed DOI: 1212/WNL.0000000000007035
- Falah Y, Al-Araji AJ. Multiple sclerosis in Iraq: History, epidemiology and the future. Iraq J Med Sci. 2014;1(1):62-8.
- Ghasemi N, Razavi S, Nikzad E. Multiple sclerosis: Pathogenesis, symptoms, diagnosis and cell-based therapy. Cell J. 2017;19(1):1-10. PubMed DOI: 22074/cellj.2016.4867
- Beard K, Srivastava S, Sharma K, Jaiswal S, Reddy SP, Lisak RP, et al. Epidemiology, epigenetics, and etiological factors in multiple sclerosis. In: Clinical Aspects of Multiple Sclerosis Essentials and Current Updates. Elsevier; 2024. p. 67-96.
- Canto E, Oksenberg JR. Multiple sclerosis genetics. Mult Scler J. 2018;24(1):75-9. PubMed DOI: 1177/1352458517737371
- Mescheriakova JY, van Nierop GP, van der Eijk AA, Kreft KL, Hintzen RQ. EBNA-1 titer gradient in families with multiple sclerosis indicates a genetic contribution. Neurol Neuroimmunol Neuroinflamm. 2020;7(6):e872. PubMed DOI: 1212/NXI.0000000000000872
- Van Langelaar J, Rijvers L, Smolders J, Van Luijn MM. B and T cells driving multiple sclerosis: Identity, mechanisms and potential triggers. Front Immunol. 2020;11:760. PubMed DOI: 3389/fimmu.2020.00760
- Zéphir H. Progress in understanding the pathophysiology of multiple sclerosis. Rev Neurol (Paris). 2018;174(6):358-63. PubMed DOI: 1016/j.neurol.2018.03.006
- Ward M, Goldman MD. Epidemiology and pathophysiology of multiple sclerosis. Continuum (Minneap Minn). 2022;28(4):988-1005. PubMed DOI: 1212/CON.0000000000001136
- Ochi H. Role of B cells in the pathogenesis of multiple sclerosis. Cen Eur J Immunol. 2021;12(4):220-7. DOI: 1111/cen3.12671
- Al-Zamali SKS, Al-Khafaji JKT, Mankhi AA. Comparison of interleukin 17A and interleukin-18 cytokines during active and latent TB infection in Iraqi patients. HIV AIDS (Auckl). 2023;23(2):012-7. Full Text
- Shah A, Panchal V, Patel K, Alimohamed Z, Kaka N, Sethi Y, et al. Pathogenesis and management of multiple sclerosis revisited. Dis Mon. 2023;69(9):101497. PubMed DOI: 1016/j.disamonth.2022.101497
- Goldschmidt C, McGinley MP. Advances in the treatment of multiple sclerosis. Neurol Clin. 2021;39(1):21-33. PubMed DOI: 1016/j.ncl.2020.09.002
- Khaleel AM, Al-Mashetah SAR. Clinical and radiological features of familial multiple sclerosis in comparison to sporadic multiple sclerosis in Iraqi patients. Basrah J Biol Sci. 2024;4(1):1-6.
- Ahmed ZO, Dema HK, Yehia OT, Noel KI. Apparent diffusion coefficient in the diagnosis and follow-up of multiple sclerosis: Role of magnetic resonance imaging. Adv J Med Sci. 2024;7(1):133-8. DOI: 54133/ajms.v7i1.1114
- Hunter SF. Overview and diagnosis of multiple sclerosis. Am J Manag Care. 2016;22(6 Suppl):S141-50. PubMed
- Schmidt S, Jöstingmeyer P. Depression, fatigue and disability are independently associated with quality of life in patients with multiple sclerosis: Results of a cross-sectional study. Mult Scler Relat Disord. 2019;35:262-9. PubMed DOI: 1016/j.msard.2019.07.029
- Hsu CY, Rheima AM, Mohammed MS, Kadhim MM, Mohammed SH, Abbas FH, et al. Application of carbon nanotubes and graphene-based nanoadsorbents in water treatment. BioNanoScience. 2023;13(4):1418-36.
- Gupta A, Roy A, Roy A, Raja V, Sharma K, Verma RJ. Role of medicinal plants in the management of multiple sclerosis. Curr Pharm Biotechnol. 2024. PubMed DOI: 2174/0113892010324850240923181408
- Goldschmidt CH, Hua LH. Re-evaluating the use of IFN-β and relapsing multiple sclerosis: Safety, efficacy and place in therapy. Degener Neurol Neuromuscul Dis. 2020;10:29-38. PubMed DOI: 2147/DNND.S224912
- Sellebjerg F, Royen L, Soelberg Sørensen P, Oturai AB, Jensen PE. Prognostic value of cerebrospinal fluid neurofilament light chain and chitinase-3-like-1 in newly diagnosed patients with multiple sclerosis. Mult Scler J. 2019;25(11):1444-51. PubMed DOI: 1177/1352458518794308
- Martin R, Sospedra M, Rosito M, Engelhardt B. Current multiple sclerosis treatments have improved our understanding of MS autoimmune pathogenesis. Eur J Immunol. 2016;46(9):2078-90. PubMed DOI: 1002/eji.201646485
- Ingram G, Pearson OR. Cannabis and multiple sclerosis. Pract Neurol. 2019;19(4):310-5. PubMed DOI: 1136/practneurol-2018-002137
- Dobson R, Giovannoni G. Multiple sclerosis–a review. Eur J Neurol. 2019;26(1):27-40. PubMed DOI: 1111/ene.13819
- Salari N, Hayati A, Kazeminia M, Rahmani A, Mohammadi M, Fatahian R, et al. The effect of exercise on balance in patients with stroke, Parkinson, and multiple sclerosis: A systematic review and meta-analysis of clinical trials. Neurol Sci. 2022;43(2):167-185. PubMed DOI: 1007/s10072-021-05689-y
- Fiorini S, Verri A, Tacchino A, Ponzio M, Brichetto G, Barla A. A machine learning pipeline for multiple sclerosis course detection from clinical scales and patient reported outcomes. Annu Int Conf IEEE Eng Med Biol Soc. 2015;2015:4443-6. PubMed DOI: 1109/EMBC.2015.7319381
- Sarbaz Y, Pourakbari H, Vojudi MH, Ghanbari A. Introducing a decision support system for multiple sclerosis based on postural tremor: A hope for separation of people who might be affected by multiple sclerosis in the future. J Biomed Eng Appl Basis Commun. 2017;29(6). DOI: 4015/S1016237217500466
- Aslam N, Khan IU, Bashamakh A, Alghool FA, Aboulnour M, Alsuwayan NM, et al. Multiple sclerosis diagnosis using machine learning and deep learning: Challenges and opportunities. Sensors (Basel). 2022;22(20):7856. PubMed DOI: 3390/s22207856
- Filippi M, Preziosa P, Arnold DL, Barkhof F, Harrison DM, Maggi P, et al. Present and future of the diagnostic work-up of multiple sclerosis: The imaging perspective. J Neurol. 2023;270(3):1286-99. PubMed DOI: 1007/s00415-022-11488-y
- Shoeibi A, Khodatars M, Jafari M, Moridian P, Rezaei M, Alizadehsani R, et al. Applications of deep learning techniques for automated multiple sclerosis detection using magnetic resonance imaging: A review. Comput Biol Med. 2021;136:104697. PubMed DOI: 1016/j.compbiomed.2021.104697 .