Motaz Amr Abu Sabaa 1, Shaimaa El Said Shaban 2, Rehab Abd El Fattah Helal 3*
Authors affiliations:
Background & objectives: Emergence agitation (EA) is a common incident following sevoflurane anesthesia, especially in children. Various pharmacological agents have been tried to prevent or at least minimize it with variable success. We aimed to evaluate the prophylactic value of oral melatonin versus oral ketamine against the incidence of EA, postoperative sedation, as well as postoperative pain in pediatric patients planned for squint surgery under sevoflurane anesthesia.
Methodology: Ninety children aged 3-5 years, of both genders, were randomized to receive either plain honey (control group or Group C), oral ketamine (Group K), or oral melatonin (Group M) before induction of anesthesia. Incidence of EA, parent separation and mask acceptance, postoperative pain scores, as well as total rescue 24 h fentanyl consumption were noted.
Results EA incidence was significantly reduced in Groups K and M compared to the control group (16.66% vs. 13.33% vs. 46.66%; P = 0.008). Pairwise comparison revealed a P value of 0.026 between Group C and Group K, and 0.011 between Group C and Group M, with no significant difference detected in the incidence of EA between Group K and Group M. Pain scores remained significantly lower up to 10 min in Groups K and M (P < 0.001), with better parent separation and mask acceptance along with significantly less fentanyl consumption in both groups as compared to the control group (P < 0.05).
Conclusions Preemptive oral administration of ketamine or melatonin, both effectively lower the incidence of emergence agitation. Furthermore, it provided better conditions for parent separation and mask acceptance and decreased rescue analgesics requirement in pediatric patients undergoing squint surgery.
Abbreviations: EA: emergence agitation, ED: emergence delirium, PSAS: Parent Separation Anxiety Scale, PAED: Pediatric Anesthesia Emergence Delirium, PACU: Post-anesthesia care unit
Clinical trial registration number: PACTR202402880294495
Keywords: Child; Ketamine; Emergence Delirium; Melatonin; Squint
Citation: Abu Sabaa MA, Shaban SES, Helal RAEF. Effect of oral melatonin versus oral ketamine on the incidence of emergence delirium in children undergoing squint surgery: a prospective randomized controlled trial. Anaesth. pain intensive care 2025;29(4):341-48. DOI: 10.35975/apic.v29i4.2819
Received: May 09, 2024; Revised: October 26, 2024; Accepted: January 01, 2025
Emergence agitation (EA) is frequently encountered in pediatric patients after sevoflurane anesthesia.1 EA is not only a painful and upsetting event for the kids and their parents, but it can also lead to unintended harm, IV cannula displacement, suture rupture, and increased intraocular pressure. The latter is especially significant after ocular surgeries, which constitute a high-risk category for EA. Insufficiently handled EA has been linked to elevated postoperative nursing care expenses, extended duration in the post-anesthesia care unit (PACU), and delayed discharge following simple procedure.2
Melatonin is a neurohormone that facilitates sleep and is produced in the pineal gland. In addition to regulating circadian rhythm, melatonin has other functions such as anti-inflammatory, antioxidant, and anticonvulsant effects.3 Melatonin is used in pediatric and neonatal populations to address many problems, including sleep disturbances, seizures, and neonatal sepsis. Additionally, less sedative effect by melatonin, has made it an attractive option for pre-operative anxiolysis. In comparison to other anxiolytics, melatonin's anxiolytic effects are attributed to its facilitative role in γ-aminobutyric acid (GABA) transmission.4 Ketamine is an efficacious agent with sedative, analgesic, and amnestic properties.5 The oral administration of ketamine has been demonstrated to decrease the incidence of EA without delaying recovery.6
This study aimed to assess the effect of oral melatonin versus oral ketamine on the incidence of emergence delirium (ED) as a primary outcome. The secondary outcomes were ease of parental separation, as well as postoperative pain in pediatric patients planned for squint surgery under sevoflurane anesthesia.
A randomized double-blinded controlled trial was carried out in a tertiary university hospital from February 2024 to January 2025 on 90 children of both genders, aged 3 to 5 years, with ASA Status I or II, who were planned for squint surgery under sevoflurane anesthesia. The study was approved by the institutional Ethics Committee (NO 36264PR351/9/23) and registered in the Pan African Clinical Trial Registry (PACTR202402880294495). A written informed consent was obtained from the patients’ parents. Participants whose parents declined to participate in the trial, those with neurological or psychiatric diseases, developmental abnormalities, allergies, or contraindications to the research medicines were excluded.
The patients were enrolled and randomized into three groups at an allocation ratio 1:1:1 to receive 5 ml of either oral plain honey (Control group), oral ketamine 5 mg/kg (K group), or oral melatonin 0.3 mg/kg (M group) with a maximum dose of 6 mg, based on a computer-generated randomization with the allocation concealed in sequentially numbered, opaque envelopes. Randomization was performed by an independent nurse who read the numbers and allocated the patients to the assigned groups. Oral ketamine was prepared by mixing the calculated dose of ketamine with plain honey to make a total volume of 5 ml; while oral melatonin is available as brown viscous syrup (6 mg / 5 ml) similar in consistency and color to honey. The calculated dose was given 45 min before induction in a 5 ml syringe by an anesthetist who was not responsible for anesthetic management or outcomes evaluation. Melatonin was mixed with plain honey if the calculated dose was less than 6 mg so as to make a volume of 5 ml.
Intraoperative monitoring was done using pulse oximetry, noninvasive blood pressure, ECG, and end tidal CO2. Anesthetic induction was achieved by sevoflurane at 5-6% via a face mask. After ensuring adequate anesthetic depth, an IV cannula was inserted, and fentanyl (1 µg/kg) was given. Anesthesia was maintained with sevoflurane in a mixture of oxygen and air. A supraglottic laryngeal mask was placed by the attending anesthesiologist. Acetaminophen (15 mg/kg) was given at the beginning of surgery, and ondansetron (0.1 mg/kg) was given IV towards the end of procedure, then sevoflurane was stopped, and the laryngeal mask was removed in the deep plane of anesthesia. Kids were transferred to the PACU for observation.
Parent Separation Anxiety Scale (PSAS),7 was the assessment method for anxiety at the time of child transfer to the operating room. The PSAS ranges from one to four, where 1 refers to easy separation; 2 equals whimpers; 3 denotes that the child cries and cannot be easily reassured but is not clinging to the parents; and 4 signifies crying and clinging to parents. A PSAS score of 1 or 2 was considered an ‘acceptable’ separation.
Grading of mask acceptance at the time of anesthetic induction was done using the 4-point Mask Acceptance Score (MAS),8 in which 1 indicates excellent acceptance without fear; 2 = good acceptance with fear that is easily reassured; 3 = fair (moderate fear but not calmed with reassurance); and 4 = poor (terrified, combative, and crying). A MAS of 1 or 2 was considered ‘satisfactory’ mask acceptance.
Pediatric Anesthesia Emergence Delirium (PAED) scale,9 was used to assess agitation level by a blinded investigator at PACU arrival and then at 10, 20, 30, 45 and 60 min after arrival.
A PAED score of 10 was considered as EA and parental contact was initiated and if the PAED score remained 10, 0.5 mg/kg IV fentanyl would be given.
Pain after surgery was assessed using the Face, Legs, Activity, Cry, and Consolability (FLACC) scale.10 It was assessed at PACU arrival, and then at 10, 20, 30, 45, and 60 min after surgery.
A FLACC score of ≥ 4 was treated with 0.5 mg/kg of IV fentanyl (the 24-h total fentanyl dose was recorded). 15 mg/kg paracetamol was given every 8 hours as our standard postoperative analgesic regimen and the fentanyl rescue boluses were limited to the first hour after surgery.
2.1. Sample size
Our primary outcome was EA incidence. According to the results of a previous study,11 the incidence of EA following ophthalmic procedures was found to be approximately 50% with sevoflurane. Assuming a reduction in EA incidence from 50% to 15% to be clinically significant, 27 subjects would be required for each group at α error of 0.05 and 80% power of the study as estimated by two tailed z test of G power program. We recruited 30 cases per group to overcome possible dropouts.
2.2. Statistical analysis
The statistical Minitab® 16 software was used for statistical analysis. Normality of data was checked with the Shapiro wilk test. The parametric data were expressed as mean ± SD and were analyzed utilizing one-way ANOVA with post hoc Tukey’s test. Non parametric data of FLACC pain scores and rescue fentanyl doses were presented as median (interquartile range) and analyzed between groups by Kruskal-Wallis test with Mann- whitney U test for pairwise comparisons between each two groups.
Categorical data were presented as patients’ number (%) and were analyzed utilizing the Chi-square test. In order to adjust the p values for multiple comparisons, Bonferroni correction was done by multiplying the p value by the number of comparisons. So, all reported P values were adjusted, and values < 0.05 were considered significant.
After a preliminary evaluation of one hundred and two patients for eligibility, twelve patients were found to be ineligible (five due to developmental problems and seven because they chose not to participate). As a result, 90 patients were ultimately enrolled and randomly divided into three groups (each with 30 subjects). Among those who were successfully recruited, there were no losses to follow-up subjects (Figure 1).
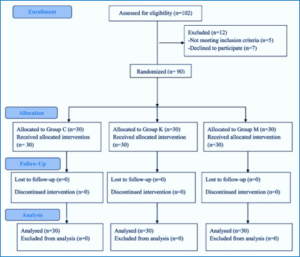
Figure 1: CONSORT Flow Diagram of participants
The demographic characteristics of the three groups were well comparable in terms of age, gender, ASA classification, weight, and duration of surgery (Table 1).
When compared to the control group, ketamine and melatonin groups had a significantly lower incidence of EA (46.66% vs. 16.66% vs. 13.33% respectively; P = 0.008). Pairwise comparison revealed a P value of 0.026 between Group C and Group K, and 0.011 between Group C and Group M, with no significant difference detected in the incidence of EA between Group K and Group M (Table 2).
There was a significant decrease in the PAED scores in groups K and M as compared to the control group on arrival to the PACU as well as at 10, 20, and 30 min (P < 0.001 for all) with comparable values noted at 45 and 60 min. In addition, lower PAED scores were found in Group M compared to Group K at 10 and 20 min (P = 0.003, 0.025 respectively) with similar scores observed at other times of measurement (Figure 2).
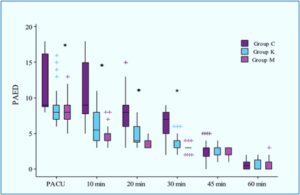
Figure 2: Pediatric Anesthesia Emergence Delirium (PAED) scale scores in the three groups. *Denotes statistically significant difference (P < 0.05)
The control group showed higher pain scores as compared to the other two groups at arrival at PACU and at 10 min (P < 0.001), with comparable pain scores at other time points. No significant differences were found in FLACC scores between Group K and Group M (Figure 3).
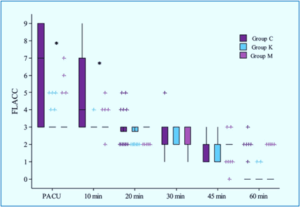
Figure 3: FLACC scale in the three groups. *Denotes statistically significant difference (P < 0.05).
Parental separation and mask acceptance conditions were better, and rescue fentanyl consumption was less in Groups K and M as compared to the control group (P < 0.05) with no difference observed between Group K and Group M as regards the three outcome measures (Table 2).
EA represents a major issue affecting up to 50% of pediatric patients recieving general anesthesia.12
Previous trials investigating oral ketamine,5,13 and oral melatonin,14,15 have shown promising results in preventing EA. To our knowledge, no studies have been conducted to compare these two drugs.
Our study demonstrated a substantial reduction in EA incidence with both ketamine (16.66%) and melatonin (13.33%) compared to the control group (46.66%). We observed significantly lower PAED scores in both ketamine and melatonin groups compared to the control group upon PACU arrival and at 10, 20, and 30 min post-arrival (P < 0.001for all).
These findings are consistent with previous research, including that of Kararmaz et al.,5 who reported an 18% incidence of EA following oral ketamine premedication (6 mg/kg) in children undergoing adenotonsillectomy with desflurane anesthesia. Our results, achieved with a lower ketamine dose of 5 mg/kg, suggest that the protective effect against EA may be attainable at lower doses, potentially minimizing dose-dependent side effects.
Our findings regarding melatonin align with those of Kain et al.,16 who found that melatonin has a dose-dependent effect on mitigating EA in pediatric patients undergoing anesthesia. Specifically, the incidence of EA was found to be 25.0% with a melatonin dose of 0.05 mg/kg, 8.3% with a dose of 0.2 mg/kg, and 5.4% with a dose of 0.4 mg/kg. Moreover, in the meta-analysis of Zhang et al.,17 melatonin doses ranging from 0.05 to 0.5 mg/kg were evaluated in studies aiming to reduce the incidence of EA in children. The incidence of EA following general anesthesia in pediatric populations is highly variable, ranging from 10% to 80%. Melatonin was found to significantly reduce EA incidence compared to placebos and midazolam, with a P < 0.01. Specifically, the EA incidence was 21.9% in the melatonin group versus 47.1% in the placebo group and 12.9% in the melatonin group versus 31.1% in the midazolam group.17
The efficacy of ketamine in EA prevention is further supported by Kuo et al.13 who found that combining ketamine and propofol was associated with the lowest incidence of postoperative EA in desflurane-based anesthesia. Regarding melatonin, Mansour et al.,18 demonstrated a dose-dependent effect on preoperative anxiety and postoperative agitation scores, with higher doses showing more significant reductions. Using an intermediate melatonin dose of 0.3 mg/kg, our study achieved significant reductions in EA incidence and severity, suggesting a potential balance between efficacy and side effects. In our study, we utilized the PAED scale to assess the severity of EA which is widely recognized for identifying EA in children older than 2 years.19 This finding aligns with earlier research by Kararmaz et al.,5 and Mason20 who reported that oral ketamine premedication administered 30 min before induction effectively reduced EA incidence.
The effectiveness of melatonin in mitigating EA is further supported by Singla et al.,21 who found that melatonin significantly decreased EA risk compared to placebo. Mansour et al.18 observed significant effects on preoperative anxiety and postoperative agitation scores when melatonin was administered two hours before admission to the operating room. This reveals that the timing of administration may play an important factor in maximizing the protective effects of both agents. Additionally, a study of Do et al.22 highlighted the importance of considering age as a factor in EA susceptibility, noting that younger children are more prone to experiencing EA.
Our study employed the FLACC scale to assess postoperative pain. We observed significantly higher pain scores in the control group compared to both ketamine and melatonin groups upon PACU arrival and at 10 min post-arrival (P < 0.001 for both time points). Notably, no significant differences in pain scores between the ketamine and melatonin groups were observed.
These findings highlight the analgesic value of both ketamine and melatonin in the early postoperative period. The analgesic effect of ketamine is well-established and can be attributed to its N-methyl-D-aspartate (NMDA) receptors antagonism.23 While less well-understood, the analgesic properties of melatonin may be related to its effects on opioid, benzodiazepine, and gamma-aminobutyric acid (GABA) receptors.24
The relationship between pain and ED is complex and often intertwined. Moore and Anghelescu2 reported that in one study, 21% of children had EA, 5% had pain, and 65% had both ED and pain simultaneously when assessed using PAED and FLACC scales. Our results, showing reduced pain scores in both treatment groups, suggest that the analgesic effects of ketamine and melatonin may contribute to their efficacy in preventing EA.
Furthermore, our study found that rescue fentanyl consumption was lower in both ketamine and melatonin groups compared to the control group (P < 0.001). This finding is particularly noteworthy when compared to the results of Singla et al.,21 who reported no significant difference in postoperative fentanyl requirements between melatonin, midazolam, and placebo groups. In their study, 9% of children in the melatonin group required postoperative fentanyl, compared to 16% in both midazolam and placebo groups.
Our study demonstrated improved parental separation and mask acceptance in ketamine and melatonin groups compared to the control group. These findings highlight the anxiolytic properties of both agents and their potential to enhance the overall perioperative experience for pediatric patients and their families.
The improved parental separation observed with melatonin in our study contrasts with the findings of Kain et al.,16 who reported that melatonin did not affect preoperative anxiety despite its benefits in reducing EA. This discrepancy may be due to differences in assessment methods, timing of administration, or patient populations.
The anxiolytic effect of ketamine observed in our study aligns with previous research. Mason20 reported that premedication with ketamine 1 mg/kg IV may be more effective than midazolam 0.1 mg/kg IV in decreasing early EA. While our study used oral ketamine at a higher dose (5 mg/kg), the observed improvements in parental separation and mask acceptance suggest that these benefits extend to the preoperative period. The comparable efficacy of ketamine and melatonin in improving these preoperative outcomes is noteworthy, and it suggests that both agents may offer viable alternatives to traditional premedication strategies.
The single-center design of the study may limit its generalizability. Although the sample size is adequate, a larger sample could provide more robust conclusions. Moreover, the study did not assess long-term outcomes or side effects and focused solely on fixed dosing.
Premedication with either oral ketamine (5 mg/kg) or oral melatonin (0.3 mg/kg) significantly decreases the incidence and severity of emergence agitation in pediatric patients undergoing squint surgery. Both drugs showed comparable efficacy in preventing emergence agitation, with melatonin demonstrating a potentially more sustained effect on PAED scores during early recovery. Additionally, both interventions improved perioperative outcomes, including pain scores, parental separation, mask acceptance, and rescue analgesic requirements.
7. Data availability
the data are available upon request from the corresponding author.
8. Ethical considerations
The study was approved by the institutional Ethics Committee (No. 36264PR351/9/23) and registered in the Pan African Clinical Trial Registry (PACTR202402880294495).
9. Funding
The study was completed out of institutional resources, and no external or industry funding was involved.
10. Conflicts of interest
The authors declare no conflicts of interest.
11. Authors' contributions
MAAS: study concept and design, data collection and interpretation, drafting the manuscript and revising it, and approved the final version of the manuscript.
SESS: data collection and analysis, drafting the manuscript and revising it, and approved the final version of the manuscript.
RA.EH: data collection, interpretation and analysis, drafted and revised the manuscript, and approved the final version of the manuscript.
Authors affiliations:
- Motaz Amr Abu Sabaa, Assistant Professor, Department of Anesthesiology, Surgical Intensive Care & Pain Medicine, Faculty of Medicine, Tanta University, Tanta, Egypt; Email: motazabusabaa@gmail.com
- Shaimaa El Said Shaban, Lecturer, Department of Anesthesiology, Surgical Intensive Care & Pain Medicine, Faculty of Medicine, Tanta University, Tanta, Egypt; Email: shimaash3ban2@gmail.com
- Rehab Abd El Fattah Helal, Lecturer, Department of Anesthesiology Surgical Intensive Care & Pain Management Faculty of Medicine, Tanta University, Tanta, Egypt; Email: rehabhelal2@gmail.com
ABSTRACT
Background & objectives: Emergence agitation (EA) is a common incident following sevoflurane anesthesia, especially in children. Various pharmacological agents have been tried to prevent or at least minimize it with variable success. We aimed to evaluate the prophylactic value of oral melatonin versus oral ketamine against the incidence of EA, postoperative sedation, as well as postoperative pain in pediatric patients planned for squint surgery under sevoflurane anesthesia.
Methodology: Ninety children aged 3-5 years, of both genders, were randomized to receive either plain honey (control group or Group C), oral ketamine (Group K), or oral melatonin (Group M) before induction of anesthesia. Incidence of EA, parent separation and mask acceptance, postoperative pain scores, as well as total rescue 24 h fentanyl consumption were noted.
Results EA incidence was significantly reduced in Groups K and M compared to the control group (16.66% vs. 13.33% vs. 46.66%; P = 0.008). Pairwise comparison revealed a P value of 0.026 between Group C and Group K, and 0.011 between Group C and Group M, with no significant difference detected in the incidence of EA between Group K and Group M. Pain scores remained significantly lower up to 10 min in Groups K and M (P < 0.001), with better parent separation and mask acceptance along with significantly less fentanyl consumption in both groups as compared to the control group (P < 0.05).
Conclusions Preemptive oral administration of ketamine or melatonin, both effectively lower the incidence of emergence agitation. Furthermore, it provided better conditions for parent separation and mask acceptance and decreased rescue analgesics requirement in pediatric patients undergoing squint surgery.
Abbreviations: EA: emergence agitation, ED: emergence delirium, PSAS: Parent Separation Anxiety Scale, PAED: Pediatric Anesthesia Emergence Delirium, PACU: Post-anesthesia care unit
Clinical trial registration number: PACTR202402880294495
Keywords: Child; Ketamine; Emergence Delirium; Melatonin; Squint
Citation: Abu Sabaa MA, Shaban SES, Helal RAEF. Effect of oral melatonin versus oral ketamine on the incidence of emergence delirium in children undergoing squint surgery: a prospective randomized controlled trial. Anaesth. pain intensive care 2025;29(4):341-48. DOI: 10.35975/apic.v29i4.2819
Received: May 09, 2024; Revised: October 26, 2024; Accepted: January 01, 2025
1. INTRODUCTION
Emergence agitation (EA) is frequently encountered in pediatric patients after sevoflurane anesthesia.1 EA is not only a painful and upsetting event for the kids and their parents, but it can also lead to unintended harm, IV cannula displacement, suture rupture, and increased intraocular pressure. The latter is especially significant after ocular surgeries, which constitute a high-risk category for EA. Insufficiently handled EA has been linked to elevated postoperative nursing care expenses, extended duration in the post-anesthesia care unit (PACU), and delayed discharge following simple procedure.2
Melatonin is a neurohormone that facilitates sleep and is produced in the pineal gland. In addition to regulating circadian rhythm, melatonin has other functions such as anti-inflammatory, antioxidant, and anticonvulsant effects.3 Melatonin is used in pediatric and neonatal populations to address many problems, including sleep disturbances, seizures, and neonatal sepsis. Additionally, less sedative effect by melatonin, has made it an attractive option for pre-operative anxiolysis. In comparison to other anxiolytics, melatonin's anxiolytic effects are attributed to its facilitative role in γ-aminobutyric acid (GABA) transmission.4 Ketamine is an efficacious agent with sedative, analgesic, and amnestic properties.5 The oral administration of ketamine has been demonstrated to decrease the incidence of EA without delaying recovery.6
This study aimed to assess the effect of oral melatonin versus oral ketamine on the incidence of emergence delirium (ED) as a primary outcome. The secondary outcomes were ease of parental separation, as well as postoperative pain in pediatric patients planned for squint surgery under sevoflurane anesthesia.
2. METHODOLOGY
A randomized double-blinded controlled trial was carried out in a tertiary university hospital from February 2024 to January 2025 on 90 children of both genders, aged 3 to 5 years, with ASA Status I or II, who were planned for squint surgery under sevoflurane anesthesia. The study was approved by the institutional Ethics Committee (NO 36264PR351/9/23) and registered in the Pan African Clinical Trial Registry (PACTR202402880294495). A written informed consent was obtained from the patients’ parents. Participants whose parents declined to participate in the trial, those with neurological or psychiatric diseases, developmental abnormalities, allergies, or contraindications to the research medicines were excluded.
The patients were enrolled and randomized into three groups at an allocation ratio 1:1:1 to receive 5 ml of either oral plain honey (Control group), oral ketamine 5 mg/kg (K group), or oral melatonin 0.3 mg/kg (M group) with a maximum dose of 6 mg, based on a computer-generated randomization with the allocation concealed in sequentially numbered, opaque envelopes. Randomization was performed by an independent nurse who read the numbers and allocated the patients to the assigned groups. Oral ketamine was prepared by mixing the calculated dose of ketamine with plain honey to make a total volume of 5 ml; while oral melatonin is available as brown viscous syrup (6 mg / 5 ml) similar in consistency and color to honey. The calculated dose was given 45 min before induction in a 5 ml syringe by an anesthetist who was not responsible for anesthetic management or outcomes evaluation. Melatonin was mixed with plain honey if the calculated dose was less than 6 mg so as to make a volume of 5 ml.
Intraoperative monitoring was done using pulse oximetry, noninvasive blood pressure, ECG, and end tidal CO2. Anesthetic induction was achieved by sevoflurane at 5-6% via a face mask. After ensuring adequate anesthetic depth, an IV cannula was inserted, and fentanyl (1 µg/kg) was given. Anesthesia was maintained with sevoflurane in a mixture of oxygen and air. A supraglottic laryngeal mask was placed by the attending anesthesiologist. Acetaminophen (15 mg/kg) was given at the beginning of surgery, and ondansetron (0.1 mg/kg) was given IV towards the end of procedure, then sevoflurane was stopped, and the laryngeal mask was removed in the deep plane of anesthesia. Kids were transferred to the PACU for observation.
Parent Separation Anxiety Scale (PSAS),7 was the assessment method for anxiety at the time of child transfer to the operating room. The PSAS ranges from one to four, where 1 refers to easy separation; 2 equals whimpers; 3 denotes that the child cries and cannot be easily reassured but is not clinging to the parents; and 4 signifies crying and clinging to parents. A PSAS score of 1 or 2 was considered an ‘acceptable’ separation.
Grading of mask acceptance at the time of anesthetic induction was done using the 4-point Mask Acceptance Score (MAS),8 in which 1 indicates excellent acceptance without fear; 2 = good acceptance with fear that is easily reassured; 3 = fair (moderate fear but not calmed with reassurance); and 4 = poor (terrified, combative, and crying). A MAS of 1 or 2 was considered ‘satisfactory’ mask acceptance.
Pediatric Anesthesia Emergence Delirium (PAED) scale,9 was used to assess agitation level by a blinded investigator at PACU arrival and then at 10, 20, 30, 45 and 60 min after arrival.
A PAED score of 10 was considered as EA and parental contact was initiated and if the PAED score remained 10, 0.5 mg/kg IV fentanyl would be given.
Pain after surgery was assessed using the Face, Legs, Activity, Cry, and Consolability (FLACC) scale.10 It was assessed at PACU arrival, and then at 10, 20, 30, 45, and 60 min after surgery.
A FLACC score of ≥ 4 was treated with 0.5 mg/kg of IV fentanyl (the 24-h total fentanyl dose was recorded). 15 mg/kg paracetamol was given every 8 hours as our standard postoperative analgesic regimen and the fentanyl rescue boluses were limited to the first hour after surgery.
2.1. Sample size
Our primary outcome was EA incidence. According to the results of a previous study,11 the incidence of EA following ophthalmic procedures was found to be approximately 50% with sevoflurane. Assuming a reduction in EA incidence from 50% to 15% to be clinically significant, 27 subjects would be required for each group at α error of 0.05 and 80% power of the study as estimated by two tailed z test of G power program. We recruited 30 cases per group to overcome possible dropouts.
2.2. Statistical analysis
The statistical Minitab® 16 software was used for statistical analysis. Normality of data was checked with the Shapiro wilk test. The parametric data were expressed as mean ± SD and were analyzed utilizing one-way ANOVA with post hoc Tukey’s test. Non parametric data of FLACC pain scores and rescue fentanyl doses were presented as median (interquartile range) and analyzed between groups by Kruskal-Wallis test with Mann- whitney U test for pairwise comparisons between each two groups.
Categorical data were presented as patients’ number (%) and were analyzed utilizing the Chi-square test. In order to adjust the p values for multiple comparisons, Bonferroni correction was done by multiplying the p value by the number of comparisons. So, all reported P values were adjusted, and values < 0.05 were considered significant.
3. RESULTS
After a preliminary evaluation of one hundred and two patients for eligibility, twelve patients were found to be ineligible (five due to developmental problems and seven because they chose not to participate). As a result, 90 patients were ultimately enrolled and randomly divided into three groups (each with 30 subjects). Among those who were successfully recruited, there were no losses to follow-up subjects (Figure 1).

Figure 1: CONSORT Flow Diagram of participants
The demographic characteristics of the three groups were well comparable in terms of age, gender, ASA classification, weight, and duration of surgery (Table 1).
| Table 1: Demographic characteristics of the studied groups | ||||
| Variable | Group C | Group K | Group M | P value |
| Age (years) | 3.867 ± 0.819 | 3.833 ± 0.834 | 3.833 ± 0.834 | 0.984 |
| Gender
· Male · Female |
23 (76.66) 7 (23.33) |
18 (60) 12 (40) |
15 (50) 15 (50) |
0.099 |
| ASA
· I · II |
25 (83.33) 5 (16.66) |
26 (86.66) 4 (13.33) |
24 (80) 6 (20) |
0.787 |
| Weight (kg) | 16.7 ± 2.168 | 16.7 ± 2.351 | 17 ± 2.691 | 0.857 |
| Duration of surgery (min) | 41.6 ± 5.97 | 42.67 ± 5.93 | 44.37 ± 5.65 | 0.188 |
| Data presented as mean ± SD or n (%); ASA; American Society of Anesthesiologists; P < 0.05 is significant. | ||||
When compared to the control group, ketamine and melatonin groups had a significantly lower incidence of EA (46.66% vs. 16.66% vs. 13.33% respectively; P = 0.008). Pairwise comparison revealed a P value of 0.026 between Group C and Group K, and 0.011 between Group C and Group M, with no significant difference detected in the incidence of EA between Group K and Group M (Table 2).
| Table 2: Perioperative data of the studied groups | |||||||
| Variable | Group C | Group K | Group M | P value | P1 | P2 | P3 |
| Incidence of ED | 14 (46.66) | 5 (16.66) | 4 (13.33) | 0.008* | 0.026* | 0.011* | 0.718 |
| Successful parental separation | 6 (20) | 26 (86.66) | 27 (90) | < 0.001* | < 0.001* | < 0.001* | 0.688 |
| Satisfactory mask acceptance | 5 (16.66) | 15 (50) | 21 (70) | <0.001* | 0.026* | < 0.001* | 0.116 |
| Post-operative rescue fentanyl (µg) | 7.5 [0-9] | 0 [0-0] | 0 [0-0] | < 0.001* | < 0.001* | < 0.001* | 0.582 |
| Data presented as median [interquartile range] or patient's number (%). ED; emergence delirium. P presented the comparison among the three groups. P1 presented the comparison between the Group C and Group K. P2 presented the comparison between Group C and Group M. P3 presented the comparison between Group K and Group M. *Denotes statistically significant difference (adjusted P < 0.05). | |||||||
There was a significant decrease in the PAED scores in groups K and M as compared to the control group on arrival to the PACU as well as at 10, 20, and 30 min (P < 0.001 for all) with comparable values noted at 45 and 60 min. In addition, lower PAED scores were found in Group M compared to Group K at 10 and 20 min (P = 0.003, 0.025 respectively) with similar scores observed at other times of measurement (Figure 2).

Figure 2: Pediatric Anesthesia Emergence Delirium (PAED) scale scores in the three groups. *Denotes statistically significant difference (P < 0.05)
The control group showed higher pain scores as compared to the other two groups at arrival at PACU and at 10 min (P < 0.001), with comparable pain scores at other time points. No significant differences were found in FLACC scores between Group K and Group M (Figure 3).

Figure 3: FLACC scale in the three groups. *Denotes statistically significant difference (P < 0.05).
Parental separation and mask acceptance conditions were better, and rescue fentanyl consumption was less in Groups K and M as compared to the control group (P < 0.05) with no difference observed between Group K and Group M as regards the three outcome measures (Table 2).
| Table 2: Perioperative data of the studied groups | |||||||
| Variable | Group C | Group K | Group M | P value | P1 | P2 | P3 |
| Incidence of ED | 14 (46.66) | 5 (16.66) | 4 (13.33) | 0.008* | 0.026* | 0.011* | 0.718 |
| Successful parental separation | 6 (20) | 26 (86.66) | 27 (90) | < 0.001* | < 0.001* | < 0.001* | 0.688 |
| Satisfactory mask acceptance | 5 (16.66) | 15 (50) | 21 (70) | <0.001* | 0.026* | < 0.001* | 0.116 |
| Post-operative rescue fentanyl (µg) | 7.5 [0-9] | 0 [0-0] | 0 [0-0] | < 0.001* | < 0.001* | < 0.001* | 0.582 |
| Data presented as median [interquartile range] or patient's number (%). ED; emergence delirium. P presented the comparison among the three groups. P1 presented the comparison between the Group C and Group K. P2 presented the comparison between Group C and Group M. P3 presented the comparison between Group K and Group M. *Denotes statistically significant difference (adjusted P < 0.05). | |||||||
4. DISCUSSION
EA represents a major issue affecting up to 50% of pediatric patients recieving general anesthesia.12
Previous trials investigating oral ketamine,5,13 and oral melatonin,14,15 have shown promising results in preventing EA. To our knowledge, no studies have been conducted to compare these two drugs.
Our study demonstrated a substantial reduction in EA incidence with both ketamine (16.66%) and melatonin (13.33%) compared to the control group (46.66%). We observed significantly lower PAED scores in both ketamine and melatonin groups compared to the control group upon PACU arrival and at 10, 20, and 30 min post-arrival (P < 0.001for all).
These findings are consistent with previous research, including that of Kararmaz et al.,5 who reported an 18% incidence of EA following oral ketamine premedication (6 mg/kg) in children undergoing adenotonsillectomy with desflurane anesthesia. Our results, achieved with a lower ketamine dose of 5 mg/kg, suggest that the protective effect against EA may be attainable at lower doses, potentially minimizing dose-dependent side effects.
Our findings regarding melatonin align with those of Kain et al.,16 who found that melatonin has a dose-dependent effect on mitigating EA in pediatric patients undergoing anesthesia. Specifically, the incidence of EA was found to be 25.0% with a melatonin dose of 0.05 mg/kg, 8.3% with a dose of 0.2 mg/kg, and 5.4% with a dose of 0.4 mg/kg. Moreover, in the meta-analysis of Zhang et al.,17 melatonin doses ranging from 0.05 to 0.5 mg/kg were evaluated in studies aiming to reduce the incidence of EA in children. The incidence of EA following general anesthesia in pediatric populations is highly variable, ranging from 10% to 80%. Melatonin was found to significantly reduce EA incidence compared to placebos and midazolam, with a P < 0.01. Specifically, the EA incidence was 21.9% in the melatonin group versus 47.1% in the placebo group and 12.9% in the melatonin group versus 31.1% in the midazolam group.17
The efficacy of ketamine in EA prevention is further supported by Kuo et al.13 who found that combining ketamine and propofol was associated with the lowest incidence of postoperative EA in desflurane-based anesthesia. Regarding melatonin, Mansour et al.,18 demonstrated a dose-dependent effect on preoperative anxiety and postoperative agitation scores, with higher doses showing more significant reductions. Using an intermediate melatonin dose of 0.3 mg/kg, our study achieved significant reductions in EA incidence and severity, suggesting a potential balance between efficacy and side effects. In our study, we utilized the PAED scale to assess the severity of EA which is widely recognized for identifying EA in children older than 2 years.19 This finding aligns with earlier research by Kararmaz et al.,5 and Mason20 who reported that oral ketamine premedication administered 30 min before induction effectively reduced EA incidence.
The effectiveness of melatonin in mitigating EA is further supported by Singla et al.,21 who found that melatonin significantly decreased EA risk compared to placebo. Mansour et al.18 observed significant effects on preoperative anxiety and postoperative agitation scores when melatonin was administered two hours before admission to the operating room. This reveals that the timing of administration may play an important factor in maximizing the protective effects of both agents. Additionally, a study of Do et al.22 highlighted the importance of considering age as a factor in EA susceptibility, noting that younger children are more prone to experiencing EA.
Our study employed the FLACC scale to assess postoperative pain. We observed significantly higher pain scores in the control group compared to both ketamine and melatonin groups upon PACU arrival and at 10 min post-arrival (P < 0.001 for both time points). Notably, no significant differences in pain scores between the ketamine and melatonin groups were observed.
These findings highlight the analgesic value of both ketamine and melatonin in the early postoperative period. The analgesic effect of ketamine is well-established and can be attributed to its N-methyl-D-aspartate (NMDA) receptors antagonism.23 While less well-understood, the analgesic properties of melatonin may be related to its effects on opioid, benzodiazepine, and gamma-aminobutyric acid (GABA) receptors.24
The relationship between pain and ED is complex and often intertwined. Moore and Anghelescu2 reported that in one study, 21% of children had EA, 5% had pain, and 65% had both ED and pain simultaneously when assessed using PAED and FLACC scales. Our results, showing reduced pain scores in both treatment groups, suggest that the analgesic effects of ketamine and melatonin may contribute to their efficacy in preventing EA.
Furthermore, our study found that rescue fentanyl consumption was lower in both ketamine and melatonin groups compared to the control group (P < 0.001). This finding is particularly noteworthy when compared to the results of Singla et al.,21 who reported no significant difference in postoperative fentanyl requirements between melatonin, midazolam, and placebo groups. In their study, 9% of children in the melatonin group required postoperative fentanyl, compared to 16% in both midazolam and placebo groups.
Our study demonstrated improved parental separation and mask acceptance in ketamine and melatonin groups compared to the control group. These findings highlight the anxiolytic properties of both agents and their potential to enhance the overall perioperative experience for pediatric patients and their families.
The improved parental separation observed with melatonin in our study contrasts with the findings of Kain et al.,16 who reported that melatonin did not affect preoperative anxiety despite its benefits in reducing EA. This discrepancy may be due to differences in assessment methods, timing of administration, or patient populations.
The anxiolytic effect of ketamine observed in our study aligns with previous research. Mason20 reported that premedication with ketamine 1 mg/kg IV may be more effective than midazolam 0.1 mg/kg IV in decreasing early EA. While our study used oral ketamine at a higher dose (5 mg/kg), the observed improvements in parental separation and mask acceptance suggest that these benefits extend to the preoperative period. The comparable efficacy of ketamine and melatonin in improving these preoperative outcomes is noteworthy, and it suggests that both agents may offer viable alternatives to traditional premedication strategies.
5. LIMITATIONS
The single-center design of the study may limit its generalizability. Although the sample size is adequate, a larger sample could provide more robust conclusions. Moreover, the study did not assess long-term outcomes or side effects and focused solely on fixed dosing.
6. CONCLUSION
Premedication with either oral ketamine (5 mg/kg) or oral melatonin (0.3 mg/kg) significantly decreases the incidence and severity of emergence agitation in pediatric patients undergoing squint surgery. Both drugs showed comparable efficacy in preventing emergence agitation, with melatonin demonstrating a potentially more sustained effect on PAED scores during early recovery. Additionally, both interventions improved perioperative outcomes, including pain scores, parental separation, mask acceptance, and rescue analgesic requirements.
7. Data availability
the data are available upon request from the corresponding author.
8. Ethical considerations
The study was approved by the institutional Ethics Committee (No. 36264PR351/9/23) and registered in the Pan African Clinical Trial Registry (PACTR202402880294495).
9. Funding
The study was completed out of institutional resources, and no external or industry funding was involved.
10. Conflicts of interest
The authors declare no conflicts of interest.
11. Authors' contributions
MAAS: study concept and design, data collection and interpretation, drafting the manuscript and revising it, and approved the final version of the manuscript.
SESS: data collection and analysis, drafting the manuscript and revising it, and approved the final version of the manuscript.
RA.EH: data collection, interpretation and analysis, drafted and revised the manuscript, and approved the final version of the manuscript.
12. REFERENCES
- Nair S, Wolf A. Emergence delirium after paediatric anaesthesia: new strategies in avoidance and treatment. BJA Educ 2018;18(1):30-3. [PubMed ] DOI: 1016/j.bjae.2017.07.001
- Moore AD, Anghelescu DL. Emergence delirium in pediatric anesthesia. Paediatr Drugs. 2017;19:11-20. [PubMed ] DOI: 1007/s40272-016-0201-5
- Gitto E, Marseglia L, Manti S, D’Angelo G, Barberi I, Salpietro C, et al. Protective role of melatonin in neonatal diseases. Oxid Med Cell Longev. 2013;2013(1):980374. [PubMed ] DOI: 1155/2013/980374
- Marseglia L, D’Angelo G, Manti S, Aversa S, Arrigo T, Reiter RJ, et al. Analgesic, anxiolytic and anaesthetic effects of melatonin: new potential uses in pediatrics. Int J Mol Sci. 2015;16(1):1209-20. [PubMed ] DOI: 3390/ijms16011209
- Kararmaz A, Kaya S, Turhanoglu S, Ozyilmaz MA. Oral ketamine premedication can prevent emergence agitation in children after desflurane anaesthesia. Paediatr Anaesth. 2004;14(6):477-82. [PubMed ] DOI: 1111/j.1460-9592.2004.01224.x
- Abu‐shahwan I, Chowdary K. Ketamine is effective in decreasing the incidence of emergence agitation in children undergoing dental repair under sevoflurane general anesthesia. Paediatr Anaesth. 2007;17(9):846-50. [PubMed ] DOI: 1111/j.1460-9592.2007.02298.x
- Dashiff CJ, Weaver M. Development and testing of a scale to measure separation anxiety of parents of adolescents. J Nurs Meas. 2008;16(1):61-80. [PubMed ] DOI: 1891/1061-3749.16.1.61
- Shukry M, Clyde MC, Kalarickal PL, Ramadhyani U. Does dexmedetomidine prevent emergence delirium in children after sevoflurane‐based general anesthesia? Paediatr Anaesth. 2005;15(12):1098-104. [PubMed ] DOI: 1111/j.1460-9592.2005.01660.x
- Sikich N, Lerman J. Development and psychometric evaluation of the pediatric anesthesia emergence delirium scale. Anesthesiology. 2004;100(5):1138-45. [PubMed ] DOI: 1097/00000542-200405000-00015
- Voepel-Lewis T, Shayevitz JR, Malviya S. The FLACC: a behavioral scale for scoring postoperative pain in young children. Pediatr Nurs. 1997;23(3):293-7. [PubMed ]
- Aouad MT, Yazbeck-Karam VG, Nasr VG, El-Khatib MF, Kanazi GE, Bleik JH. A single dose of propofol at the end of surgery for the prevention of emergence agitation in children undergoing strabismus surgery during sevoflurane anesthesia. Anesthesiology. 2007;107(5):733-8. [PubMed ] DOI: 1097/01.anes.0000287009.46896.a7
- Vlajkovic GP, Sindjelic RP. Emergence delirium in children: many questions, few answers. Anesth Analg. 2007;104(1):84-91. [PubMed ] DOI: 1213/01.ane.0000250914.91881.a8
- Kuo HC, Hung KC, Wang HY, Zeng BS, Chen TY, Li DJ, et al. Prophylaxis for paediatric emergence delirium in desflurane-based anaesthesia: a network meta-analysis. J Anesth. 2024;38(2):155-66. [PubMed ] DOI: 1007/s00540-023-03219-y
- Khalifa OSM, Hassanin AAM. Melatonin, ketamine and their combination in half doses for management of sevoflurane agitation in children undergoing adenotonsillectomy. Egypt J Anaesth. 2013;29(4):337-41. DOI: 1016/j.egja.2013.05.006
- Komazaki M, Mihara T, Nakamura N, Ka K, Goto T. Preventive effect of ramelteon on emergence agitation after general anaesthesia in paediatric patients undergoing tonsillectomy: a randomised, placebo-controlled clinical trial. Sci Rep. 2020;10(1):21996. [PubMed ] DOI: 1038/s41598-020-79078-4
- Kain ZN, MacLaren JE, Herrmann L, Mayes L, Rosenbaum A, Hata J, et al. Preoperative melatonin and its effects on induction and emergence in children undergoing anesthesia and surgery. Anesthesiology. 2009;111(1):44-9. [PubMed ] DOI: 1097/ALN.0b013e3181a91870
- Zhang D, Jia X, Lin D, Ma J. Melatonin or its analogs as premedication to prevent emergence agitation in children: a systematic review and meta-analysis. BMC Anesthesiol. 2023;23(1):392. [PubMed ] DOI: 1186/s12871-023-02356-x
- Mansour MS, Sedky AM, Sultan AA. Evaluation of preoperative melatonin on emergence agitation after herniorrhaphy surgeries in pediatrics. Res Opin Anaesth Intensive Care. 2024;11(2):132-9. DOI: 4103/roaic.roaic_7_24
- Shin S, Evans F, Mason K. Emergence delirium in pediatric patients. Pediatr Anesth. 2021;5:1-6. Full Text
- Mason K. Paediatric emergence delirium: a comprehensive review and interpretation of the literature. Br J Anaesth. 2017;118(3):335-43. [PubMed ] DOI: 1093/bja/aew477
- Singla L, Mathew PJ, Jain A, Yaddanapudi S, Peters NJ. Oral melatonin as part of multimodal anxiolysis decreases emergence delirium in children whereas midazolam does not: A randomised, double-blind, placebo-controlled study. Eur J Anaesthesiol. 2021;38(11):1130-7. [PubMed ] DOI: 1097/EJA.0000000000001561
- Do W, Kim H-S, Kim SH, Kang H, Lee D, Baik J, et al. Sleep quality and emergence delirium in children undergoing strabismus surgery: a comparison between preschool-and school-age patients. BMC Anesthesiol. 2021;21:290. [PubMed ] DOI: 1186/s12871-021-01507-2
- Schoevers RA, Chaves TV, Balukova SM, Kortekaas R. Oral ketamine for the treatment of pain and treatment-resistant depression. Br J Psychiatry. 2016;208(2):108-13. [PubMed ] DOI: 1192/bjp.bp.115.165498
- Mellor K, Papaioannou D, Thomason A, Bolt R, Evans C, Wilson M, et al. Melatonin for pre-medication in children: a systematic review. BMC Pediatr. 2022;22(1):107. [PubMed ] DOI: 1186/s12887-022-03149-w