Amany Mohamed Abotaleb 1, Wafaa Madhy Atia Abdelwahed 2, Asmaa Fawzy Amer 3
Authors affiliations:
Background & Objective: laparoscopic cholecystectomy (LC) is routinely performed under general anesthesia, but relieving postoperative pain poses real challenge, with the current trends of avoiding narcotic analgesics, due to their serious side effects. Addition of regional nerve blocks offers a more flexible analgesic approach. We hypnotized that addition of dexmedetomidine to the local anesthetic solution will prolong the duration of the blocks. This study aimed to assess the effectiveness of erector spinae plane block (ESPB) using bupivacaine and dexmedetomidine (DEX) for pain controlling in LC.
Methods: This randomized, controlled, double-masked trial included 60 cases of 18 to 65 years, both genders, scheduled for LC. Cases were randomized equally into two groups: Group I received ESPB with 20 mL of bupivacaine 0.25% on each side, and Group II received ESPB with 18 mL of bupivacaine 0.25% plus 2 mL of DEX 0.5 µg/kg on each side. Blocks were performed before the induction of general anesthesia. Ultrasonography was used to guide the ESPB.
Results: The morphine consumption in 1st 24 hours postoperatively was reduced significantly in group II contrasted to group I (10.59 ± 3.51 vs. 16.41 ± 5.43 mg, P < 0.001). The time to first rescue analgesia was delayed significantly in group II than in Group I (16.97 ± 4.66 vs 9.63 ± 1.77 h, P < 0.001). Intraoperative fentanyl consumption was insignificantly different between both groups. Postoperative visual analog scale, heart rate, and mean blood pressure measurements at post-anesthesia care unit 1, 3, 6, 12, and 18 h were reduced significantly in Group II than in Group I (P < 0.001). The 4-point sedation scale was significantly better in Group II than Group I (P = 0.031).
Conclusion: DEX as an adjuvant to ESPB during LC improved analgesia by reducing the need for morphine postoperatively, as well as the pain level with relatively better hemodynamics.
Abbreviations: ESPB: erector spinae plane block, LC: Laparoscopic Cholecystectomy, VAS: visual analog scale,
Keywords: Analgesia; Dexmedetomidine; Laparoscopic Cholecystectomy; Erector Spinae Plane Block; Bupivacaine
Citation: Abotaleb AM, Abdelwahed WMA, Amer AF. Analgesic efficacy of adding dexmedetomidine to bupivacaine in ultrasound-guided erector spinae plane block for laparoscopic cholecystectomy: a randomized, controlled, double-blind trial. Anaesth. pain intensive care 2025;29(4):318-25. DOI: 10.35975/apic.v29i4.2816
Received: October 22, 2024; Revised: January 27, 2025; Accepted: February 03, 2025
Laparoscopic cholecystectomy (LC) is a common ambulatory surgery. Even though LC is considered a minimally invasive procedure, postoperative pain is evident.1 Post-LC pain can manifest in a spectrum of intensity, from mild to severe. The pain experience typically comprises three distinct components: incisional pain arising from the trocar site, visceral pain arising from the surgical intervention, and shoulder discomfort, which is thought to be a manifestation of visceral pain transmitted to the referred pain area.2
Efficient management of postoperative pain control is essential in optimizing recovery outcomes in laparoscopic surgeries, as it enables patients to experience reduced discomfort, improved mobility, and a more rapid return to normal activities.3
Instead, pain management via multimodal analgesic approaches is necessary to achieve pain relief. Regional anesthesia techniques, such as transverse abdominis plane blocks,4 epidural anesthesia,5 paravertebral block6 and erector spinae plane (ESPB)7 block, have gained popularity for the management of pain associated with abdominal surgeries.
ESPB block, interfacial plane block, involves injecting local anesthetic into the fascial plane situated between the erector spinae muscle and the transverse process of the spine.8 ESPB is a unique regional anesthetic method regarded as an effective block of peripheral nerve for postoperative analgesia due to its improved technical simplicity, decreased hypotension rate, and avoidance of hematoma.9
Dexmedetomidine (DEX) is an alpha 2-adrenoceptor agonist that exhibits remarkable selectivity. It possesses sympatholytic, sedative, anxiolytic, and analgesic-sparing properties while also causing minimal respiratory function impairment.10
We hypothesized that adding DEX 0.5 µg/kg to bupivacaine 0.25% in ultrasound guided ESPB block can improve postoperative analgesia during LC. Hence, this work was conducted to study the effectiveness of adding DEX to ESPB in pain control in LC.
This controlled, randomized, double-blinded trial included 60 cases of 18 to 65 years, both sexes, physical status I-II based on the American Society of Anesthesiologists (ASA), scheduled for LC. The study was started from January 2022 to November 2022 after approval by the Tanta Faculty of Medicine Research Ethics Committee, Tanta, Egypt (Approval Code: 35225/1/22), and registration at clinicaltrials.gov (ID: NCT05369468). The patient’s informed written consent was acquired.
Exclusion criteria were spine or chest wall deformity, contamination of the injection location, coagulopathy, allergies to trial medications, and uncooperative patients who could not use a visual analog scale (VAS) to describe their pain.
2.1. Sample size calculation
This sample size was calculated using G*Power 3.1.9.2 (Universitat Kiel, Germany). The mean ± SD total amount of morphine consumption 24h postoperative was 6.08 ± 3.66 mg in ESPB, as reported by Cesur et al.11 To detect a 50% difference between groups with α error of 0.05 and 80% power of the study, a minimum of 24 patients per group was calculated for statistical significance. To account for potential dropouts, 30 patients were included per group.
2.2. Randomization and Blindness
Using a computer-generated random number sequence, 60 cases were assigned between two groups via opaque, sealed envelopes in a parallel manner. Group I: received bilateral ESPB (20 mL bupivacaine 0.25% on each side). Group II: received bilateral ESPB (18mL of bupivacaine 0.25% plus 2 mL DEX 0.5 ug/kg diluted in saline on each side). The investigator and outcomes assessor were blinded. Blind nurses grouped patients by random numbers. Without trial involvement, a devoted pharmacist prepared the research solutions. An anesthesiologist who was uninformed of the group assignment took intraoperative and postoperative measurements.
Pre-operatively, all cases were informed about VAS instructions and underwent the assessment of history taking, general examination, and laboratory investigations.
A cannula was placed, and 7 mL/kg of lactated ringer solution was infused intravenously (IV). Pulse oximetry, non-invasive blood pressure, a three-lead electrocardiogram, a temperature probe, and capnography monitored the patients. Under complete aseptic conditions, the blocks were performed before inducing general anesthesia (GA).
2.3. Ultrasound Guided ESPB
In the sitting position, the ESPB was done under US direction (Philips® CX50 extreme edition, USA). A longitudinal parasagittal transducer probe (6–12 MHz) was placed lateral to the T9 spinous process by three cm; hence, the rhomboid major, trapezius, and ES muscles became visible. A 22-gauge, 50-mm blunt in-plane catheter (B. Braun Medical Inc., Bethlehem, PA) was inserted. The needle tip entered the fascial plane on the deep (anterior) ES muscle. The evident fluid spread elevated the ES muscle off the transverse process, confirming the needle position. The LA was injected according to the group allocation.
Thirty minutes after administration, the absence of a pinprick sensation at the injection site confirmed the effectiveness of the block. Cases with unsuccessful blocks were excluded from the study and replaced with others.
To induce GA, propofol 2 mg/kg, fentanyl 1 µg/kg, and rocuronium bromide 0.6 mg/kg were used IV. A bispectral index of 40-60 was maintained with oxygen and 2% sevoflurane. 0.3 mg of supplemental rocuronium maintained neuromuscular relaxation. When heart rate (HR) or mean arterial pressure (MAP) increased by more than 20% from baseline, further 1 µg/kg IV doses of fentanyl were given. The total amount of fentanyl consumed intraoperatively was recorded. On mechanical ventilation, the end-tidal CO2 was sustained between 35-40 mmHg.
Finally, all anesthetics were stopped. Reversing the neuromuscular blocking was done using 1.0 mg atropine and 2.5 mg neostigmine IV. Patients were moved to the post-anesthesia care unit (PACU) and discharged when they reached modified Aldrete scores ≥ 9.
HR and MAP were assessed at baseline, 5, 10, and 15 min after block and 30, 60, and 90 min intraoperative and at PACU, 1, 3, 6, 12, 18, and 24h postoperative.
VAS score was assessed at discharge, 20 min, 1, 3, 6, 10, 12, 18, and 24h postoperative. In patients with a VAS score >3, IV 0.1 mg/kg morphine was injected as a rescue analgesic. The total 24h postoperative consumption of morphine and time to 1st analgesic request were recorded. 4-point scale (severe, moderate, mild, or none) was used for sedation assessment.
Postoperative complications were recorded, including bradycardia (HR< 50 beats/min; treated with IV 0.6 mg atropine), hypotension (MAP <65mmHg or < 20% of baseline; treated with IV 5 mg ephedrine and/or normal saline), and PONV (treated with IV 4 mg ondansetron).
Total morphine consumption 24 h postoperatively was the 1ry outcome. The pain score, total intraoperative consumption of fentanyl, the first time to request analgesia, and sedation were the 2ry outcomes.
Fisher's exact test assessed the qualitative data which were expressed as frequency and percentage (%). A two-tailed P < 0.05 was significant.
2.4. Statistical analysis
SPSS v27 (IBM, Armonk, NY, USA) analyzed data. Histograms and the Shapiro-Wilks test determined data normality. The unpaired student t-test compared parametric data which were expressed as mean and SD.
The Mann-Whitney test compared non-parametric data which were expressed as median and IQR. Chi-square or
In our study, 84 patients were screened for inclusion in the trial; 18 did not meet the criteria, and six declined. The remaining 60 patients were equally randomized between the two groups. All assigned patients were analyzed and followed up (Figure 1).
Figure 1: CONSORT flowchart of the enrolled patients.
The characteristics of patients and surgical duration were similar between both groups (Table 1).
HR was significantly lower in Group II in comparison with Group I at 60min and 90min intraoperatively (P = 0.045 and 0.007 respectively) and at PACU, 1, 3, 6, 12 h, and 18 h postoperatively (P = 0.006, 0.014, 0.002, 0.010, 0.024, and < 0.001 respectively.
MAP was significantly lower in Group II in comparison with Group I at 60min and 90min intraoperatively (P = 0.002 and <0.001 respectively) and at PACU,1h, 3 h, 6 h, 12 h and 18 h postoperatively (P < 0.001).
VAS measurements at discharge, 20 min, 1h, 3h, and 6h were comparable among groups and were significantly lower at 10h, 12h, and 18h in Group II than in Group I (P = 0.003, 0.002 and 0.010 respectively) (Figure 2).
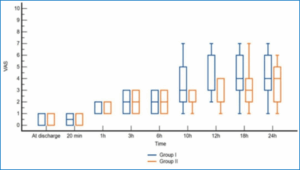
Figure 2: Comparative Visual Analog Scale of the studied groups.
The intraoperative fentanyl consumption was 154.33 ± 32.98 µg in Group I and 144 ± 17.73 µg in Group II. The mean consumption of morphine in first 24 h postoperative was 16.41 ± 5.43 mg in Group I and 10.59 ± 3.51 mg in Group II. The time to first rescue analgesia was 9.63 ± 1.77 h in Group I and 16.97 ± 4.66 h in Group II. Intraoperative fentanyl consumption was insignificantly different between both groups. 24h postoperative morphine consumed was reduced significantly in Group II than in Group I (P < 0.001). The first time to analgesic request was delayed significantly in Group II than in Group I (P < 0.001) (Table 2).
The 4-point sedation scale was significantly better in Group II contrasted to Group I (P = 0.031). Bradycardia, hypotension, and PONV were similar between groups (Table 3).
The major finding in our study demonstrated that combined DEX to US-guided ESPB improved the analgesia and decreased the consumption of morphine postoperatively, and pain score with prolonged analgesia and better hemodynamics and sedation scale.
The spinal nerve’s anatomical location and the spinal column's different anatomy may play an important role in determining the surgery's outcome following the ESPB placement.12 bupivacaine is the most used LA for nerve blocks. However, its duration of action is a major limiting factor, so adding adjuvants like epinephrine, dexamethasone, midazolam, ketamine, and DEX.13
DEX is selective for the beta-2 adrenoceptors, making it α2 -adrenoceptor agonist. The effect of LA was augmented by the influence of DEX as central nervous system (CNS) presynaptic activation of alpha 2 adrenoceptor and its postsynaptic activation and terminating the propagation of pain signals.\14 Prolonged postoperative analgesia and improved pain management have been shown when DEX added to LA in neuraxial and peripheral nerve blocks.15
The analgesic action of perineuronal DEX refers to the increase of cation channels triggered via hyperpolarization, which restricts the nerve's membrane potential from reverting to its resting status for future discharge following hyperpolarization.16
In agreement with our results, Shaker et al.17 found that DEX as an additive to 0.25% bupivacaine in US-guided ESPB leads to reducing overall opioid use and delaying the need for rescue analgesia and VAS score following modified radical mastectomy.
Wang et al.18 showed that adding 0.5 µg/kg DEX to 0.5% ropivacaine significantly prolonged the analgesic period of ESPB without increasing the risk of adverse effects in patients having open thoracotomy.
Moreover, a longer duration of analgesia and decreased pain score with stable hemodynamics after shoulder arthroscopy were found when DEX was added to US-guided ESPB, as well as a decrease in intraoperative fentanyl and postoperative morphine consumption, as evidenced by Hamed et al.19
Yang et al.20 reported delayed time to initial postoperative analgesia, as well as reduced opioid intake during the surgery. Analgesic dose and pain score in DEX were added to the ESPB group than ESPB alone.
Ahmed et al.21 showed that DEX was shown to be an effective adjuvant to ESPB, extending the analgesia's duration and decreasing pain ratings both at rest and during movement.
Harmoniously, the addition of 0.5 µg/kg DEX to ESPB did not result in noticeable alterations in HR or MAP. That is mainly because the dosage of DEX was low, and the average age of the trial participants was not particularly high. There was also no significant variation in the occurrence of other side effects such hypotension, bradycardia, or PONV between groups.
When combined with LA, DEX may increase the ESPB duration in thoracoscopy patients by 120% (about 18 h).22 The addition of DEX to LA has been shown by Wang et al.18 to increase the duration of the nerve block, improve intraoperative hemodynamic stability, decrease postoperative pain rating, and decrease the need for postoperative analgesics. In a study comparing two doses of DEX (50 and 75 μg), Abdulatif et al.23 found that although the lower dosage (50 μg) decreased postoperative morphine usage, sensory block duration, and rescue analgesia time, the higher dose (75 μg) increased the risk of hypotension. Consequences such as bradycardia or hypotension should be tracked when using large doses of DEX as an adjunct for nerve block. Therefore, a DEX dose of 0.5 μg/kg was used in this study.
Nevertheless, Esmaoglu et al.24 highlighted that after adding 100 µg of DEX to the LA, the HR and MAP reduced dramatically within two hours. Previous studies25 have reported considerable hypotension and bradycardia after DEX administration. That was not observed in our as we decreased the dosage of DEX (0.5 µg/kg).
Additionally, Jian et al.26 concluded that DEX combined as an additive to ESPB is more effective than ESPB alone regarding overall analgesia with lower pain score and rehabilitation after thoracoscopic wedge resection.
Adding 0.5 µg/kg DEX to LA decreased the overall opioid use on the first day postoperatively, as shown by Xu et al.27 Adding DEX during the first 48 hours after surgery dramatically reduced overall postoperative opioid usage, according to the findings by Wang et al. [18]. We suggested that the addition of DEX in ESPB led to the reduction of opioid use.
Our research was limited by involving a limited sample size to demonstrate the effectiveness of the block. Also, no control group received systemic analgesia. Different additives with different volumes and concentrations in further studies are recommended.
As an adjuvant to ESPB during LC, DEX improved analgesia by decreasing postoperative morphine consumption and pain score with relatively better hemodynamics.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
AMA: Conceptualization, First draft of the manuscript
WMAA: Data collection
AFA: Data analysis
All authors approved this manuscript for submission.
Authors affiliations:
- Amany Mohamed Abotaleb, Lecturer, Department of Anesthesiology, Surgical Intensive Care & Pain Medicine, Faculty of Medicine, Tanta University, Tanta, Egypt; Email: amanyabotaleb111@gmail.com
- Wafaa Madhy Atia Abdelwahed, Lecturer, Department of Anesthesiology, Surgical Intensive Care & Pain Medicine, Faculty of Medicine, Tanta University, Tanta, Egypt; Email; wafaa.madhy@gmail.com
- Asmaa Fawzy Amer, Assistant Professor. Department of Anesthesiology, Surgical Intensive Care & Pain Medicine, Faculty of Medicine, Tanta University, Tanta, Egypt; Email: asmaafawzyamer@yahoo.com
ABSTRACT
Background & Objective: laparoscopic cholecystectomy (LC) is routinely performed under general anesthesia, but relieving postoperative pain poses real challenge, with the current trends of avoiding narcotic analgesics, due to their serious side effects. Addition of regional nerve blocks offers a more flexible analgesic approach. We hypnotized that addition of dexmedetomidine to the local anesthetic solution will prolong the duration of the blocks. This study aimed to assess the effectiveness of erector spinae plane block (ESPB) using bupivacaine and dexmedetomidine (DEX) for pain controlling in LC.
Methods: This randomized, controlled, double-masked trial included 60 cases of 18 to 65 years, both genders, scheduled for LC. Cases were randomized equally into two groups: Group I received ESPB with 20 mL of bupivacaine 0.25% on each side, and Group II received ESPB with 18 mL of bupivacaine 0.25% plus 2 mL of DEX 0.5 µg/kg on each side. Blocks were performed before the induction of general anesthesia. Ultrasonography was used to guide the ESPB.
Results: The morphine consumption in 1st 24 hours postoperatively was reduced significantly in group II contrasted to group I (10.59 ± 3.51 vs. 16.41 ± 5.43 mg, P < 0.001). The time to first rescue analgesia was delayed significantly in group II than in Group I (16.97 ± 4.66 vs 9.63 ± 1.77 h, P < 0.001). Intraoperative fentanyl consumption was insignificantly different between both groups. Postoperative visual analog scale, heart rate, and mean blood pressure measurements at post-anesthesia care unit 1, 3, 6, 12, and 18 h were reduced significantly in Group II than in Group I (P < 0.001). The 4-point sedation scale was significantly better in Group II than Group I (P = 0.031).
Conclusion: DEX as an adjuvant to ESPB during LC improved analgesia by reducing the need for morphine postoperatively, as well as the pain level with relatively better hemodynamics.
Abbreviations: ESPB: erector spinae plane block, LC: Laparoscopic Cholecystectomy, VAS: visual analog scale,
Keywords: Analgesia; Dexmedetomidine; Laparoscopic Cholecystectomy; Erector Spinae Plane Block; Bupivacaine
Citation: Abotaleb AM, Abdelwahed WMA, Amer AF. Analgesic efficacy of adding dexmedetomidine to bupivacaine in ultrasound-guided erector spinae plane block for laparoscopic cholecystectomy: a randomized, controlled, double-blind trial. Anaesth. pain intensive care 2025;29(4):318-25. DOI: 10.35975/apic.v29i4.2816
Received: October 22, 2024; Revised: January 27, 2025; Accepted: February 03, 2025
1. INTRODUCTION
Laparoscopic cholecystectomy (LC) is a common ambulatory surgery. Even though LC is considered a minimally invasive procedure, postoperative pain is evident.1 Post-LC pain can manifest in a spectrum of intensity, from mild to severe. The pain experience typically comprises three distinct components: incisional pain arising from the trocar site, visceral pain arising from the surgical intervention, and shoulder discomfort, which is thought to be a manifestation of visceral pain transmitted to the referred pain area.2
Efficient management of postoperative pain control is essential in optimizing recovery outcomes in laparoscopic surgeries, as it enables patients to experience reduced discomfort, improved mobility, and a more rapid return to normal activities.3
Instead, pain management via multimodal analgesic approaches is necessary to achieve pain relief. Regional anesthesia techniques, such as transverse abdominis plane blocks,4 epidural anesthesia,5 paravertebral block6 and erector spinae plane (ESPB)7 block, have gained popularity for the management of pain associated with abdominal surgeries.
ESPB block, interfacial plane block, involves injecting local anesthetic into the fascial plane situated between the erector spinae muscle and the transverse process of the spine.8 ESPB is a unique regional anesthetic method regarded as an effective block of peripheral nerve for postoperative analgesia due to its improved technical simplicity, decreased hypotension rate, and avoidance of hematoma.9
Dexmedetomidine (DEX) is an alpha 2-adrenoceptor agonist that exhibits remarkable selectivity. It possesses sympatholytic, sedative, anxiolytic, and analgesic-sparing properties while also causing minimal respiratory function impairment.10
We hypothesized that adding DEX 0.5 µg/kg to bupivacaine 0.25% in ultrasound guided ESPB block can improve postoperative analgesia during LC. Hence, this work was conducted to study the effectiveness of adding DEX to ESPB in pain control in LC.
2. METHODOLOGY
This controlled, randomized, double-blinded trial included 60 cases of 18 to 65 years, both sexes, physical status I-II based on the American Society of Anesthesiologists (ASA), scheduled for LC. The study was started from January 2022 to November 2022 after approval by the Tanta Faculty of Medicine Research Ethics Committee, Tanta, Egypt (Approval Code: 35225/1/22), and registration at clinicaltrials.gov (ID: NCT05369468). The patient’s informed written consent was acquired.
Exclusion criteria were spine or chest wall deformity, contamination of the injection location, coagulopathy, allergies to trial medications, and uncooperative patients who could not use a visual analog scale (VAS) to describe their pain.
2.1. Sample size calculation
This sample size was calculated using G*Power 3.1.9.2 (Universitat Kiel, Germany). The mean ± SD total amount of morphine consumption 24h postoperative was 6.08 ± 3.66 mg in ESPB, as reported by Cesur et al.11 To detect a 50% difference between groups with α error of 0.05 and 80% power of the study, a minimum of 24 patients per group was calculated for statistical significance. To account for potential dropouts, 30 patients were included per group.
2.2. Randomization and Blindness
Using a computer-generated random number sequence, 60 cases were assigned between two groups via opaque, sealed envelopes in a parallel manner. Group I: received bilateral ESPB (20 mL bupivacaine 0.25% on each side). Group II: received bilateral ESPB (18mL of bupivacaine 0.25% plus 2 mL DEX 0.5 ug/kg diluted in saline on each side). The investigator and outcomes assessor were blinded. Blind nurses grouped patients by random numbers. Without trial involvement, a devoted pharmacist prepared the research solutions. An anesthesiologist who was uninformed of the group assignment took intraoperative and postoperative measurements.
Pre-operatively, all cases were informed about VAS instructions and underwent the assessment of history taking, general examination, and laboratory investigations.
A cannula was placed, and 7 mL/kg of lactated ringer solution was infused intravenously (IV). Pulse oximetry, non-invasive blood pressure, a three-lead electrocardiogram, a temperature probe, and capnography monitored the patients. Under complete aseptic conditions, the blocks were performed before inducing general anesthesia (GA).
2.3. Ultrasound Guided ESPB
In the sitting position, the ESPB was done under US direction (Philips® CX50 extreme edition, USA). A longitudinal parasagittal transducer probe (6–12 MHz) was placed lateral to the T9 spinous process by three cm; hence, the rhomboid major, trapezius, and ES muscles became visible. A 22-gauge, 50-mm blunt in-plane catheter (B. Braun Medical Inc., Bethlehem, PA) was inserted. The needle tip entered the fascial plane on the deep (anterior) ES muscle. The evident fluid spread elevated the ES muscle off the transverse process, confirming the needle position. The LA was injected according to the group allocation.
Thirty minutes after administration, the absence of a pinprick sensation at the injection site confirmed the effectiveness of the block. Cases with unsuccessful blocks were excluded from the study and replaced with others.
To induce GA, propofol 2 mg/kg, fentanyl 1 µg/kg, and rocuronium bromide 0.6 mg/kg were used IV. A bispectral index of 40-60 was maintained with oxygen and 2% sevoflurane. 0.3 mg of supplemental rocuronium maintained neuromuscular relaxation. When heart rate (HR) or mean arterial pressure (MAP) increased by more than 20% from baseline, further 1 µg/kg IV doses of fentanyl were given. The total amount of fentanyl consumed intraoperatively was recorded. On mechanical ventilation, the end-tidal CO2 was sustained between 35-40 mmHg.
Finally, all anesthetics were stopped. Reversing the neuromuscular blocking was done using 1.0 mg atropine and 2.5 mg neostigmine IV. Patients were moved to the post-anesthesia care unit (PACU) and discharged when they reached modified Aldrete scores ≥ 9.
HR and MAP were assessed at baseline, 5, 10, and 15 min after block and 30, 60, and 90 min intraoperative and at PACU, 1, 3, 6, 12, 18, and 24h postoperative.
VAS score was assessed at discharge, 20 min, 1, 3, 6, 10, 12, 18, and 24h postoperative. In patients with a VAS score >3, IV 0.1 mg/kg morphine was injected as a rescue analgesic. The total 24h postoperative consumption of morphine and time to 1st analgesic request were recorded. 4-point scale (severe, moderate, mild, or none) was used for sedation assessment.
Postoperative complications were recorded, including bradycardia (HR< 50 beats/min; treated with IV 0.6 mg atropine), hypotension (MAP <65mmHg or < 20% of baseline; treated with IV 5 mg ephedrine and/or normal saline), and PONV (treated with IV 4 mg ondansetron).
Total morphine consumption 24 h postoperatively was the 1ry outcome. The pain score, total intraoperative consumption of fentanyl, the first time to request analgesia, and sedation were the 2ry outcomes.
Fisher's exact test assessed the qualitative data which were expressed as frequency and percentage (%). A two-tailed P < 0.05 was significant.
2.4. Statistical analysis
SPSS v27 (IBM, Armonk, NY, USA) analyzed data. Histograms and the Shapiro-Wilks test determined data normality. The unpaired student t-test compared parametric data which were expressed as mean and SD.
The Mann-Whitney test compared non-parametric data which were expressed as median and IQR. Chi-square or
3. RESULTS
In our study, 84 patients were screened for inclusion in the trial; 18 did not meet the criteria, and six declined. The remaining 60 patients were equally randomized between the two groups. All assigned patients were analyzed and followed up (Figure 1).

Figure 1: CONSORT flowchart of the enrolled patients.
The characteristics of patients and surgical duration were similar between both groups (Table 1).
| Table 1: Characteristics of patients and surgical duration of the studied groups | |||||
| Variables | Group I
(n = 30) |
Group II
(n = 30) |
95% CI | P value | |
| Age (years) | 44.27 ± 12.95 | 41.1 ± 13.34 | -3.63 - 9.96 | 0.355 | |
| Sex | Male | 13 (43.33) | 12 (40) | 0.59-1.97 | 0.793 |
| Female | 17 (56.67) | 18 (60) | |||
| Weight (kg) | 71.6 ± 9.53 | 69.57 ± 7.39 | -2.37 - 6.44 | 0.360 | |
| Height (m) | 1.73 ± 0.08 | 1.72 ± 0.08 | -0.04 -0.05 | 0.829 | |
| BMI (kg/m2) | 24.17 ± 4.22 | 23.55 ± 3.24 | -1.32 - 2.57 | 0.524 | |
| ASA physical status | I | 22 (73.33) | 19 (63.33) | 0.53-4.77 | 0.405 |
| II | 8 (26.67) | 11 (36.67) | |||
| Surgical duration (min) | 83.8 ± 13.32 | 81.1 ± 14 | -4.39 -9.72 | 0.453 | |
| Data are expressed as mean ± SD or frequency (%). ASA: American Society of Anesthesiologists, BMI: Body mass index. | |||||
HR was significantly lower in Group II in comparison with Group I at 60min and 90min intraoperatively (P = 0.045 and 0.007 respectively) and at PACU, 1, 3, 6, 12 h, and 18 h postoperatively (P = 0.006, 0.014, 0.002, 0.010, 0.024, and < 0.001 respectively.
MAP was significantly lower in Group II in comparison with Group I at 60min and 90min intraoperatively (P = 0.002 and <0.001 respectively) and at PACU,1h, 3 h, 6 h, 12 h and 18 h postoperatively (P < 0.001).
VAS measurements at discharge, 20 min, 1h, 3h, and 6h were comparable among groups and were significantly lower at 10h, 12h, and 18h in Group II than in Group I (P = 0.003, 0.002 and 0.010 respectively) (Figure 2).

Figure 2: Comparative Visual Analog Scale of the studied groups.
The intraoperative fentanyl consumption was 154.33 ± 32.98 µg in Group I and 144 ± 17.73 µg in Group II. The mean consumption of morphine in first 24 h postoperative was 16.41 ± 5.43 mg in Group I and 10.59 ± 3.51 mg in Group II. The time to first rescue analgesia was 9.63 ± 1.77 h in Group I and 16.97 ± 4.66 h in Group II. Intraoperative fentanyl consumption was insignificantly different between both groups. 24h postoperative morphine consumed was reduced significantly in Group II than in Group I (P < 0.001). The first time to analgesic request was delayed significantly in Group II than in Group I (P < 0.001) (Table 2).
| Table 2: Consumption of fentanyl intraoperatively and morphine postoperatively among group | ||||
| Narcotics used | Group I (n = 30) |
Group II (n = 30) |
95% CI | P value |
| Intraoperative fentanyl consumption (µg) | 154.33 ± 32.98 | 144 ± 17.73 | 41.69 - 63.91 | 0.136 |
| 1st 24h postop morphine consumption (mg) | 16.41 ± 5.43 | 10.59 ± 3.51 | 3.46 - 8.18 | <0.001* |
| Time to first analgesic request (h) | 9.63 ± 1.77 | 16.97 ± 4.66 | -9.15 - -5.51 | <0.001* |
| Data are expressed as mean ± SD or frequency (%), *: Significant as P ≤ 0.05. | ||||
The 4-point sedation scale was significantly better in Group II contrasted to Group I (P = 0.031). Bradycardia, hypotension, and PONV were similar between groups (Table 3).
| Table 3: Sedation scale and complications | ||||
| Parameters | Group I
(n = 30) |
Group II (n = 30) |
P value | |
| 4-point sedation scale | None | 28 (93.33) | 20 (66.67) | 0.031* |
| Mild | 2 (6.67) | 8 (26.67) | ||
| Moderate | 0 (0) | 2 (6.67) | ||
| Severe | 0 (0) | 0 (0) | ||
| Complications | Bradycardia | 2 (6.67) | 3 (10) | 1 |
| Hypotension | 1 (3.33) | 2 (6.67) | 1 | |
| PONV | 4 (13.33) | 1 (3.33) | 0.353 | |
| Data are expressed as mean ± SD or frequency (%), *: Significant as P ≤ 0.05. PONV: postoperative nausea and vomiting. | ||||
4. DISCUSSION
The major finding in our study demonstrated that combined DEX to US-guided ESPB improved the analgesia and decreased the consumption of morphine postoperatively, and pain score with prolonged analgesia and better hemodynamics and sedation scale.
The spinal nerve’s anatomical location and the spinal column's different anatomy may play an important role in determining the surgery's outcome following the ESPB placement.12 bupivacaine is the most used LA for nerve blocks. However, its duration of action is a major limiting factor, so adding adjuvants like epinephrine, dexamethasone, midazolam, ketamine, and DEX.13
DEX is selective for the beta-2 adrenoceptors, making it α2 -adrenoceptor agonist. The effect of LA was augmented by the influence of DEX as central nervous system (CNS) presynaptic activation of alpha 2 adrenoceptor and its postsynaptic activation and terminating the propagation of pain signals.\14 Prolonged postoperative analgesia and improved pain management have been shown when DEX added to LA in neuraxial and peripheral nerve blocks.15
The analgesic action of perineuronal DEX refers to the increase of cation channels triggered via hyperpolarization, which restricts the nerve's membrane potential from reverting to its resting status for future discharge following hyperpolarization.16
In agreement with our results, Shaker et al.17 found that DEX as an additive to 0.25% bupivacaine in US-guided ESPB leads to reducing overall opioid use and delaying the need for rescue analgesia and VAS score following modified radical mastectomy.
Wang et al.18 showed that adding 0.5 µg/kg DEX to 0.5% ropivacaine significantly prolonged the analgesic period of ESPB without increasing the risk of adverse effects in patients having open thoracotomy.
Moreover, a longer duration of analgesia and decreased pain score with stable hemodynamics after shoulder arthroscopy were found when DEX was added to US-guided ESPB, as well as a decrease in intraoperative fentanyl and postoperative morphine consumption, as evidenced by Hamed et al.19
Yang et al.20 reported delayed time to initial postoperative analgesia, as well as reduced opioid intake during the surgery. Analgesic dose and pain score in DEX were added to the ESPB group than ESPB alone.
Ahmed et al.21 showed that DEX was shown to be an effective adjuvant to ESPB, extending the analgesia's duration and decreasing pain ratings both at rest and during movement.
Harmoniously, the addition of 0.5 µg/kg DEX to ESPB did not result in noticeable alterations in HR or MAP. That is mainly because the dosage of DEX was low, and the average age of the trial participants was not particularly high. There was also no significant variation in the occurrence of other side effects such hypotension, bradycardia, or PONV between groups.
When combined with LA, DEX may increase the ESPB duration in thoracoscopy patients by 120% (about 18 h).22 The addition of DEX to LA has been shown by Wang et al.18 to increase the duration of the nerve block, improve intraoperative hemodynamic stability, decrease postoperative pain rating, and decrease the need for postoperative analgesics. In a study comparing two doses of DEX (50 and 75 μg), Abdulatif et al.23 found that although the lower dosage (50 μg) decreased postoperative morphine usage, sensory block duration, and rescue analgesia time, the higher dose (75 μg) increased the risk of hypotension. Consequences such as bradycardia or hypotension should be tracked when using large doses of DEX as an adjunct for nerve block. Therefore, a DEX dose of 0.5 μg/kg was used in this study.
Nevertheless, Esmaoglu et al.24 highlighted that after adding 100 µg of DEX to the LA, the HR and MAP reduced dramatically within two hours. Previous studies25 have reported considerable hypotension and bradycardia after DEX administration. That was not observed in our as we decreased the dosage of DEX (0.5 µg/kg).
Additionally, Jian et al.26 concluded that DEX combined as an additive to ESPB is more effective than ESPB alone regarding overall analgesia with lower pain score and rehabilitation after thoracoscopic wedge resection.
Adding 0.5 µg/kg DEX to LA decreased the overall opioid use on the first day postoperatively, as shown by Xu et al.27 Adding DEX during the first 48 hours after surgery dramatically reduced overall postoperative opioid usage, according to the findings by Wang et al. [18]. We suggested that the addition of DEX in ESPB led to the reduction of opioid use.
5. LIMITATIONS
Our research was limited by involving a limited sample size to demonstrate the effectiveness of the block. Also, no control group received systemic analgesia. Different additives with different volumes and concentrations in further studies are recommended.
6. CONCLUSION
As an adjuvant to ESPB during LC, DEX improved analgesia by decreasing postoperative morphine consumption and pain score with relatively better hemodynamics.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
AMA: Conceptualization, First draft of the manuscript
WMAA: Data collection
AFA: Data analysis
All authors approved this manuscript for submission.
11. REFERENCES
- Tulgar S, Kapakli MS, Senturk O, Selvi O, Serifsoy TE, Ozer Z. Evaluation of ultrasound-guided erector spinae plane block for postoperative analgesia in laparoscopic cholecystectomy: a prospective, randomized, controlled clinical trial. J Clin Anesth. 2018;49:101-6. [PubMed] DOI: 1016/j.jclinane.2018.06.019
- Olsen SW, Lehmkuhl L, Hamborg LW, Torkov AKA, Fog-Nielsen R, Lauridsen J. Postoperative pain relief after ambulatory laparoscopic surgery: a nonmatched case-control study. J Perianesth Nurs. 2024;39:254-62. [PubMed] DOI: 1016/j.jopan.2023.08.002
- Sia CJY, Wee SL, Au-Yong PS, Lie SA, Tan WJ, Foo FJ, et al. Analgesia efficacy of erector spinae plane block in laparoscopic abdominal surgeries: a systemic review and meta-analysis. Int J Surg. 2024;10:97. [PubMed] DOI: 1097/JS9.0000000000001421
- Elamin G, Waters PS, Hamid H, O'Keeffe HM, Waldron RM, Duggan M, et al. Efficacy of a laparoscopically delivered transversus abdominis plane block technique during elective laparoscopic cholecystectomy: a prospective, double-blind randomized trial. J Am Coll Surg. 2015;221:335-44. [PubMed] DOI: 1016/j.jamcollsurg.2015.03.030
- Gramatica L, Brasesco O, Luna AM, Martinessi V, Panebianco G, Labaque F, et al. Laparoscopic cholecystectomy performed under regional anesthesia in patients with chronic obstructive pulmonary disease. Surg Endosc. 2002;16:472-5. [PubMed] DOI: 1007/s00464-001-8148-0
- Aydin G, Aydin O. The efficacy of ultrasound-guided paravertebral block in laparoscopic cholecystectomy. Medicina. 2018;54:75. [PubMed] DOI: 3390/medicina54050075
- Mahrous MAS, Kamal MM, Elhennawy AM, Youssef AMA. Comparative study between erector spinae plane block and transversus abdominis plane block for postoperative pain management of patients undergoing laparoscopic cholecystectomy. QJM. 2023;116. DOI: 1093/qjmed/hcad069.061
- Daghmouri MA, Akremi S, Chaouch MA, Mesbahi M, Amouri N, Jaoua H, et al. Bilateral erector spinae plane block for postoperative analgesia in laparoscopic cholecystectomy: a systematic review and meta-analysis of randomized controlled trials. Pain Pract. 2021;21:357-65. [PubMed] DOI: 1111/papr.12953
- Zhang Z, Kong H, Li Y, Xu ZZ, Li X, Ma JH, et al. Erector spinae plane block versus quadratus lumborum block for postoperative analgesia after laparoscopic nephrectomy: a randomized controlled trial. J Clin Anesth. 2024;96:111466. [PubMed] DOI: 1016/j.jclinane.2024.111466
- Memiş D, Turan A, Karamanlioğlu B, Pamukçu Z, Kurt I. Adding dexmedetomidine to lidocaine for intravenous regional anesthesia. Anesth Analg. 2004;98:835-40. [PubMed] DOI: 1213/01.ane.0000100680.77978.66
- Cesur S, Hu YRKL, Aksu C, Ku A. Bilateral versus unilateral erector spinae plane block for postoperative analgesia in laparoscopic cholecystectomy: a randomized controlled study. Braz J Anesthesiol. 2023;73:72-7. [PubMed] DOI: 1016/j.bjane.2021.04.020
- Kendall MC, Alves L, Traill LL, De Oliveira GS. The effect of ultrasound-guided erector spinae plane block on postsurgical pain: a meta-analysis of randomized controlled trials. BMC Anesthesiol. 2020;20:99-108. [PubMed] DOI: 1186/s12871-020-01016-8
- Laiq N, Khan MN, Arif M, Khan S. Midazolam with bupivacaine for improving analgesia quality in brachial plexus block for upper limb surgeries. J Coll Physicians Surg Pak. 2008;18:674-8. [PubMed]
- El-Tahawy M, Shaaban A, Ahmad A. Comparison between dexmedetomidine and magnesium sulfate as adjuvants for intravenous regional anesthesia. Ain Shams J Anesthesiol. 2015;8:129-36.
- Castillo RL, Ibacache M, Cortínez I, Carrasco-Pozo C, Farías JG, Carrasco RA, et al. Dexmedetomidine improves cardiovascular and ventilatory outcomes in critically ill patients: basic and clinical approaches. Front Pharmacol. 2020;10:16-24. [PubMed] DOI: 3389/fphar.2019.01641
- Wang X, Ran G, Chen X, Xie C, Wang J, Liu X, et al. The effect of ultrasound-guided erector spinae plane block combined with dexmedetomidine on postoperative analgesia in patients undergoing modified radical mastectomy: a randomized controlled trial. Pain Ther. 2021;10:475-84. [PubMed] DOI: 1007/s40122-020-00234-9
- Shaker EH, Soliman AM, Bedewy AAE, Adlan S. Dexmedetomidine versus ketamine in erector spinae plane block for postoperative analgesia following modified radical mastectomy: a prospective randomized controlled blinded study. Egypt J Anaesth. 2024;40:184-92. DOI: 1080/11101849.2024.2331351
- Wang Q, Li H, Wei S, Zhang G, Ni C, Sun L, et al. Dexmedetomidine added to ropivacaine for ultrasound-guided erector spinae plane block prolongs analgesia duration and reduces perioperative opioid consumption after thoracotomy: a randomized, controlled clinical study. Clin J Pain. 2021;38:8-14. [PubMed] DOI: 1097/AJP.0000000000000992
- Hamed MA, Fargaly OS, Abdelghaffar RA, Moussa MA, Algyar MF. Dexmedetomidine as an adjuvant for high-thoracic erector spinae plane block for analgesia in shoulder arthroscopy: a randomized controlled study. BMC Anesthesiol. 2023;23:53-61. [PubMed] DOI: 1186/s12871-023-02014-2
- Yang J, Zhao M, Zhang XR, Wang XR, Wang ZH, Feng XY, et al. Ropivacaine with dexmedetomidine or dexamethasone in a thoracic paravertebral nerve block combined with an erector spinae plane block for thoracoscopic lobectomy analgesia: a randomized controlled trial. Drug Des Devel Ther. 2022;16:1561-71. [PubMed] DOI: 2147/DDDT.S366428
- Ahmed HI, Ali AR, El Emam NH, Magdy M, Gamal RM. Dexmedetomidine vs dexamethasone as adjuvants to levobupivacaine in ultrasound-guided erector spinae plane block for patients undergoing modified radical mastectomy: a randomized double-blind study. Anaesth Pain Intensive Care. 2023;27:65-72. DOI: 35975/apic.v27i1.2122
- Gao Z, Xiao Y, Wang Q, Li Y. Dexmedetomidine vs dexamethasone as adjuvant for ropivacaine in ultrasound-guided erector spinae plane block for thoracoscopic lobectomy: a randomized, double-blind, placebo-controlled trial. Ann Transl Med. 2019;7:668. [PubMed] DOI: 21037/atm.2019.10.74
- Abdulatif M, Fawzy M, Nassar H, Hasanin A, Ollaek M, Mohamed H. Perineural dexmedetomidine in femoral nerve block: a dose-finding randomized, controlled, double-blind study. Egypt J Anaesth. 2016;71:1177-85. [PubMed] DOI: 1111/anae.13603
- Esmaoglu A, Yegenoglu F, Akin A, Turk CY. Dexmedetomidine added to levobupivacaine prolongs axillary brachial plexus block. Anesth Analg. 2010;111:1548-51. [PubMed] DOI: 1213/ANE.0b013e3181fa3095
- Gad M, Elmetwally M. Dexmedetomidine with levobupivacaine in ultrasound-guided serratus plane block for modified radical mastectomy surgery. Res Opin Anesth Intensive Care. 2019;6:234-42. DOI: 4103/roaic.roaic_23_19
- Jian C, Shen Y, Fu H, Yu L. Erector spinae plane block with dexmedetomidine and ropivacaine of same dose, different concentrations: analgesic and rehabilitation effect in thoracoscopic wedge resection. BMC Anesthesiol. 2022;22:225-31. [PubMed] DOI: 1186/s12871-022-01768-5
- Xu L, Hu Z, Shen J, McQuillan PM. US-guided transversus abdominis and rectus sheath block with ropivacaine and dexmedetomidine in elderly high-risk patients. Minerva Anestesiol. 2018;84:18-24. [PubMed] DOI: 23736/S0375-9393.17.11538-5