Mahmoud A. Kamel 1, Eman Abdulfattah Mohammed Ismael 2, Somia Mohammed Abdel Aziz El Sheikh 3, Mai M. Elrawas 4, Ehab Hanafy Shaker Gendy 5
Authors affiliations:
Background & objectives: Mastectomy has been associated with moderate to severe postoperative pain, and radical mastectomy has always been cumbersome for the parients due to it being difficult to manage. During the recent past regional anesthesia techniques have gained popularity to be used alone or as a part of balanced anesthesia. We evaluated the safety and efficacy of ultrasound-guided erector spinae plane block (ESPB) in contrast to thoracic paravertebral block (TPVB) for postoperative analgesia following modified radical mastectomy (MRM).
Methodology: In this prospective, randomized, single-blind research, 44 women, scheduled for MRM, were enrolled. Patients were randomly assigned into two groups: Group TPVB recieved TPVB, and the second group received ESPB at the T5 level. Both blocks were performed prior to the induction of general anesthesia (GA) using 25 mL of 0.25 bupivacaine 0.25%, under aseptic conditions. Standard GA technique was used in both groups. Inj morphine IV was used as a rescue analgesic. Time to first analgesic, total analgesic dose and the number of patients requiring analgesia were noted.
Results: Both groups needed analgesia for a time that was comparable to one another. In the first twenty-four hours following the operation, between intraoperative fentanyl consumption, the number of cases requiring rescue morphine, total morphine consumption, pain VAS scores at rest, and movement did not show significant differences between the two groups. The hemodynamic variables remained comparable in both of the groups. Compared to TPVB group, the level of patient satisfaction was significantly higher with the ESPB group.
Conclusion: ESPB is an acceptable analgesic alternative to paravertebral block after MRM. ESPB is equally effective as TPVB in the duration of analgesia and postoperative pain and morphine usage during the first postoperative day.
Abbreviations: ESPB: Erector Spinae Plane Block, MRM: modified radical mastectomy, NCI: National Cancer Institute, TPVB: Thoracic Paravertebral Block, VAS: Visual Analogue Scale
Keywords: Analgesia; Erector Spinae Plane Block; Modified Radical Mastectomy; Paravertebral Block; Pain; Postoperative Pain; Ultrasound
Citation: Kamel MA, Ismael EAM, El Sheikh SMAZ, Elrawas MM, Gendy EHS. Ultrasound guided erector spinae plane block versus thoracic paravertebral block for perioperative analgesia for modified radical mastectomy: a randomized trial. Anaesth. pain intensive care 2025;29(4):292-300. DOI: 10.35975/apic.v29i4.2814
Received: January 27, 2025; Revised: May 01, 2025; Accepted: May 21, 2025
Around twelve percent of the world’s cancer burden is attributable to breast cancer (BC), making it the most frequent malignancy. Surgery is the predominant treatment modality, ranging from breast-conserving therapy to mastectomy with axillary clearance.1 Postoperative pain is a significant issue in breast cancer surgery, with over half of patients experiencing moderate to severe acute pain and 28% later developing chronic pain.2 Pain may result in delayed wound healing, hemodynamic complications, increased stress, respiratory depression, as well as a reduced quality of life by compromising psychological, physical, and social functioning if pain left untreated. The pain is exacerbated by the extensive tissue destruction that occurs throughout modified radical mastectomy (MRM)3 The MRM procedure involves the complete removal of the breast as well as the underlying fascia of the pectoralis major muscle, as well as the excision of level I and II axillary lymph nodes.4 Consequently, it is imperative to give precedence to the avoidance of postoperative pain in these patients.
Diverse analgesic techniques are employed to mitigate postoperative pain in BC surgery. Currently, opioids are still in use for pain management after BC surgery despite their potential adverse effects, including respiratory depression, drowsiness, constipation, nausea, and vomiting. Various regional methods, including paravertebral blocks and thoracic epidural anesthesia, offer superior pain management, enhance pulmonary function, and diminish perioperative opioid use.5
The thoracic paravertebral block (TPVB)6 has historically been the anesthesiologist’s preferred method for alleviating pain throughout and after MRM.7 However, TPVB is linked to many drawbacks, including pneumothorax, hypotension, central spread of local anesthetic, sympathetic block, or failure block, which may impose restrictions on the method. The utilization of fascial plane blocks in oncological breast surgery has been supported by evidence-based medicine in recent years. Erector spinae plane block (ESPB), serratus anterior plane blocks, and pectoral nerve blocks can enhance pain management, improve pulmonary function, and reduce perioperative opioid consumption.5
The objective of this investigation was to assess the safety and efficacy of ultrasound-guided ESPB contrasted with TPVB for postoperative analgesia following MRM.
ESPB and PVB groups were compared in a 1:1 ratio to investigate the duration of analgesia. In a previous study,9 with a pooled standard deviation of 6.8 hours, there was a variation of 1.7 hours in the duration of analgesia. We needed to enroll 22 patients in each group to reject the null hypothesis that the two groups’ population means are identical with a power of 0.8, according to these results. The probability of a Type I error in this test of the null hypothesis is 0.05.
The NCI’s Institutional Research Board approved this research (Code: AP1811-30104). Before enrolling in the trial, patients or their guardians gave written informed consent.
Forty- four women with breast cancer, scheduled for MRM, were randomly selected at the National Cancer Institute, Cairo University, for this prospective randomized, single-blind research between June 2021 and June 2023. Female people between the ages of 18 and 65 years. who had a history of breast cancer, undergoing elective unilateral mastectomy with general anesthesia (GA), and had a current American Society of Anesthesiologists physical status of I or II were included.
Exclusion criteria were coagulopathy, severe obesity (BMI > 40 kg/m2), history of narcotic pain medication use, major cardiac disorder, renal dysfunction, allergies to local anesthetics, decreased pulmonary reserve, psychiatric illness, preexisting neurological deficits, or infection at the block site.
Parallel computer-generated random numbers were utilized to divide patients into two equal groups, and their allocation codes were sealed in opaque envelopes. The TPVB was administered to the individuals in the TPVB Group (n = 22) by injecting 25 milliliters of 0.25 percent bupivacaine into the space between the pleura and the costotransverse ligament. The 22 patients who made up the ESPB Group were each given a 25-milliliter injection of 0.25 percent bupivacaine in a cephalic-to-caudal direction until the needle reached the T5 transverse process, which is located below the erector-spinae muscle.
The research was single-blinded, as the outcome assessors were blinded to group allocation. An anesthesiologist with experience in ultrasound-guided blocks, who did not take part in data collection or analysis, conducted all blocks prior to induction of GA.
All patients underwent meticulous history-taking and clinical evaluation. Laboratory assessments comprised a CBC, hepatic and renal function tests, and coagulation profiles. Cases were directed to assess their pain using the 10 centimeter Visual Analogue Scale (VAS) (0: no pain to ten: highest conceivable suffering).
All Cases received premedication with 2 mg of midazolam and 150 mg of ranitidine orally the preceding night. Two hours before surgery, 500 ml lactated Ringer’s solutions were infused before induction of GA and were fully monitored by ECG, blood pressure, and pulse oximetry. Using the same ultrasound machine with a 13-16 MHz linear ultrasonic array probe (Fujifilm Sonosite M-Turbo Ultrasound equipment), the blocks were conducted 30 min before surgery under full aseptic precautions using a 22-gauge (50 mm) echogenic needle.
2.1. TPVB Technique
The procedure was conducted laterally at the T3 level with the individual in a seated position. The transverse process, which is 2.5 centimeters laterally to the T3 spinous process, was reached after 2% lidocaine was injected into the skin. The transverse process and parietal pleura were located by positioning the ultrasonic probe 5 cm from the midline and moving it medially in craniocaudal direction. Throughout the transverse processes, the superior costotransverse ligament was seen as a series of uniform echogenic bands interrupted by areas of low echo intensity. 25 mL of 0.25 percent bupivacaine was injected into the area between pleura as well as the costotransverse ligament.
2.2. ESPB Technique
The operation site was elevated, and the patients were positioned in a lateral decubitus position. The linear probe was positioned transversely on the spinous process of the T5 vertebra, and its ultrasonographic picture was recognized. The probe was subsequently repositioned laterally to examine the lamina. Upon seeing and centering the T5 TP, the probe was turned 90° clockwise. The subcutaneous tissue was identified above three muscular layers: the trapezius muscle superiorly, the rhomboid major muscle medially, and the ESM inferiorly. The needle was placed in-plane, cephalic to caudal, until the tip came into touch with the T5 TP under the ESM. At the side of operation bupivacaine 0. 25% was injected in 25 ml total.
After the block was performed, the patients were monitored for a period of thirty min.
The sensory level of the block was evaluated by an observer who was blindfolded, using a pin-prick sensation throughout each dermatomal distribution from T1 to T8 at regular intervals of five min. It was determined that a block failure had occurred if the pin-prick sensation did not alleviate in any of the segments investigated.
2.3. General anesthesia
Initiated GA with 1 µg/kg fentanyl IV, followed by 1.5-2 mg/kg propofol. Atracurium 0.5 mg/kg helped tracheal intubation. Maintaining GA was done with sevoflurane (2%) and 50% oxygen. The patient’s lungs were ventilated with positive pressure to maintain 4.0–4.5 kPa end-tidal CO2. During surgery, ECG, noninvasive blood pressure, end-tidal CO2, and SpO2 were measured. HR and MAP were monitored before induction, throughout tracheal intubation, during skin incision, and every 15 min till operation. Throughout surgery, all patients received 5-8 mL/kg/hr of normal saline. If the MAP exceeded 20% of baseline for 2 consecutive measurements, a 0.1 µg/kg bolus was given. Atropine and neostigmine reversed neuromuscular blockage. Patient consciousness allowed trachea extubation.
All cases received intraoperative analgesia in the form of ketorolac 30 mg, and paracetamol IV infusion 1 gm. The patients were monitored for intraoperative fentanyl consumption.
Hypotension (MAP < 80% of baseline) was managed with normal saline boluses and, if necessary, 3 mg of ephedrine (IV). Bradycardia (heart rate < 50 beats per min) was managed with atropine 0.4 mg. Each individual was administered antiemetic prophylaxis with ondansetron 0.1 mg/kg prior to the conclusion of surgery.
2.4. Postoperative Care
In the post-anesthesia care unit (PACU), patients’ vital signs and pain levels were closely monitored after they were transported from the operating room. Ketorolac 30 mg/8 hours was administered as analgesia. If VAS was 4 or more, morphine 3 mg was given as rescue analgesia. Adverse effects were monitored including postoperative nausea and vomiting (PONV), local anesthetic (LA) toxicity, procedure-induced pneumothorax,hematoma in the puncture sites, as well as respiratory depression.
The duration of analgesia was quantified as the time from extubation to the initial rescue morphine analgesic dose Degree of patient satisfaction was assessed on a 4-point Likert scale (1 - not satisfied; 2 - partially satisfied; 3 - satisfied; 4 - very satisfied).8
The primary outcome was the duration of analgesia. The secondary outcomes were the intraoperative fentanyl consumption, total morphine consumption in 24 hours after surgery, postoperative pain scores, and patient satisfaction.
2.5. Statistical analysis
Data was analyzed using SPSS v26, which was developed by IBM Inc. Chicago, IL, USA. Histograms and the Shapiro-Wilk test were employed to determine if the data had a normal distribution. Mean and standard deviation (SD) were the quantitative parametric variables used to compare the two groups using Student’s t-test. For quantitative non-parametric data, we determined the median and interquartile range (IQR) using the
Mann-Whitney U test.... For each qualitative variable, we collected the frequency and percentage (%) and, based on their relevance, used either Fisher’s exact test or a Chi-square test to assess the data. Any two-tailed P value below 0.05 was deemed significant.
Nine individuals did not meet the eligibility criteria, and four people declined to participate. A total of fifty-seven individuals were assessed for eligibility. The others were randomly assigned to two comparable groups of twenty-two individuals each. Statistical analysis was conducted on all assigned individuals (Figure 1).
Figure 1: CONSORT flowchart of the enrolled individuals
The two groups were comparable in terms of age, weight, duration of surgery, ASA physical status, as well as side of surgery (Table 1).
The analgesic duration was comparable between the two groups (P = 0.122). No significant variance in intraoperative fentanyl consumption was seen between the two groups (P = 0.818). No significant distinction was seen in the number of cases requiring rescue morphine between the two groups (P = 0.540). Furthermore, there was no significant distinction in total morphine consumption between the two groups (P = 0.829) (Table 2).
There was no significant distinction in the VAS scores between the two groups at rest and with movement throughout the postoperative period up to 24 hours (Table 3).
The intraoperative HR was comparable between the two groups from baseline to one hour. The HR of the ESPB group was significantly greater than that of the TPVB group from between two and four hours (Figure 2, A).
The intraoperative MAP was comparable between the two groups at baseline and for a period of up to 2 hours. The ESPB group exhibited a significantly decreased MAP than the TPVB group after 4 hours (Figure 2, B).
Postoperative heart rate was significantly higher after 1 hour in the ESPB group than the TPVB group (P = 0.025). After that, there was no significant variance between groups in HR from two to twenty-four hours (Figure 2, C). Postoperative MAP was significantly higher in the ESPB group after 1, 2, and 4 hours than in the TPVB group. Then the MAP was comparable between the two groups from 6 to 24 hours (Figure 2, D).
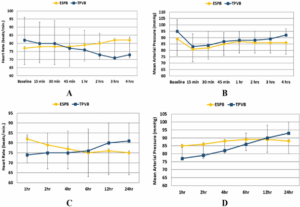
Figure 2: A) Intraoperative HR; B) MAP of the two groups; C) Postoperative HR and D) MAP of the two groups
Patient satisfaction was significantly higher in the ESPB group in contrast to the TPVB group (P < 0.001) (Table 4).
No complications were reported for patients including LA allergy or toxicity, or infection at the site of injection.
The current prospective randomized trial demonstrated that ESPB was equally effective as TPVB in the duration of analgesia and in postoperative pain and morphine usage during the first postoperative day after MRM. Both techniques were associated with hemodynamic stability and minor adverse effects. However, patients’ satisfaction was significantly higher with ESPB.
ESPB was proved effective as an analgesic modality after breast and thoracic surgery in contrast to non-block control groups in randomized trials and meta-analyses.10,11
The present study’s equivalent analgesic effect on PVB is in line with previous research that suggested ESPB as an alternative analgesic approach to PVB in breast surgery. In individuals who were undergoing breast surgery, a retrospective propensity-matched research was performed to compare ESPB and TPVB. The postoperative fentanyl consumption as well as pain scores following ESPB were not inferior to those following TPVB, as reported by the authors.12
Previous prospective trials showed similar opioid-sparing effects between the two techniques in women undergoing MRM.12 A recent meta-analysis confirmed the comparable analgesic profile of ESPB in addition to PVB in breast surgery. The study included 4 RCTs comprising 310 patients. Pooled analysis revealed no significant variances between the two techniques in perioperative pain intensity, analgesic consumption, duration of analgesia, hemodynamic response, and adverse effects. Nevertheless, procedure time was significantly shorter, and block success was more frequent in ESPB.13 On the contrary, a recent prospective RCT reported comparable postoperative VAS scores with ESPB and TPVB, but the 24-hour morphine consumption was significantly higher in the ESPB group.14
The PVB has been the gold standard in oncological breast surgery for a long time and continues to be the preferred method for massive procedures.15 However, finding an alternative to this technique was a field of extensive research. Prior studies have established that PVB is a sophisticated regional anesthetic procedure necessitating specialized expertise to maneuver the needle under ultrasound guidance into paravertebral space. El-Boghdadly16 stated that the TPVB is a highly demanding method that necessitates extensive training, making it impracticable in the now-saturated ultrasound training workshops. According to the example evaluation of the thoracic paravertebral area by Krediet et al.17 there are nine different procedures available for the TPVB, and each one is technically challenging in its way. The practitioner may encounter difficulty in locating an aperture to guide the needle between the transverse processes as well as the thoracic paravertebral region if they are employing a parasagittal in-plane view. In addition, even with ultrasonography, TPVB consequences such as epidural or intrathecal dispersion and accidental pleural puncture are always a risk.18
The comparable analgesic efficacy of ESPB to PVB is reinforced by its ease of execution, devoid of significant technical challenges compared to TPVB, which necessitates less expertise. Some authors regard ESPB as a technical modification of PVB,19 which may elucidate the analogous analgesic effects of both procedures. Local anesthetics were initially postulated to exert their effects by extending anteriorly into the paravertebral space, where they could affect the ventral rami and spinal nerve roots.20 Nonetheless, the precise mechanism of action of ESPB remains unclear due to the complications associated with the multidirectional dispersion of the injectate.21
The anatomical dispersion of local anesthetics following ESP injection is predominantly cephalocaudal from the injection site, according to Forero et al.20 This dissemination could occur from T1 to T11 as a result of an injection at the T5 level. The ventral rami and sympathetic nerves may be involved in ESPB, which may result in analgesia for visceral pain as well as certain sympathetically mediated symptoms, in addition to potential motor inhibition, as indicated by an increasing body of clinical data.22
The most likely significant action is the block of neuronal targets. The probable neurological targets encompass neurons situated inside the erector spinae plane (ESP), the erector spinae muscle, and neighboring compartments, including spinal nerve roots, ventral rami, and the brachial plexus.21 A negligible proportion of the delivered local anesthetic attains the paravertebral and epidural regions. Nevertheless, the selective but substantial effects on nociceptive transmission and processing are a result of the low concentration surrounding neuronal targets in these compartments.23
In a cadaveric model, Ivanusic et al.24 indicated that the ventral rami of the intercostal neurons were not stained by the dye that was injected deep into the ESP.24 The injectate’s substantial cephalad and lateral dispersion to the lateral cutaneous branches of the intercostal nerves may be related to an additional mechanism of breast analgesia, which they proposed.
The two techniques also provided a comparable analgesic effect in other types of surgery. Zhao et al. conducted a non-inferiority RCT that demonstrated no distinction in the pain score and morphine consumption at 24 hours between an ESPB as well as PVB following video-assisted thoracic surgery.25 Another similar study reported similar efficacy of continuous ESPB to continuous PVB regarding pain intensity and opioid consumption in the first 48 hours.26 Fang et al. confirmed the comparable analgesic profile of the two techniques in patients with open thoracotomy and reported fewer complications in the ESPB group.27 A retrospective study evaluated the analgesic impact of ESPB after video or robot-assisted thoracic surgery for lung cancer compared to PVB. ESPB was related to less postoperative pain, but comparable morphine consumption and respiratory complications at 24 hours.28
In order to assess the analgesic efficacy and safety of ESPB versus PVB for pain management following thoracic surgery, a meta-analysis of RCTs was used. The investigation comprised 10 studies that enrolled a total of 624 individuals. Pain scores and opioid consumption did not exhibit any significant variations.29
The results of the current trial together with the findings of previous trials and meta-analyses encourage using ESPB as an alternative to PVB in breast & thoracic surgery. The comparable efficacy and hemodynamic safety support the advantage of ESPB as an easier technique, which could balance the estimated 4%-10% PVB failure rate [12]. Moreover, ESPB appears to be a good substitute when PVB is contraindicated or risky in such situations as coagulation problems, high BMI, and patients receiving vitamin K antagonists or anticoagulants.
This work has limitations. This study presented a single-center experience involving a limited patient cohort, grounded on local practice regimens, hence constraining the generalizability of our findings to other forms of breast surgery at different institutions. The follow-up was restricted to the initial 24 hours, precluding an assessment of the effect of ESPB on chronic pain at the 3-month mark, a subject of significant interest in breast surgery.
ESPB provided a similar duration of analgesia, pain scores, and total postoperative morphine consumption compared to PVB after MRM. Both techniques maintained stable hemodynamics during and after surgery. However, patient satisfaction was significantly higher with ESPB. Given its comparable efficacy, easier performance, and suitability for cases where PVB is contraindicated, ESPB is a safe and acceptable alternative to PVB for women undergoing MRM.
Authors affiliations:
- Mahmoud A. Kamel, Assistant Professor Anesthesia, Intensive Care and Pain Management, National Cancer Institute (NCI), Cairo University, Cairo, Egypt. Email: Mkamel_76@hotmail.com
- Eman Abdulfattah Mohammed Ismael, MD Anesthesia, Intensive Care and Pain Management, National Cancer Institute (NCI), Cairo University, Cairo, Egypt. Email: emanismael305@gmail.com
- Somia Mohammed Abdel Aziz El Sheikh, Assistant Professor Anesthesia, Intensive Care and Pain Management, National Cancer Institute (NCI), Cairo University, Cairo, Egypt. Email: somaya_elshaikh@outlook.com
- Mai M. Elrawas, Lecturer of Anesthesia, Intensive Care and Pain Management, National Cancer Institute (NCI), Cairo University, Cairo, Egypt. Email: mai.elrawas@nci.cu.edu.eg
- Ehab Hanafy Shaker Gendy, Assistant Professor Anesthesia, Intensive Care and Pain Management, National Cancer Institute (NCI), Cairo University, Cairo, Egypt. Email: ehab.gendy@nci.cu.edu.eg
ABSTRACT
Background & objectives: Mastectomy has been associated with moderate to severe postoperative pain, and radical mastectomy has always been cumbersome for the parients due to it being difficult to manage. During the recent past regional anesthesia techniques have gained popularity to be used alone or as a part of balanced anesthesia. We evaluated the safety and efficacy of ultrasound-guided erector spinae plane block (ESPB) in contrast to thoracic paravertebral block (TPVB) for postoperative analgesia following modified radical mastectomy (MRM).
Methodology: In this prospective, randomized, single-blind research, 44 women, scheduled for MRM, were enrolled. Patients were randomly assigned into two groups: Group TPVB recieved TPVB, and the second group received ESPB at the T5 level. Both blocks were performed prior to the induction of general anesthesia (GA) using 25 mL of 0.25 bupivacaine 0.25%, under aseptic conditions. Standard GA technique was used in both groups. Inj morphine IV was used as a rescue analgesic. Time to first analgesic, total analgesic dose and the number of patients requiring analgesia were noted.
Results: Both groups needed analgesia for a time that was comparable to one another. In the first twenty-four hours following the operation, between intraoperative fentanyl consumption, the number of cases requiring rescue morphine, total morphine consumption, pain VAS scores at rest, and movement did not show significant differences between the two groups. The hemodynamic variables remained comparable in both of the groups. Compared to TPVB group, the level of patient satisfaction was significantly higher with the ESPB group.
Conclusion: ESPB is an acceptable analgesic alternative to paravertebral block after MRM. ESPB is equally effective as TPVB in the duration of analgesia and postoperative pain and morphine usage during the first postoperative day.
Abbreviations: ESPB: Erector Spinae Plane Block, MRM: modified radical mastectomy, NCI: National Cancer Institute, TPVB: Thoracic Paravertebral Block, VAS: Visual Analogue Scale
Keywords: Analgesia; Erector Spinae Plane Block; Modified Radical Mastectomy; Paravertebral Block; Pain; Postoperative Pain; Ultrasound
Citation: Kamel MA, Ismael EAM, El Sheikh SMAZ, Elrawas MM, Gendy EHS. Ultrasound guided erector spinae plane block versus thoracic paravertebral block for perioperative analgesia for modified radical mastectomy: a randomized trial. Anaesth. pain intensive care 2025;29(4):292-300. DOI: 10.35975/apic.v29i4.2814
Received: January 27, 2025; Revised: May 01, 2025; Accepted: May 21, 2025
1. INTRODUCTION
Around twelve percent of the world’s cancer burden is attributable to breast cancer (BC), making it the most frequent malignancy. Surgery is the predominant treatment modality, ranging from breast-conserving therapy to mastectomy with axillary clearance.1 Postoperative pain is a significant issue in breast cancer surgery, with over half of patients experiencing moderate to severe acute pain and 28% later developing chronic pain.2 Pain may result in delayed wound healing, hemodynamic complications, increased stress, respiratory depression, as well as a reduced quality of life by compromising psychological, physical, and social functioning if pain left untreated. The pain is exacerbated by the extensive tissue destruction that occurs throughout modified radical mastectomy (MRM)3 The MRM procedure involves the complete removal of the breast as well as the underlying fascia of the pectoralis major muscle, as well as the excision of level I and II axillary lymph nodes.4 Consequently, it is imperative to give precedence to the avoidance of postoperative pain in these patients.
Diverse analgesic techniques are employed to mitigate postoperative pain in BC surgery. Currently, opioids are still in use for pain management after BC surgery despite their potential adverse effects, including respiratory depression, drowsiness, constipation, nausea, and vomiting. Various regional methods, including paravertebral blocks and thoracic epidural anesthesia, offer superior pain management, enhance pulmonary function, and diminish perioperative opioid use.5
The thoracic paravertebral block (TPVB)6 has historically been the anesthesiologist’s preferred method for alleviating pain throughout and after MRM.7 However, TPVB is linked to many drawbacks, including pneumothorax, hypotension, central spread of local anesthetic, sympathetic block, or failure block, which may impose restrictions on the method. The utilization of fascial plane blocks in oncological breast surgery has been supported by evidence-based medicine in recent years. Erector spinae plane block (ESPB), serratus anterior plane blocks, and pectoral nerve blocks can enhance pain management, improve pulmonary function, and reduce perioperative opioid consumption.5
The objective of this investigation was to assess the safety and efficacy of ultrasound-guided ESPB contrasted with TPVB for postoperative analgesia following MRM.
2. METHODOLOGY
ESPB and PVB groups were compared in a 1:1 ratio to investigate the duration of analgesia. In a previous study,9 with a pooled standard deviation of 6.8 hours, there was a variation of 1.7 hours in the duration of analgesia. We needed to enroll 22 patients in each group to reject the null hypothesis that the two groups’ population means are identical with a power of 0.8, according to these results. The probability of a Type I error in this test of the null hypothesis is 0.05.
The NCI’s Institutional Research Board approved this research (Code: AP1811-30104). Before enrolling in the trial, patients or their guardians gave written informed consent.
Forty- four women with breast cancer, scheduled for MRM, were randomly selected at the National Cancer Institute, Cairo University, for this prospective randomized, single-blind research between June 2021 and June 2023. Female people between the ages of 18 and 65 years. who had a history of breast cancer, undergoing elective unilateral mastectomy with general anesthesia (GA), and had a current American Society of Anesthesiologists physical status of I or II were included.
Exclusion criteria were coagulopathy, severe obesity (BMI > 40 kg/m2), history of narcotic pain medication use, major cardiac disorder, renal dysfunction, allergies to local anesthetics, decreased pulmonary reserve, psychiatric illness, preexisting neurological deficits, or infection at the block site.
Parallel computer-generated random numbers were utilized to divide patients into two equal groups, and their allocation codes were sealed in opaque envelopes. The TPVB was administered to the individuals in the TPVB Group (n = 22) by injecting 25 milliliters of 0.25 percent bupivacaine into the space between the pleura and the costotransverse ligament. The 22 patients who made up the ESPB Group were each given a 25-milliliter injection of 0.25 percent bupivacaine in a cephalic-to-caudal direction until the needle reached the T5 transverse process, which is located below the erector-spinae muscle.
The research was single-blinded, as the outcome assessors were blinded to group allocation. An anesthesiologist with experience in ultrasound-guided blocks, who did not take part in data collection or analysis, conducted all blocks prior to induction of GA.
All patients underwent meticulous history-taking and clinical evaluation. Laboratory assessments comprised a CBC, hepatic and renal function tests, and coagulation profiles. Cases were directed to assess their pain using the 10 centimeter Visual Analogue Scale (VAS) (0: no pain to ten: highest conceivable suffering).
All Cases received premedication with 2 mg of midazolam and 150 mg of ranitidine orally the preceding night. Two hours before surgery, 500 ml lactated Ringer’s solutions were infused before induction of GA and were fully monitored by ECG, blood pressure, and pulse oximetry. Using the same ultrasound machine with a 13-16 MHz linear ultrasonic array probe (Fujifilm Sonosite M-Turbo Ultrasound equipment), the blocks were conducted 30 min before surgery under full aseptic precautions using a 22-gauge (50 mm) echogenic needle.
2.1. TPVB Technique
The procedure was conducted laterally at the T3 level with the individual in a seated position. The transverse process, which is 2.5 centimeters laterally to the T3 spinous process, was reached after 2% lidocaine was injected into the skin. The transverse process and parietal pleura were located by positioning the ultrasonic probe 5 cm from the midline and moving it medially in craniocaudal direction. Throughout the transverse processes, the superior costotransverse ligament was seen as a series of uniform echogenic bands interrupted by areas of low echo intensity. 25 mL of 0.25 percent bupivacaine was injected into the area between pleura as well as the costotransverse ligament.
2.2. ESPB Technique
The operation site was elevated, and the patients were positioned in a lateral decubitus position. The linear probe was positioned transversely on the spinous process of the T5 vertebra, and its ultrasonographic picture was recognized. The probe was subsequently repositioned laterally to examine the lamina. Upon seeing and centering the T5 TP, the probe was turned 90° clockwise. The subcutaneous tissue was identified above three muscular layers: the trapezius muscle superiorly, the rhomboid major muscle medially, and the ESM inferiorly. The needle was placed in-plane, cephalic to caudal, until the tip came into touch with the T5 TP under the ESM. At the side of operation bupivacaine 0. 25% was injected in 25 ml total.
After the block was performed, the patients were monitored for a period of thirty min.
The sensory level of the block was evaluated by an observer who was blindfolded, using a pin-prick sensation throughout each dermatomal distribution from T1 to T8 at regular intervals of five min. It was determined that a block failure had occurred if the pin-prick sensation did not alleviate in any of the segments investigated.
2.3. General anesthesia
Initiated GA with 1 µg/kg fentanyl IV, followed by 1.5-2 mg/kg propofol. Atracurium 0.5 mg/kg helped tracheal intubation. Maintaining GA was done with sevoflurane (2%) and 50% oxygen. The patient’s lungs were ventilated with positive pressure to maintain 4.0–4.5 kPa end-tidal CO2. During surgery, ECG, noninvasive blood pressure, end-tidal CO2, and SpO2 were measured. HR and MAP were monitored before induction, throughout tracheal intubation, during skin incision, and every 15 min till operation. Throughout surgery, all patients received 5-8 mL/kg/hr of normal saline. If the MAP exceeded 20% of baseline for 2 consecutive measurements, a 0.1 µg/kg bolus was given. Atropine and neostigmine reversed neuromuscular blockage. Patient consciousness allowed trachea extubation.
All cases received intraoperative analgesia in the form of ketorolac 30 mg, and paracetamol IV infusion 1 gm. The patients were monitored for intraoperative fentanyl consumption.
Hypotension (MAP < 80% of baseline) was managed with normal saline boluses and, if necessary, 3 mg of ephedrine (IV). Bradycardia (heart rate < 50 beats per min) was managed with atropine 0.4 mg. Each individual was administered antiemetic prophylaxis with ondansetron 0.1 mg/kg prior to the conclusion of surgery.
2.4. Postoperative Care
In the post-anesthesia care unit (PACU), patients’ vital signs and pain levels were closely monitored after they were transported from the operating room. Ketorolac 30 mg/8 hours was administered as analgesia. If VAS was 4 or more, morphine 3 mg was given as rescue analgesia. Adverse effects were monitored including postoperative nausea and vomiting (PONV), local anesthetic (LA) toxicity, procedure-induced pneumothorax,hematoma in the puncture sites, as well as respiratory depression.
The duration of analgesia was quantified as the time from extubation to the initial rescue morphine analgesic dose Degree of patient satisfaction was assessed on a 4-point Likert scale (1 - not satisfied; 2 - partially satisfied; 3 - satisfied; 4 - very satisfied).8
The primary outcome was the duration of analgesia. The secondary outcomes were the intraoperative fentanyl consumption, total morphine consumption in 24 hours after surgery, postoperative pain scores, and patient satisfaction.
2.5. Statistical analysis
Data was analyzed using SPSS v26, which was developed by IBM Inc. Chicago, IL, USA. Histograms and the Shapiro-Wilk test were employed to determine if the data had a normal distribution. Mean and standard deviation (SD) were the quantitative parametric variables used to compare the two groups using Student’s t-test. For quantitative non-parametric data, we determined the median and interquartile range (IQR) using the
Mann-Whitney U test.... For each qualitative variable, we collected the frequency and percentage (%) and, based on their relevance, used either Fisher’s exact test or a Chi-square test to assess the data. Any two-tailed P value below 0.05 was deemed significant.
3. RESULTS
Nine individuals did not meet the eligibility criteria, and four people declined to participate. A total of fifty-seven individuals were assessed for eligibility. The others were randomly assigned to two comparable groups of twenty-two individuals each. Statistical analysis was conducted on all assigned individuals (Figure 1).

Figure 1: CONSORT flowchart of the enrolled individuals
The two groups were comparable in terms of age, weight, duration of surgery, ASA physical status, as well as side of surgery (Table 1).
| Table 1: Demographic data, duration, and side of surgery of the two groups | ||||
| Variables | ESPB group (n = 22) |
TPVB group
(n = 22) |
p-value | |
| Age (years) | 57.4 ± 9.0 | 60.7 ± 8.3 | 0.204 | |
| Weight (kg) | 76.9 ± 10.3 | 70.8 ± 11.4 | 0.070 | |
| ASA physical status (I/II) | 9/13 (40.9) | 10/12 (45.5) | 0.761 | |
| Duration of surgery (min) | 213 ± 31 | 216 ± 29 | 0.641 | |
| Side of surgery | Right | 11 (50) | 10 (45.5) | 0.763 |
| Left | 11 (50) | 12 (54.5) | ||
| Data presented as mean ± SD or frequency (%). ASA: American Society of Anesthesiologists.
TPVB: thoracic paravertebral block. ESPB: erector spinae plane block. |
||||
The analgesic duration was comparable between the two groups (P = 0.122). No significant variance in intraoperative fentanyl consumption was seen between the two groups (P = 0.818). No significant distinction was seen in the number of cases requiring rescue morphine between the two groups (P = 0.540). Furthermore, there was no significant distinction in total morphine consumption between the two groups (P = 0.829) (Table 2).
| Table 2: Analgesic outcomes of the two groups | |||
| Analgesic status | ESPB group (n = 22) |
TPVB group
(n = 22) |
p-value |
| Duration of analgesia (hours) | 6 (4-12) | 6 (6-12) | 0.122 |
| Intraoperative fentanyl consumption (µg) | 99.3 ± 13.2 | 98.4 ± 12.8 | 0.818 |
| No. of patients needing rescue analgesia | 10 (45.5) | 8 (36.4) | 0.540 |
| Morphine consumption in 1st 24 h
postoperative (mg) |
6.0 (6.0-9.0) | 6.0 (3.0-9.0) | 0.829 |
| Data presented as mean ± standard deviation, median (IQR) or frequency (%).
TPVB: thoracic paravertebral block. ESPB: erector spinae plane block. |
|||
There was no significant distinction in the VAS scores between the two groups at rest and with movement throughout the postoperative period up to 24 hours (Table 3).
| Table 3: The postoperative visual analog scale scores at rest and with
movements in the two groups |
||||
| VAS Scores | ESPB group (n = 22) |
TPVB group
(n = 22) |
p-value | |
| At rest | After 1 hr | 0 (0-3) | 0 (0-2) | 0.402 |
| After 2 hrs | 2 (0-3) | 1 (0-3) | 0.098 | |
| After 4 hrs | 2 (0-5) | 2 (1-3) | 0.457 | |
| After 6 hrs | 3 (1-5) | 3 (1-5) | 0.921 | |
| After 12 hrs | 3 (0-5) | 3 (2-4) | 0.961 | |
| After 24 hrs | 3 (0-4) | 3 (2-4) | 0.343 | |
| With movement | After 1 hr | 1 (1-3) | 1 (1-3) | 0.421 |
| After 2 hrs | 3 (1-3) | 2 (1-3) | 0.062 | |
| After 4 hrs | 3 (1-6) | 3 (2-3) | 0.566 | |
| After 6 hrs | 3 (2-5) | 3 (2-6) | 0.500 | |
| After 12 hrs | 3 (1-7) | 3 (2-5) | 0.874 | |
| After 24 hrs | 3 (1-5) | 3 (2-5) | 0.082 | |
| Data presented as median (IQR). TPVB: thoracic paravertebral block.
ESPB: erector spinae plane block. |
||||
The intraoperative HR was comparable between the two groups from baseline to one hour. The HR of the ESPB group was significantly greater than that of the TPVB group from between two and four hours (Figure 2, A).
The intraoperative MAP was comparable between the two groups at baseline and for a period of up to 2 hours. The ESPB group exhibited a significantly decreased MAP than the TPVB group after 4 hours (Figure 2, B).
Postoperative heart rate was significantly higher after 1 hour in the ESPB group than the TPVB group (P = 0.025). After that, there was no significant variance between groups in HR from two to twenty-four hours (Figure 2, C). Postoperative MAP was significantly higher in the ESPB group after 1, 2, and 4 hours than in the TPVB group. Then the MAP was comparable between the two groups from 6 to 24 hours (Figure 2, D).

Figure 2: A) Intraoperative HR; B) MAP of the two groups; C) Postoperative HR and D) MAP of the two groups
Patient satisfaction was significantly higher in the ESPB group in contrast to the TPVB group (P < 0.001) (Table 4).
No complications were reported for patients including LA allergy or toxicity, or infection at the site of injection.
| Table 4: Patient satisfaction of the two groups. | |||
| Satisfaction level | ESPB group (n = 22) |
TPVB group (n = 22) |
p-value |
| Very satisfied | 12 (54.5) | 4 (18.2) | < 0.001 |
| Satisfied | 8 (36.4) | 7 (31.8) | |
| Partially satisfied | 1 (4.5) | 11 (50.0) | |
| Not satisfied | 1 (4.5) | 0 (0.0) | |
| Data presented as frequency (%). TPVB: thoracic paravertebral block.
ESPB: erector spinae plane block. |
|||
4. DISCUSSION
The current prospective randomized trial demonstrated that ESPB was equally effective as TPVB in the duration of analgesia and in postoperative pain and morphine usage during the first postoperative day after MRM. Both techniques were associated with hemodynamic stability and minor adverse effects. However, patients’ satisfaction was significantly higher with ESPB.
ESPB was proved effective as an analgesic modality after breast and thoracic surgery in contrast to non-block control groups in randomized trials and meta-analyses.10,11
The present study’s equivalent analgesic effect on PVB is in line with previous research that suggested ESPB as an alternative analgesic approach to PVB in breast surgery. In individuals who were undergoing breast surgery, a retrospective propensity-matched research was performed to compare ESPB and TPVB. The postoperative fentanyl consumption as well as pain scores following ESPB were not inferior to those following TPVB, as reported by the authors.12
Previous prospective trials showed similar opioid-sparing effects between the two techniques in women undergoing MRM.12 A recent meta-analysis confirmed the comparable analgesic profile of ESPB in addition to PVB in breast surgery. The study included 4 RCTs comprising 310 patients. Pooled analysis revealed no significant variances between the two techniques in perioperative pain intensity, analgesic consumption, duration of analgesia, hemodynamic response, and adverse effects. Nevertheless, procedure time was significantly shorter, and block success was more frequent in ESPB.13 On the contrary, a recent prospective RCT reported comparable postoperative VAS scores with ESPB and TPVB, but the 24-hour morphine consumption was significantly higher in the ESPB group.14
The PVB has been the gold standard in oncological breast surgery for a long time and continues to be the preferred method for massive procedures.15 However, finding an alternative to this technique was a field of extensive research. Prior studies have established that PVB is a sophisticated regional anesthetic procedure necessitating specialized expertise to maneuver the needle under ultrasound guidance into paravertebral space. El-Boghdadly16 stated that the TPVB is a highly demanding method that necessitates extensive training, making it impracticable in the now-saturated ultrasound training workshops. According to the example evaluation of the thoracic paravertebral area by Krediet et al.17 there are nine different procedures available for the TPVB, and each one is technically challenging in its way. The practitioner may encounter difficulty in locating an aperture to guide the needle between the transverse processes as well as the thoracic paravertebral region if they are employing a parasagittal in-plane view. In addition, even with ultrasonography, TPVB consequences such as epidural or intrathecal dispersion and accidental pleural puncture are always a risk.18
The comparable analgesic efficacy of ESPB to PVB is reinforced by its ease of execution, devoid of significant technical challenges compared to TPVB, which necessitates less expertise. Some authors regard ESPB as a technical modification of PVB,19 which may elucidate the analogous analgesic effects of both procedures. Local anesthetics were initially postulated to exert their effects by extending anteriorly into the paravertebral space, where they could affect the ventral rami and spinal nerve roots.20 Nonetheless, the precise mechanism of action of ESPB remains unclear due to the complications associated with the multidirectional dispersion of the injectate.21
The anatomical dispersion of local anesthetics following ESP injection is predominantly cephalocaudal from the injection site, according to Forero et al.20 This dissemination could occur from T1 to T11 as a result of an injection at the T5 level. The ventral rami and sympathetic nerves may be involved in ESPB, which may result in analgesia for visceral pain as well as certain sympathetically mediated symptoms, in addition to potential motor inhibition, as indicated by an increasing body of clinical data.22
The most likely significant action is the block of neuronal targets. The probable neurological targets encompass neurons situated inside the erector spinae plane (ESP), the erector spinae muscle, and neighboring compartments, including spinal nerve roots, ventral rami, and the brachial plexus.21 A negligible proportion of the delivered local anesthetic attains the paravertebral and epidural regions. Nevertheless, the selective but substantial effects on nociceptive transmission and processing are a result of the low concentration surrounding neuronal targets in these compartments.23
In a cadaveric model, Ivanusic et al.24 indicated that the ventral rami of the intercostal neurons were not stained by the dye that was injected deep into the ESP.24 The injectate’s substantial cephalad and lateral dispersion to the lateral cutaneous branches of the intercostal nerves may be related to an additional mechanism of breast analgesia, which they proposed.
The two techniques also provided a comparable analgesic effect in other types of surgery. Zhao et al. conducted a non-inferiority RCT that demonstrated no distinction in the pain score and morphine consumption at 24 hours between an ESPB as well as PVB following video-assisted thoracic surgery.25 Another similar study reported similar efficacy of continuous ESPB to continuous PVB regarding pain intensity and opioid consumption in the first 48 hours.26 Fang et al. confirmed the comparable analgesic profile of the two techniques in patients with open thoracotomy and reported fewer complications in the ESPB group.27 A retrospective study evaluated the analgesic impact of ESPB after video or robot-assisted thoracic surgery for lung cancer compared to PVB. ESPB was related to less postoperative pain, but comparable morphine consumption and respiratory complications at 24 hours.28
In order to assess the analgesic efficacy and safety of ESPB versus PVB for pain management following thoracic surgery, a meta-analysis of RCTs was used. The investigation comprised 10 studies that enrolled a total of 624 individuals. Pain scores and opioid consumption did not exhibit any significant variations.29
The results of the current trial together with the findings of previous trials and meta-analyses encourage using ESPB as an alternative to PVB in breast & thoracic surgery. The comparable efficacy and hemodynamic safety support the advantage of ESPB as an easier technique, which could balance the estimated 4%-10% PVB failure rate [12]. Moreover, ESPB appears to be a good substitute when PVB is contraindicated or risky in such situations as coagulation problems, high BMI, and patients receiving vitamin K antagonists or anticoagulants.
5. LIMITATIONS
This work has limitations. This study presented a single-center experience involving a limited patient cohort, grounded on local practice regimens, hence constraining the generalizability of our findings to other forms of breast surgery at different institutions. The follow-up was restricted to the initial 24 hours, precluding an assessment of the effect of ESPB on chronic pain at the 3-month mark, a subject of significant interest in breast surgery.
6. CONCLUSION
ESPB provided a similar duration of analgesia, pain scores, and total postoperative morphine consumption compared to PVB after MRM. Both techniques maintained stable hemodynamics during and after surgery. However, patient satisfaction was significantly higher with ESPB. Given its comparable efficacy, easier performance, and suitability for cases where PVB is contraindicated, ESPB is a safe and acceptable alternative to PVB for women undergoing MRM.
7. REFERENCES
- Bray F, Laversanne M, Sung H, Ferlay J, Soerjomataram I, Jemal A, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-63. [PubMed] DOI: 3322/caac.21834
- Villa G, Mandarano R, Scirè-Calabrisotto C, De Rosa S, Martucci G, Antonelli M, et al. Chronic pain after breast surgery: incidence, associated factors, and impact on quality of life, an observational prospective study. Perioper Med. 2021;10:6. [PubMed] DOI: 1186/s13741-021-00176-6
- Li T, Zhang P, Zhang Y, Liu X, Wang H, Zhao Y, et al. Efficacy of breast-conserving surgery versus modified radical surgery in the treatment of early breast cancer: a meta-analysis. Altern Ther Health Med. 2025;31:542-875. [PubMed]
- Manouras A, Markogiannakis H, Genetzakis M, Lagoudianakis E, Papadima A, Bramis K, et al. Modified radical mastectomy with axillary dissection using the electrothermal bipolar vessel sealing system. Arch Surg. 2008;143:575-80. [PubMed] DOI: 1001/archsurg.143.6.575
- Plunkett A, Scott TL, Tracy E. Regional anesthesia for breast cancer surgery: which block is best? A review of the current literature. Pain Manag. 2022;12:943-50. [PubMed] DOI: 2217/pmt-2022-0048
- Moharam SA, Elshikh A, Abdelbadie M, Hassan R, Shaban A, Ghoneim M, et al. Efficacy of adding ketamine to levobupivacaine in paravertebral block on acute and chronic pain in thoracotomy: a randomized controlled double-blinded trial. Pain Rep. 2024;9:1206-8. [PubMed] DOI: 1097/PR9.0000000000001206
- Amr SA, Othman AH, Ahmed EH, Samir M, Farouk A, Mahmoud A, et al. Comparison between ultrasound guided erector spinae plane block and paravertebral block on acute and chronic post mastectomy pain after modified radical mastectomy: randomized controlled trial. BMC Anesthesiol. 2024;24:420. [PubMed] DOI: 1186/s12871-024-02810-4
- Fujiwara T, Kondo M, Yamada H, Tanaka E, Matsumoto T, Saito K, et al. Factors affecting patient satisfaction related to cost and treatment effectiveness in rheumatoid arthritis: results from the multicenter observational cohort study, FRANK Registry. Arthritis Res Ther. 2022;24:53-8. [PubMed] DOI: 1186/s13075-022-02746-5
- Swisher MW, Wallace AM, Sztain JF, Said ET, Khatibi B, Gabriel RA, et al. Erector spinae plane versus paravertebral nerve blocks for postoperative analgesia after breast surgery: a randomized clinical trial. Reg Anesth Pain Med. 2020;45:260-6. [PubMed] DOI: 1136/rapm-2019-101013
- Singh S, Kumar G, Akhileshwar. Ultrasound-guided erector spinae plane block for postoperative analgesia in modified radical mastectomy: a randomised control study. Indian J Anaesth. 2019;63:200-4. [PubMed] DOI: 4103/ija.IJA_758_18
- Weng WT, Wang CJ, Li CY, Huang YF, Chen HH, Chang CL, et al. Erector spinae plane block similar to paravertebral block for perioperative pain control in breast surgery: a meta-analysis study. Pain Physician. 2021;24:203-13. [PubMed]
- Elewa AM, Faisal M, Sjöberg F, Nabil A, Osman H, Tawfik M, et al. Comparison between erector spinae plane block and paravertebral block regarding postoperative analgesic consumption following breast surgery: a randomized controlled study. BMC Anesthesiol. 2022;22:189. [PubMed] DOI: 1186/s12871-022-01724-3
- Chen W, Zhou X, Li H, Zhang L, Jiang Y, Sun Q, et al. Analgesic effectiveness of preoperative ultrasound-guided erector spinae plane block versus paravertebral nerve block for breast surgery: a systematic review and meta-analysis of four randomized controlled trials. Pain Pract. 2023;23:511-22. [PubMed] DOI: 1111/papr.13208
- Jayakrishnan S, Dua A, Kumar A. Comparison of fascial plane blocks (ESPB vs. TPVB) for pain relief following modified radical mastectomy. J Anaesthesiol Clin Pharmacol. 2024;40:410-5. [PubMed]
- Jacobs A, Lemoine A, Joshi GP, Van de Velde M, Bonnet F, Chelly JE, et al. PROSPECT guideline for oncological breast surgery: a systematic review and procedure-specific postoperative pain management recommendations. Anaesthesia. 2020;75:664-73. [PubMed] DOI: 1111/anae.14964
- El-Boghdadly K, Pawa A. The erector spinae plane block: plane and simple. Anaesthesia. 2017;72:434-8. [PubMed] DOI: 1111/anae.13830
- Krediet AC, Moayeri N, van Geffen GJ, Bruhn J, Renes SH, Gielen MJ, et al. Different approaches to ultrasound-guided thoracic paravertebral block: an illustrated review. Anesthesiology. 2015;123:459-74. [PubMed] DOI: 1097/ALN.0000000000000747
- Kus A, Gurkan Y, Gul Akgul A, Solak M, Toker K. Pleural puncture and intrathoracic catheter placement during ultrasound guided paravertebral block. J Cardiothorac Vasc Anesth. 2013;27:e11-2. [PubMed] DOI: 1053/j.jvca.2012.10.018
- Gadsden J. The erector spinae plane block: the case of the elusive mechanism of action. Can J Anaesth. 2021;68:288-92. [PubMed] DOI: 1007/s12630-020-01876-1
- Forero M, Adhikary SD, Lopez H, Tsui C, Chin KJ. The erector spinae plane block: a novel analgesic technique in thoracic neuropathic pain. Reg Anesth Pain Med. 2016;41:621-7. [PubMed] DOI: 1097/AAP.0000000000000451
- Chin KJ, El-Boghdadly K. Mechanisms of action of the erector spinae plane (ESP) block: a narrative review. Can J Anaesth. 2021;68:387-408. [PubMed] DOI: 1007/s12630-020-01875-2
- Hong JH, Park JH, Park KB, Choi S, Kim JH, Lee S, et al. Sympatholytic effect of the high thoracic erector spinae plane block. Pain Physician. 2024;27:43-9. [PubMed]
- Diwan S, Nair A. Is paravertebral-epidural spread the underlying mechanism of action of erector spinae plane block? Turk J Anaesthesiol Reanim. 2020;48:86-7. [PubMed] DOI: 5152/TJAR.2019.81226
- Ivanusic J, Konishi Y, Barrington MJ. A cadaveric study investigating the mechanism of action of erector spinae blockade. Reg Anesth Pain Med. 2018;43:567-71. [PubMed] DOI: 1097/AAP.0000000000000789
- Zhao H, Xin L, Feng Y. The effect of preoperative erector spinae plane vs. paravertebral blocks on patient-controlled oxycodone consumption after video-assisted thoracic surgery: a prospective randomized, blinded, non-inferiority study. J Clin Anesth. 2020;62:109737. [PubMed] DOI: 1016/j.jclinane.2020.109737
- Taketa Y, Irisawa Y, Fujitani T. Comparison of ultrasound-guided erector spinae plane block and thoracic paravertebral block for postoperative analgesia after video-assisted thoracic surgery: a randomized controlled non-inferiority clinical trial. Reg Anesth Pain Med. 2019:rapm-2019-100827. [PubMed] DOI: 1136/rapm-2019-100827
- Fang B, Wang Z, Huang X. Ultrasound-guided preoperative single-dose erector spinae plane block provides comparable analgesia to thoracic paravertebral block following thoracotomy: a single center randomized controlled double-blind study. Ann Transl Med. 2019;7:174. [PubMed] DOI: 21037/atm.2019.03.53
- Durey B, Djerada Z, Boujibar F, Girard C, Filaire M, Guenoun T, et al. Erector spinae plane block versus paravertebral block after thoracic surgery for lung cancer: a propensity score study. Cancers (Basel). 2023;15(8):2306. [PubMed] DOI: 3390/cancers15082306
- Capuano P, Hileman BA, Martucci G, Del Sorbo L, Gregoretti C, Navalesi P, et al. Erector spinae plane block versus paravertebral block for postoperative pain management in thoracic surgery: a systematic review and meta-analysis. Minerva Anestesiol. 2023;89:1042-50. [PubMed] DOI: 23736/S0375-9393.23.17510-9