Andy Nugroho, Dewi Yulianti Bisri 2, Iwan Abdul Rachman 3
Authors affiliations:
Subdural empyema (SDE) is a critical intracranial infection requiring urgent surgical intervention. Anesthetic management in infants with SDE involves challenges such as elevated intracranial pressure (ICP), seizures, and hemodynamic instability. Effective management, including careful monitoring and appropriate anesthesia technique, is essential to improve outcomes and reduce the complications.
A 3-months-old male infant, 6 kg, was admitted with recurrent fever and seizures, accompanied by a history of thrombocytopenia. Neuroimaging revealed extensive subdural empyema, prompting urgent craniotomy for empyema evacuation. General anesthesia was successfully administered, maintaining hemodynamic stability throughout the procedure. Patient’s clinical condition was significantly improved and extubated on the next day, then was transferred to the pediatric ward for continued care.
Preoperative management of an infant with subdural empyema and elevated ICP, focuses on reducing ICP, managing seizures, stabilizing hemodynamics and addressing infection. A Total Intravenous Anesthesia (TIVA) based approach, combined with fluid and ventilation management and neuroprotective agents ensure stable perioperative conditions and support recovery. Postoperatively, a multidisciplinary approach involving neurological monitoring, seizure prophylaxis with midazolam, and antimicrobial therapy was implemented to optimize recovery, control ICP, and prevent complications. This comprehensive management strategy aims to prevent secondary brain injury and promotes a stable recovery.
Abbreviations: CPP: Cerebral perfusion pressure, DBP: Diastolic blood pressure, EtCO2: End tidal CO2, ETT: endotracheal tube, HR: Heart Rate, ICP: intracranial pressure, pGCS: pediatric Glasgow Coma Scale, SBP: Systolic blood pressure, SDE: Subdural empyema, SpO2: Venous capillary oxygen saturation; Temp: Temperature, TIVA: Total Intravenous Anesthesia
Keyword: anesthesia; craniotomy; ICP; infant; subdural empyema; TIVA
Citation: Nugroho A, Bisri DY, Rachman IA. Navigating the anesthetic challenges for craniotomy in infant with bilateral subdural empyema: A case report. Anaesth. pain intensive care 2025;29(3):715-719.
DOI: 10.35975/apic.v29i3.2781
Received: March 10, 2025; Revised: March 22, 2025; Accepted: March 22, 2025
Subdural empyema (SDE) is a critical intracranial infection characterized by pus accumulation between the dura mater and the arachnoid membrane, requires urgent surgical intervention.1 Anesthetic management in infants with SDE poses significant challenges due to the occurred elevated intracranial pressure (ICP), ongoing seizures, and hemodynamic instability. Preoperative assessment is crucial to identify these risks and optimize the patient's condition. Adequate airway management is crucial, especially with neurological compromise. Intensive vital signs and neurological status monitoring are necessary throughout the procedure. Anesthesia induction should use minimally cardiovascular effects agents. Intraoperative management should focus on hemodynamic stability and ICP control, often requiring intravenous fluids and medications to support blood pressure. Postoperative monitoring in critical care is vital for early complication detection. Effective anesthetic management improves outcomes in infants with SDE, reduces long-term neurological deficits, and increase survival rates.
A 3-months-old male infant, 6 kgs, was admitted with recurrent fever and seizures. The infant had been healthy until three weeks prior, until prolonged fever developed alongside two generalized seizures. The most recent seizure lasted approximately three minutes and was characterized by generalized tonic posturing, blank staring, and peripheral cyanosis. Resolution of this seizure occurred following the administration of a 5 mg rectal diazepam. For three weeks prior to admission, the patient experienced recurrent episodes of fever and seizures.
Physical examination revealed clear airway, a respiratory rate of 36 breaths per minute, and oxygen saturation of 99% on 1 L/min nasal cannula, without signs of respiratory distress. Cardiovascular assessment showed a heart rate of 186 bpm, a strong and regular pulse, with normal heart sounds and no murmurs. The body temperature was 39.6°C, and he was alert with pediatric Glasgow Coma Scale (pGCS) of E4V5M6, although exhibiting fever and lethargy. Primitive reflexes were present and appropriate regarding his developmental age.
Neuroimaging (Figure 1) demonstrated extensive subdural empyema with mass effect, characterized by bilateral fronto-temporo-parietal hypodense fluid collections with rim contrast enhancement, leading to compression and displacement of the anterior horn of the left lateral ventricle, dilation of the temporal horns of both ventricles, and rightward subfalcine herniation (the most common type of brain herniation, occurring when brain tissue is pushed under the falx cerebri).
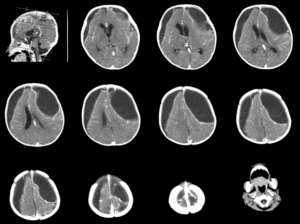
Figure 1: Brain CT Scan demonstrated extensive subdural empyema with mass effect, characterized by bilateral fronto-temporo-parietal hypodense fluid collections with rim contrast enhancement, leading to compression and displacement of the anterior horn of the left lateral ventricle, dilation of the temporal horns of both lateral ventricles, and rightward subfalcine herniation.
The patient also exhibited microcephaly, with persistent cranial suture patency and open anterior and posterior fontanelles. Chest radiography (Figure 2) revealed left-sided pneumonia, despite the absence of respiratory distress.
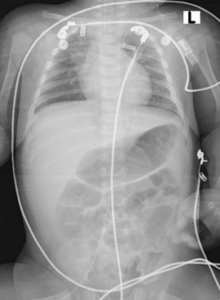
Figure 2: Chest radiography revealed left-sided pneumonia
Laboratory findings indicated a leukocytosis of 42,600/µL and thrombocytosis of 810,000/µL, suggesting an ongoing infectious and inflammatory process. Other significant parameters included hemoglobin 10.4 g/dL and hematocrit 29%. Coagulation and electrolytes studies were within normal limits (PT 15.1 sec, APTT 21.1 sec, INR 1.13, sodium 146 mmol/L, potassium 5.5 mmol/L and calcium 1.25 mmol/L). Hepatic and renal function tests were normal (SGOT 35 U/L, SGPT 20 U/L, creatinine 0.2 mg/dL, urea at 9 mg/dL). Hepatitis B surface antigen (HBsAg) was negative. Prompt treatment was initiated with ceftriaxone 300 mg q12h IV, metronidazole 50 mg q8h IV, dexamethasone 1 mg q6h IV, and paracetamol 90 mg q6h IV. Due to neurological decline, neuroimaging findings of subdural empyema with mass effect, and hematological complications, the patient was classified as ASA III E, necessitating urgent craniotomy for empyema evacuation.
General anesthesia was induced with fentanyl 10 µg IV, propofol 10 mg IV, and atracurium 5 mg IV. A cuffed endotracheal tube (ETT) size 3.5 was inserted to the depth of 11.5 cm. Hemodynamic parameters were maintained throughout the surgical procedure which lasted for three hours. Intraoperative fluid management included Inj Ringerfundin® at 25 mL/hour, and additional medications included ondansetron 0.8 mg IV for nausea prophylaxis and paracetamol 90 mg IV for analgesia. Anesthesia was maintained via TIVA with propofol at 50 µg/kg/min, while dobutamine was administered at 2 µg/kg/min IV infusion via Central Venous Catheter (CVC) to counteract propofol-induced cardiovascular depression. Inj atracurium 0.3 mg IV was injected on an intermittent basis. Prior to the incision of the skin and the drilling of the cranium, additional fentanyl 2 µg IV were administered. Ventilatory support was provided using volume-controlled ventilation, with 40% O2 in air, a tidal volume of 35 mL, a positive end-expiratory pressure (PEEP) of 4 cmH₂O, and inspiratory-to-expiratory (I:E) ratio of 1:2.
Intraoperative hemodynamics remained stable. Invasive Blood Pressure (IBP) monitoring showed systolic blood pressure (SBP) between 88–96 mmHg, diastolic blood pressure between 43–53 mmHg, heart rate between 176–121 bpm, oxygen saturation of 98–99%, and end-tidal CO₂ (EtCO₂) of 26–27 mmHg. Total intraoperative fluid administration was Ringerfundin® 150 mL, with estimated blood loss of 70 mL and urine output of 200 mL. The patient was subsequently transferred to the PICU for monitoring.
Postoperative analgesia was maintained with paracetamol (90 mg q6h IV), and sedation was continued with midazolam (0.5 mg/kg/hour IV). Postoperative laboratory analysis revealed hemoglobin at 6.7 g/dL, hematocrit at 19%, thrombocytosis at 573,000/µL, and leukocytosis at 10,900/µL. Arterial blood gas analysis demonstrated a pH of 7.453, base excess of -3.1, pCO₂ of 29.7 mmHg, pO₂ of 172.7 mmHg, HCO₃⁻ of 21 mmol/L, total CO₂ of 21.9 mmol/L, O₂ saturation of 99.6%, and arterial lactate of 2.2 mmol/L. Electrolyte values remained stable, with sodium at 138 mmol/L, potassium at 3.9 mmol/L, chloride at 109 mmol/L, magnesium at 0.6 mmol/L, and albumin at 3.4 g/dL. The patient continued to receive ceftriaxone, metronidazole, dexamethasone and paracetamol, with the addition of ranitidine 6 mg q12h IV for gastrointestinal prophylaxis. The patient continued to be supported by a ventilator, but was successfully weaned and extubated on the first postoperative day. His hemodynamics were stable and he did not experience any respiratory compromise. Packed red blood cells (PRCs) 80 mL were administered due to significant anemia. By postoperative day two, the patient demonstrated clinical improvement and was transferred to the pediatric ward.
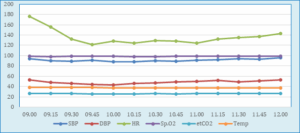
Figure 3: Intraoperative vital signs monitoring
Note: SBP: Systolic Blood Pressure; DBP: Diastolic Blood Pressure; HR: Heart Rate; SpO2: Venous capillary oxygen saturation; EtCO2: End tidal CO2; Temp: Temperature
The management of an infant with SDE and mass effect is challenging due to the complexity of neurological, infection, and systemic complications. Main concerns included elevated ICP, seizures, systemic infection, and hemodynamic instability. The infant presented with recurrent seizures, fever, and lethargy, required urgent evaluation of ICP and cerebral perfusion. pGCS score was E4V5M6, indicating preserved consciousness but subtle neurological compromise. Signs of elevated ICP included lethargy and bulging fontanelles.
Normocapnia was maintained with PaCO₂ levels between 35-40 mmHg. Fluid administration was carefully managed to prevent cerebral edema, avoiding hypotonic solutions.2 Dexamethasone was administered to reduce perilesional edema and inflammatory cytokines.3 Given the history of seizures, careful selection of anesthetic agents is important. Electrolyte and hematological disturbances were monitored and corrected. A transfusion plan was prepared for potential intraoperative blood loss. Empirical broad-spectrum antibiotics, including ceftriaxone and metronidazole, were initiated due to the high likelihood of systemic infection.4 Paracetamol was used for antipyresis and analgesia.
Intraoperative neuro-anesthesia aimed to maintain adequate cerebral perfusion pressure (CPP), prevent intracranial hypertension, and ensure hemodynamic stability. CPP was defined as mean arterial pressure (MAP) minus ICP, with careful control of factors affecting ICP, such as hypercapnia and hypoxia.5 Controlled hypocapnia was targeted to reduce ICP.6 TIVA using propofol minimizes the risk of proconvulsant agents, provides a safer anesthetic approach for infants while maintain effective responsiveness to various potentially serious adverse events.7,8 A balanced fluid strategy, without hypo-osmolar fluid, was employed to maintain circulation without worsening cerebral edema.2 Fentanyl and propofol were selected for induction to minimize ICP fluctuations.9,10 Continuous propofol infusion at 50 µg/kg/min, which has neuroprotective and anticonvulsant effects, was maintained.7 Intraoperative monitoring included IBP to regulate MAP and adequate CPP. CVC was utilized during the surgery to facilitate rapid fluid administration, vasoactive medications, and manage venous air embolism intraoperatively.2 A continuous dobutamine infusion was used to counteract hypotension. Ventilation was volume-controlled, with a tidal volume of 35 mL and PEEP at 4 cmH₂O. EtCO₂ was maintained at 26–27 mmHg to promote cerebral vasoconstriction.6
Postoperative care in the PICU focused on monitoring neurological status through GCS assessments. Head positioning was optimized to minimize ICP, and controlled ventilation was maintained. Dexamethasone was continued to reduce cerebral inflammation.11 Seizure prophylaxis was critical, with midazolam infusion initiated for sedation and anticonvulsant effects.12 Hemodynamically stable, the patient was weaned off sedation and extubated on day 1 postoperatively. MAP was maintained within 50–150 mmHg to minimize the risk of ischemia or hemorrhage,2 and fluid management was guided by intraoperative losses. Postoperative anemia was treated with transfusion of 80 mL of PRCs to provide adequate cerebral perfusion.13 Empirical antibiotic was continued and inflammatory markers were monitored. Pain control was achieved with IV paracetamol, avoiding opioids to prevent respiratory depression.14
Optimized perioperative management in an infant with SDE and mass effect provide cerebral protection, hemodynamic stability, seizure prevention, and infection control. Preoperative strategies focus on reducing ICP, stabilizing metabolism, and administering broad-spectrum antibiotics. Intraoperative management prioritizes controlled ventilation, hemodynamic stabilization, TIVA, and fluid balance. Postoperative monitoring should be done intensively to ensure recovery.
5. Patients Consent
Written consent was obtained from the patient’s parents for the publication of this case report for academic purpose.
6. Ethical Approval
This Case Report Study was approved by the Ethical Committee of Moewardi General Regional Hospital, Surakarta, Central Java, Indonesia, following the institution's ethical guidelines for patient care and research. The approval ensures compliance with standards for patient privacy, consent, and proper use of clinical data.
7. Funding
The authors declare that there was no funding or grant support.
8. Conflict of interests
The authors declare that there was no conflict of interests related to the content of this article.
9. Authors Contribution
AN: Performing anesthesia; Concepting the study; Acquisiting data; Paper writing; Editing; Agreement to be accountable of all aspects of the work.
DYB, IAR: Supervising anesthesia; Collating discussions; Revising for important intellectual content; Editing; Agreement to be accountable of all aspects of the work.
Authors affiliations:
- Andy Nugroho, Department of Anesthesiology & Intensive Therapy, Medical Faculty of Universitas Padjadjaran, Bandung, Indonesia; Email: andy23002@mail.unpad.ac.id; {ORCID: 0000-0003-3709-5114}
- Dewi Yulianti Bisri, Department of Anesthesiology & Intensive Therapy, Medical Faculty of Universitas Padjadjaran, Bandung, Indonesia; Email: dewi.yulianti@unpad.ac.id
- Iwan Abdul Rachman, Department of Anesthesiology & Intensive Therapy, Medical Faculty of Universitas Padjadjaran, Bandung, Indonesia; Email: iwan.abdul@unpad.ac.id
ABSTRACT
Subdural empyema (SDE) is a critical intracranial infection requiring urgent surgical intervention. Anesthetic management in infants with SDE involves challenges such as elevated intracranial pressure (ICP), seizures, and hemodynamic instability. Effective management, including careful monitoring and appropriate anesthesia technique, is essential to improve outcomes and reduce the complications.
A 3-months-old male infant, 6 kg, was admitted with recurrent fever and seizures, accompanied by a history of thrombocytopenia. Neuroimaging revealed extensive subdural empyema, prompting urgent craniotomy for empyema evacuation. General anesthesia was successfully administered, maintaining hemodynamic stability throughout the procedure. Patient’s clinical condition was significantly improved and extubated on the next day, then was transferred to the pediatric ward for continued care.
Preoperative management of an infant with subdural empyema and elevated ICP, focuses on reducing ICP, managing seizures, stabilizing hemodynamics and addressing infection. A Total Intravenous Anesthesia (TIVA) based approach, combined with fluid and ventilation management and neuroprotective agents ensure stable perioperative conditions and support recovery. Postoperatively, a multidisciplinary approach involving neurological monitoring, seizure prophylaxis with midazolam, and antimicrobial therapy was implemented to optimize recovery, control ICP, and prevent complications. This comprehensive management strategy aims to prevent secondary brain injury and promotes a stable recovery.
Abbreviations: CPP: Cerebral perfusion pressure, DBP: Diastolic blood pressure, EtCO2: End tidal CO2, ETT: endotracheal tube, HR: Heart Rate, ICP: intracranial pressure, pGCS: pediatric Glasgow Coma Scale, SBP: Systolic blood pressure, SDE: Subdural empyema, SpO2: Venous capillary oxygen saturation; Temp: Temperature, TIVA: Total Intravenous Anesthesia
Keyword: anesthesia; craniotomy; ICP; infant; subdural empyema; TIVA
Citation: Nugroho A, Bisri DY, Rachman IA. Navigating the anesthetic challenges for craniotomy in infant with bilateral subdural empyema: A case report. Anaesth. pain intensive care 2025;29(3):715-719.
DOI: 10.35975/apic.v29i3.2781
Received: March 10, 2025; Revised: March 22, 2025; Accepted: March 22, 2025
1. INTRODUCTION
Subdural empyema (SDE) is a critical intracranial infection characterized by pus accumulation between the dura mater and the arachnoid membrane, requires urgent surgical intervention.1 Anesthetic management in infants with SDE poses significant challenges due to the occurred elevated intracranial pressure (ICP), ongoing seizures, and hemodynamic instability. Preoperative assessment is crucial to identify these risks and optimize the patient's condition. Adequate airway management is crucial, especially with neurological compromise. Intensive vital signs and neurological status monitoring are necessary throughout the procedure. Anesthesia induction should use minimally cardiovascular effects agents. Intraoperative management should focus on hemodynamic stability and ICP control, often requiring intravenous fluids and medications to support blood pressure. Postoperative monitoring in critical care is vital for early complication detection. Effective anesthetic management improves outcomes in infants with SDE, reduces long-term neurological deficits, and increase survival rates.
2. CASE REPORT
A 3-months-old male infant, 6 kgs, was admitted with recurrent fever and seizures. The infant had been healthy until three weeks prior, until prolonged fever developed alongside two generalized seizures. The most recent seizure lasted approximately three minutes and was characterized by generalized tonic posturing, blank staring, and peripheral cyanosis. Resolution of this seizure occurred following the administration of a 5 mg rectal diazepam. For three weeks prior to admission, the patient experienced recurrent episodes of fever and seizures.
Physical examination revealed clear airway, a respiratory rate of 36 breaths per minute, and oxygen saturation of 99% on 1 L/min nasal cannula, without signs of respiratory distress. Cardiovascular assessment showed a heart rate of 186 bpm, a strong and regular pulse, with normal heart sounds and no murmurs. The body temperature was 39.6°C, and he was alert with pediatric Glasgow Coma Scale (pGCS) of E4V5M6, although exhibiting fever and lethargy. Primitive reflexes were present and appropriate regarding his developmental age.
Neuroimaging (Figure 1) demonstrated extensive subdural empyema with mass effect, characterized by bilateral fronto-temporo-parietal hypodense fluid collections with rim contrast enhancement, leading to compression and displacement of the anterior horn of the left lateral ventricle, dilation of the temporal horns of both ventricles, and rightward subfalcine herniation (the most common type of brain herniation, occurring when brain tissue is pushed under the falx cerebri).

Figure 1: Brain CT Scan demonstrated extensive subdural empyema with mass effect, characterized by bilateral fronto-temporo-parietal hypodense fluid collections with rim contrast enhancement, leading to compression and displacement of the anterior horn of the left lateral ventricle, dilation of the temporal horns of both lateral ventricles, and rightward subfalcine herniation.
The patient also exhibited microcephaly, with persistent cranial suture patency and open anterior and posterior fontanelles. Chest radiography (Figure 2) revealed left-sided pneumonia, despite the absence of respiratory distress.

Figure 2: Chest radiography revealed left-sided pneumonia
Laboratory findings indicated a leukocytosis of 42,600/µL and thrombocytosis of 810,000/µL, suggesting an ongoing infectious and inflammatory process. Other significant parameters included hemoglobin 10.4 g/dL and hematocrit 29%. Coagulation and electrolytes studies were within normal limits (PT 15.1 sec, APTT 21.1 sec, INR 1.13, sodium 146 mmol/L, potassium 5.5 mmol/L and calcium 1.25 mmol/L). Hepatic and renal function tests were normal (SGOT 35 U/L, SGPT 20 U/L, creatinine 0.2 mg/dL, urea at 9 mg/dL). Hepatitis B surface antigen (HBsAg) was negative. Prompt treatment was initiated with ceftriaxone 300 mg q12h IV, metronidazole 50 mg q8h IV, dexamethasone 1 mg q6h IV, and paracetamol 90 mg q6h IV. Due to neurological decline, neuroimaging findings of subdural empyema with mass effect, and hematological complications, the patient was classified as ASA III E, necessitating urgent craniotomy for empyema evacuation.
General anesthesia was induced with fentanyl 10 µg IV, propofol 10 mg IV, and atracurium 5 mg IV. A cuffed endotracheal tube (ETT) size 3.5 was inserted to the depth of 11.5 cm. Hemodynamic parameters were maintained throughout the surgical procedure which lasted for three hours. Intraoperative fluid management included Inj Ringerfundin® at 25 mL/hour, and additional medications included ondansetron 0.8 mg IV for nausea prophylaxis and paracetamol 90 mg IV for analgesia. Anesthesia was maintained via TIVA with propofol at 50 µg/kg/min, while dobutamine was administered at 2 µg/kg/min IV infusion via Central Venous Catheter (CVC) to counteract propofol-induced cardiovascular depression. Inj atracurium 0.3 mg IV was injected on an intermittent basis. Prior to the incision of the skin and the drilling of the cranium, additional fentanyl 2 µg IV were administered. Ventilatory support was provided using volume-controlled ventilation, with 40% O2 in air, a tidal volume of 35 mL, a positive end-expiratory pressure (PEEP) of 4 cmH₂O, and inspiratory-to-expiratory (I:E) ratio of 1:2.
Intraoperative hemodynamics remained stable. Invasive Blood Pressure (IBP) monitoring showed systolic blood pressure (SBP) between 88–96 mmHg, diastolic blood pressure between 43–53 mmHg, heart rate between 176–121 bpm, oxygen saturation of 98–99%, and end-tidal CO₂ (EtCO₂) of 26–27 mmHg. Total intraoperative fluid administration was Ringerfundin® 150 mL, with estimated blood loss of 70 mL and urine output of 200 mL. The patient was subsequently transferred to the PICU for monitoring.
Postoperative analgesia was maintained with paracetamol (90 mg q6h IV), and sedation was continued with midazolam (0.5 mg/kg/hour IV). Postoperative laboratory analysis revealed hemoglobin at 6.7 g/dL, hematocrit at 19%, thrombocytosis at 573,000/µL, and leukocytosis at 10,900/µL. Arterial blood gas analysis demonstrated a pH of 7.453, base excess of -3.1, pCO₂ of 29.7 mmHg, pO₂ of 172.7 mmHg, HCO₃⁻ of 21 mmol/L, total CO₂ of 21.9 mmol/L, O₂ saturation of 99.6%, and arterial lactate of 2.2 mmol/L. Electrolyte values remained stable, with sodium at 138 mmol/L, potassium at 3.9 mmol/L, chloride at 109 mmol/L, magnesium at 0.6 mmol/L, and albumin at 3.4 g/dL. The patient continued to receive ceftriaxone, metronidazole, dexamethasone and paracetamol, with the addition of ranitidine 6 mg q12h IV for gastrointestinal prophylaxis. The patient continued to be supported by a ventilator, but was successfully weaned and extubated on the first postoperative day. His hemodynamics were stable and he did not experience any respiratory compromise. Packed red blood cells (PRCs) 80 mL were administered due to significant anemia. By postoperative day two, the patient demonstrated clinical improvement and was transferred to the pediatric ward.

Figure 3: Intraoperative vital signs monitoring
Note: SBP: Systolic Blood Pressure; DBP: Diastolic Blood Pressure; HR: Heart Rate; SpO2: Venous capillary oxygen saturation; EtCO2: End tidal CO2; Temp: Temperature
3. DISCUSSION
The management of an infant with SDE and mass effect is challenging due to the complexity of neurological, infection, and systemic complications. Main concerns included elevated ICP, seizures, systemic infection, and hemodynamic instability. The infant presented with recurrent seizures, fever, and lethargy, required urgent evaluation of ICP and cerebral perfusion. pGCS score was E4V5M6, indicating preserved consciousness but subtle neurological compromise. Signs of elevated ICP included lethargy and bulging fontanelles.
Normocapnia was maintained with PaCO₂ levels between 35-40 mmHg. Fluid administration was carefully managed to prevent cerebral edema, avoiding hypotonic solutions.2 Dexamethasone was administered to reduce perilesional edema and inflammatory cytokines.3 Given the history of seizures, careful selection of anesthetic agents is important. Electrolyte and hematological disturbances were monitored and corrected. A transfusion plan was prepared for potential intraoperative blood loss. Empirical broad-spectrum antibiotics, including ceftriaxone and metronidazole, were initiated due to the high likelihood of systemic infection.4 Paracetamol was used for antipyresis and analgesia.
Intraoperative neuro-anesthesia aimed to maintain adequate cerebral perfusion pressure (CPP), prevent intracranial hypertension, and ensure hemodynamic stability. CPP was defined as mean arterial pressure (MAP) minus ICP, with careful control of factors affecting ICP, such as hypercapnia and hypoxia.5 Controlled hypocapnia was targeted to reduce ICP.6 TIVA using propofol minimizes the risk of proconvulsant agents, provides a safer anesthetic approach for infants while maintain effective responsiveness to various potentially serious adverse events.7,8 A balanced fluid strategy, without hypo-osmolar fluid, was employed to maintain circulation without worsening cerebral edema.2 Fentanyl and propofol were selected for induction to minimize ICP fluctuations.9,10 Continuous propofol infusion at 50 µg/kg/min, which has neuroprotective and anticonvulsant effects, was maintained.7 Intraoperative monitoring included IBP to regulate MAP and adequate CPP. CVC was utilized during the surgery to facilitate rapid fluid administration, vasoactive medications, and manage venous air embolism intraoperatively.2 A continuous dobutamine infusion was used to counteract hypotension. Ventilation was volume-controlled, with a tidal volume of 35 mL and PEEP at 4 cmH₂O. EtCO₂ was maintained at 26–27 mmHg to promote cerebral vasoconstriction.6
Postoperative care in the PICU focused on monitoring neurological status through GCS assessments. Head positioning was optimized to minimize ICP, and controlled ventilation was maintained. Dexamethasone was continued to reduce cerebral inflammation.11 Seizure prophylaxis was critical, with midazolam infusion initiated for sedation and anticonvulsant effects.12 Hemodynamically stable, the patient was weaned off sedation and extubated on day 1 postoperatively. MAP was maintained within 50–150 mmHg to minimize the risk of ischemia or hemorrhage,2 and fluid management was guided by intraoperative losses. Postoperative anemia was treated with transfusion of 80 mL of PRCs to provide adequate cerebral perfusion.13 Empirical antibiotic was continued and inflammatory markers were monitored. Pain control was achieved with IV paracetamol, avoiding opioids to prevent respiratory depression.14
4. CONCLUSION
Optimized perioperative management in an infant with SDE and mass effect provide cerebral protection, hemodynamic stability, seizure prevention, and infection control. Preoperative strategies focus on reducing ICP, stabilizing metabolism, and administering broad-spectrum antibiotics. Intraoperative management prioritizes controlled ventilation, hemodynamic stabilization, TIVA, and fluid balance. Postoperative monitoring should be done intensively to ensure recovery.
5. Patients Consent
Written consent was obtained from the patient’s parents for the publication of this case report for academic purpose.
6. Ethical Approval
This Case Report Study was approved by the Ethical Committee of Moewardi General Regional Hospital, Surakarta, Central Java, Indonesia, following the institution's ethical guidelines for patient care and research. The approval ensures compliance with standards for patient privacy, consent, and proper use of clinical data.
7. Funding
The authors declare that there was no funding or grant support.
8. Conflict of interests
The authors declare that there was no conflict of interests related to the content of this article.
9. Authors Contribution
AN: Performing anesthesia; Concepting the study; Acquisiting data; Paper writing; Editing; Agreement to be accountable of all aspects of the work.
DYB, IAR: Supervising anesthesia; Collating discussions; Revising for important intellectual content; Editing; Agreement to be accountable of all aspects of the work.
10. REFERENCES
- French H, Schaefer N, Keijzers G, Barison D, Olson S. Intracranial subdural empyema: a 10-year case series. West J Emerg Med. 2012;13(6):509–11. PubMed
- Kalita N, Goswami A, Goswami P. Making pediatric neuroanesthesia safer. J Pediatr Neurosci. 2017;12:305–12. PubMed DOI: 4103/jpn.JPN_173_17
- Ye Y, Xu J, Han Y. Dexamethasone for delayed edema after intracerebral hemorrhage: to be or not to be? Heliyon. 2023;9(7). PubMed DOI: 1016/j.heliyon.2023.e17621
- Bruner DI, Littlejohn L, Pritchard A. Subdural empyema presenting with seizure, confusion, and focal weakness. West J Emerg Med. 2012;13(6):509–11. PubMed DOI: 5811/westjem.2012.5.11727
- Butterworth JF, Mackey DC, Wasnick JD. Morgan & Mikhail’s Clinical Anesthesiology. 7th ed. New York: McGraw Hill; 2022.
- Kukreti V, Mohseni-Bod H, Drake J. Management of raised intracranial pressure in children with traumatic brain injury. J Pediatr Neurosci. 2014;9:207–15. PubMed DOI: 4103/1817-1745.147572
- Shetty A, Pardeshi S, Shah VM, Kulkarni A. Anesthesia considerations in epilepsy surgery. Int J Surg. 2016;36:454–9. PubMed DOI: 1016/j.ijsu.2015.07.006
- Filho EM, Riechelmann MB. Propofol use in newborns and children: is it safe? A systematic review. J Pediatr (Rio J). 2020;96:289–309. PubMed DOI: 1016/j.jped.2019.08.011
- Mazandi VM, Lang SS, Rahman RK, Nishisaki A, Beaulieu F, Zhang B, et al. Co-administration of ketamine in pediatric patients with neurologic conditions at risk for intracranial hypertension. Neurocrit Care. 2023;38(2):242–53. PubMed DOI: 1007/s12028-022-01611-2
- Zanza C, Piccolella F, Racca F, Romenskaya T, Longhitano Y, Franceschi F, et al. Ketamine in acute brain injury: current opinion following cerebral circulation and electrical activity. Healthcare (Basel). 2022;10. PubMed DOI: 3390/healthcare10030566
- Vazquez S, Gold J, Spirollari E, Akmal S, Hanft SJ. The story of dexamethasone and how it became one of the most widely used drugs in neurosurgery. J Neurosurg. 2024;140(4):1191–7. PubMed DOI: 3171/2023.9.JNS231099
- Pacifici GM. Clinical pharmacology of midazolam in neonates and children: effect of disease—a review. Int J Pediatr. 2014;2014:1–20. PubMed DOI: 1155/2014/309342
- Naik S, Nirale A, Bharadwaj S, Sangeetha RP, Shukla D, Kamath S. Post-operative anemia in children undergoing elective neurosurgery: an analysis of incidence, risk factors, and outcomes. J Neurosci Rural Pract. 2024;15(1):29–33. PubMed DOI: 25259/JNRP_338_2023
- Niesters M, Overdyk F, Smith T, Aarts L, Dahan A. Opioid-induced respiratory depression in paediatrics: a review of case reports. Br J Anaesth. 2013;110:175–82. PubMed DOI: 1093/bja/aes447