Kiran Reddy Atla 1, Vinith Kumar Kumaran 2, Brindha Rathnasabapathy 3,
Prabhu Thilaak 4, Panneerselvam Periasamy 5, Arbind Kumar Choudhary 6
Authors affiliations:
Background & objective: Preoperative anxiety in pediatric patients is associated with poor cooperation, increased anesthetic requirements, and adverse postoperative outcomes. Oral premedication with midazolam is common, but its effectiveness may be suboptimal when used alone. This study aimed to evaluate the efficacy and safety of a combination of oral midazolam and ketamine compared to oral midazolam alone in children aged 1 to 6 years undergoing elective surgery.
Methodology: A prospective, randomized, double-blind clinical trial was conducted involving 62 ASA I/II children scheduled for elective surgery under general anesthesia. Participants were randomized into two groups: Group M received oral midazolam 0.5 mg/kg, and Group MK received midazolam 0.25 mg/kg with ketamine 3 mg/kg, both diluted in paracetamol syrup. Sedation scores were recorded at 5-minute intervals up to 20 minutes post-administration. Behavioral responses were assessed at parental separation and during mask induction. Heart rate and oxygen saturation were monitored, and adverse events were noted. Statistical analysis was performed using SPSS v22.0, with P < 0.05 considered significant.
Results: Group MK demonstrated significantly lower sedation scores at 5, 15, and 20 minutes (P = 0.004, 0.036, 0.001 respectively). Behavioral scores at parental separation and induction were also significantly better in Group MK (P < 0.001, P = 0.001). Mild but significant increases in heart rate were noted in Group MK at 5, 10, and 20 minutes (P < 0.05), with no respiratory compromise in either group. Adverse events were minimal, with three transient cases of nystagmus and two instances of drug rejection noted in the combination group.
Conclusion: The combination of oral midazolam and ketamine provides earlier onset, deeper sedation, and improved behavioral responses compared to midazolam alone, with a comparable safety profile. This regimen offers a superior alternative for pediatric premedication in elective surgical settings.
Keywords: Pediatric Sedation; Midazolam; Ketamine; Premedication; Parental Separation Anxiety Scale
Citation: Atla KR, Kumaran VK, Rathnasabapathy B, Thilaak P, Periasamy P, Choudhary AK. A clinical comparison of oral midazolam vs. oral midazolam-ketamine for premedication in pediatric patients undergoing elective surgeries: efficacy and safety assessment. Anaesth. pain intensive care 2025;29(3):466-472
DOI: 10.35975/apic.v29i3.2760
Received: February 20, 2025; Revised: March 15, 2025; Accepted: March 22, 2025
Preoperative anxiety is a common and impactful concern in pediatric anesthesia, particularly among children aged 1 to 6 years. This anxiety often manifests as agitation, crying, resistance to separation from parents, and poor compliance during induction, potentially leading to increased anesthetic requirements, postoperative behavioral disturbances, and reduced parental satisfaction. The perioperative period, especially the moment of separation and mask induction, represents a significant psychological stressor for young children due to limited coping mechanisms and unfamiliar surroundings.1,2
Pharmacologic premedication is widely employed to alleviate such anxiety, facilitate smooth separation, and ensure a calm induction. Among the commonly used agents, midazolam, a short-acting benzodiazepine, is preferred for its rapid onset, anxiolytic effect, and anterograde amnesia. However, midazolam alone may not consistently produce satisfactory sedation in all children and is sometimes associated with paradoxical reactions, poor mask acceptance, or inadequate anxiolysis.3,4
To enhance the sedative effect, combination regimens have gained attention. Ketamine, an NMDA receptor antagonist with strong sedative and analgesic properties, preserves airway reflexes and supports hemodynamic stability. When used in low oral doses, it synergizes with midazolam to deepen sedation without significantly increasing adverse effects.5 Several studies have demonstrated that combining midazolam with ketamine improves the depth and quality of sedation, facilitates easier separation, and enhances cooperation at induction compared to midazolam alone.6
The oral route remains the most favored in pediatric practice due to ease of administration and high acceptability. Moreover, palatability enhancers such as paracetamol syrup can improve compliance and reduce the likelihood of vomiting or refusal to ingest the medication. Despite growing interest in combination premedication strategies, clinical data comparing midazolam and midazolam-ketamine combinations in Indian pediatric populations remain limited.7,8
This study was conducted to compare the efficacy and safety of oral midazolam alone with a combination of midazolam and ketamine in children aged 1–6 years undergoing elective surgery. The primary objective was to compare sedation scores at multiple time intervals post-administration. Secondary outcomes included behavioral scores at parent separation and induction, hemodynamic responses, and the incidence of adverse events.
This prospective, randomized, double-blind clinical trial was conducted in the Department of Anesthesiology at a tertiary care teaching hospital over a six-month period from August 2018 to January 2019. Ethical clearance was obtained from the Institutional Ethics Committee (Ref: VMKVMC&H/IEC/19/039), and written informed consent was obtained from the parents or legal guardians of all participating children. The study enrolled 70 pediatric patients aged between 1 and 6 years, all classified as American Society of Anesthesiologists (ASA) physical status I or II and scheduled for elective surgeries under general anesthesia. Children with upper respiratory tract infections, known drug allergies, developmental disorders, or ASA grade III or higher were excluded. After eligibility screening, 8 children were excluded—4 did not meet inclusion criteria and 4 declined participation. A total of 62 participants were randomized into two groups (Group M and Group MK) using a computer-generated block randomization sequence with allocation concealment ensured through opaque sealed envelopes. Group M received oral midazolam 0.5 mg/kg, while Group MK received oral midazolam 0.25 mg/kg combined with ketamine 3 mg/kg. The total volume was adjusted to 0.5 mL/kg and administered in paracetamol syrup to enhance palatability. Drug preparation and administration were carried out by an anesthesiologist not involved in observation or outcome assessment, maintaining double-blinding throughout.
Sedation scores were assessed at baseline and at 5-minute intervals for 20 minutes using a standardized 4-point scale. Acceptable sedation was defined as a score of 1 or 2. At 20 minutes, children were separated from parents and transferred to the operating room, where behavioral response was assessed using the Parental Separation Anxiety Scale. Mask acceptance during induction was also evaluated. Hemodynamic parameters, including heart rate and oxygen saturation, were recorded at baseline and every 5 minutes for 20 minutes following premedication. Adverse events such as vomiting, bradycardia, hypoxia, and medication rejection were monitored.
As shown in Figure 1: CONSORT Flow Diagram, of the 62 children randomized, all 31 children in Group M received the intervention and were included in the final analysis. In Group MK, 31 children were allocated and received the medication; however, 2 were excluded from analysis as they spat out the drug, leaving 29 children analyzed in that group. The primary outcome was sedation score at 5-minute intervals, while secondary outcomes included behavioral scores, hemodynamic stability, and adverse event incidence.
Statistical analysis was conducted using SPSS version 22.0 (IBM Corp., Armonk, NY, USA). Continuous variables were expressed as mean ± standard deviation and compared using Student’s t-test or the Mann–Whitney U test, depending on data distribution. Categorical variables were analyzed using the chi-square or Fisher’s exact test. A two-tailed P-value of less than 0.05 was considered statistically significant.
A total of 62 pediatric patients were initially enrolled. After excluding two participants from the Midazolam + Ketamine group (Group MK) who expelled the study drug, the final analysis included 60 children: 31 in Group M (Midazolam alone) and 29 in Group MK (Midazolam + Ketamine). The outcomes are presented under the following subsections.
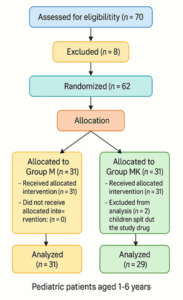
Figure 1: CONSORT Flow Diagram
3.1 Demographic and Baseline Characteristics
Baseline parameters including age, weight, gender distribution, and type of surgery were assessed to ensure comparability between the two groups. As shown in Table 1, the groups were well-matched with no statistically significant differences across these variables.
Age and weight are expressed as mean ± standard deviation and were compared using the independent t-test. Gender distribution was analyzed using the Chi-square test. No statistically significant differences were found between the two groups in any baseline characteristic (P > 0.05), indicating effective randomization. Type of surgery data was aggregated for overall case mix description and not included in statistical comparison There were no statistically significant differences between Group M and Group MK in terms of age, weight, or gender distribution (P > 0.05). Urological procedures were the most common surgeries
performed (48%), followed by ENT (25%), plastic (18%), general (5%), and others (3%), as depicted in Figure 2. The balanced demographic and surgical characteristics confirm the effectiveness of randomization and enhance the generalizability of the study findings.
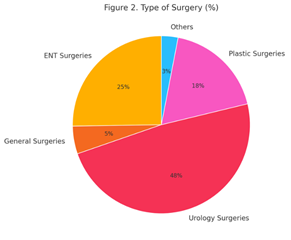
Figure 2: Type of Surgery (%)
3.2 Sedation Scores Over Time
Sedation levels were monitored at baseline and every five minutes up to 20 minutes following drug administration. As shown in Table 2, children in Group MK (Midazolam + Ketamine) exhibited significantly lower sedation scores—indicating deeper sedation—at 5, 15, and 20 minutes compared to Group M (Midazolam alone).
Sedation scores were assessed using a standardized 4-point scale. Values are presented as mean ± standard deviation. Comparisons between the two groups at each time point were conducted using the independent t-test. Statistically significant differences were observed at 5, 15, and 20 minutes (P < 0.05), with Group MK demonstrating significantly deeper sedation than Group M at these intervals.
3.3 Behavioral Scores at Parent Separation and
Induction
Behavioral responses during parent separation and anesthesia induction were evaluated using standardized scoring tools. As detailed in Table 3, children in Group MK demonstrated significantly better behavior than those in Group M.
At the time of induction, the mean behavioral score in Group MK was 1.07 ± 0.54, compared to 2.00 ± 0.68 in Group M (P < 0.001). Similarly, during separation from parents, Group MK showed more favorable responses (1.07 ± 0.26 vs. 1.52 ± 0.63; P = 0.001). These findings highlight the superior anxiolytic and cooperative effect of the Midazolam-Ketamine combination in the pediatric population.
3.4 Hemodynamic Parameters and Oxygen Saturation
Heart rate (HR) and peripheral oxygen saturation (SpO₂) were monitored at baseline and at 5-minute intervals up to 20 minutes following oral premedication. These parameters were assessed to evaluate the cardiovascular and respiratory safety profiles of the sedative regimens. As shown in Table 4, significant differences were noted in HR between the two groups at certain time points, while SpO₂ values remained stable and comparable throughout the observation period.
Heart rate and oxygen saturation values are expressed as mean ± standard deviation. The independent t-test was used for intergroup comparisons at each time point. Statistically significant differences in HR were observed at 5, 10, and 20 minutes (*P < 0.05), with Group MK showing transient elevations, which remained within physiologically acceptable limits. No significant differences in SpO₂ were observed, and all values remained ≥98%, indicating preserved respiratory function and comparable safety profiles.
Adverse Events:
No adverse events were reported in Group M; whereas, three children experienced transient nystagmus, and two children expelled the medication before full ingestion in Group MK. All incidents were self-limiting and required no clinical intervention.
Preoperative anxiety remains a pervasive challenge in pediatric anesthesia, often manifesting as agitation, resistance, and poor cooperation during induction. Such psychological distress, particularly in children aged 1 to 6 years, can negatively impact the anesthetic course and postoperative recovery. Pharmacologic premedication remains a cornerstone strategy to alleviate anxiety, enhance patient compliance, and ensure a smoother perioperative experience.9,10 In this randomized,
double-blind trial, we evaluated the clinical efficacy of oral midazolam alone compared with a combination of oral midazolam and ketamine. The results support the superiority of the combination regimen in terms of sedation depth, onset time, behavioral control, and safety profile.
Children in the midazolam-ketamine group (Group MK) demonstrated significantly lower sedation scores at 5, 15, and 20 minutes post-administration compared to those in the midazolam-only group. These differences reflect a more rapid onset and sustained sedation, which is clinically advantageous in high-volume surgical environments where efficiency is critical. Our findings align with those reported by Funk et al., who observed faster onset and deeper sedation with the midazolam-ketamine combination in pediatric dental patients undergoing minor procedures. Similarly, Ghai et al. and Thakur et al. reported enhanced anxiolysis and improved cooperation during induction when ketamine was used as an adjunct to midazolam, highlighting the synergistic action of the two agents.11,12
The improved behavioral scores observed in Group MK at the time of parent separation and mask acceptance further underscore the benefit of the combination therapy in reducing procedural distress. Given the developmental vulnerability and emotional dependency of children in this age group, such improvements are particularly meaningful from both clinical and parental perspectives. Oral ketamine, even at low doses, is known to produce dissociative sedation while preserving airway reflexes and spontaneous respiration—an ideal complement to midazolam's anxiolytic and amnestic properties.13
Hemodynamic monitoring revealed statistically significant elevations in heart rate at 5, 10, and 20 minutes in Group MK, consistent with ketamine’s sympathomimetic effects. However, these elevations remained within normal physiological limits and did not require intervention. These findings corroborate the data from Darlong et al., who noted transient increases in heart rate with oral ketamine administration, which were clinically insignificant.14
Oxygen saturation levels remained stable throughout the observation period, with no instances of desaturation, airway obstruction, or bradycardia in either group. This confirms the respiratory safety of both regimens when administered under close monitoring. Previous studies have similarly shown that oral midazolam and ketamine—when used in appropriate dosages—rarely cause respiratory compromise in ASA I and II pediatric populations.15
Adverse events were limited and non-serious. In Group MK, three children experienced transient nystagmus, and two were excluded due to spitting out the medication. Such effects are consistent with the pharmacodynamic profile of ketamine and have been reported in earlier trials as mild and self-limiting¹⁶. Importantly, no cases of vomiting, hallucinations, emergence delirium, or other serious adverse effects were recorded, reinforcing the safety of the studied regimen.16,17
The surgical case mix included predominantly urological and ENT procedures, which reflects a representative pediatric surgical population. The two study groups were demographically comparable, with no significant differences in baseline age, weight, or gender distribution, thereby affirming the integrity of the randomization process.18,19
While the study provides valuable insights, it has certain limitations. The relatively small sample size and single-center design may affect the generalizability of the findings. Additionally, the exclusion of patients with ASA grade III or higher limits the applicability to more complex clinical scenarios. Moreover, this trial focused exclusively on intraoperative sedation and did not capture postoperative recovery variables such as pain, emergence agitation, or parent satisfaction. Future research should expand upon these dimensions and include alternative adjuncts such as dexmedetomidine or clonidine for comparative effectiveness studies.20
In conclusion, the combination of oral midazolam and ketamine offers significant clinical advantages over midazolam alone in terms of sedation depth, behavioral control, and tolerability. Its use in pediatric elective surgeries appears both effective and safe, making it a valuable option in routine anesthetic practice.
The combination of oral midazolam and ketamine proved to be significantly more effective than oral midazolam alone in providing early-onset, deeper sedation and improved behavioral compliance during both parental separation and anesthesia induction in children aged 1 to 6 years undergoing elective surgery. The combination regimen was well-tolerated, with minimal and self-limiting adverse effects, and no clinically significant respiratory or hemodynamic compromise. These findings suggest that oral midazolam-ketamine, when administered in appropriate doses, is a safe and superior alternative to midazolam monotherapy for pediatric premedication. Its use may enhance perioperative efficiency and patient comfort in routine pediatric anesthetic practice.
7. Data availability
The dataset presented in the study is available on request from the corresponding author during submission or after publication.
8. Conflict of Interests
The authors declare that they have no conflicts of interest regarding the publication of this manuscript.
9. Ethical issues
Ethical clearance was obtained from the Institutional Ethics Committee (Ref: VMKVMC&H/IEC/19/039), and written informed consent was obtained from the parents or legal guardians of all participating children.
10. Funding/Support
The authors declare that this research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
11. Authors contribution
KRA: Conceptualization, Literature search, Data Collection, Manuscript drafting
VKK: Study Design, Data Analysis, Manuscript Review
RB: Supervision, Critical Review, Approval of the Final Manuscript
DHS: Data Collection, Statistical Analysis, Manuscript Editing
PT: Methodology Development, Interpretation of Results, Manuscript Review
AKC: Conceptualization, Data Interpretation, Corresponding Author
Prabhu Thilaak 4, Panneerselvam Periasamy 5, Arbind Kumar Choudhary 6
Authors affiliations:
- Kiran Reddy Atla, Postgraduate, Department of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College and Hospital, VMRF, Salem, Tamil Nadu, India: Email: kiranatla35@gmail.com, {ORCID:0009-0005-8075-3348}
- Vinith Kumar Kumaran, Vinayaka Mission's Kirupananda Variyar Medical College and Hospital, VMRF, Salem, Tamil Nadu, India: Email: vinithkumar1403@gmail.com, {ORCID:0009-0009-9376-9161}
- Brindha Rathnasabapathy, Department of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College and Hospital, VMRF, Salem, Tamil Nadu, India: Email: mskbrins63@gmail.com, {ORCID:0000-0002-9776-2700}
- Prabhu Thilaak, Department of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College and Hospital, Salem, Tamil Nadu, India: Email: prabhuthilaak@gmail.com, {ORCID:0009-0003-7452-2602}
- Panneerselvam Periasamy, Assistant Professor of Physiology, Government Erode Medical College and Hospital, Erode, Tamil Nadu, India; E-mail: pannphysio@gmail.com; {ORCID:0000-0002-3358-313X}
- Arbind Kumar Choudhary, Assistant Professor of Pharmacology, Government Erode Medical College and Hospital, Tamil Nadu, India: Email: arbindkch@gmail.com, {ORCID:0000-0001-8910-1745}
ABSTRACT
Background & objective: Preoperative anxiety in pediatric patients is associated with poor cooperation, increased anesthetic requirements, and adverse postoperative outcomes. Oral premedication with midazolam is common, but its effectiveness may be suboptimal when used alone. This study aimed to evaluate the efficacy and safety of a combination of oral midazolam and ketamine compared to oral midazolam alone in children aged 1 to 6 years undergoing elective surgery.
Methodology: A prospective, randomized, double-blind clinical trial was conducted involving 62 ASA I/II children scheduled for elective surgery under general anesthesia. Participants were randomized into two groups: Group M received oral midazolam 0.5 mg/kg, and Group MK received midazolam 0.25 mg/kg with ketamine 3 mg/kg, both diluted in paracetamol syrup. Sedation scores were recorded at 5-minute intervals up to 20 minutes post-administration. Behavioral responses were assessed at parental separation and during mask induction. Heart rate and oxygen saturation were monitored, and adverse events were noted. Statistical analysis was performed using SPSS v22.0, with P < 0.05 considered significant.
Results: Group MK demonstrated significantly lower sedation scores at 5, 15, and 20 minutes (P = 0.004, 0.036, 0.001 respectively). Behavioral scores at parental separation and induction were also significantly better in Group MK (P < 0.001, P = 0.001). Mild but significant increases in heart rate were noted in Group MK at 5, 10, and 20 minutes (P < 0.05), with no respiratory compromise in either group. Adverse events were minimal, with three transient cases of nystagmus and two instances of drug rejection noted in the combination group.
Conclusion: The combination of oral midazolam and ketamine provides earlier onset, deeper sedation, and improved behavioral responses compared to midazolam alone, with a comparable safety profile. This regimen offers a superior alternative for pediatric premedication in elective surgical settings.
Keywords: Pediatric Sedation; Midazolam; Ketamine; Premedication; Parental Separation Anxiety Scale
Citation: Atla KR, Kumaran VK, Rathnasabapathy B, Thilaak P, Periasamy P, Choudhary AK. A clinical comparison of oral midazolam vs. oral midazolam-ketamine for premedication in pediatric patients undergoing elective surgeries: efficacy and safety assessment. Anaesth. pain intensive care 2025;29(3):466-472
DOI: 10.35975/apic.v29i3.2760
Received: February 20, 2025; Revised: March 15, 2025; Accepted: March 22, 2025
1. INTRODUCTION
Preoperative anxiety is a common and impactful concern in pediatric anesthesia, particularly among children aged 1 to 6 years. This anxiety often manifests as agitation, crying, resistance to separation from parents, and poor compliance during induction, potentially leading to increased anesthetic requirements, postoperative behavioral disturbances, and reduced parental satisfaction. The perioperative period, especially the moment of separation and mask induction, represents a significant psychological stressor for young children due to limited coping mechanisms and unfamiliar surroundings.1,2
Pharmacologic premedication is widely employed to alleviate such anxiety, facilitate smooth separation, and ensure a calm induction. Among the commonly used agents, midazolam, a short-acting benzodiazepine, is preferred for its rapid onset, anxiolytic effect, and anterograde amnesia. However, midazolam alone may not consistently produce satisfactory sedation in all children and is sometimes associated with paradoxical reactions, poor mask acceptance, or inadequate anxiolysis.3,4
To enhance the sedative effect, combination regimens have gained attention. Ketamine, an NMDA receptor antagonist with strong sedative and analgesic properties, preserves airway reflexes and supports hemodynamic stability. When used in low oral doses, it synergizes with midazolam to deepen sedation without significantly increasing adverse effects.5 Several studies have demonstrated that combining midazolam with ketamine improves the depth and quality of sedation, facilitates easier separation, and enhances cooperation at induction compared to midazolam alone.6
The oral route remains the most favored in pediatric practice due to ease of administration and high acceptability. Moreover, palatability enhancers such as paracetamol syrup can improve compliance and reduce the likelihood of vomiting or refusal to ingest the medication. Despite growing interest in combination premedication strategies, clinical data comparing midazolam and midazolam-ketamine combinations in Indian pediatric populations remain limited.7,8
This study was conducted to compare the efficacy and safety of oral midazolam alone with a combination of midazolam and ketamine in children aged 1–6 years undergoing elective surgery. The primary objective was to compare sedation scores at multiple time intervals post-administration. Secondary outcomes included behavioral scores at parent separation and induction, hemodynamic responses, and the incidence of adverse events.
2. METHODOLOGY
This prospective, randomized, double-blind clinical trial was conducted in the Department of Anesthesiology at a tertiary care teaching hospital over a six-month period from August 2018 to January 2019. Ethical clearance was obtained from the Institutional Ethics Committee (Ref: VMKVMC&H/IEC/19/039), and written informed consent was obtained from the parents or legal guardians of all participating children. The study enrolled 70 pediatric patients aged between 1 and 6 years, all classified as American Society of Anesthesiologists (ASA) physical status I or II and scheduled for elective surgeries under general anesthesia. Children with upper respiratory tract infections, known drug allergies, developmental disorders, or ASA grade III or higher were excluded. After eligibility screening, 8 children were excluded—4 did not meet inclusion criteria and 4 declined participation. A total of 62 participants were randomized into two groups (Group M and Group MK) using a computer-generated block randomization sequence with allocation concealment ensured through opaque sealed envelopes. Group M received oral midazolam 0.5 mg/kg, while Group MK received oral midazolam 0.25 mg/kg combined with ketamine 3 mg/kg. The total volume was adjusted to 0.5 mL/kg and administered in paracetamol syrup to enhance palatability. Drug preparation and administration were carried out by an anesthesiologist not involved in observation or outcome assessment, maintaining double-blinding throughout.
Sedation scores were assessed at baseline and at 5-minute intervals for 20 minutes using a standardized 4-point scale. Acceptable sedation was defined as a score of 1 or 2. At 20 minutes, children were separated from parents and transferred to the operating room, where behavioral response was assessed using the Parental Separation Anxiety Scale. Mask acceptance during induction was also evaluated. Hemodynamic parameters, including heart rate and oxygen saturation, were recorded at baseline and every 5 minutes for 20 minutes following premedication. Adverse events such as vomiting, bradycardia, hypoxia, and medication rejection were monitored.
As shown in Figure 1: CONSORT Flow Diagram, of the 62 children randomized, all 31 children in Group M received the intervention and were included in the final analysis. In Group MK, 31 children were allocated and received the medication; however, 2 were excluded from analysis as they spat out the drug, leaving 29 children analyzed in that group. The primary outcome was sedation score at 5-minute intervals, while secondary outcomes included behavioral scores, hemodynamic stability, and adverse event incidence.
Statistical analysis was conducted using SPSS version 22.0 (IBM Corp., Armonk, NY, USA). Continuous variables were expressed as mean ± standard deviation and compared using Student’s t-test or the Mann–Whitney U test, depending on data distribution. Categorical variables were analyzed using the chi-square or Fisher’s exact test. A two-tailed P-value of less than 0.05 was considered statistically significant.
3. RESULTS
A total of 62 pediatric patients were initially enrolled. After excluding two participants from the Midazolam + Ketamine group (Group MK) who expelled the study drug, the final analysis included 60 children: 31 in Group M (Midazolam alone) and 29 in Group MK (Midazolam + Ketamine). The outcomes are presented under the following subsections.

Figure 1: CONSORT Flow Diagram
3.1 Demographic and Baseline Characteristics
Baseline parameters including age, weight, gender distribution, and type of surgery were assessed to ensure comparability between the two groups. As shown in Table 1, the groups were well-matched with no statistically significant differences across these variables.
| Table 1: Demographic and baseline characteristics of study participants | |||
| Parameter | Group M
(n = 31) |
Group MK
(n = 29) |
P-value |
| Age (years) | 3.13 ± 1.41 | 2.72 ± 1.53 | 0.16 t |
| Weight (kg) | 14.13 ± 2.13 | 13.52 ± 3.16 | 0.31 t |
| Gender Distribution | |||
| Male | 13 (41.9) | 19 (65.5) | 0.08 ꭓ2 |
| Female | 18 (58.1) | 10 (34.5) | |
| Data presented as mean ± SD or n (%); P > 0.05 considered as significant;
t - independent t-test; ꭓ2 - Chi-square test |
|||
performed (48%), followed by ENT (25%), plastic (18%), general (5%), and others (3%), as depicted in Figure 2. The balanced demographic and surgical characteristics confirm the effectiveness of randomization and enhance the generalizability of the study findings.

Figure 2: Type of Surgery (%)
3.2 Sedation Scores Over Time
Sedation levels were monitored at baseline and every five minutes up to 20 minutes following drug administration. As shown in Table 2, children in Group MK (Midazolam + Ketamine) exhibited significantly lower sedation scores—indicating deeper sedation—at 5, 15, and 20 minutes compared to Group M (Midazolam alone).
| Table 2: Comparison of sedation scores over time between the groups | |||
| Time (min) | Group M
(n = 31) |
Group MK
(n = 29) |
P-value |
| Before Premed | 3.26 ± 0.51 | 2.61 ± 0.50 | 0.54 |
| 0 | 3.26 ± 0.51 | 2.10 ± 0.47 | 0.865 |
| 5 | 3.34 ± 0.48 * | 1.97 ± 0.41 * | 0.004 |
| 10 | 3.21 ± 0.68 | 1.72 ± 0.46 | 0.209 |
| 15 | 2.24 ± 0.44 * | 1.77 ± 0.43 * | 0.036 |
| 20 | 1.97 ± 0.33 * | 1.52 ± 0.51 * | 0.001 |
| Data presented as mean ± SD; *Indicates statistically significant difference (P < 0.05). | |||
3.3 Behavioral Scores at Parent Separation and
Induction
Behavioral responses during parent separation and anesthesia induction were evaluated using standardized scoring tools. As detailed in Table 3, children in Group MK demonstrated significantly better behavior than those in Group M.
| Table 3: Comparative behavioral scores at parental separation and anesthesia induction | |||
| Parameter | Group M
(n = 31) |
Group MK
(n = 29) |
P-value |
| At Induction | 2.00 ± 0.68 * | 1.07 ± 0.54 * | < 0.001 |
| At Parental Separation | 1.52 ± 0.63 * | 1.07 ± 0.26 * | 0.001 |
| Data presented as mean ± SD. Comparisons between groups were made using the independent t-test; *P < 0.05 considered statistically significant | |||
3.4 Hemodynamic Parameters and Oxygen Saturation
Heart rate (HR) and peripheral oxygen saturation (SpO₂) were monitored at baseline and at 5-minute intervals up to 20 minutes following oral premedication. These parameters were assessed to evaluate the cardiovascular and respiratory safety profiles of the sedative regimens. As shown in Table 4, significant differences were noted in HR between the two groups at certain time points, while SpO₂ values remained stable and comparable throughout the observation period.
| Table 4: Comparative heart rate and oxygen saturation data over time in the groups | ||||||
| Time (min) | Heart Rate (bpm) | SpO₂ (%) | ||||
| Group M | Group MK | P-value | Group M | Group MK | P-value | |
| Before Premed | 103.16 ± 7.35 | 104.00 ± 8.52 | 0.84 | 98.6 ± 0.9 | 98.5 ± 1.0 | 0.78 |
| 0 | 103.35 ± 8.02 | 105.72 ± 8.25 | 0.12 | 98.4 ± 0.8 | 98.6 ± 0.9 | 0.42 |
| 5 | 102.81 ± 7.52 * | 107.16 ± 9.18 * | 0.04 | 98.2 ± 0.7 | 98.3 ± 0.6 | 0.35 |
| 10 | 102.72 ± 7.71 * | 108.25 ± 7.95 * | 0.03 | 98.3 ± 0.6 | 98.4 ± 0.7 | 0.29 |
| 15 | 98.45 ± 8.76 | 100.43 ± 9.52 | 0.18 | 98.5 ± 0.8 | 98.4 ± 0.6 | 0.76 |
| 20 | 102.55 ± 7.40 * | 99.87 ± 6.88 * | 0.02 | 98.7 ± 0.7 | 98.6 ± 0.5 | 0.65 |
| Data presented as mean ± SD. Comparisons between groups were made using the independent t-test; *P < 0.05 considered statistically significant | ||||||
Adverse Events:
No adverse events were reported in Group M; whereas, three children experienced transient nystagmus, and two children expelled the medication before full ingestion in Group MK. All incidents were self-limiting and required no clinical intervention.
4. DISCUSSION
Preoperative anxiety remains a pervasive challenge in pediatric anesthesia, often manifesting as agitation, resistance, and poor cooperation during induction. Such psychological distress, particularly in children aged 1 to 6 years, can negatively impact the anesthetic course and postoperative recovery. Pharmacologic premedication remains a cornerstone strategy to alleviate anxiety, enhance patient compliance, and ensure a smoother perioperative experience.9,10 In this randomized,
double-blind trial, we evaluated the clinical efficacy of oral midazolam alone compared with a combination of oral midazolam and ketamine. The results support the superiority of the combination regimen in terms of sedation depth, onset time, behavioral control, and safety profile.
Children in the midazolam-ketamine group (Group MK) demonstrated significantly lower sedation scores at 5, 15, and 20 minutes post-administration compared to those in the midazolam-only group. These differences reflect a more rapid onset and sustained sedation, which is clinically advantageous in high-volume surgical environments where efficiency is critical. Our findings align with those reported by Funk et al., who observed faster onset and deeper sedation with the midazolam-ketamine combination in pediatric dental patients undergoing minor procedures. Similarly, Ghai et al. and Thakur et al. reported enhanced anxiolysis and improved cooperation during induction when ketamine was used as an adjunct to midazolam, highlighting the synergistic action of the two agents.11,12
The improved behavioral scores observed in Group MK at the time of parent separation and mask acceptance further underscore the benefit of the combination therapy in reducing procedural distress. Given the developmental vulnerability and emotional dependency of children in this age group, such improvements are particularly meaningful from both clinical and parental perspectives. Oral ketamine, even at low doses, is known to produce dissociative sedation while preserving airway reflexes and spontaneous respiration—an ideal complement to midazolam's anxiolytic and amnestic properties.13
Hemodynamic monitoring revealed statistically significant elevations in heart rate at 5, 10, and 20 minutes in Group MK, consistent with ketamine’s sympathomimetic effects. However, these elevations remained within normal physiological limits and did not require intervention. These findings corroborate the data from Darlong et al., who noted transient increases in heart rate with oral ketamine administration, which were clinically insignificant.14
Oxygen saturation levels remained stable throughout the observation period, with no instances of desaturation, airway obstruction, or bradycardia in either group. This confirms the respiratory safety of both regimens when administered under close monitoring. Previous studies have similarly shown that oral midazolam and ketamine—when used in appropriate dosages—rarely cause respiratory compromise in ASA I and II pediatric populations.15
Adverse events were limited and non-serious. In Group MK, three children experienced transient nystagmus, and two were excluded due to spitting out the medication. Such effects are consistent with the pharmacodynamic profile of ketamine and have been reported in earlier trials as mild and self-limiting¹⁶. Importantly, no cases of vomiting, hallucinations, emergence delirium, or other serious adverse effects were recorded, reinforcing the safety of the studied regimen.16,17
The surgical case mix included predominantly urological and ENT procedures, which reflects a representative pediatric surgical population. The two study groups were demographically comparable, with no significant differences in baseline age, weight, or gender distribution, thereby affirming the integrity of the randomization process.18,19
5. LIMITATIONS
While the study provides valuable insights, it has certain limitations. The relatively small sample size and single-center design may affect the generalizability of the findings. Additionally, the exclusion of patients with ASA grade III or higher limits the applicability to more complex clinical scenarios. Moreover, this trial focused exclusively on intraoperative sedation and did not capture postoperative recovery variables such as pain, emergence agitation, or parent satisfaction. Future research should expand upon these dimensions and include alternative adjuncts such as dexmedetomidine or clonidine for comparative effectiveness studies.20
In conclusion, the combination of oral midazolam and ketamine offers significant clinical advantages over midazolam alone in terms of sedation depth, behavioral control, and tolerability. Its use in pediatric elective surgeries appears both effective and safe, making it a valuable option in routine anesthetic practice.
6. CONCLUSION
The combination of oral midazolam and ketamine proved to be significantly more effective than oral midazolam alone in providing early-onset, deeper sedation and improved behavioral compliance during both parental separation and anesthesia induction in children aged 1 to 6 years undergoing elective surgery. The combination regimen was well-tolerated, with minimal and self-limiting adverse effects, and no clinically significant respiratory or hemodynamic compromise. These findings suggest that oral midazolam-ketamine, when administered in appropriate doses, is a safe and superior alternative to midazolam monotherapy for pediatric premedication. Its use may enhance perioperative efficiency and patient comfort in routine pediatric anesthetic practice.
7. Data availability
The dataset presented in the study is available on request from the corresponding author during submission or after publication.
8. Conflict of Interests
The authors declare that they have no conflicts of interest regarding the publication of this manuscript.
9. Ethical issues
Ethical clearance was obtained from the Institutional Ethics Committee (Ref: VMKVMC&H/IEC/19/039), and written informed consent was obtained from the parents or legal guardians of all participating children.
10. Funding/Support
The authors declare that this research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
11. Authors contribution
KRA: Conceptualization, Literature search, Data Collection, Manuscript drafting
VKK: Study Design, Data Analysis, Manuscript Review
RB: Supervision, Critical Review, Approval of the Final Manuscript
DHS: Data Collection, Statistical Analysis, Manuscript Editing
PT: Methodology Development, Interpretation of Results, Manuscript Review
AKC: Conceptualization, Data Interpretation, Corresponding Author
11. REFERENCES
- Funk W, Jakob W, Riedl T, Taeger K. Oral preanesthetic medication for children: double-blind randomized study of a combination of midazolam and ketamine vs midazolam or ketamine alone. Br J Anaesth. 2000;84(3):335–40. PubMed DOI: 1093/oxfordjournals.bja.a013435
- Ghai B, Grandhe RP, Kumar A, Chari P. Comparative evaluation of midazolam and ketamine with midazolam alone as oral premedication. Paediatr Anaesth. 2005;15(7):554–9. PubMed DOI: 1111/j.1460-9592.2004.01523.x
- Darlong V, Shende D, Subramanyam M, Pandey R, Garg R. Low- versus high-dose combination of midazolam-ketamine for oral premedication in children. Anaesth Analg. 2004;98(4):952–7. PubMed
- Kumar A, Sharma S, Sethi A, Kumar A. Comparison of oral midazolam and midazolam-ketamine combination for sedation in pediatric dental patients. J Clin Pediatr Dent. 2010;34(4):365–70.
- Walia C, Pankaj, Prabhu M, Thomas D. Oral premedication in children: Comparison of combination of midazolam-ketamine and oral midazolam—A randomized trial. Indian J Clin Anaesth. 2018;5(2):249–54. DOI: 18231/2394-4994.2018.0046
- Khatavkar SS, Bakhshi RG. Comparative study between oral midazolam and oral ketamine as premedication in pediatric patients. Indian J Clin Anaesth. 2016;3(2):242–5. DOI: 18231/2394-4994.2018.0036
- Bakhshi RG, Khatavkar SS. A comparative study of efficacy and safety of oral ketamine and oral midazolam as premedication in pediatric patients. Indian J Clin Anaesth. 2017;4(2):193–7.
- Bagheri M, Soltani AE, Qorbani M, Sureda A, Faghihi T. Efficacy and safety of low dose oral ketamine for controlling pain and distress during intravenous cannulation in children: a double-blind, randomized, placebo-controlled trial. Korean J Pain. 2022;35(3):311–7. PubMed DOI: 3344/kjp.2022.35.3.311
- Osama NA, Mahmoud SR, Salem AS, Tawfik DS. Intranasal midazolam alone versus midazolam/ketamine combination for preoperative sedation in pediatric patients undergoing ophthalmic procedures: a randomized controlled trial. Ain-Shams J Anaesthesiol. 2022;14:12. DOI: 1186/s42077-022-00212-7
- Oliveira Filho GR, Silva C, Lima RM, Lira F, Holanda A, Garcia JB. Comparison between oral midazolam versus oral ketamine plus midazolam as preanesthetic medication in children: a randomized clinical trial. Braz J Anesthesiol. 2022;72(5):573–9. PubMed DOI: 1016/j.bjane.2022.09.003
- Banerjee B, Bose A, Pahari S, Dan AK. A comparative study of paediatric oral premedication: midazolam, ketamine and low dose combination of midazolam and ketamine. J Indian Med Assoc. 2011;109(6):386–8. PubMed
- Raja SG, Sarate GS. A comparative study of efficacy and safety of oral ketamine and oral midazolam as premedicant for paediatric cardiac catheterization. Indian J Clin Anaesth. 2018;5(3):354–60. DOI: 18231/2394-4994.2018.0067
- Lin YC, Moynihan RJ, Hackel A. A comparison of oral midazolam, oral ketamine and oral midazolam combined with ketamine as preanesthetic medication for pediatric outpatients. Anesthesiology. 1993;79:A1177.
- Singh N, Pandey RK, Saksena AK, Jaiswal JN. A comparative evaluation of oral midazolam with other sedative as premedication in pediatric dentistry. J Clin Pediatr Dent. 2002;26(2):161–4. PubMed DOI: 17796/jcpd.26.2.j714x4795474mr2p
- Koirala B, Pandey RK, Saksena AK, Kumar R, Sharma S. A comparative evaluation of newer sedatives in conscious sedation. J Clin Pediatr Dent. 2006;30:273–6. PubMed DOI: 17796/jcpd.30.4.540025283p827511
- Warner DL, Cabaret J, Velling D. Ketamine plus midazolam, a most effective paediatric oral premedicant. Paediatr Anaesth. 1995;5:293–5. PubMed DOI: 1111/j.1460-9592.1995.tb00307.x
- Hasani M. Comparison of oral premedication with combination of midazolam with ketamine vs midazolam ketamine alone in children. Tehran Univ Med J. 2002;60:423–8. FullText
- Ramakrishna R, Hemanth KJ, Sunil BV, Reddy KR, Reddy AK, Reddy RK, et al. Oral premedication in children: A randomized study of a combination of oral midazolam, ketamine with atropine vs midazolam and atropine vs ketamine and atropine. Indian J Clin Anaesth. 2018;5(3):261–5. DOI: 18231/2394-4994.2018.0048
- Singh N, Pandey RK, Saksena AK, Jaiswal JN. A comparative evaluation of oral midazolam with other sedative as premedication in pediatric dentistry. J Clin Pediatr Dent. 2002;26(2):161–4. PubMed DOI: 17796/jcpd.26.2.j714x4795474mr2p
- Koirala B, Pandey RK, Saksena AK, Kumar R, Sharma S. A comparative evaluation of newer sedatives in conscious sedation. J Clin Pediatr Dent. 2006;30:273–6. PubMed DOI: 17796/jcpd.30.4.540025283p827511