Hussein Hamdy Gomaa Seleem 1, Gehan Fouad Kamel 2, Sherif Samir Wahba 3, Mohamed Abd-Elsalam El-Gendy 4, Amr Fouad Hafez 5
Authors affiliations:
Background & objective: Peribulbar block is a widely used technique for vitreoretinal surgeries due to its safety and efficacy. Adding adjuvants to local anesthetics is known to enhance the quality of the block and prolong the duration of its effect. In this study, we compared the effectiveness of dexmedetomidine and neostigmine as adjuvants to local anesthetic in peribulbar blocks for vitreoretinal surgeries.
Methodology: This prospective, randomized, controlled, blinded study was conducted on patients undergoing vitreoretinal surgery under peribulbar block. Patients were randomized into three groups: Group D received dexmedetomidine 25 µg, Group N received neostigmine 0.5 mg, and Group C served as control group with normal saline.
Results: The duration of analgesia was significantly longer with Group D (182.4 ± 4.9 min), followed by Group N (171.7 ± 4.2 min), and Group C (175.0 ± 9.2 min) (P < 0.001). Also, the duration of motor block was significantly longer with Group D (160.0 ± 4.2 min), followed by Group N (156.3 ± 3.3 min) and Group C (151.3 ± 7.2 min) (P <.001).
Conclusion: Dexmedetomidine as an adjuvant to local anesthetic in peribulbar blocks provides a faster onset and prolonged duration of sensory and motor block compared to neostigmine.
Keywords: Peribulbar Block; Dexmedetomidine; Neostigmine; Local Anesthesia; Vitreoretinal Surgery; Regional Anesthesia
Citation: Seleem HHG, Kamel GF, Wahba SS, El-Gendy MA, Hafez AF. Comparative study between dexmedetomidine vs. neostigmine as adjuvants to local anesthetic mixture for peribulbar block for vitreoretinal surgeries. Anaesth. pain intensive care 2025;29(3):444-450. DOI: 10.35975/apic.v29i3.2756
Received: February 18, 2025; Revised: March 20, 2025; Accepted: March 23, 2025
Ophthalmic regional anesthesia is a cornerstone in eye surgeries, offering effective analgesia with minimal complications. Peribulbar anesthesia, a safer alternative to retrobulbar block, is commonly used for vitreoretinal surgeries due to its lower risk of complications such as retrobulbar haemorrhage or perforation and also more suitable for elderly people with chronic illness which is better for them than general anesthesia. It doesn’t only provide safety, but also comfort for both patient and surgeon.1-3
Vitreoretinal surgeries often have longer durations compared to anterior chamber procedures, necessitating prolonged anesthesia with minimal complications. The addition of adjuvants to local anesthetics aims to enhance the quality of the block by improving onset, duration, and postoperative analgesia.
Adjuvants also help to reduce cumulative local anesthetic dosage thereby limiting potential dose-dependent adverse effects thus increase patient and surgeon satisfaction.4,5
Dexmedetomidine, a selective α2-adrenoceptor agonist, has shown efficacy in prolonging sensory and motor block duration as an adjuvant while providing sedation and analgesia.6,7
Neostigmine, a reversible acetylcholinesterase inhibitor, has also been reported to enhance analgesic effects by increasing acetylcholine concentration at nerve endings. It also activates both nicotinic and muscarinic acetylcholine receptors at the same time, so must be combined with anticholinergic drugs (such as atropine) to avoid its muscarinic side effects (arrhythmia, increased secretion, nausea, or vomiting, etc.)
As adjuvant it enhances the quality of the block by increasing onset, duration, and helping in reduction of total local anesthetic dosage thereby limiting potential dose-dependent adverse effects.8,9
This study aims to compare the effectiveness of dexmedetomidine and neostigmine as adjuvants to a local anesthetic mixture in peribulbar blocks for vitreoretinal surgeries.
The primary outcome was to evaluate the onset and duration of both sensory and motor blocks, while the secondary outcomes were pain assessment using VAS score, vital data, safety of the block and occurrence of complications including nausea, vomiting, allergy and hematoma.
This prospective, randomized, controlled, blinded study was conducted at the ophthalmic surgery unit of Ain Shams University Hospitals from March 2023 to March 2024. The trial was approved by the Ethical Committee of the Faculty of Medicine Ain Shams University (FMASU MD 314/2022) on December 8, 2022 REC-FMASU@med.asu.edu.eg. The study was registered in ClinicalTrials.gov database under ID number NCT06501352 https://clinicaltrials.gov/study/NCT06501352
2.1. Randomization and Patient Allocation
The inclusion criteria were adult patients aged 21 to 60 years old, with axial globe length < 26 mm, ASA I and II, undergoing vitreoretinal surgery under peribulbar block. Exclusion criteria were patient refusal, bleeding disorders, allergy to the study drugs or presence of infection at injection site. A total of 81 adult patients from both genders were recruited.
2.2. Study interventions
Patients were randomly assigned into three equal groups of 27 patients by using computer-generated numbers, routine preoperative investigations were done to all patients including laboratory investigations, e.g., complete blood picture, coagulation profile, and electrocardiogram. Demographic data including age, weight, and gender were recorded. The patients were fasted for 6-8 hours preoperatively. On admission to operating room, sedation was done using 0.05 mg/kg midazolam IV. The patients were monitored during the procedure, using pulse oximetry, non-invasive blood pressure and ECG. An intravenous drip was established.
Our basic anesthetic mixture was 4 mL of bupivacaine 0.5% + 3 mL of mixture of Lidocaine 2% + hyaluronidase + 1 mL of the study drug (total 8 mL) was put in 10 mL syringe. Topical anesthetic drops - benoxinate hydrochloride 0.4%, were instilled to all patients as three drops in the eyes for two times 1 minute in-between. Sterilization was done with iodine.
The patients were divided into three groups: Group D received peribulbar block with local anesthetic mixture + 25 mcg dexmedetomidine, Group N received peribulbar block with local anesthetic mixture + 0.5 mg neostigmine, and Group C received peribulbar block with local anesthetic mixture + 1 mL normal saline (control). The block was done by using a 25G needle that was inserted transcutaneously through the inferotemporal approach at the junction between the medial two thirds and the lateral third of the lower orbital margin with the bevel facing the globe and the tip towards the floor of the orbit until the needle passed through the orbital septum, it was directed then posteriorly tangential to the floor of the orbit for 25mm length and medial epicanthus approach at which the 8 mL local anesthetic mixture was injected 4 mL in each site. Then, ocular compression was applied for 5 minutes by Honan intraocular pressure IOP reducer adjusted at 20mmHg.
Postoperatively, the patients were kept under observation at PACU for one hour to monitor vital signs (consciousness level, blood pressure, heart rate, respiratory rate) then discharged to ward and observed for returning of pain. The patients were also observed for the occurrence of any adverse effect and/or complication related to the procedure (e.g. hematoma, nausea, vomiting or reaction to the study drugs).
Assessment of sensory blockage was done by using cotton wool on cornea after injection and every 30 seconds till loss of sensation, then measuring the duration of sensory blockage by return of corneal sensation. Motor blockade (akinesia) was used as the main index of anesthesia effectiveness. It was assessed using a 12-point score (12-point akinesia score) as: 0 (no movement), 1 (flicker), 2 (full movement) for the four recti muscles, levator palpebrae and orbicularis oculi (total score 12). Successful block was defined as an ocular motility score between 0 and 6. This score was compared between the three groups 1, 2 and 5 min after injection and at the end of the surgical procedures. Post procedure pain assessment was done by visual analogue score (VAS) till 12 hours after beginning of procedure, at 2, 4, 8, and 12 hours post-operative. Rescue analgesia was given if VAS between (2and4) 1 gm of IV paracetamol. If VAS more than 4, nalbuphine was given according to body weight. Occurrence of complications like allergy, hematoma, nausea and vomiting was assessed and recorded. Also, vital data (heart rate, mean arterial blood pressure and Oxygen saturation) were assessed and recorded as a baseline reading, then every 15 minutes intra-operative till end of the procedure, VAS score was assessed at 2, 4, 8, and 12 hours post-procedure.
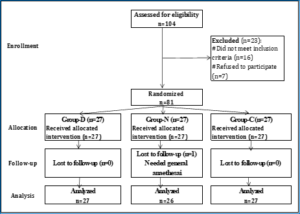
Figure 1: Study flow diagram
2.3. Sample size
Since there is no previous study comparing dexmedetomidine and neostigmine as an adjuvant to local anesthetic mixture in peribulbar block in Vitreoretinal Surgeries. A sample size of 27 cases per group -totaling 81 cases in three groups- achieve 80% power to reject the null hypothesis of zero effect size when the population effect size is 0.4 (a relatively large effect size) and the significance level (alpha) is 0.05 using one-way ANOVA to compare the means of the outcomes of the three groups. The sample was inflated by 20% to compensate for the dropouts.
2.4. Statistical analysis
The collected data were coded, tabulated, and statistically analyzed using IBM SPSS statistics (Statistical Package for Social Sciences) software version 28.0, IBM Corp., Chicago, USA, 2021. Quantitative data tested for normality using Shapiro-Wilk test, then described as mean ± SD (standard deviation) as well as minimum and maximum of the range, and then compared using ANOVA test. Qualitative data described as number and percentage and then compared using Chi square test and Fisher’s Exact test. Bonferoni test used for post hoc comparisons. The level of significance was taken at p-value ≤0.050 was significant, otherwise was non-significant.
Table (1) showed that: No statistically significant difference between the studied groups regarding age, weight, BMI, ASA grade and axial length.
No statistically significant differences were found between the studied groups regarding onset of sensory and motor block. Duration of sensory block was significantly longest in Group D with no significant difference between significant difference between Group N and Group C. Duration of motor block was longest in Group D, followed by Group N and shortest in Group C, the differences between the study group were statistically significant (Table 2).
Table 3 and Figures 2 and 3 show that there was no statistically significant differences between the studied groups regarding the heart rate and mean arterial pressure at different time periods.
Table 4 and Figure 4 show that pain scores at 4 and 8 hours was significantly lowest in Group D with no significant difference between Group N and Group C.
Need to rescue analgesia was non-significantly less frequent in Group D.
Table 5 shows that nausea and vomiting was less frequent in Group D compared to Group N and C, but the differences were not significant.
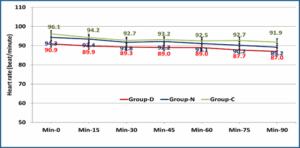
Figure 2: Comparative heart rates in the studied groups.
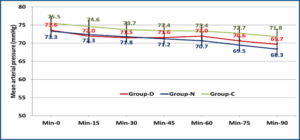
Figure 3: Comparative mean arterial pressure in the studied groups
In ophthalmic surgeries, the planned procedure and estimated time influence appropriate choice of local and regional anesthetic. In search for the optimum local anesthetic mixture, we compared dexmedetomidine 25 µg versus neostigmine 0.5 mg to standard local anesthetic mixture for peribulbar block in vitreoretinal surgeries. We assessed the effect of this mixture regarding the onset and duration of sensory and motor block.
We found that adding dexmedetomidine to local anesthetic mixture enhances ocular anesthesia and akinesia by increasing the duration of both sensory and motor block significantly with the least postoperative rescue analgesia needed compared to other groups. On the other hand, adding neostigmine to local anesthetic mixture was associated with a significantly higher intensity/ duration of the motor block while sensory block was comparable with control group.
The ability of dexmedetomidine to fasten the onset and prolong the duration of peripheral nerve block is explained to be due to its peripheral effect by activating α2 adrenoceptors in peripheral blood vessels, constricting blood vessels around the injection site, delaying the absorption of local anesthetics, and prolonging their block time.10
In terms of side effects, dexmedetomidine was associated with less nausea and vomiting than other groups.
In agreement with our study, Ahmed and his colleagues compared dexmedetomidine against neostigmine added to local anesthetics mixture in peribulbar block in cataract surgery.10 They found that dexmedetomidine was associated with significantly faster onset and insignificantly longer duration of the block. It should be noted that they used a higher dose of dexmedetomidine (50 µg) which might have influenced the performance of dexmedetomidine.
When compared to placebo, 20 µg of dexmedetomidine provided better block quality with no hemodynamic affection. It was concluded that dexmedetomidine was a useful and safe adjunct to the peribulbar block in vitreoretinal surgeries increasing the surgeons’ satisfaction by providing efficient pain relief.11
Interestingly, Subramanian and his colleagues compared 25 µg of dexmedetomidine to 50 µg when added to ropivacaine 0.75% in peribulbar block for patients undergoing vitreoretinal surgeries.12 They found that there was no statistically significant difference between either dose regarding the quality of the block and patient’s satisfaction. However, there was a statistically significant difference between the high dose and the low dose group in terms of hemodynamics; the 50-µg dose was associated with a statistically significant decrease in both HR and MAP. Perhaps, their results go with our findings regarding the good safety profile of lower dose of dexmedetomidine.
In contrast to our study, Mohamed and Genidy Mahmoud didn’t record any side effects for adding neostigmine to LA mixture in peribulbar block.13 They also found that neostigmine was associated with significantly faster onset and longer duration of sensory block than placebo.
In another study by Aboul Fetouh and his colleagues studied the effect of different doses of neostigmine (0.25 mg and 0.5 mg) when added to local anesthetic in peribulbar block for cataract surgery.14 Their research found that adding 0.5 mg of neostigmine to lidocaine 2% enhanced the onset of sensory and motor block and prolonged the duration of the block. Thus, its use produced more suitable surgical conditions for cataract surgery. Moreover, it showed effective postoperative analgesia, with prolonged time to the first analgesic dose without any side effects. Neostigmine at 0.5 mg was safe and exhibited a non-significant increase in the incidence of postoperative nausea and vomiting.
In search for better outcomes, Mohamed and his colleagues added neostigmine with ketorolac to local anesthetic mixture in peribulbar block for vitreoretinal surgeries.15 They found that the combination induced more rapid onset and longer duration of both sensory and motor blocks and lengthened the time to first analgesic requirement. Furthermore, the use of these adjuvants was associated with non-significant adverse effects, better safety, hemodynamic stability, and significant patients’ and surgeons’ satisfaction.
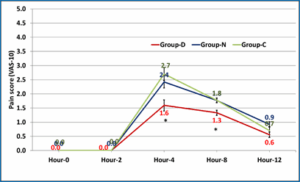
Figure 4: Comparative pain score (VAS-10) in the study groups (*significant).
Our study has some limitations. Firstly, the sample size was small. Secondly, we used a single dose of dexmedetomidine and may be, more doses would have impacted our results. Thirdly, we didn’t use anti-emitics.
Adding 25 µg of dexmedetomidine to local anesthetic mixture in peribulbar block for vitreoretinal surgeries enhances ocular anesthesia and akinesia by increasing the duration of both sensory and motor block significantly with the least postoperative rescue analgesia needed compared to neostigmine and the control groups.
Also, adding 0.5 mg of neostigmine to local anesthetic mixture enhances ocular anesthesia by increasing motor block significantly in comparison with the control group and the duration of sensory block, but insignificantly.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
HH: Conduction of the study work.
GF: Manuscript editing
SS: Statistical analysis and review
MA, AF: Literature search
Authors affiliations:
- Hussein Hamdy Gomaa Seleem, Assistant Lecturer, Department of Anesthesia, Intensive Care and Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: husseinhamdy@med.asu.edu.eg, {ORCID:0009-0000-2538-2162}
- Gehan Fouad Kamel, Professor of Anesthesia, Intensive Care and Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: Drgehan_youssif@med.asu.edu.eg
- Sherif Samir Wahba, Professor of Anesthesia, Intensive Care and Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: Sherifwahba2012@yahoo.com
- Mohamed Abd-Elsalam El-Gendy, Associate Professor of Anesthesia, Intensive Care and Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: MohammadAlgendy@med.asu.edu.eg
- Amr Fouad Hafez, Associate Professor of Anesthesia, Intensive Care and Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: Amr_icu@hotmail.com
ABSTRACT
Background & objective: Peribulbar block is a widely used technique for vitreoretinal surgeries due to its safety and efficacy. Adding adjuvants to local anesthetics is known to enhance the quality of the block and prolong the duration of its effect. In this study, we compared the effectiveness of dexmedetomidine and neostigmine as adjuvants to local anesthetic in peribulbar blocks for vitreoretinal surgeries.
Methodology: This prospective, randomized, controlled, blinded study was conducted on patients undergoing vitreoretinal surgery under peribulbar block. Patients were randomized into three groups: Group D received dexmedetomidine 25 µg, Group N received neostigmine 0.5 mg, and Group C served as control group with normal saline.
Results: The duration of analgesia was significantly longer with Group D (182.4 ± 4.9 min), followed by Group N (171.7 ± 4.2 min), and Group C (175.0 ± 9.2 min) (P < 0.001). Also, the duration of motor block was significantly longer with Group D (160.0 ± 4.2 min), followed by Group N (156.3 ± 3.3 min) and Group C (151.3 ± 7.2 min) (P <.001).
Conclusion: Dexmedetomidine as an adjuvant to local anesthetic in peribulbar blocks provides a faster onset and prolonged duration of sensory and motor block compared to neostigmine.
Keywords: Peribulbar Block; Dexmedetomidine; Neostigmine; Local Anesthesia; Vitreoretinal Surgery; Regional Anesthesia
Citation: Seleem HHG, Kamel GF, Wahba SS, El-Gendy MA, Hafez AF. Comparative study between dexmedetomidine vs. neostigmine as adjuvants to local anesthetic mixture for peribulbar block for vitreoretinal surgeries. Anaesth. pain intensive care 2025;29(3):444-450. DOI: 10.35975/apic.v29i3.2756
Received: February 18, 2025; Revised: March 20, 2025; Accepted: March 23, 2025
1. INTRODUCTION
Ophthalmic regional anesthesia is a cornerstone in eye surgeries, offering effective analgesia with minimal complications. Peribulbar anesthesia, a safer alternative to retrobulbar block, is commonly used for vitreoretinal surgeries due to its lower risk of complications such as retrobulbar haemorrhage or perforation and also more suitable for elderly people with chronic illness which is better for them than general anesthesia. It doesn’t only provide safety, but also comfort for both patient and surgeon.1-3
Vitreoretinal surgeries often have longer durations compared to anterior chamber procedures, necessitating prolonged anesthesia with minimal complications. The addition of adjuvants to local anesthetics aims to enhance the quality of the block by improving onset, duration, and postoperative analgesia.
Adjuvants also help to reduce cumulative local anesthetic dosage thereby limiting potential dose-dependent adverse effects thus increase patient and surgeon satisfaction.4,5
Dexmedetomidine, a selective α2-adrenoceptor agonist, has shown efficacy in prolonging sensory and motor block duration as an adjuvant while providing sedation and analgesia.6,7
Neostigmine, a reversible acetylcholinesterase inhibitor, has also been reported to enhance analgesic effects by increasing acetylcholine concentration at nerve endings. It also activates both nicotinic and muscarinic acetylcholine receptors at the same time, so must be combined with anticholinergic drugs (such as atropine) to avoid its muscarinic side effects (arrhythmia, increased secretion, nausea, or vomiting, etc.)
As adjuvant it enhances the quality of the block by increasing onset, duration, and helping in reduction of total local anesthetic dosage thereby limiting potential dose-dependent adverse effects.8,9
This study aims to compare the effectiveness of dexmedetomidine and neostigmine as adjuvants to a local anesthetic mixture in peribulbar blocks for vitreoretinal surgeries.
The primary outcome was to evaluate the onset and duration of both sensory and motor blocks, while the secondary outcomes were pain assessment using VAS score, vital data, safety of the block and occurrence of complications including nausea, vomiting, allergy and hematoma.
2. METHODOLOGY
This prospective, randomized, controlled, blinded study was conducted at the ophthalmic surgery unit of Ain Shams University Hospitals from March 2023 to March 2024. The trial was approved by the Ethical Committee of the Faculty of Medicine Ain Shams University (FMASU MD 314/2022) on December 8, 2022 REC-FMASU@med.asu.edu.eg. The study was registered in ClinicalTrials.gov database under ID number NCT06501352 https://clinicaltrials.gov/study/NCT06501352
2.1. Randomization and Patient Allocation
The inclusion criteria were adult patients aged 21 to 60 years old, with axial globe length < 26 mm, ASA I and II, undergoing vitreoretinal surgery under peribulbar block. Exclusion criteria were patient refusal, bleeding disorders, allergy to the study drugs or presence of infection at injection site. A total of 81 adult patients from both genders were recruited.
2.2. Study interventions
Patients were randomly assigned into three equal groups of 27 patients by using computer-generated numbers, routine preoperative investigations were done to all patients including laboratory investigations, e.g., complete blood picture, coagulation profile, and electrocardiogram. Demographic data including age, weight, and gender were recorded. The patients were fasted for 6-8 hours preoperatively. On admission to operating room, sedation was done using 0.05 mg/kg midazolam IV. The patients were monitored during the procedure, using pulse oximetry, non-invasive blood pressure and ECG. An intravenous drip was established.
Our basic anesthetic mixture was 4 mL of bupivacaine 0.5% + 3 mL of mixture of Lidocaine 2% + hyaluronidase + 1 mL of the study drug (total 8 mL) was put in 10 mL syringe. Topical anesthetic drops - benoxinate hydrochloride 0.4%, were instilled to all patients as three drops in the eyes for two times 1 minute in-between. Sterilization was done with iodine.
The patients were divided into three groups: Group D received peribulbar block with local anesthetic mixture + 25 mcg dexmedetomidine, Group N received peribulbar block with local anesthetic mixture + 0.5 mg neostigmine, and Group C received peribulbar block with local anesthetic mixture + 1 mL normal saline (control). The block was done by using a 25G needle that was inserted transcutaneously through the inferotemporal approach at the junction between the medial two thirds and the lateral third of the lower orbital margin with the bevel facing the globe and the tip towards the floor of the orbit until the needle passed through the orbital septum, it was directed then posteriorly tangential to the floor of the orbit for 25mm length and medial epicanthus approach at which the 8 mL local anesthetic mixture was injected 4 mL in each site. Then, ocular compression was applied for 5 minutes by Honan intraocular pressure IOP reducer adjusted at 20mmHg.
Postoperatively, the patients were kept under observation at PACU for one hour to monitor vital signs (consciousness level, blood pressure, heart rate, respiratory rate) then discharged to ward and observed for returning of pain. The patients were also observed for the occurrence of any adverse effect and/or complication related to the procedure (e.g. hematoma, nausea, vomiting or reaction to the study drugs).
Assessment of sensory blockage was done by using cotton wool on cornea after injection and every 30 seconds till loss of sensation, then measuring the duration of sensory blockage by return of corneal sensation. Motor blockade (akinesia) was used as the main index of anesthesia effectiveness. It was assessed using a 12-point score (12-point akinesia score) as: 0 (no movement), 1 (flicker), 2 (full movement) for the four recti muscles, levator palpebrae and orbicularis oculi (total score 12). Successful block was defined as an ocular motility score between 0 and 6. This score was compared between the three groups 1, 2 and 5 min after injection and at the end of the surgical procedures. Post procedure pain assessment was done by visual analogue score (VAS) till 12 hours after beginning of procedure, at 2, 4, 8, and 12 hours post-operative. Rescue analgesia was given if VAS between (2and4) 1 gm of IV paracetamol. If VAS more than 4, nalbuphine was given according to body weight. Occurrence of complications like allergy, hematoma, nausea and vomiting was assessed and recorded. Also, vital data (heart rate, mean arterial blood pressure and Oxygen saturation) were assessed and recorded as a baseline reading, then every 15 minutes intra-operative till end of the procedure, VAS score was assessed at 2, 4, 8, and 12 hours post-procedure.

Figure 1: Study flow diagram
2.3. Sample size
Since there is no previous study comparing dexmedetomidine and neostigmine as an adjuvant to local anesthetic mixture in peribulbar block in Vitreoretinal Surgeries. A sample size of 27 cases per group -totaling 81 cases in three groups- achieve 80% power to reject the null hypothesis of zero effect size when the population effect size is 0.4 (a relatively large effect size) and the significance level (alpha) is 0.05 using one-way ANOVA to compare the means of the outcomes of the three groups. The sample was inflated by 20% to compensate for the dropouts.
2.4. Statistical analysis
The collected data were coded, tabulated, and statistically analyzed using IBM SPSS statistics (Statistical Package for Social Sciences) software version 28.0, IBM Corp., Chicago, USA, 2021. Quantitative data tested for normality using Shapiro-Wilk test, then described as mean ± SD (standard deviation) as well as minimum and maximum of the range, and then compared using ANOVA test. Qualitative data described as number and percentage and then compared using Chi square test and Fisher’s Exact test. Bonferoni test used for post hoc comparisons. The level of significance was taken at p-value ≤0.050 was significant, otherwise was non-significant.
3. RESULTS
Table (1) showed that: No statistically significant difference between the studied groups regarding age, weight, BMI, ASA grade and axial length.
| Table 1: Demographic characteristics between the studied groups. | |||||
| Variables | Group-D
(n=27) |
Group-N
(n=26) |
Group-C
(n=27) |
^P-value | |
| Age (years) | 49.4 ± 10.7 | 46.9 ± 9.9 | 48.9 ± 8.5 | ^0.632 | |
| Gender
(n, %) |
Male | 16 (59.3%) | 15 (57.7%) | 16 (59.3%) | #0.991 |
| Female | 11 (40.7%) | 11 (42.3%) | 11 (40.7%) | ||
| Weight (kg) | 77.6 ± 7.9 | 80.2 ± 7.1 | 78.0 ± 7.1 | ^0.393 | |
| ASA
(n, %) |
I | 8 (29.6%) | 10 (38.5%) | 7 (25.9%) | #0.601 |
| II | 19 (70.4%) | 16 (61.5%) | 20 (74.1%) | ||
| Axial length (mm) | 23.5 ± 1.2 | 23.9 ± 1.2 | 23.8 ± 1.0 | ^0.358 | |
| BMI: Body Mass Index. ASA: American Association of Anesthesiologists. ^ANOVA test. #Chi square test. | |||||
| Table 2: Sensory and motor block between the studied groups | ||||
| Variables | Group-D
(n=27) |
Group-N
(n=26) |
Group-C
(n=27) |
^P-value |
| Sensory block | ||||
| Onset (min) | 1.0 ± 0.1 | 1.0 ± 0.1 | 1.1 ± 0.2 | 0.450 |
| Duration (min) | 182.4 ± 4.9a | 171.7 ± 4.2b | 175.0 ± 9.2b | < 0.001* |
| Motor block | ||||
| Onset (min) | 2.1 ± 0.2 | 2.0 ± 0.2 | 2.1 ± 0.3 | 0.167 |
| Duration (min) | 160.0 ± 4.2a | 156.3 ± 3.3b | 151.3 ± 7.2c | < 0.001* |
| Score | 0.6 ± 0.8 | 0.5 ± 0.6 | 0.7 ± 0.8 | 0.493 |
| ^ANOVA test. *Significant. Homogenous groups had the same symbol “a, b or c” based on pot hoc Bonferroni test.
Data presented as mean ± SD; P < 0.05 considered as significant |
||||
| Table 3: Heart rate and mean arterial pressure between the studied groups. | ||||
| Time | Group-D
(n=27) |
Group-N
(n=26) |
Group-C
(n=27) |
^P-value |
| Heart rate (beat/min) | ||||
| Min-0 | 73.6 ± 6.3 | 73.3 ± 7.4 | 75.5 ± 10.0 | 0.548 |
| Min-15 | 72.0 ± 6.2 | 72.3 ± 7.4 | 74.6 ± 9.8 | 0.427 |
| Min-30 | 71.5 ± 5.8 | 71.8 ± 7.1 | 73.7 ± 9.7 | 0.519 |
| Min-45 | 71.6 ± 6.0 | 71.2 ± 7.1 | 73.4 ± 10.1 | 0.546 |
| Min-60 | 72.0 ± 6.2 | 70.7 ± 7.3 | 73.4 ± 10.3 | 0.490 |
| Min-75 | 70.6 ± 6.1 | 69.5 ± 7.0 | 72.7 ± 9.9 | 0.308 |
| Min-90 | 69.7 ± 6.2 | 68.3 ± 7.1 | 71.8 ± 9.9 | 0.283 |
| Mean arterial pressure (mmHg) | ||||
| Min-0 | 90.9 ± 9.0 | 94.3 ± 9.9 | 96.1 ± 9.0 | 0.119 |
| Min-15 | 89.9 ± 9.4 | 93.4 ± 9.6 | 94.2 ± 8.9 | 0.193 |
| Min-30 | 89.3 ± 9.3 | 91.8 ± 9.7 | 92.7 ± 8.5 | 0.359 |
| Min-45 | 89.0 ± 9.2 | 92.2 ± 9.9 | 93.2 ± 8.6 | 0.229 |
| Min-60 | 89.0 ± 9.3 | 91.1 ± 9.8 | 92.5 ± 8.8 | 0.371 |
| Min-75 | 87.7 ± 9.5 | 90.2 ± 10.1 | 92.7 ± 8.8 | 0.160 |
| Min-90 | 87.0 ± 9.5 | 89.2 ± 10.1 | 91.9 ± 8.9 | 0.174 |
| ^ANOVA test. *Significant. Homogenous groups had the same symbol “a, b” based on pot hoc Bonferroni test. Data presented as mean ± SD | ||||
| Table 4: Pain score (VAS-10) and need to rescue analgesia between the studied groups. | ||||
| Time | Group-D
(n=27) |
Group-N
(n=26) |
Group-C
(n=27) |
P-value |
| Hour-0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | NA |
| Hour-2 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | NA |
| Hour-4 | 1.6 ± 1.0a | 2.4 ± 1.1b | 2.7 ± 1.2b | ^0.001* |
| Hour-8 | 1.3 ± 0.5a | 1.8 ± 0.4b | 1.8 ± 0.4b | ^< 0.001* |
| Hour-12 | 0.6 ± 0.5 | 0.9 ± 0.5 | 0.7 ± 0.8 | ^0.091 |
| Rescue analgesia | 3 (11.1) | 6 (23.1) | 9 (33.3) | #0.147 |
| Data presented as mean ± SD or n (%); NA: Not applicable. #Chi square test. ^ANOVA test. *Significant. Homogenous groups had the same symbol “a, b” based on pot hoc Bonferroni test. | ||||
Table 5 shows that nausea and vomiting was less frequent in Group D compared to Group N and C, but the differences were not significant.

Figure 2: Comparative heart rates in the studied groups.

Figure 3: Comparative mean arterial pressure in the studied groups
4. DISCUSSION
In ophthalmic surgeries, the planned procedure and estimated time influence appropriate choice of local and regional anesthetic. In search for the optimum local anesthetic mixture, we compared dexmedetomidine 25 µg versus neostigmine 0.5 mg to standard local anesthetic mixture for peribulbar block in vitreoretinal surgeries. We assessed the effect of this mixture regarding the onset and duration of sensory and motor block.
We found that adding dexmedetomidine to local anesthetic mixture enhances ocular anesthesia and akinesia by increasing the duration of both sensory and motor block significantly with the least postoperative rescue analgesia needed compared to other groups. On the other hand, adding neostigmine to local anesthetic mixture was associated with a significantly higher intensity/ duration of the motor block while sensory block was comparable with control group.
The ability of dexmedetomidine to fasten the onset and prolong the duration of peripheral nerve block is explained to be due to its peripheral effect by activating α2 adrenoceptors in peripheral blood vessels, constricting blood vessels around the injection site, delaying the absorption of local anesthetics, and prolonging their block time.10
In terms of side effects, dexmedetomidine was associated with less nausea and vomiting than other groups.
In agreement with our study, Ahmed and his colleagues compared dexmedetomidine against neostigmine added to local anesthetics mixture in peribulbar block in cataract surgery.10 They found that dexmedetomidine was associated with significantly faster onset and insignificantly longer duration of the block. It should be noted that they used a higher dose of dexmedetomidine (50 µg) which might have influenced the performance of dexmedetomidine.
When compared to placebo, 20 µg of dexmedetomidine provided better block quality with no hemodynamic affection. It was concluded that dexmedetomidine was a useful and safe adjunct to the peribulbar block in vitreoretinal surgeries increasing the surgeons’ satisfaction by providing efficient pain relief.11
Interestingly, Subramanian and his colleagues compared 25 µg of dexmedetomidine to 50 µg when added to ropivacaine 0.75% in peribulbar block for patients undergoing vitreoretinal surgeries.12 They found that there was no statistically significant difference between either dose regarding the quality of the block and patient’s satisfaction. However, there was a statistically significant difference between the high dose and the low dose group in terms of hemodynamics; the 50-µg dose was associated with a statistically significant decrease in both HR and MAP. Perhaps, their results go with our findings regarding the good safety profile of lower dose of dexmedetomidine.
In contrast to our study, Mohamed and Genidy Mahmoud didn’t record any side effects for adding neostigmine to LA mixture in peribulbar block.13 They also found that neostigmine was associated with significantly faster onset and longer duration of sensory block than placebo.
In another study by Aboul Fetouh and his colleagues studied the effect of different doses of neostigmine (0.25 mg and 0.5 mg) when added to local anesthetic in peribulbar block for cataract surgery.14 Their research found that adding 0.5 mg of neostigmine to lidocaine 2% enhanced the onset of sensory and motor block and prolonged the duration of the block. Thus, its use produced more suitable surgical conditions for cataract surgery. Moreover, it showed effective postoperative analgesia, with prolonged time to the first analgesic dose without any side effects. Neostigmine at 0.5 mg was safe and exhibited a non-significant increase in the incidence of postoperative nausea and vomiting.
In search for better outcomes, Mohamed and his colleagues added neostigmine with ketorolac to local anesthetic mixture in peribulbar block for vitreoretinal surgeries.15 They found that the combination induced more rapid onset and longer duration of both sensory and motor blocks and lengthened the time to first analgesic requirement. Furthermore, the use of these adjuvants was associated with non-significant adverse effects, better safety, hemodynamic stability, and significant patients’ and surgeons’ satisfaction.

Figure 4: Comparative pain score (VAS-10) in the study groups (*significant).
5. LIMITATIONS
Our study has some limitations. Firstly, the sample size was small. Secondly, we used a single dose of dexmedetomidine and may be, more doses would have impacted our results. Thirdly, we didn’t use anti-emitics.
6. CONCLUSION
Adding 25 µg of dexmedetomidine to local anesthetic mixture in peribulbar block for vitreoretinal surgeries enhances ocular anesthesia and akinesia by increasing the duration of both sensory and motor block significantly with the least postoperative rescue analgesia needed compared to neostigmine and the control groups.
Also, adding 0.5 mg of neostigmine to local anesthetic mixture enhances ocular anesthesia by increasing motor block significantly in comparison with the control group and the duration of sensory block, but insignificantly.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
HH: Conduction of the study work.
GF: Manuscript editing
SS: Statistical analysis and review
MA, AF: Literature search
11. REFERENCES
- Siqueira HC, De Siqueira CM, De Lima MM, Lins LT. Ultrasound-guided peribulbar block with blunt canula for cataract surgery: a review of historical case-series. Open J Ophthalmol. 2022;12(3):322–34.DOI: 4236/ojoph.2022.123029
- Fawal SM, Nofal WH, Sabek EA, Abdelaal WA. Minimum effective volume of local anesthetic in peribulbar block: does it differ with the eyeball axial length? Braz J Anesthesiol. 2021;71:635–41. PubMed DOI: 1016/j.bjane.2021.09.001
- Kalaiyarasan R, Shende D, Parthiban M, Lomi N. Comparison of peribulbar block and subtenon infiltration in pediatric enucleation for retinoblastoma. Indian J Ophthalmol. 2021;69(8):2106–10. PubMed DOI: 4103/ijo.IJO_2829_20
- Ribeiro JA, Ribeiro DS, Scott IU, Abrão J, Jorge R. Pain during pars plana vitrectomy following sub-Tenon versus peribulbar anesthesia: a randomized trial. PLoS One. 2020;15(8):e0236624. PubMed DOI: 1371/journal.pone.0236624
- Gouveia B, Ferreira L, Maia P. Anesthetic approach in ambulatory vitrectomy: peribulbar block vs. balanced general anesthesia. Anesthesiol Res Pract. 2022;2022(1):3838222. PubMed DOI: 1155/2022/3838222 DOI: 10.4103/njms.NJMS_7_20
- Nalawade SH, Nilesh K, Kumar CM, Punde PA, Patil PB. A clinical comparative study of dexmedetomidine as an adjuvant to 2% plain lignocaine and 2% lignocaine with 1:200,000 adrenaline as local anesthetic agents for surgical removal of impacted mandibular third molars. Natl J Maxillofac Surg. 2021;12(2):255–61. PubMed DOI: 4103/njms.NJMS_7_20
- Reel B, Maani CV. Dexmedetomidine. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK513303/
- Si S, Zhao X, Su F, Lu H, Zhang D, Sun L, et al. New advances in clinical application of neostigmine: no longer focusing solely on increasing skeletal muscle strength. Front Pharmacol. 2023;14:1227496. PubMed DOI: 3389/fphar.2023.1227496
- Chen Z, Liu Z, Feng C, Jin Y, Zhao X. Dexmedetomidine as an adjuvant in peripheral nerve block. Drug Des Devel Ther. 2023;17:1463–84. PubMed DOI: 2147/DDDT.S405294
- Ahmed AG, Ali MA, Kassim DY, Ibrahim MS, Hussein HA. Comparative randomized double-blind study between neostigmine and dexmedetomidine as additives to local anesthetic mixture in peribulbar anesthesia in cataract operations. Egypt J Med Res. 2023;4(3):7–23. DOI: 21608/ejmr.2022.178565.1289
- Gujral GS, Agarwal M, Gautam P, Shrivastav A, Singh S. Evaluation of the effect and safety of dexmedetomidine as an additive to local anesthesia in peribulbar block for vitreoretinal surgery. Indian J Ophthalmol. 2019;67(5):636–40. PubMed DOI: 4103/ijo.IJO_1386_18
- Subramanian S, Bapat M, Braganza S, Thirumalesh MB. Effect of adding dexmedetomidine to 0.75% ropivacaine in peribulbar block for vitreoretinal surgery. J Anaesthesiol Clin Pharmacol. 2022;38(3):458–63. PubMed DOI: 4103/joacp.JOACP_384_20
- Mohamed AZ, Genidy MM. Evaluation of the effect of neostigmine as adjuvant to local anesthetic mixture in peribulbar anesthesia. Minia J Med Res. 2014;25(2):211–9. DOI: 21608/mjmr.2024.262020.1621
- Aboul Fetouh IS, Sherif NA, Osama NA, Mohamad MK. Safety and efficacy of adding different doses of neostigmine as an adjuvant in peribulbar block for cataract surgery: a randomized controlled trial. Egypt J Anaesth. 2021;37(1):349–55. DOI: 1080/11101849.2021.1959256
- Mohamad MK, Sherif NA, Khattab RS, Osama NA, Aboul Fetouh IS. Neostigmine and ketorolac as adjuvants to local anesthetic through peribulbar block in patients undergoing vitrectomy surgeries: a randomized controlled trial. Egypt J Anaesth. 2022;38(1):550–8. DOI: 1080/11101849.2022.2127649