Mohamed Ali Mahmoud 1, Haitham Abdullah M. Osman 2, Kareem Abd Elaal Hamed 3
Authors affiliations:
Background & objective: Sepsis and septic shock are almost always associated with severe hypotension. Various vasopressors are being used to treat hypotension. Sometimes process of weaning off intravenous (IV) vasopressors becomes really problematic. We assessed the effect of midodrine administration to facilitate weaning.
Methodology: This randomized controlled trial involved 150 patients, eighteen years of age or older, both sexes, who required a vasopressor for a period exceeding 24 hours due to septic shock. Randomly, patients were assigned to one of the two groups: Group M received midodrine 10 mg orally or by the Ryle’s tube every 8 hours; Group C did not receive midodrine.
Results: The mean arterial blood pressure (MAP) of both groups did not exhibit a significant difference when IV vasopressors were initiated and were markedly increased in Group M compared to Group C when IV vasopressors were discontinued (P < 0.001). The MAP percentage change was substantially increased in Group M compared to Group C (42.33 ± 22.01 vs 31.16 ± 17.36, P < 0.001). The number of IV vasopressors used was comparable between both groups. The duration of IV norepinephrine use was notably reduced in Group M than in Group C (P = 0.003). ICU duration, hospital stay, and hospital mortality differed insignificantly between the two groups.
Conclusion: Midodrine effectively increases blood pressure and reduces IV norepinephrine use, while ICU stay, hospital stay, and the hospital mortality were comparable in both groups in septic shock patients.
Abbreviations: APACHE: Acute Physiology and Chronic Health Evaluation, FDA: Food and Drug Administration, ICU: intensive care unit, IVV: Intravenous vasopressors, LOS: length of stay, MAP: mean arterial pressure
Keywords: Midodrine; Vasopressor; Septic shock; Intensive Care Unit
Citation: Mahmoud MA, Osman HAM, Hamed KAE. Impact of using midodrine for vasopressor weaning in septic shock patients: a randomized controlled trial. Anaesth. pain intensive care 2025;29(3):520-526. DOI: 10.35975/apic.v29i3.2752
Received: October 15, 2024; Revised: February 26, 2025; Accepted: March 24, 2025
About 2-3% of patients in intensive care unit (ICU) suffer from septic shock, and It is reported that the mortality rate is as high as 50%.1 Septic shock is a prevalent cause of ICU admission, and it frequently necessitates intravenous vasopressor support.2 Intravenous vasopressors (IVV) are indispensable for shock management subsequent to adequate fluid resuscitation.3,4 In order to accomplish the blood pressure objectives, a variety of guidelines are available to facilitate the management of vasopressor therapy.5,6 Nevertheless, the persistence of vasoplegia can make the cessation of IVV challenging.7 In these instances, the ongoing requirement for IVV therapy may impede patients' discharge from the ICU by necessitating close hemodynamic monitoring, thereby extending their hospital length of stay (LOS).8
Midodrine, an oral alpha-1 agonist, was initially approved by the United States Food and Drug Administration (FDA) for the treatment of orthostatic hypotension. Nevertheless, it has been used as a medication that is not approved for use in the treatment of a wide range of other conditions, such as hypotension brought on by dialysis,9 hepatocellular carcinoma,10 neurogenic hypotension,11 and hypotension that is linked to carotid artery stenting.12 The utilization of midodrine as an adjunctive agent has been on the rise in order to wean IVV in patients with shock, thereby reducing their ICU and hospital LOS.13
Midodrine has a higher safety profile than vasopressors, as it is associated with sympathomimetic adverse effects, such as bradyarrhythmias of the vagal reflex, paraesthesia, supine hypertension, and the most prevalent.14 The use of midodrine to wean vasopressors in medical ICU patients diagnosed with sepsis is supported by limited clinical trial data. Therefore, the aim of this trial was to assess the effect of midodrine administration on the process of weaning off IV vasopressors.
This randomized controlled trial was conducted on a group of 150 patients, eighteen years of age or older, both sexes, who had septic shock and needed vasopressor for more than 24 hours. The study carried out at Al Azhar University Hospital, Assiut, Egypt from January 2024 to September 2024.
2.1. Ethical considerations
The study is submitted to the research ethics committee of the Faculty of Medicine, Al-Azhar University, Assiut, for approval before starting the study. After approval of the local medical ethical committee of faculty of medicine (Assiut) at Al Azhar University code number (AZAST/Research/13/14-DEC-2023). Informed written consent was obtained from the patient.
The criteria for exclusion were as follows: patients who passed away within 24 hours of being admitted to the ICU, moved from a different facility, received concurrent IV inotrope support, or midodrine was administered prior to an admission and patients with hypovolemic shock and known allergy to midodrine.
2.2. Randomization and blindness
Participants were randomly assigned using computer-generated randomization numbers, and the code of each patient was stored in an opaque sealed envelope. Each patient was allocated to one of two groups. according to a 1:1 allocation ratio in a parallel manner: Group M: Patients received midodrine 10 mg orally or by the Ryle every 8 hours, and Group C: Patients not received midodrine. All patients received vasopressor and other medications according to the guideline.15
The demographic variables, The Acute Physiology and Chronic Health Evaluation (APACHE) II and the source of the infection were documented. The liver and kidney function tests were conducted, as well as a complete blood count.
The mean arterial pressure (MAP) readings were documented during the initiation and discontinuation of IV vasopressor therapy.
Early administration of 4 mL/kg of IV crystalloids boluses over 20 min with continuous fluid responsiveness monitoring. Fluid can be repeated up to 30 ml/kg within the first 3 h of resuscitation until MAP > 65 mmHg, or the patient becomes a fluid non-responder (SV increase less than 10%). Norepinephrine infusion will start when the patient fails to achieve MAP ≥ 65 mmHg after fluid resuscitation.
The infusion of norepinephrine (NE) was initiated at 10 µg/min and subsequently adjusted to ensure that the MAP remained above 65 mmHg. Oral midodrine (10 mg, three times daily) was administered to the midodrine group patients in addition to NE for a maximum of five days.
The phenylephrine is used combined in patients who were concurrently administered two or more intravenous vasopressors. Broad-spectrum antimicrobials were implemented within the initial three hours of the investigation due to the potential sources of infection. The microbial culture results in the subsequent phases were consistent with the de-escalation of the antimicrobials in accordance with the patient's clinical status.
The primary outcome was MAP when IV vasopressors discontinued. The secondary outcomes included the percentage change of MAP, the number of IV vasopressors used, duration of IV norepinephrine, ICU stays, hospital stays and hospital mortality.
2.3. Sample size
G*Power 3.1.9.2 (Universitat Kiel, Germany) was employed to calculate the sample size. A pilot study was conducted, with five cases in each group, the mean (± SD) of MAP when IV vasopressors discontinued (the primary outcome) was 74.2 ± 5.35 in Group M and 71.8 ± 4.7 mmHg in Group C. The following parameters were used to determine the sample size: The study had a group ratio of 1:1, an effect size of 0.476, a 95% confidence limit, 80% power, and In order to mitigate attrition, each cohort was supplemented with four cases. Consequently, we recruited 75 patients for each group.
2.4. Statistical analysis
SPSS v26 (IBM Inc., Chicago, IL, USA) was employed to conduct the statistical analysis. The quantitative variables were compared and contrasted between the two groups using an unpaired Student's t-test and were presented as mean and standard deviation. The qualitative variables were analyzed using the Chi-square test or Fisher's exact test and were presented as a percentage and frequency. Statistical significance was defined as a two-tailed P value that was less than 0.05.
In this trial, eligibility assessments were conducted for 176 patients. Seventeen patients did not satisfy the criteria, and nine patients declined to participate. Randomly, the patients that remained were divided into two equal groups, each consisting of 75 patients. The statistical analysis and follow-up of all allocated patients were conducted (Figure 1).
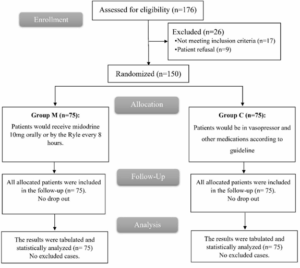
Figure 1: CONSORT flowchart of the enrolled patients
The two groups did not exhibit any significant differences in patients’ demographic characteristics or the source of infection (Table 1).
There were no significant differences in laboratory investigation results regarding hemoglobin, albumin, AST, ALT, serum creatinine, or serum urea in the groups as shown in Table 2.
The two groups did not exhibit a substantial difference in MAP when IV vasopressors were initiated. When IV vasopressors were discontinued, Group M exhibited a significant increase in MAP compared to Group C (P < 0.001). Group M demonstrated a significantly increased percentage change of MAP than Group C (P < 0.001) as shown in Table 3.
Significant differences between the two groups were not observed in the IV vasopressors used, dopamine, or epinephrine. Norepinephrine was used in all patients in both groups. In comparison to Group C, the duration of IV norepinephrine use was significantly shorter in Group M (P = 0.003). Phenylephrine and vasopressin weren’t used in any patient in both groups.
The mean duration of midodrine was 3.69 ± 1.46 days (Table 4).
The duration of ICU stay and total hospital stay were comparable in both groups; as was the hospital mortality (Table 5).
Midodrine has become increasingly popular in recent years as an adjunct to the cessation of IV vasopressors, with the intention of reducing the risk of severe adverse effects that are associated with vasopressor medication. It induces a predictable, dose-dependent rise in blood pressure by decreasing LOS and increasing systemic vascular resistance.14 The utilization of midodrine is prevalent due to its cost-effectiveness, superior results, and cost-saving potential in the context of budget impact analysis.16
In our result, Group M exhibited a significantly higher MAP than Group C when IV vasopressors were discontinued. The percentage change of MAP notably higher in Group M compared to Group C. Congruent with our conclusion, Poveromo et al. reported that midodrine's capacity to elevate the MAP and improve hemodynamic stability may enable an earlier discharge from the ICU.17 Moreover, Wright et al. reported that standing systolic blood pressure (SBP) was substantially elevated by midodrine.18 Compared to the control group, there was a substantial linear
relationship between the mean SBP and the midodrine dosage in neurogenic orthostatic hypotension.
In contrast, Levine et al. found that the MAP was insignificantly different between before midodrine and following midodrine administration.19 The reason for this discrepancy is that their study included ICU patients for a variety of reasons while our investigation concentrated on patients who were admitted to ICU due to septic shock. Moreover, Poveromo et al. showed that the MAP of the midodrine and control groups was not significantly different when IV vasopressors were discontinued.17
According to our results, there was no statistically significant difference between the two categories. With respect to the duration of hospital stays, hospital mortality, and the duration of ICU stays.
These results were consistent with Santer et al., who found that in patients with persistent hypotension in the ICU, there was negligible distinction in the duration of hospital stay, ICU stay, or time in the midodrine and placebo groups.8 furthermore, Ellena et al. found that patients in the midodrine group were notably shorter in ICU stays and hospital LOS compared to the vasopressor-only group.14 This difference may be due to different doses of midodrine. Also, Poveromo et al. demonstrated that the midodrine group had a significantly lengthier hospital LOS than the control group, and that the midodrine group had a lower hospital mortality rate.17 This difference may be due to their study, which included patients admitted to ICU due to several causes and different vasopressors used.
In the current study, the number of IV vasopressors used insignificantly differed between groups. The duration of IV norepinephrine was markedly reduced in Group M than in Group C. This is supported by Saad et al., in septic shock patients, who reported that the midodrine group experienced a significantly shortened duration of IV norepinephrine than the control group.20 Moreover, Poveromo et al. found that compared to the previous study, which lasted three days, the extent of IV vasopressor use before midodrine initiation was reduced to 1.6 days in their study.17 On the other hand, Santer et al. revealed that between the midodrine and placebo groups, there was no discernible difference in the latency to vasopressor discontinuation.8 This difference can be attributed to different midodrine doses used (20 mg); our study was conducted on septic shock patients admitted to the ICU, but their study included persistent hypotension patients admitted to the ICU. Also, Ellena et al. revealed that the duration of vasopressor therapy in the midodrine group was slightly differ compared to vasopressor-only group.14
Our trial had some limitations; the research was conducted at a single centre with a small sample size; the patients were not monitored following their discharge from the ICU, and a fixed midodrine drug dosage regimen was chosen.
Midodrine effectively increases blood pressure and reduces IV norepinephrine duration while ICU stays, hospital stays, and hospital mortality were comparable in both groups in septic shock patients.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
MAM: Conceptualization
HAMO: Data collection
KAEH: Data analysis
All authors approved this manuscript for submission.
Authors affiliations:
- Mohamed Ali Mahmoud, Lecturer, Department of Anesthesiology, Intensive Care & Pain Medicine, Faculty of Medicine, Al Azhar University, Assiut, Egypt; Email: mohamedali.226@azhar.edu.eg; {ORCID: 0009-0009-1282-5628}
- Haitham Abdullah M. Osman, Lecturer, Department of Anesthesiology, Intensive Care & Pain Medicine, Faculty of Medicine, Al Azhar University, Assiut, Egypt; Email: haithamosman.4423@azhar.edu.eg; {ORCID:0009-0000-8050-8565}
- Kareem Abd Elaal Hamed, Lecturer, Department of Anesthesiology, Intensive Care & Pain Medicine, Faculty of Medicine, Al Azhar University, Assiut, Egypt; Email: KareemAbdElal.2344@azhar.edu.eg
ABSTRACT
Background & objective: Sepsis and septic shock are almost always associated with severe hypotension. Various vasopressors are being used to treat hypotension. Sometimes process of weaning off intravenous (IV) vasopressors becomes really problematic. We assessed the effect of midodrine administration to facilitate weaning.
Methodology: This randomized controlled trial involved 150 patients, eighteen years of age or older, both sexes, who required a vasopressor for a period exceeding 24 hours due to septic shock. Randomly, patients were assigned to one of the two groups: Group M received midodrine 10 mg orally or by the Ryle’s tube every 8 hours; Group C did not receive midodrine.
Results: The mean arterial blood pressure (MAP) of both groups did not exhibit a significant difference when IV vasopressors were initiated and were markedly increased in Group M compared to Group C when IV vasopressors were discontinued (P < 0.001). The MAP percentage change was substantially increased in Group M compared to Group C (42.33 ± 22.01 vs 31.16 ± 17.36, P < 0.001). The number of IV vasopressors used was comparable between both groups. The duration of IV norepinephrine use was notably reduced in Group M than in Group C (P = 0.003). ICU duration, hospital stay, and hospital mortality differed insignificantly between the two groups.
Conclusion: Midodrine effectively increases blood pressure and reduces IV norepinephrine use, while ICU stay, hospital stay, and the hospital mortality were comparable in both groups in septic shock patients.
Abbreviations: APACHE: Acute Physiology and Chronic Health Evaluation, FDA: Food and Drug Administration, ICU: intensive care unit, IVV: Intravenous vasopressors, LOS: length of stay, MAP: mean arterial pressure
Keywords: Midodrine; Vasopressor; Septic shock; Intensive Care Unit
Citation: Mahmoud MA, Osman HAM, Hamed KAE. Impact of using midodrine for vasopressor weaning in septic shock patients: a randomized controlled trial. Anaesth. pain intensive care 2025;29(3):520-526. DOI: 10.35975/apic.v29i3.2752
Received: October 15, 2024; Revised: February 26, 2025; Accepted: March 24, 2025
1. INTRODUCTION
About 2-3% of patients in intensive care unit (ICU) suffer from septic shock, and It is reported that the mortality rate is as high as 50%.1 Septic shock is a prevalent cause of ICU admission, and it frequently necessitates intravenous vasopressor support.2 Intravenous vasopressors (IVV) are indispensable for shock management subsequent to adequate fluid resuscitation.3,4 In order to accomplish the blood pressure objectives, a variety of guidelines are available to facilitate the management of vasopressor therapy.5,6 Nevertheless, the persistence of vasoplegia can make the cessation of IVV challenging.7 In these instances, the ongoing requirement for IVV therapy may impede patients' discharge from the ICU by necessitating close hemodynamic monitoring, thereby extending their hospital length of stay (LOS).8
Midodrine, an oral alpha-1 agonist, was initially approved by the United States Food and Drug Administration (FDA) for the treatment of orthostatic hypotension. Nevertheless, it has been used as a medication that is not approved for use in the treatment of a wide range of other conditions, such as hypotension brought on by dialysis,9 hepatocellular carcinoma,10 neurogenic hypotension,11 and hypotension that is linked to carotid artery stenting.12 The utilization of midodrine as an adjunctive agent has been on the rise in order to wean IVV in patients with shock, thereby reducing their ICU and hospital LOS.13
Midodrine has a higher safety profile than vasopressors, as it is associated with sympathomimetic adverse effects, such as bradyarrhythmias of the vagal reflex, paraesthesia, supine hypertension, and the most prevalent.14 The use of midodrine to wean vasopressors in medical ICU patients diagnosed with sepsis is supported by limited clinical trial data. Therefore, the aim of this trial was to assess the effect of midodrine administration on the process of weaning off IV vasopressors.
2. METHODOLOGY
This randomized controlled trial was conducted on a group of 150 patients, eighteen years of age or older, both sexes, who had septic shock and needed vasopressor for more than 24 hours. The study carried out at Al Azhar University Hospital, Assiut, Egypt from January 2024 to September 2024.
2.1. Ethical considerations
The study is submitted to the research ethics committee of the Faculty of Medicine, Al-Azhar University, Assiut, for approval before starting the study. After approval of the local medical ethical committee of faculty of medicine (Assiut) at Al Azhar University code number (AZAST/Research/13/14-DEC-2023). Informed written consent was obtained from the patient.
The criteria for exclusion were as follows: patients who passed away within 24 hours of being admitted to the ICU, moved from a different facility, received concurrent IV inotrope support, or midodrine was administered prior to an admission and patients with hypovolemic shock and known allergy to midodrine.
2.2. Randomization and blindness
Participants were randomly assigned using computer-generated randomization numbers, and the code of each patient was stored in an opaque sealed envelope. Each patient was allocated to one of two groups. according to a 1:1 allocation ratio in a parallel manner: Group M: Patients received midodrine 10 mg orally or by the Ryle every 8 hours, and Group C: Patients not received midodrine. All patients received vasopressor and other medications according to the guideline.15
The demographic variables, The Acute Physiology and Chronic Health Evaluation (APACHE) II and the source of the infection were documented. The liver and kidney function tests were conducted, as well as a complete blood count.
The mean arterial pressure (MAP) readings were documented during the initiation and discontinuation of IV vasopressor therapy.
Early administration of 4 mL/kg of IV crystalloids boluses over 20 min with continuous fluid responsiveness monitoring. Fluid can be repeated up to 30 ml/kg within the first 3 h of resuscitation until MAP > 65 mmHg, or the patient becomes a fluid non-responder (SV increase less than 10%). Norepinephrine infusion will start when the patient fails to achieve MAP ≥ 65 mmHg after fluid resuscitation.
The infusion of norepinephrine (NE) was initiated at 10 µg/min and subsequently adjusted to ensure that the MAP remained above 65 mmHg. Oral midodrine (10 mg, three times daily) was administered to the midodrine group patients in addition to NE for a maximum of five days.
The phenylephrine is used combined in patients who were concurrently administered two or more intravenous vasopressors. Broad-spectrum antimicrobials were implemented within the initial three hours of the investigation due to the potential sources of infection. The microbial culture results in the subsequent phases were consistent with the de-escalation of the antimicrobials in accordance with the patient's clinical status.
The primary outcome was MAP when IV vasopressors discontinued. The secondary outcomes included the percentage change of MAP, the number of IV vasopressors used, duration of IV norepinephrine, ICU stays, hospital stays and hospital mortality.
2.3. Sample size
G*Power 3.1.9.2 (Universitat Kiel, Germany) was employed to calculate the sample size. A pilot study was conducted, with five cases in each group, the mean (± SD) of MAP when IV vasopressors discontinued (the primary outcome) was 74.2 ± 5.35 in Group M and 71.8 ± 4.7 mmHg in Group C. The following parameters were used to determine the sample size: The study had a group ratio of 1:1, an effect size of 0.476, a 95% confidence limit, 80% power, and In order to mitigate attrition, each cohort was supplemented with four cases. Consequently, we recruited 75 patients for each group.
2.4. Statistical analysis
SPSS v26 (IBM Inc., Chicago, IL, USA) was employed to conduct the statistical analysis. The quantitative variables were compared and contrasted between the two groups using an unpaired Student's t-test and were presented as mean and standard deviation. The qualitative variables were analyzed using the Chi-square test or Fisher's exact test and were presented as a percentage and frequency. Statistical significance was defined as a two-tailed P value that was less than 0.05.
3. RESULTS
In this trial, eligibility assessments were conducted for 176 patients. Seventeen patients did not satisfy the criteria, and nine patients declined to participate. Randomly, the patients that remained were divided into two equal groups, each consisting of 75 patients. The statistical analysis and follow-up of all allocated patients were conducted (Figure 1).

Figure 1: CONSORT flowchart of the enrolled patients
The two groups did not exhibit any significant differences in patients’ demographic characteristics or the source of infection (Table 1).
| Table 1: Patient characteristics and source of infection of the studied groups | ||||
| Variables | Group M
(n = 75) |
Group C
(n = 75) |
P-value | |
| Age (years) | 62.16 ± 12.4 | 61.27 ± 12.32 | 0.659 | |
| Sex | Male | 48 (64) | 52 (69.33) | 0.603 |
| Female | 27 (36) | 23 (30.67) | ||
| Weight (kg) | 85.47 ± 11.11 | 89.32 ± 13.98 | 0.068 | |
| APACHE II | 20.53 ± 3.55 | 19.88 ± 4.35 | 0.315 | |
| Source of infection | Pneumonia | 38 (50.67) | 36 (48) | 0.955 |
| UTI | 20 (26.67) | 17 (22.67) | ||
| Peritonitis | 12 (16) | 9 (12) | ||
| Others | 31 (41.33) | 30 (40) | ||
| Data given as mean ± SD or frequency (%). APACHE: Acute physiology and chronic health evaluation. UTI: Urinary tract infection. | ||||
There were no significant differences in laboratory investigation results regarding hemoglobin, albumin, AST, ALT, serum creatinine, or serum urea in the groups as shown in Table 2.
| Table 2: The laboratory parameters of the studied groups | |||
| Parameter | Group M (n = 75) |
Group C (n = 75) |
P value |
| Hemoglobin (g/dl) | 10.38 ± 1.95 | 10.05 ± 1.81 | 0.292 |
| Albumin (g/dl) | 2.95 ± 0.65 | 3.12 ± 0.5 | 0.087 |
| AST (units/L) | 50(26−94.5) | 54(39.5−89) | 0.349 |
| ALT (units/L) | 38(22−58.5) | 37(25−57.5) | 0.458 |
| Serum creatinine (mg/dl) | 1.7(0.9−2.85) | 1.8(1.5−2.3) | 0.148 |
| Serum urea (mg/dl) | 40.64 ± 24.98 | 33.96 ± 18.44 | 0.064 |
| Data are shown as mean ± SD and median (IQR). AST: Aspartate aminotransferase, ALT: Alanine transaminase. | |||
The two groups did not exhibit a substantial difference in MAP when IV vasopressors were initiated. When IV vasopressors were discontinued, Group M exhibited a significant increase in MAP compared to Group C (P < 0.001). Group M demonstrated a significantly increased percentage change of MAP than Group C (P < 0.001) as shown in Table 3.
| Table 3: Mean arterial pressure when IV vasopressors were initiated and discontinued in the studied groups | ||||
| Mean arterial pressure | Group M
(n = 75) |
Group C
(n = 75) |
P−value | |
| When IV vasopressors are initiated (mmHg) | 52.77 ± 5.9 | 54.39 ± 6.56 | 0.115 | |
| When IV vasopressors discontinued (mmHg) | 73.11 ± 4.33 | 70.29 ± 3.3 | < 0.001 | |
| Change (%) | 42.33 ± 22.01 | 31.16 ± 17.36 | < 0.001 | |
| Data are shown as mean ± SD; P < 0.05 considered as significant. | ||||
Significant differences between the two groups were not observed in the IV vasopressors used, dopamine, or epinephrine. Norepinephrine was used in all patients in both groups. In comparison to Group C, the duration of IV norepinephrine use was significantly shorter in Group M (P = 0.003). Phenylephrine and vasopressin weren’t used in any patient in both groups.
The mean duration of midodrine was 3.69 ± 1.46 days (Table 4).
| Table 4: Vasopressors used, duration of midodrine and IV norepinephrine of the studied groups | ||||
| Vasopressors | Group M(n = 75) | Group C(n = 75) | P value | |
| Norepinephrine | 75 (100) | 75 (100) | --- | |
| Dopamine | 12 (16) | 12 (16) | 1 | |
| Epinephrine | 9 (12) | 7 (9.33) | 0.792 | |
| Phenylephrine | 0 (0) | 0 (0) | --- | |
| Vasopressin | 0 (0) | 0 (0) | --- | |
| Number of IV vasopressors used | One | 51 (68) | 57 (76) | 0.168 |
| Two | 21 (28) | 18 (24) | ||
| Duration of midodrine (days) | 3.69 ± 1.46 | --- | --- | |
| Duration of IV norepinephrine (hrs) | 71.23 ± 32.15 | 88.12 ± 36.14 | 0.003 | |
| Data are presented as mean ± SD or frequency (%); P < 0.05 considered as significant. | ||||
The duration of ICU stay and total hospital stay were comparable in both groups; as was the hospital mortality (Table 5).
| Table 5: ICU duration, hospital stay and mortality of the studied groups | |||
| Variable | Group M
(n = 75) |
Group C
(n = 75) |
P−value |
| ICU stays (days) | 5.71 ± 1.92 | 5.79 ± 2.02 | 0.804 |
| Hospital stays (days) | 9.56 ± 3.31 | 9.47 ± 3.68 | 0.871 |
| Hospital mortality | 21 (28%) | 17 (22.67%) | 0.573 |
| Data are presented as mean ± SD or frequency (%); P < 0.05 considered as significant; ICU: intensive care unit. | |||
4. DISCUSSION
Midodrine has become increasingly popular in recent years as an adjunct to the cessation of IV vasopressors, with the intention of reducing the risk of severe adverse effects that are associated with vasopressor medication. It induces a predictable, dose-dependent rise in blood pressure by decreasing LOS and increasing systemic vascular resistance.14 The utilization of midodrine is prevalent due to its cost-effectiveness, superior results, and cost-saving potential in the context of budget impact analysis.16
In our result, Group M exhibited a significantly higher MAP than Group C when IV vasopressors were discontinued. The percentage change of MAP notably higher in Group M compared to Group C. Congruent with our conclusion, Poveromo et al. reported that midodrine's capacity to elevate the MAP and improve hemodynamic stability may enable an earlier discharge from the ICU.17 Moreover, Wright et al. reported that standing systolic blood pressure (SBP) was substantially elevated by midodrine.18 Compared to the control group, there was a substantial linear
relationship between the mean SBP and the midodrine dosage in neurogenic orthostatic hypotension.
In contrast, Levine et al. found that the MAP was insignificantly different between before midodrine and following midodrine administration.19 The reason for this discrepancy is that their study included ICU patients for a variety of reasons while our investigation concentrated on patients who were admitted to ICU due to septic shock. Moreover, Poveromo et al. showed that the MAP of the midodrine and control groups was not significantly different when IV vasopressors were discontinued.17
According to our results, there was no statistically significant difference between the two categories. With respect to the duration of hospital stays, hospital mortality, and the duration of ICU stays.
These results were consistent with Santer et al., who found that in patients with persistent hypotension in the ICU, there was negligible distinction in the duration of hospital stay, ICU stay, or time in the midodrine and placebo groups.8 furthermore, Ellena et al. found that patients in the midodrine group were notably shorter in ICU stays and hospital LOS compared to the vasopressor-only group.14 This difference may be due to different doses of midodrine. Also, Poveromo et al. demonstrated that the midodrine group had a significantly lengthier hospital LOS than the control group, and that the midodrine group had a lower hospital mortality rate.17 This difference may be due to their study, which included patients admitted to ICU due to several causes and different vasopressors used.
In the current study, the number of IV vasopressors used insignificantly differed between groups. The duration of IV norepinephrine was markedly reduced in Group M than in Group C. This is supported by Saad et al., in septic shock patients, who reported that the midodrine group experienced a significantly shortened duration of IV norepinephrine than the control group.20 Moreover, Poveromo et al. found that compared to the previous study, which lasted three days, the extent of IV vasopressor use before midodrine initiation was reduced to 1.6 days in their study.17 On the other hand, Santer et al. revealed that between the midodrine and placebo groups, there was no discernible difference in the latency to vasopressor discontinuation.8 This difference can be attributed to different midodrine doses used (20 mg); our study was conducted on septic shock patients admitted to the ICU, but their study included persistent hypotension patients admitted to the ICU. Also, Ellena et al. revealed that the duration of vasopressor therapy in the midodrine group was slightly differ compared to vasopressor-only group.14
5. LIMITATIONS
Our trial had some limitations; the research was conducted at a single centre with a small sample size; the patients were not monitored following their discharge from the ICU, and a fixed midodrine drug dosage regimen was chosen.
6. CONCLUSIONS
Midodrine effectively increases blood pressure and reduces IV norepinephrine duration while ICU stays, hospital stays, and hospital mortality were comparable in both groups in septic shock patients.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
MAM: Conceptualization
HAMO: Data collection
KAEH: Data analysis
All authors approved this manuscript for submission.
11. REFERENCES
- Schoenberg MH, Weiss M, Radermacher P. Outcome of patients with sepsis and septic shock after ICU treatment. Langenbecks Arch Surg. 1998;383:44-8. PubMed DOI: 1007/s004230050090
- Kamaleldin M, Kilcommons S, Opgenorth D, Fiest K, Karvellas CJ, Kutsogiannis J, et al. Midodrine therapy for vasopressor dependent shock in the intensive care unit: A protocol for a systematic review and meta-analysis. BMJ Open. 2022;12:640-60. PubMed DOI: 1136/bmjopen-2022-064060
- Müllner M, Urbanek B, Havel C, Losert H, Waechter F, Gamper G. Vasopressors for shock. Cochrane Database Syst Rev. 2004:37-49. PubMed DOI: 1002/14651858.CD003709.pub2
- Herget-Rosenthal S, Saner F, Chawla LS. Approach to hemodynamic shock and vasopressors. Clin J Am Soc Nephrol. 2008;3:546-53. PubMed DOI: 2215/CJN.01820407
- Rossaint R, Bouillon B, Cerny V, Coats TJ, Duranteau J, Fernández-Mondéjar E, et al. The European guideline on management of major bleeding and coagulopathy following trauma: fourth edition. Crit Care. 2016;20:100-24. PubMed DOI: 1186/s13054-016-1265-x
- Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med. 2017;43:304-77. PubMed DOI: 1007/s00134-017-4683-6
- Levy B, Fritz C, Tahon E, Jacquot A, Auchet T, Kimmoun A. Vasoplegia treatments: The past, the present, and the future. Crit Care. 2018;22:52-61. PubMed DOI: 1186/s13054-018-1967-3
- Santer P, Anstey MH, Patrocínio MD, Wibrow B, Teja B, Shay D, et al. Effect of midodrine versus placebo on time to vasopressor discontinuation in patients with persistent hypotension in the intensive care unit (MIDAS): an international randomised clinical trial. Intensive Care Med. 2020;46:1884-93. PubMed DOI: 1007/s00134-020-06216-x
- Prakash S, Garg AX, Heidenheim AP, House AA. Midodrine appears to be safe and effective for dialysis-induced hypotension: a systematic review. Nephrol Dial Transplant. 2004;19:2553-8. PubMed DOI: 1093/ndt/gfh420
- Singh V, Dhungana SP, Singh B, Vijayverghia R, Nain CK, Sharma N, et al. Midodrine in patients with cirrhosis and refractory or recurrent ascites: a randomized pilot study. J Hepatol. 2012;56:348-54. PubMed DOI: 1016/j.jhep.2011.04.027
- Low PA, Gilden JL, Freeman R, Sheng KN, McElligott MA. Efficacy of midodrine vs placebo in neurogenic orthostatic hypotension. A randomized, double-blind multicenter study. Midodrine Study Group. JAMA. 1997;277:1046-51. PubMed
- Sharma S, Lardizabal JA, Bhambi B. Oral midodrine is effective for the treatment of hypotension associated with carotid artery stenting. J Cardiovasc Pharmacol Ther. 2008;13:94-7. PubMed DOI: 1177/1074248408317709
- Rizvi MS, Trivedi V, Nasim F, Lin E, Kashyap R, Andrijasevic N, et al. Trends in use of midodrine in the icu: A single-center retrospective case series. Crit Care Med. 2018;46:628-33. PubMed DOI: 1097/CCM.0000000000003121
- Ellena KJ, Combs JL, Draper HM, Belfer JJ. Role of midodrine on vasopressor duration in patients with sepsis. J Shock Hemodyn. 2023;2:202-13. DOI: 58464/2836-0605.1041
- Levine AR, Meyer MJ, Bittner EA, Berg S, Kalman R, Stanislaus AB, et al. Oral midodrine treatment accelerates the liberation of intensive care unit patients from intravenous vasopressor infusions. J Crit Care. 2013;28:756-62. PubMed DOI: 1016/j.jcrc.2013.05.021
- Adly DHE, Bazan NS, El Borolossy RM, Anan IF, Fakher MA, El Wakeel LM. Midodrine improves clinical and economic outcomes in patients with septic shock: a randomized controlled clinical trial. Ir J Med Sci. 2022;191:2785-95. PubMed DOI: 1007/s11845-021-02903-w
- Poveromo L, Michalets E, Sutherland S. Midodrine for the weaning of vasopressor infusions. J Clin Pharm Ther. 2016;41:260-5. PubMed DOI: 1111/jcpt.12375
- Wright R, Kaufmann H, Perera R, Opfer-Gehrking T, McElligott M, Sheng K, et al. A double-blind, dose-response study of midodrine in neurogenic orthostatic hypotension. Neurology. 1998;51:120-4. PubMed https://pubmed.ncbi.nlm.nih.gov/9674789/
- Levine AR, Meyer MJ, Bittner EA, Berg S, Kalman R, Stanislaus AB, et al. Oral midodrine treatment accelerates the liberation of intensive care unit patients from intravenous vasopressor infusions. J Crit Care. 2013;28:756-62. PubMed DOI: 1016/j.jcrc.2013.05.021
- A Elmorsy M, I Saad S, MS El Wahdan M. Oral midodrine use in weaning of intravenous vasopressor infusions in septic shock patients. Benha J Appl Sci. 2024;9:159-66. DOI: 21608/bjas.2024.280053.1378