Huda F. Ghazaly1, Mahmoud M. Khalefa2, Mohamed A. Sedeek3, Mohamed K. Hasanen4
Author affiliations:
Background & objective: The benefits of sphenopalatine ganglion (SPG) block during endoscopic septoplasty are yet unclear due to a lack of adequate data. We investigated the efficacy of SPG block (SPGB) with lidocaine versus bupivacaine for minimizing intraoperative bleeding and providing a bloodless surgical field in patients having endoscopic septoplasty under general anesthesia.
Methodology: In this double-blind, randomized controlled trial, 60 patients with ASA I and II were scheduled for endoscopic septoplasty. Following standard general anesthesia, patients were randomly assigned to one of three groups. Group C (n = 20): SPGB was performed using normal saline in the control group; Group L (n = 20); patients received SPGB with 2% lidocaine, and Group B (n = 20); patients received SPGB with 0.5% bupivacaine. Intraoperative blood loss, surgical field quality, postoperative pain-free duration, postoperative analgesic requirements, and the patient satisfaction were recorded. The collected data were verified, coded by the researcher, and analyzed using the Statistical Package for Social Sciences (IBM-SPSS/PC/VER 21). Statistical analysis was done with the Shapiro-Wilk test, one-way ANOVA test and two-way repeated measure ANOVA test as required. The Kaplan–Meier curve was plotted to explore the differences in the post-operative pain-free durations among the studied groups using the log-rank test. A p-value less than 0.05 was considered significant.
Results: The intraoperative blood loss and surgical field grading were lower in the groups treated with lidocaine (44.9 ± 3.7 ml, P < 0.001) or bupivacaine (41.4 ± 2.9 ml, P < 0.001) in comparison to the control group (89.95 ± 7.8 ml). There was no difference between the two groups. Postoperative meperidine requirement in 24 h was higher in the control group (36.25 ± 2.9 mg) than the lidocaine group (18.75 ± 2.5 mg; P < 0.001) and the bupivacaine group (16.25 ± 2.7 mg; P < 0.001).
Conclusion: Sphenopalatine ganglion blockade with bupivacaine or lidocaine decreased intraoperative blood loss and improved surgical field visibility compared to placebo during endoscopic septoplasty under general anesthesia. There were statistically no significant differences in sphenopalatine ganglion blocks with either lidocaine or bupivacaine.
Abbreviations: SPG- Sphenopalatine Ganglion; SPGB- Sphenopalatine Ganglion Block; VAS- Visual Analog Scale
Keywords: Bupivacaine; Endoscopic Septoplasty; Lidocaine; Postoperative Analgesia; Sphenopalatine Ganglion Block
Citation: Ghazaly HF, Khalefa MM, Sedeek MA, Hasanen MK. Sphenopalatine ganglion block with lidocaine versus bupivacaine for optimizing intraoperative bleeding and surgical field visibility during endoscopic septoplasty: a randomized double-blind controlled trial. Anaesth. pain intensive care 2024;28(1):92-99; DOI: 10.35975/apic.v28i1.2393
Received: August 29, 2023; Reviewed: December 29, 2023; Accepted: December 31, 2023
Nasal septum surgeries are among the most common operations performed by otolaryngologists worldwide.1-2 Lanza and colleagues were the first to describe the use of endoscopic techniques to improve visualization during septal surgery.3 However, the new technique also posed several challenges to the anesthesiologists during endoscopic septoplasty, including maintaining hemodynamic stability, providing a bloodless surgical field, and producing satisfactory perioperative analgesic requirements. The sphenopalatine ganglion (SPG) is located in the pterygopalatine fossa, immediately behind the middle turbinate. It has somatosensory effects via the trigeminal and facial nerves, visceral motor parasympathetic activity via the superficial petrosal nerve, and sympathetic roles via its connection to the cervical sympathetic chain via the deep petrosal nerve.4 Thus, sphenopalatine ganglion block (SPGB) promotes mucosal vasoconstriction and reduces mucosal blood flow in the nasal cavity, resulting in a clear surgical field during nasal surgery. Moreover, SPGB anesthetizes the posterior septum, middle turbinate, sphenoid sinus, and posterior ethmoid cavity, and hence potentially prolonging postoperative analgesia.5 In previous clinical studies, the combination of SPGB with general anesthesia has been reported to reduce intraoperative blood loss and postoperative pain in patients undergoing functional endoscopic sinus surgery (FESS).6,7 However, there is a lack of evidence for the benefit of SPGB during endoscopic septoplasty.8 Furthermore, although both the long-acting bupivacaine and the short-acting lidocaine are widely available and inexpensive amide-based local anesthetics, no data indicates which one is more effective than the other when used for SPGB. This double-blind, randomized, controlled trial compared the efficacy of lidocaine versus bupivacaine for SPGB with regard to optimizing intraoperative blood loss and surgical field quality during endoscopic septoplasty under general anesthesia.
The study was approved by the Ethics Committee of Aswan University Hospital (approval number: aswu/506/1/21) (date of registration: 05/01/2021) and registered with ClinicalTrials.gov (NCT05269147) dated 07/03/2022. Participants were informed about the study's goals and risks before signing a written informed consent form. All procedures were carried out following the Helsinki Declaration and subsequent amendments.
Sixty patients of 18-60 y of age, of both genders, with an ASA class I or II and planned to undergo endoscopic septoplasty were included in the study. Patients, who had additional surgery with endoscopic septoplasty, had systemic or bleeding disorders or were unwilling to participate in the study were excluded.
Patients were randomly assigned to one of three equal groups using computer-generated randomization tables. The group allocation was hidden in serially numbered, sealed, opaque envelopes. The patients, the surgeon, and the data collectors were unaware of the group assignment. An investigator who was not involved in the block performance or outcome evaluation prepared a fixed volume of study drugs based on group assignment. SPG block (SPGB) was conducted using normal saline in the control group (Group C); lidocaine 2% was used in the Group L, and 0.5% bupivacaine in the Group B.
No premedication or sedation was given to participants. Peripheral intravenous access was secured. Perioperative monitoring was established, including ECG, non-invasive arterial blood pressure, and pulse oximetry. Mean blood pressure (MBP) and heart rate (HR) were measured before the block (0 min.) and then every 5 min after the block until recovery time.
All patients were given a standardized general anesthesia protocol including fentanyl 2 µg/kg, propofol 2 mg/kg and atracurium 0.5 mg/kg and endotracheal tube inserted. Isoflurane in a mixture of 50% oxygen and 50% air was utilized to maintain anesthesia and keep mean arterial blood pressure and heart rate within 20% of the baseline values. Mechanical ventilation was designed to maintain an end-tidal carbon dioxide partial pressure (EtCO2) of 35 to 40 mmHg. At the end of the surgery, isoflurane was discontinued, and the residual neuromuscular block was reversed with neostigmine and atropine. Patients were then taken to the postoperative anesthesia care unit (PACU) for close observation, hemodynamic and oxygen saturation (SpO2) monitoring, and, if necessary, oxygen therapy. Patients were discharged from the PACU using Aldrete criteria.
Patients were positioned in a 15-degree reverse Trendelenburg position to aid venous drainage. The surgeon, who was blind to the study drugs, conducted the SPGB via a transnasal endoscopic technique. Endoscopic nasal SPGB allows for precise anatomy of the sphenopalatine foramen and direct injection of the target ganglion. The transnasal injection was performed with a 25-gauge spinal needle, and the injectates, containing 4 ml study drug plus one ml (4 mg) dexamethasone, were given according to the patient's study group assignment; 2.5 ml was injected into each side of the nose.
The primary endpoint was the surgical field quality using the Fromme ordinal scale, the operating surgeon graded the operating field as; 0 = no bleeding, almost bloodless field), 1 = Mild bleeding but not a surgical annoyance), 2 = Moderate bleeding, annoying but not impeding correct dissection), 3 - Moderate bleeding that compromised surgical dissection, 4 = Heavy but controlled bleeding that considerably impeded dissection, and 5 = severe uncontrolled bleeding. Intraoperative bleeding was measured using volumetric and gravimetric methods. Visual Analog pain score (VAS) was assessed at 30 min, 1 h, 4 h, 12 h, and 24 h after surgery. At the end of the operation, each patient received 1 g of paracetamol and 30 mg of ketorolac intravenously, and this combination was repeated every 8 h. The time to first postoperative request for rescue analgesia was noted. Meperidine 15 mg/dose was used as rescue analgesic when VAS ≥ 3. Total 24-h meperidine needed was recorded. Postoperative nausea and vomiting was noted. Patient satisfaction with pain control was noted 24 h after surgery as follows: 0 = poor/worse than expected, 1 = good/as expected, or 2 = excellent/better than expected. The surgical time was measured from incision to wound closure and duration of anesthesia was measured from anesthesia induction to anesthetic drug discontinuation.
Statistical analysis
The sample size was calculated using G*Power 3 software, with a power of 80% and type I error of 5% (α = 0.05 and β = 80%) on two-tailed tests to detect an effect size of 0.4 in the surgical field quality and the intraoperative bleeding volume between the three study groups based on a previous study.9 The minimum required sample was 54 participants, which were divided into three equal groups (18 participants needed for each group). The sample size was raised to include 20 in each group to compensate for drop-outs.
The collected data were verified, coded by the researcher, and analyzed using the Statistical Package for Social Sciences (IBM-SPSS/PC/VER 21). Descriptive statistics: means, standard deviations, medians, ranges, and percentages were calculated. Test of significance: Chi-square/Monte Carlo Exact test was used to compare the difference in the distribution of frequencies among different groups. The Shapiro-Wilk test was used to test data normality. For continuous variables with more than two categories, a one-way ANOVA test was calculated to test the mean differences of the data that follow a normal distribution; a post-hoc test was calculated using Tukey’s corrections for pairwise comparisons between the study groups. Also, a two-way repeated measure ANOVA test was calculated to test the mean differences of the data that followed a normal distribution and had repeated measures (between groups, within groups, and overall difference); the post-hoc test was calculated using Tukey’s corrections for pairwise comparisons between the two study groups. The Kaplan–Meier curve was plotted to explore the differences in the post-operative pain-free durations among the studied groups using the log-rank test. P < 0.05 was considered significant.
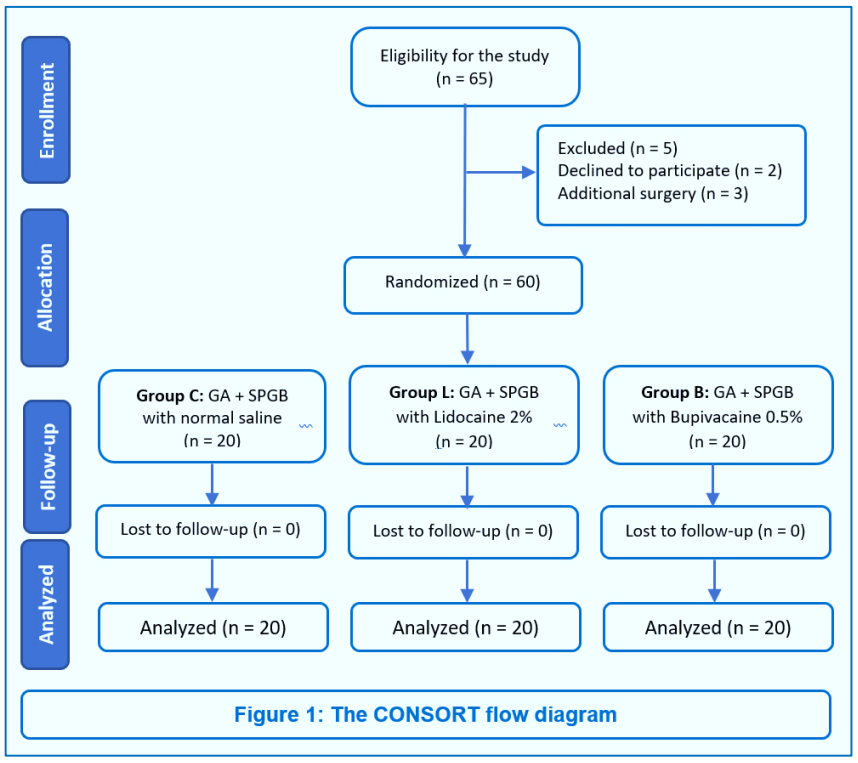
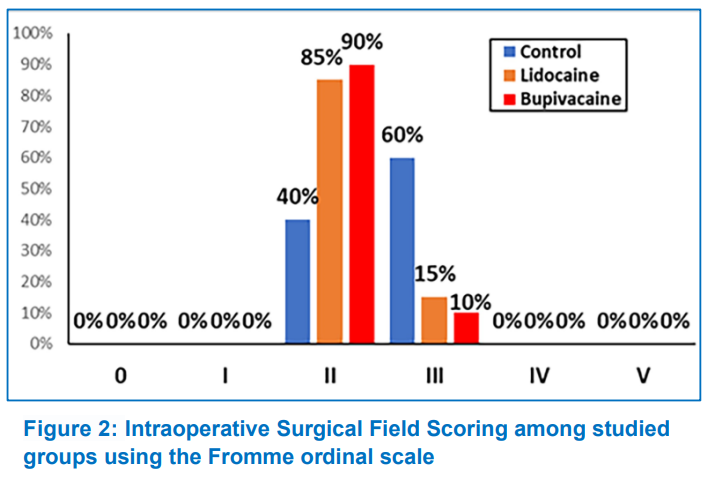
Sixty-five patients were eligible for the study. Five patients were excluded, two declined to participate, and three planned to receive additional surgery with endoscopic septoplasty. Sixty patients met the inclusion criteria and completed the study (Figure 1). The baseline patient characteristics data, length of surgery, and duration of anesthesia were comparable among the three study groups (Table 1). The mean amount of blood loss was lower in the Group L (P < 0.001) and Group B (P < 0.001) compared with the Group C, no difference was observed between the Group L and Group B (P = 0.640). The surgical field grading based on the Fromme ordinal scale was significantly lower in the Group L (P = 0.001) and Group B (P = 0.001) compared with the Group C; however, no difference was noted between the Group L and Group B (Table 2). Also, we found a significant association between surgical field scoring and the study groups (P = 0.001). In the control group, the surgeon reported moderate bleeding that compromised surgical dissection (grade 3) in 60% of patients and moderate bleeding that was annoying but did not impede correct dissection (grade 2) in 40% of patients. The Group L and Group B had a majority in grade 2 (85% and 90%, respectively) and a minority in grade 3 (15% and 10%, respectively) (Figure 2).
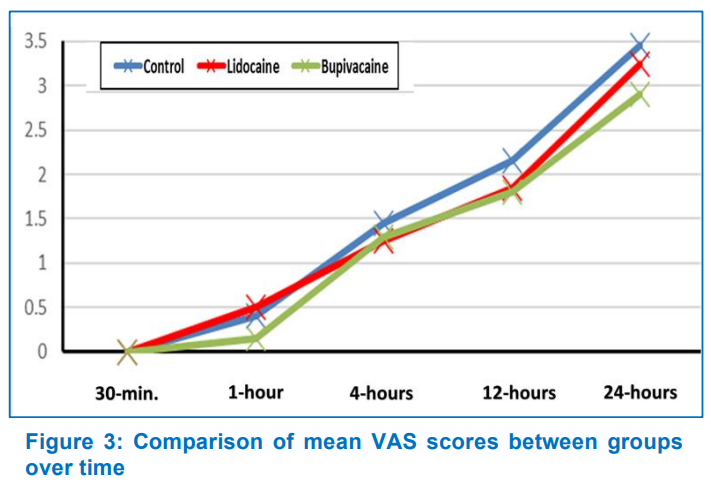
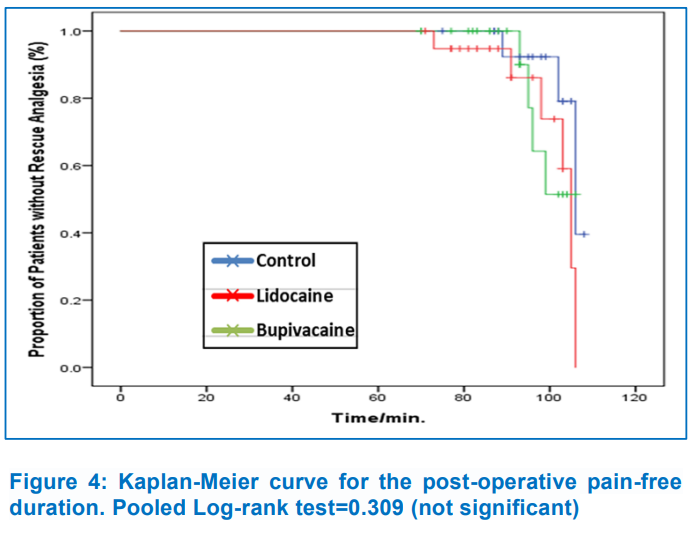
The mean VAS score had no significant difference (P > 0.05) over the study period between the groups (Figure 3). The postoperative pain-free duration in Group B (218.8 ± 17.2 min) was longer, but did not reach statistical significance when compared to Group C (179.2 ± 23.2 min, P = 0.686) and Group L (166.7 ± 23.8 min, P = 0.094) (Figure. 4). The total postoperative meperidine requirement was higher (P < 0.001) in the control group than in the Group L and Group B (Table 2).
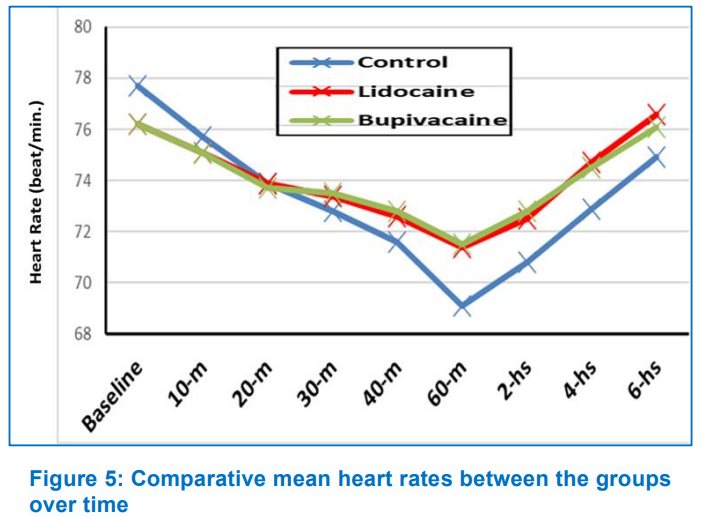
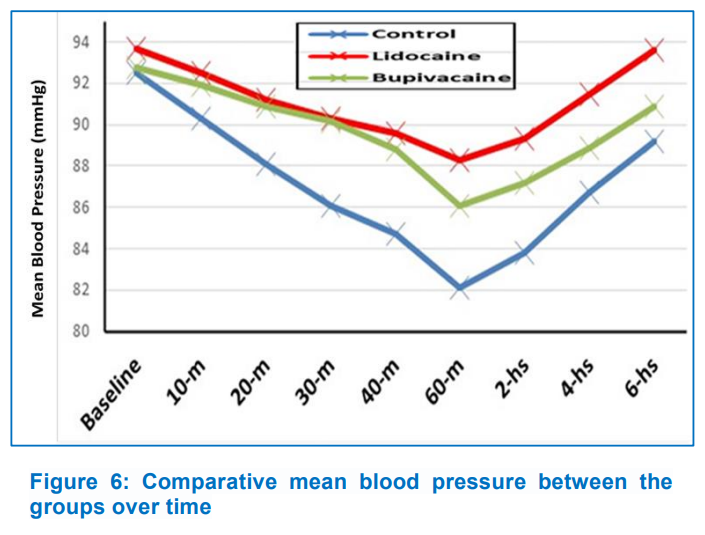
Heart rate (HR) and mean blood pressure (MBP) showed no significant difference (P > 0.05) between groups during the study period. In the interaction between time and treatment group throughout the trial, HR reduction in Group B was significantly greater (P < 0.001) than in the other two groups, and MBP reduction was significantly higher (P < 0.001) in Group C than in the other two groups (Figures 5, 6). After surgery, seven patients in Group B (35%) expressed satisfied with their pain control, followed by five patients in Group L (25%) and four patients (20%) in Group C. Postoperative nausea and vomiting were comparable among groups (Group C, 15%; Group L, 20%; and Group B, 30%) (Table 2).
The most common problem associated with septoplasty is excessive bleeding, which reduces the visibility of the operative field and increases the risk of complications.10 Our clinical trial is probably the first one to assess the efficacy of SPGB with lidocaine versus bupivacaine for optimizing intraoperative bleeding and surgical field visibility during endoscopic septoplasty under general anesthesia. We found that the mean amount of intraoperative blood loss and surgical field grading based on the Fromme ordinal scale was significantly lower in the Group L and Group B compared with the Group C, but no difference was found between the two.
Moreover, there was a significant relationship between surgical field scoring as determined by the surgeon and the studied groups; 12 patients (60%) in the Group C had moderate bleeding that moderately compromised surgical dissection (grade 3), while three patients (15%) in the Group L and two patients (10%) in the bupivacaine group had the same. Total postoperative meperidine requirement was higher in the control group than in both the lidocaine and bupivacaine groups.
Our findings are generally in line with an earlier study, which reported that intraoperative blood loss was significantly (P = 0.024) lower in patients, who received SPGB with bupivacaine 0.5% (44.73 ± 7.73 ml) compared to the control group (49.25 ± 7.20 ml) during septal surgery.11 The intraoperative blood loss was also significantly lower in patients, who received SPGB with 1% xylocaine local anesthetic than in patients who received a placebo (125.00 ± 41.52 ml,158.33 ± 37.33 ml, respectively; P = 0.0018) during endonasal endoscopic surgeries.7 Furthermore, Bhattacharyya et al. found that patients who received bilateral SPGB during endoscopic sinus surgery under general anesthesia had significantly lower blood loss (75.1667 ± 12.7633; P < 0.0001) and intraoperative surgical field category scale (1.4667 ± 0.681; P < 0.0001) than the control group.13 In contrast to the previous results, Rezaeian et al. revealed no statistically significant differences in intraoperative blood loss between SPGB and non-SPGB patients for FESS under general anesthesia. They explained that the surgeries were all performed by the same surgeon and that the cases were adjusted to account for variables such as blood loss.12
We found that the mean VAS score showed no significant difference (P > 0.05) between groups over the study period. This finding contradicts a recent clinical study that evaluated the effect of bilateral endoscopic SPGB for the management of postoperative pain in patients undergoing septoplasty and reported that the VAS scores were significantly (P < 0.001) lower in the patients who received SPGB with bupivacaine 0.5% compared to the control group upon arrival to the PACU and 2, 6, 12, and 24 h after septal surgery.15 This discrepancy could be explained by their use of lidocaine (40 mg/mL) in combination with adrenaline (0.025 mg) for topical nasal anesthesia following SPGB. Additionally, the various septoplasty techniques may have played a role. Notably, in 60 patients having general anesthesia for FESS, Cho et al. did not achieve statistically significant reductions in postoperative pain after performing SPGB with 0.25% bupivacaine.13 Sethi et al., on the other hand, investigated the efficacy of SPGB with 0.5% bupivacaine for postoperative analgesia in patients undergoing FESS under general anesthesia. They discovered that VAS scores were significantly lower in the bupivacaine group than in the saline group. Five patients (25%) in the bupivacaine group and fifteen patients (75%) in the saline group required rescue analgesia.14 This disparity may be attributed to the author’s use of different doses and concentrations of local anesthetics.
Bhattacharyya et al. investigated the effects of bilateral SPGB with levobupivacaine on 60 individuals having FESS under general anesthesia. They discovered that intraoperative MAP was significantly lower (P < 0.001) in the block group (65.5833 ± 1.0593 mmHg) than in the control group (81.0790 ± 8.1541 mmHg). Intraoperative HR was lower in the block group (74.0633 ± 3.9959 beats/min) than in the control group (78.2600 ± 5.7590 beats/min), which was statistically significant (P = 0.0018).9 Throughout the study, however, we found that the Group B had a significantly lower mean heart rate than the Group L and Group C, and that the Group C had a significantly lower mean blood pressure than the Group L and B. This could be due to the Group C's modified intraoperative isoflurane consumption for blood pressure control and increased postoperative meperidine needs for pain control. In addition, Ekici and Alagoz investigated the effect of bilateral endoscopic SPGB on patient satisfaction with postoperative pain control after septoplasty. 70% of patients in the SPGB group rated their pain control as excellent, compared to 16.7% in the Group C.15 However, We found no significant differences in patient satisfaction between the study groups; seven patients (35%) in Group B were satisfied with their pain control after surgery, followed by five patients (25%) in Group L, and four patients (20%) in Group C. This could be related to the fact that we used a multimodal analgesic regimen augmented with intravenous meperidine as rescue analgesia to maintain postoperative pain scores of 3 (as measured by a visual analog scale).
The limitations in our study include; a small sample size; secondly, the consumption of isoflurane was not measured, which could explain the lower mean blood pressure in the Group C. Third, VAS scores were examined 24 h following surgery; however, this should be prolonged to 72 h. Fourth, We didn't account for the incidence of laryngospasm, which is quite common after septoplasty. And finally, dexamethasone was utilized in all study groups, which can be a source of conflict in data analysis, particularly on secondary outcomes such as PONV.
In conclusion, sphenopalatine ganglion block groups utilizing bupivacaine or lidocaine revealed a significant decrease in intraoperative blood loss, surgical field grading, and postoperative opioid demand in patients having endoscopic septoplasty under general anesthesia as compared to the control group. Sphenopalatine ganglion block with bupivacaine or lidocaine showed no statistically significant alterations.
7. Trial registration
The study was approved by the Ethics Committee of Aswan University Hospital (approval number: aswu/506/1/21) (date of registration: 05/01/2021) and registered on ClinicalTrials.gov (NCT05269147) (07/03/2022).
8. Acknowledgments
The authors want to thank all doctors and staff at the otorhinolaryngology department for their assistance throughout the study. Moreover, we are grateful to the general OT anesthesiology technicians and nursing staff.
9. Competing interests
The authors declare that they have no competing interests. The authors received no outside funding.
10. Authors’ contributions
HG: Designed the study and wrote the manuscript.
MK: Performed the surgery and the SPGB; analyzed and interpreted the patient data
MS: Observed the patient intraoperatively and assessed the patient's elapsed time to the first postoperative request for rescue analgesia as well as postoperative analgesic needs in the first 24 hours.
All authors have read and approved the final version of the manuscript.
Author affiliations:
- Huda F. Ghazaly, PhD, Assistant Professor of Anesthesia & Intensive Care; Faculty of Medicine, Aswan University, Aswan, Egypt; E-mail: hfa737@gmail.com
- Mahmoud M. Khalefa, PhD, Lecturer of Otolaryngology; Faculty of Medicine, Aswan University, Aswan, Egypt; E-mail: mahmoudkhalefa7791@gmail.com
- Mohamed A. Sedeek, MSc, Resident of Anesthesia & Intensive Care; Faculty of Medicine, Aswan University, Aswan, Egypt; E-mail: hanaafahmy7@gmail.com
- Mohamed K. Hasanen, PhD, Lecturer of Anesthesia & Intensive Care; Faculty of Medicine, Aswan University, Aswan, Egypt; E-mail: drmohamedkamel85@gmail.com
ABSTRACT
Background & objective: The benefits of sphenopalatine ganglion (SPG) block during endoscopic septoplasty are yet unclear due to a lack of adequate data. We investigated the efficacy of SPG block (SPGB) with lidocaine versus bupivacaine for minimizing intraoperative bleeding and providing a bloodless surgical field in patients having endoscopic septoplasty under general anesthesia.
Methodology: In this double-blind, randomized controlled trial, 60 patients with ASA I and II were scheduled for endoscopic septoplasty. Following standard general anesthesia, patients were randomly assigned to one of three groups. Group C (n = 20): SPGB was performed using normal saline in the control group; Group L (n = 20); patients received SPGB with 2% lidocaine, and Group B (n = 20); patients received SPGB with 0.5% bupivacaine. Intraoperative blood loss, surgical field quality, postoperative pain-free duration, postoperative analgesic requirements, and the patient satisfaction were recorded. The collected data were verified, coded by the researcher, and analyzed using the Statistical Package for Social Sciences (IBM-SPSS/PC/VER 21). Statistical analysis was done with the Shapiro-Wilk test, one-way ANOVA test and two-way repeated measure ANOVA test as required. The Kaplan–Meier curve was plotted to explore the differences in the post-operative pain-free durations among the studied groups using the log-rank test. A p-value less than 0.05 was considered significant.
Results: The intraoperative blood loss and surgical field grading were lower in the groups treated with lidocaine (44.9 ± 3.7 ml, P < 0.001) or bupivacaine (41.4 ± 2.9 ml, P < 0.001) in comparison to the control group (89.95 ± 7.8 ml). There was no difference between the two groups. Postoperative meperidine requirement in 24 h was higher in the control group (36.25 ± 2.9 mg) than the lidocaine group (18.75 ± 2.5 mg; P < 0.001) and the bupivacaine group (16.25 ± 2.7 mg; P < 0.001).
Conclusion: Sphenopalatine ganglion blockade with bupivacaine or lidocaine decreased intraoperative blood loss and improved surgical field visibility compared to placebo during endoscopic septoplasty under general anesthesia. There were statistically no significant differences in sphenopalatine ganglion blocks with either lidocaine or bupivacaine.
Abbreviations: SPG- Sphenopalatine Ganglion; SPGB- Sphenopalatine Ganglion Block; VAS- Visual Analog Scale
Keywords: Bupivacaine; Endoscopic Septoplasty; Lidocaine; Postoperative Analgesia; Sphenopalatine Ganglion Block
Citation: Ghazaly HF, Khalefa MM, Sedeek MA, Hasanen MK. Sphenopalatine ganglion block with lidocaine versus bupivacaine for optimizing intraoperative bleeding and surgical field visibility during endoscopic septoplasty: a randomized double-blind controlled trial. Anaesth. pain intensive care 2024;28(1):92-99; DOI: 10.35975/apic.v28i1.2393
Received: August 29, 2023; Reviewed: December 29, 2023; Accepted: December 31, 2023
1. INTRODUCTION
Nasal septum surgeries are among the most common operations performed by otolaryngologists worldwide.1-2 Lanza and colleagues were the first to describe the use of endoscopic techniques to improve visualization during septal surgery.3 However, the new technique also posed several challenges to the anesthesiologists during endoscopic septoplasty, including maintaining hemodynamic stability, providing a bloodless surgical field, and producing satisfactory perioperative analgesic requirements. The sphenopalatine ganglion (SPG) is located in the pterygopalatine fossa, immediately behind the middle turbinate. It has somatosensory effects via the trigeminal and facial nerves, visceral motor parasympathetic activity via the superficial petrosal nerve, and sympathetic roles via its connection to the cervical sympathetic chain via the deep petrosal nerve.4 Thus, sphenopalatine ganglion block (SPGB) promotes mucosal vasoconstriction and reduces mucosal blood flow in the nasal cavity, resulting in a clear surgical field during nasal surgery. Moreover, SPGB anesthetizes the posterior septum, middle turbinate, sphenoid sinus, and posterior ethmoid cavity, and hence potentially prolonging postoperative analgesia.5 In previous clinical studies, the combination of SPGB with general anesthesia has been reported to reduce intraoperative blood loss and postoperative pain in patients undergoing functional endoscopic sinus surgery (FESS).6,7 However, there is a lack of evidence for the benefit of SPGB during endoscopic septoplasty.8 Furthermore, although both the long-acting bupivacaine and the short-acting lidocaine are widely available and inexpensive amide-based local anesthetics, no data indicates which one is more effective than the other when used for SPGB. This double-blind, randomized, controlled trial compared the efficacy of lidocaine versus bupivacaine for SPGB with regard to optimizing intraoperative blood loss and surgical field quality during endoscopic septoplasty under general anesthesia.
2. METHODOLOGY
The study was approved by the Ethics Committee of Aswan University Hospital (approval number: aswu/506/1/21) (date of registration: 05/01/2021) and registered with ClinicalTrials.gov (NCT05269147) dated 07/03/2022. Participants were informed about the study's goals and risks before signing a written informed consent form. All procedures were carried out following the Helsinki Declaration and subsequent amendments.
Sixty patients of 18-60 y of age, of both genders, with an ASA class I or II and planned to undergo endoscopic septoplasty were included in the study. Patients, who had additional surgery with endoscopic septoplasty, had systemic or bleeding disorders or were unwilling to participate in the study were excluded.
Patients were randomly assigned to one of three equal groups using computer-generated randomization tables. The group allocation was hidden in serially numbered, sealed, opaque envelopes. The patients, the surgeon, and the data collectors were unaware of the group assignment. An investigator who was not involved in the block performance or outcome evaluation prepared a fixed volume of study drugs based on group assignment. SPG block (SPGB) was conducted using normal saline in the control group (Group C); lidocaine 2% was used in the Group L, and 0.5% bupivacaine in the Group B.
No premedication or sedation was given to participants. Peripheral intravenous access was secured. Perioperative monitoring was established, including ECG, non-invasive arterial blood pressure, and pulse oximetry. Mean blood pressure (MBP) and heart rate (HR) were measured before the block (0 min.) and then every 5 min after the block until recovery time.
All patients were given a standardized general anesthesia protocol including fentanyl 2 µg/kg, propofol 2 mg/kg and atracurium 0.5 mg/kg and endotracheal tube inserted. Isoflurane in a mixture of 50% oxygen and 50% air was utilized to maintain anesthesia and keep mean arterial blood pressure and heart rate within 20% of the baseline values. Mechanical ventilation was designed to maintain an end-tidal carbon dioxide partial pressure (EtCO2) of 35 to 40 mmHg. At the end of the surgery, isoflurane was discontinued, and the residual neuromuscular block was reversed with neostigmine and atropine. Patients were then taken to the postoperative anesthesia care unit (PACU) for close observation, hemodynamic and oxygen saturation (SpO2) monitoring, and, if necessary, oxygen therapy. Patients were discharged from the PACU using Aldrete criteria.
Patients were positioned in a 15-degree reverse Trendelenburg position to aid venous drainage. The surgeon, who was blind to the study drugs, conducted the SPGB via a transnasal endoscopic technique. Endoscopic nasal SPGB allows for precise anatomy of the sphenopalatine foramen and direct injection of the target ganglion. The transnasal injection was performed with a 25-gauge spinal needle, and the injectates, containing 4 ml study drug plus one ml (4 mg) dexamethasone, were given according to the patient's study group assignment; 2.5 ml was injected into each side of the nose.
The primary endpoint was the surgical field quality using the Fromme ordinal scale, the operating surgeon graded the operating field as; 0 = no bleeding, almost bloodless field), 1 = Mild bleeding but not a surgical annoyance), 2 = Moderate bleeding, annoying but not impeding correct dissection), 3 - Moderate bleeding that compromised surgical dissection, 4 = Heavy but controlled bleeding that considerably impeded dissection, and 5 = severe uncontrolled bleeding. Intraoperative bleeding was measured using volumetric and gravimetric methods. Visual Analog pain score (VAS) was assessed at 30 min, 1 h, 4 h, 12 h, and 24 h after surgery. At the end of the operation, each patient received 1 g of paracetamol and 30 mg of ketorolac intravenously, and this combination was repeated every 8 h. The time to first postoperative request for rescue analgesia was noted. Meperidine 15 mg/dose was used as rescue analgesic when VAS ≥ 3. Total 24-h meperidine needed was recorded. Postoperative nausea and vomiting was noted. Patient satisfaction with pain control was noted 24 h after surgery as follows: 0 = poor/worse than expected, 1 = good/as expected, or 2 = excellent/better than expected. The surgical time was measured from incision to wound closure and duration of anesthesia was measured from anesthesia induction to anesthetic drug discontinuation.
Statistical analysis
The sample size was calculated using G*Power 3 software, with a power of 80% and type I error of 5% (α = 0.05 and β = 80%) on two-tailed tests to detect an effect size of 0.4 in the surgical field quality and the intraoperative bleeding volume between the three study groups based on a previous study.9 The minimum required sample was 54 participants, which were divided into three equal groups (18 participants needed for each group). The sample size was raised to include 20 in each group to compensate for drop-outs.
The collected data were verified, coded by the researcher, and analyzed using the Statistical Package for Social Sciences (IBM-SPSS/PC/VER 21). Descriptive statistics: means, standard deviations, medians, ranges, and percentages were calculated. Test of significance: Chi-square/Monte Carlo Exact test was used to compare the difference in the distribution of frequencies among different groups. The Shapiro-Wilk test was used to test data normality. For continuous variables with more than two categories, a one-way ANOVA test was calculated to test the mean differences of the data that follow a normal distribution; a post-hoc test was calculated using Tukey’s corrections for pairwise comparisons between the study groups. Also, a two-way repeated measure ANOVA test was calculated to test the mean differences of the data that followed a normal distribution and had repeated measures (between groups, within groups, and overall difference); the post-hoc test was calculated using Tukey’s corrections for pairwise comparisons between the two study groups. The Kaplan–Meier curve was plotted to explore the differences in the post-operative pain-free durations among the studied groups using the log-rank test. P < 0.05 was considered significant.

| Table 1: Baseline patient characteristics and surgical data | ||||
| Parameter | Group C
(n = 20) |
Group L
(n = 20) |
Group B
(n = 20) |
P-value |
| Age (y) | 34.35 ± 11.8 | 35.50 ± 12.4 | 35.10 ± 12.3 | 0.955 |
| Gender / Male | 13 (65%) | 11 (55%) | 13 (65%) | 0.754 |
| BMI (kg/m2) | 27.51 ± 2.1 | 27.23 ± 2.2 | 26.98 ± 1.9 | 0.720 |
| ASA | ||||
| · ASA I | 16 (80%) | 17 (85%) | 15 (75%) | 0.732 |
| · ASA II | 4 (20%) | 3 (15%) | 5 (25%) | |
| Length of Surgery (min) | 58.70 ± 4.2 | 57.30 ± 4.3 | 59.85 ± 3.4 | 0.909 |
| Anesthesia duration (min) | 94.95 ± 8.7 | 89.85 ± 10.6 | 90.15 ± 11.4 | 0.223 |
| Continuous variables are presented as mean ± SD. Categorical variables are presented as numbers (%). Group C, SPGB with normal saline; Group L, SPGB with lidocaine 2%; Group B, SPGB with bupivacaine 0.5%; ASA, American Society of Anesthesiologists; SD, standard deviation. | ||||
| Table 2: Primary and secondary outcomes | ||||
| Variable | Group C
(n = 20) |
Group L
(n = 20) |
Group B
(n = 20) |
P-value |
| Amount of blood loss (ml) | 89.95 ± 7.8 | 44.85 ± 3.7 * | 41.35 ± 2.9 * | < 0.001 |
| Surgical Field Scoring (Median (IQR)) | 3 (1) | 2 (0) * | 2 (0) * | 0.001 |
| Total meperidine (mg)/24 h (Mean ± SD) | 36.25 ± 2.9 | 18.75 ± 2.5 * | 16.25 ± 2.7 * | < 0.001 |
| Postoperative nausea/vomiting | 3 (15%) | 4 (20%) | 6 (30%) | 0.196 |
| Patient satisfaction | ||||
| · Poor | 1 (5%) | 0 (0%) | 0 (0%) | 0.124 |
| · Good | 15 (75%) | 15 (75%) | 13 (65%) | |
| · Excellent | 4 (20%) | 5 (25%) | 7 (35%) | |
| Group C, SPGB with normal saline; Group L, SPGB with lidocaine 2%; Group B, SPGB with bupivacaine 0.5%. *Statistical significance compared with the control group | ||||
3. RESULTS
Sixty-five patients were eligible for the study. Five patients were excluded, two declined to participate, and three planned to receive additional surgery with endoscopic septoplasty. Sixty patients met the inclusion criteria and completed the study (Figure 1). The baseline patient characteristics data, length of surgery, and duration of anesthesia were comparable among the three study groups (Table 1). The mean amount of blood loss was lower in the Group L (P < 0.001) and Group B (P < 0.001) compared with the Group C, no difference was observed between the Group L and Group B (P = 0.640). The surgical field grading based on the Fromme ordinal scale was significantly lower in the Group L (P = 0.001) and Group B (P = 0.001) compared with the Group C; however, no difference was noted between the Group L and Group B (Table 2). Also, we found a significant association between surgical field scoring and the study groups (P = 0.001). In the control group, the surgeon reported moderate bleeding that compromised surgical dissection (grade 3) in 60% of patients and moderate bleeding that was annoying but did not impede correct dissection (grade 2) in 40% of patients. The Group L and Group B had a majority in grade 2 (85% and 90%, respectively) and a minority in grade 3 (15% and 10%, respectively) (Figure 2).



The mean VAS score had no significant difference (P > 0.05) over the study period between the groups (Figure 3). The postoperative pain-free duration in Group B (218.8 ± 17.2 min) was longer, but did not reach statistical significance when compared to Group C (179.2 ± 23.2 min, P = 0.686) and Group L (166.7 ± 23.8 min, P = 0.094) (Figure. 4). The total postoperative meperidine requirement was higher (P < 0.001) in the control group than in the Group L and Group B (Table 2).
Heart rate (HR) and mean blood pressure (MBP) showed no significant difference (P > 0.05) between groups during the study period. In the interaction between time and treatment group throughout the trial, HR reduction in Group B was significantly greater (P < 0.001) than in the other two groups, and MBP reduction was significantly higher (P < 0.001) in Group C than in the other two groups (Figures 5, 6). After surgery, seven patients in Group B (35%) expressed satisfied with their pain control, followed by five patients in Group L (25%) and four patients (20%) in Group C. Postoperative nausea and vomiting were comparable among groups (Group C, 15%; Group L, 20%; and Group B, 30%) (Table 2).
4. DISCUSSION
The most common problem associated with septoplasty is excessive bleeding, which reduces the visibility of the operative field and increases the risk of complications.10 Our clinical trial is probably the first one to assess the efficacy of SPGB with lidocaine versus bupivacaine for optimizing intraoperative bleeding and surgical field visibility during endoscopic septoplasty under general anesthesia. We found that the mean amount of intraoperative blood loss and surgical field grading based on the Fromme ordinal scale was significantly lower in the Group L and Group B compared with the Group C, but no difference was found between the two.


Moreover, there was a significant relationship between surgical field scoring as determined by the surgeon and the studied groups; 12 patients (60%) in the Group C had moderate bleeding that moderately compromised surgical dissection (grade 3), while three patients (15%) in the Group L and two patients (10%) in the bupivacaine group had the same. Total postoperative meperidine requirement was higher in the control group than in both the lidocaine and bupivacaine groups.
Our findings are generally in line with an earlier study, which reported that intraoperative blood loss was significantly (P = 0.024) lower in patients, who received SPGB with bupivacaine 0.5% (44.73 ± 7.73 ml) compared to the control group (49.25 ± 7.20 ml) during septal surgery.11 The intraoperative blood loss was also significantly lower in patients, who received SPGB with 1% xylocaine local anesthetic than in patients who received a placebo (125.00 ± 41.52 ml,158.33 ± 37.33 ml, respectively; P = 0.0018) during endonasal endoscopic surgeries.7 Furthermore, Bhattacharyya et al. found that patients who received bilateral SPGB during endoscopic sinus surgery under general anesthesia had significantly lower blood loss (75.1667 ± 12.7633; P < 0.0001) and intraoperative surgical field category scale (1.4667 ± 0.681; P < 0.0001) than the control group.13 In contrast to the previous results, Rezaeian et al. revealed no statistically significant differences in intraoperative blood loss between SPGB and non-SPGB patients for FESS under general anesthesia. They explained that the surgeries were all performed by the same surgeon and that the cases were adjusted to account for variables such as blood loss.12
We found that the mean VAS score showed no significant difference (P > 0.05) between groups over the study period. This finding contradicts a recent clinical study that evaluated the effect of bilateral endoscopic SPGB for the management of postoperative pain in patients undergoing septoplasty and reported that the VAS scores were significantly (P < 0.001) lower in the patients who received SPGB with bupivacaine 0.5% compared to the control group upon arrival to the PACU and 2, 6, 12, and 24 h after septal surgery.15 This discrepancy could be explained by their use of lidocaine (40 mg/mL) in combination with adrenaline (0.025 mg) for topical nasal anesthesia following SPGB. Additionally, the various septoplasty techniques may have played a role. Notably, in 60 patients having general anesthesia for FESS, Cho et al. did not achieve statistically significant reductions in postoperative pain after performing SPGB with 0.25% bupivacaine.13 Sethi et al., on the other hand, investigated the efficacy of SPGB with 0.5% bupivacaine for postoperative analgesia in patients undergoing FESS under general anesthesia. They discovered that VAS scores were significantly lower in the bupivacaine group than in the saline group. Five patients (25%) in the bupivacaine group and fifteen patients (75%) in the saline group required rescue analgesia.14 This disparity may be attributed to the author’s use of different doses and concentrations of local anesthetics.
Bhattacharyya et al. investigated the effects of bilateral SPGB with levobupivacaine on 60 individuals having FESS under general anesthesia. They discovered that intraoperative MAP was significantly lower (P < 0.001) in the block group (65.5833 ± 1.0593 mmHg) than in the control group (81.0790 ± 8.1541 mmHg). Intraoperative HR was lower in the block group (74.0633 ± 3.9959 beats/min) than in the control group (78.2600 ± 5.7590 beats/min), which was statistically significant (P = 0.0018).9 Throughout the study, however, we found that the Group B had a significantly lower mean heart rate than the Group L and Group C, and that the Group C had a significantly lower mean blood pressure than the Group L and B. This could be due to the Group C's modified intraoperative isoflurane consumption for blood pressure control and increased postoperative meperidine needs for pain control. In addition, Ekici and Alagoz investigated the effect of bilateral endoscopic SPGB on patient satisfaction with postoperative pain control after septoplasty. 70% of patients in the SPGB group rated their pain control as excellent, compared to 16.7% in the Group C.15 However, We found no significant differences in patient satisfaction between the study groups; seven patients (35%) in Group B were satisfied with their pain control after surgery, followed by five patients (25%) in Group L, and four patients (20%) in Group C. This could be related to the fact that we used a multimodal analgesic regimen augmented with intravenous meperidine as rescue analgesia to maintain postoperative pain scores of 3 (as measured by a visual analog scale).
5. LIMITATIONS
The limitations in our study include; a small sample size; secondly, the consumption of isoflurane was not measured, which could explain the lower mean blood pressure in the Group C. Third, VAS scores were examined 24 h following surgery; however, this should be prolonged to 72 h. Fourth, We didn't account for the incidence of laryngospasm, which is quite common after septoplasty. And finally, dexamethasone was utilized in all study groups, which can be a source of conflict in data analysis, particularly on secondary outcomes such as PONV.
6. CONCLUSION
In conclusion, sphenopalatine ganglion block groups utilizing bupivacaine or lidocaine revealed a significant decrease in intraoperative blood loss, surgical field grading, and postoperative opioid demand in patients having endoscopic septoplasty under general anesthesia as compared to the control group. Sphenopalatine ganglion block with bupivacaine or lidocaine showed no statistically significant alterations.
7. Trial registration
The study was approved by the Ethics Committee of Aswan University Hospital (approval number: aswu/506/1/21) (date of registration: 05/01/2021) and registered on ClinicalTrials.gov (NCT05269147) (07/03/2022).
8. Acknowledgments
The authors want to thank all doctors and staff at the otorhinolaryngology department for their assistance throughout the study. Moreover, we are grateful to the general OT anesthesiology technicians and nursing staff.
9. Competing interests
The authors declare that they have no competing interests. The authors received no outside funding.
10. Authors’ contributions
HG: Designed the study and wrote the manuscript.
MK: Performed the surgery and the SPGB; analyzed and interpreted the patient data
MS: Observed the patient intraoperatively and assessed the patient's elapsed time to the first postoperative request for rescue analgesia as well as postoperative analgesic needs in the first 24 hours.
All authors have read and approved the final version of the manuscript.
11. REFERENCES
- Thomas A, Alt J, Gale C, Vijayakumar S, Padia R, Peters M, et al. Surgeon and hospital cost variability for septoplasty and inferior turbinate reduction. Int Forum Allergy Rhinol. 2016;6(10):1069-1074. [PubMed] DOI: 1002/alr.21775
- Kulkarni SV, Kulkarni VP, Burse K, Bharath M, Bharadwaj C, Sancheti V. Endoscopic Septoplasty: A Retrospective Analysis of 415 Cases. Indian J Otolaryngol Head Neck Surg. 2015;67(3):248-54. [PubMed] DOI: 1007/s12070-015-0880-1
- Stewart MG, Witsell DL, Smith TL, Weaver EM, Yueh B, Hannley MT. Development and validation of the Nasal Obstruction Symptom Evaluation (NOSE) scale. Otolaryngol Head Neck Surg. 2004;130(2):157-63. [PubMed] DOI: 1016/j.otohns.2003.09.016
- Robbins MS, Robertson CE, Kaplan E, Ailani J, Charleston L 4th, Kuruvilla D, et al. The Sphenopalatine Ganglion: Anatomy, Pathophysiology, and Therapeutic Targeting in Headache. Headache. 2016;56(2):240-58. [PubMed] DOI: 1111/head.12729
- Ah-See K. Rhinology: Diseases Of The Nose, Sinuses, And Skull Base. DW Kennedy , PH Hwang, eds. Thieme Medical Publishers. J Laryngol Otol. 2012;126(11):1194-1194. DOI: 1007/s00405-012-2206-1
- Kesimci E, Öztürk L, Bercin S, Kırış M, Eldem A, Kanbak O. Role of sphenopalatine ganglion block for postoperative analgesia after functional endoscopic sinus surgery. Eur Arch Otorhinolaryngol. 2012;269(1):165-9. [PubMed] DOI: 1007/s00405-011-1702-z
- Naik SM, Naik SS. Regional Anesthesia as Supplementation to General Anesthesia in Endonasal Surgeries. J Recent Adv Pain. 2019;5(2):40-44. [FreeFullText]
- Fujiwara T, Kuriyama A, Kato Y, Fukuoka T, Ota E. Perioperative local anaesthesia for reducing pain following septal surgery. Cochrane Database Syst Rev. 2018;8(8):CD012047. [PubMed] DOI: 1002/14651858.CD012047.pub2
- Bhattacharyya S, Tewari M, Ghosh S, Chakroborty J, Das W, Barman U. Evaluation of the efficacy of bilateral sphenopalatine ganglion block in endoscopic sinus surgery under general anesthesia: a randomized prospective controlled trial. Res Opin Anesth Intensive Care. 2016;3(4):173. [FreeFullText]
- Dąbrowska-Bień J, Skarżyński PH, Gwizdalska I, Łazęcka K, Skarżyński H. Complications in septoplasty based on a large group of 5639 patients. Eur Arch Otorhinolaryngol. 2018;275(7):1789-1794. [PubMed] DOI: 1007/s00405-018-4990-8
- Ekici NY, Alagöz S. The effectiveness of endoscopic sphenopalatine ganglion block in management of postoperative pain after septal surgery. Int Forum Allergy Rhinol. 2019;9(12):1521-1525. [PubMed] DOI: 1002/alr.22411
- Rezaeian A, Hashemi SM, Dokhanchi ZS. Effect of Sphenopalatine Ganglion Block With Bupivacaine on Postoperative Pain in Patients Undergoing Endoscopic Sinus Surgery. Allergy Rhinol. 2019;10:215265671882128. [PubMed] DOI: 1177/2152656718821282
- Cho DY, Drover DR, Nekhendzy V, Butwick AJ, Collins J, Hwang PH. The effectiveness of preemptive sphenopalatine ganglion block on postoperative pain and functional outcomes after functional endoscopic sinus surgery. Int Forum Allergy Rhinol. 2011;1(3):212-218. [PubMed] DOI: 1002/alr.20040
- Sethi S, Mahesh PV, Malhotra SK, Maitra S, Gupta AK. Comparison ropivacaine and bupivacaine in sphenopalatine ganglion block for postoperative analgesia after functional endoscopic sinus surgery: A randomized controlled trial. Acta Anaesthesiol Belg. 2017;68(3):137-142. [FreeFullText]