Marwa M. Mohamed, MD1, Ahmed F. Abdelraof, MD2, Mohamed A. Noser, MD3
Author affiliations:
Background & objective: Cleft palate is a congenital anomaly of the palate of the infants, and it may or may not be associated with cleft lip. The only remedy is surgical repair under general anesthesia. Postoperative pain relief in cleft palate repair surgery is very important to keep the infant or child calm and pain-free. It can only be achieved by use of analgesic drugs. We compared ketodex - a combination of ketamine and dexmedetomidine, versus opioid based anesthesia in cleft palate surgery as regards postoperative respiratory depression, need for oxygenation, and the emergence agitation.
Methodology: Fifty children, aged 6 months to 5 y, undergoing cleft palate surgery under general anesthesia, were randomly assigned to either ketodex group (Group KD) or opioids group (Group OP). Group KD received ketamine 1mg/kg loading dose followed by infusion @ 0.1 mg/kg/h, and dexmedetomidine loading dose 1 µg/kg followed by infusion @ 0.2 µg/kg/h], isoflurane anesthesia. Group OP received fentanyl in a loading dose of 2 µg/kg followed by infusion @ 0.5 µg/kg/h, followed by isoflurane anesthesia. Heart rate, mean blood pressure, emergence agitation, extubation time, FLACC score for pain, surgeons’ satisfaction, and the adverse effects were noted.
Results: Emergence agitation score was significantly lower in Group KD compared to Group OP. FLACC score showed insignificant difference between the two groups. There was no need for opioid administration in the PACU. Three patients in Group OP desaturated and needed oxygen supply. Incidence of vomiting was significantly higher in Group OP compared to Group KD (P = 0.022).
Conclusion: Ketodex used in opioid free anesthesia for cleft palate repair surgery in children provides adequate pain relief without respiratory depression, and is a good alternative of routine opioid based general anesthesia.
Key words: Ketamine; Dexmedetomidine; Opioids; Anesthesia; Recovery; Cleft Palate Surgery; Pediatrics
Citation: Mohamed MM, Abdelraof AF, Noser MA. Ketodex versus opioid based anesthesia in cleft palate. Anaesth. pain intensive care 2024;28(1):74-80; DOI: 10.35975/apic.v28i1.2239
Received: May 31, 2023; Reviewed: June 06, 2023; Accepted: January 02, 2024
Cleft palate abnormalities are common in pediatric patients. all patients with cleft palate must be anesthetized and have surgery to correct this anomaly. The incidence of airway obstruction reaches up to18% in isolated cleft palate cases and this risk increases in symptomatic children.1The risk of postoperative airway compromise needs an ideal perioperative anesthetic technique that results in adequate pain relief without respiratory depression. It is also favorable if such anesthetic technique could provide good postoperative analgesia, so decreasing the need for opioids and other analgesics that may result in respiratory depression. In addition to that, postoperative pain can lead to vigorous crying, which in turn increases the risk of wound dehiscence and pulmonary complications that lead to delayed hospital discharge.2
Dexmedetomidine is a selective α2 receptor agonist with sympatholytic, sedative, analgesic, opioid, and anesthetic-sparing properties without inducing significant respiratory depression. It attenuates the stress response to surgery and ensures a stable hemodynamics.3-4
Ketamine is an N-methyl-D-aspartate (NMDA) receptor antagonist. It is an I.V anesthetic and it has analgesic and sedative effects.5 It causes stimulation of the cardiorespiratory systems. It leads to an increase in cerebral blood flow, cerebral metabolic rate and intracranial pressure.6
When combined with ketamine, dexmedetomidine may reduce the tachycardia, hypertension, salivation, and emergence events that occur when ketamine is used. Ketamine may mitigate the tachycardia and hypotension caused by dexmedetomidine. A combination of ketamine and dexmedetomidine [ Ketodex ] has low incidence of adverse effects, good analgesic, predictable recovery and no cardiorespiratory depression.7 So here in this study, our goal was to compare Ketodex versus opioid based anesthesia in cleft palate repair surgeries as regard postoperative respiratory depression and emergence agitation and need for oxygen therapy.
Ethical approval for this study (No. FMASU R 10 2022) by the Research Ethics Committee of the
Faculty of Medicine, Ain Shams University, was obtained. The study was registered at ClinicalTrials.gov (No. NCT05402189). Written informed consent was obtained from all subjects.
Fifty patients of both genders, aged 6 months-5 y, ASA grade 1 and 2, scheduled for cleft palate repair under general anesthesia (GA) without other congenital anomalies were included. Patients who had an allergy to any of the study drugs, history of prematurity, delayed motor, mental or developmental milestones were excluded.
Patients were divided into 2 groups; Group KD and Group OP. Group KD patients received 1 mg/kg ketamine and 1 µg/kg dexmedetomidine diluted in 20 ml normal saline as a loading dose, followed by ketamine 0.1 mg/kg/h and dexmedetomidine 0.2 µg/kg/h as infusion.
Group OP patients received fentanyl 2 µg/kg diluted in 20 ml normal saline as a loading dose and then 0.5 µg/kg/h fentanyl as infusion.
2.1. Anesthesia and surgical procedure
In the operating room monitoring of pulse oximetry, non-invasive blood pressure, and an electrocardiogram was initiated.
An anesthesiologist, unaware of the patient's randomization status, administered study medications that had been prepared and sealed in an envelope by an anesthesiologist who had no further involvement in the research.
The study medication was given at induction and continued intraoperatively. Induction of anesthesia with sevoflurane inhalational anesthetic was done through a face mask and an IV line was secured. Suxamethonium 1.5 mg/kg IV was administered, an appropriate-sized ETT was inserted, and the throat was packed. A standard ventilator (Datex-Ohmeda, Helsinki, Finland) with a pediatric circle circuit was used for ventilation with appropriate settings. GA was maintained with isoflurane in 50% O2, with atracurium, titrated according to the hemodynamic parameters. Dexamethasone 0.2 mg/kg was given as an antiemetic and to reduce airway edema. Post-intubation, the loading doses of the study drugs were injected and infusions started. The urinary bladder of all patients was emptied with a sterile catheter before recovery.
At the end of surgery, patients were reversed with 0.05 mg/kg IV neostigmine and 0.02 mg/kg atropine, and extubated after meeting the standard extubation criteria.
2.2. Monitoring
Hemodynamic parameters, i.e., HR and MAP, were noted at following time points; pre-induction, pre-intubation, post-intubation and start of infusion, start of surgical dissection and every 15 min thereafter, end of surgery, extubation, and at 0.5, 1, and 2 h after shifting to PACU.
SpO2, EtCO2, end-tidal isoflurane concentration, and blood loss were monitored. Body temperature was stabilized using a warming mattress. The time taken from stopping the infusion to extubation was noted.
The PACU emergence agitation score was noted on the 0–4 point Watcha Emergence Agitation Scale.8
Pain was assessed with the FLACC (Faces, Legs, Activity, Cry and Consolability) score and Behavioral Pain Assessment Scale pain score.9
Time to requirement of first postoperative analgesic was noted. If the FLACC pain score was ≥ 4, then 1 µg/kg IV fentanyl was given as a rescue analgesic. The number of patients requiring rescue fentanyl in both groups was noted.
Surgeons' satisfaction score (0–10), based on bleeding from the surgical field, was noted at the end of surgery.
The children were shifted to the ward after 2 h. In the ward, children aged less than one year received a paracetamol suppository and children more than one year received a diclofenac suppository 3 times per day.
Hypertension and tachycardia were diagnosed intraoperatively if there was > 20% rise in MAP or HR above baseline on two or more readings in 2-3 min, which were managed by increasing isoflurane up to the maximum of 1% end-tidal concentration, followed by 1 µg/kg IV fentanyl if not controlled. Hypotension was defined as a reduction in SBP as per age-specific guidelines,10 and was corrected as required. Bradycardia was managed by stopping infusions and administering 0.01 mg/kg atropine. Postoperative desaturation, nausea, and vomiting were also noted.
The primary outcome of this study was the incidence of postoperative respiratory depression, while the secondary outcomes were demographic characteristics like age, weight, height, gestation week, incidence of emergence agitation during recovery from anesthesia, the incidence of the requirement of postoperative oxygen therapy, and the need of postoperative opioids and paracetamol.
2.3. Statistical methods
Sample size was calculated by using G power program, setting power at 80% alpha error at 5% and assuming large effect size difference of 0.5. It was estimated that the sample of 25 patients per group can detect a statistically significant difference between 2 groups. Based on that band after considering 20% dropout rate a sample of at least 50 patients (25 in each group) would be sufficient to achieve study objectives.
The Data was collected and statistical analysis was done using Statistical Package for Social Sciences (SPSS version 24) software. Arithmetic means, standard deviations, numbers and percentages were calculated as appropriate. For categorized parameters chai square test was used while for numerical data Student’s t-test was used to compare two groups. The level of significance was 0.05.
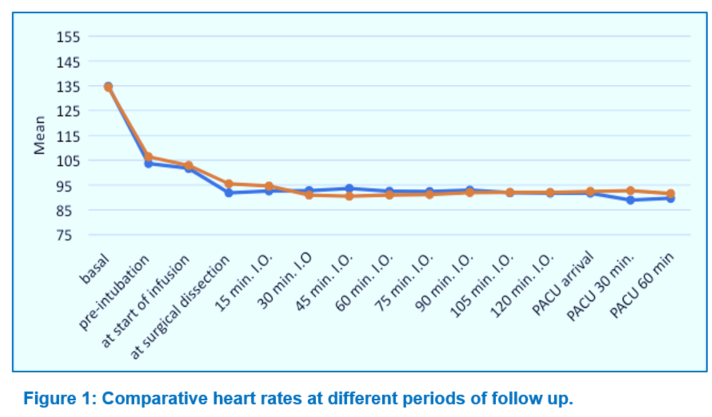
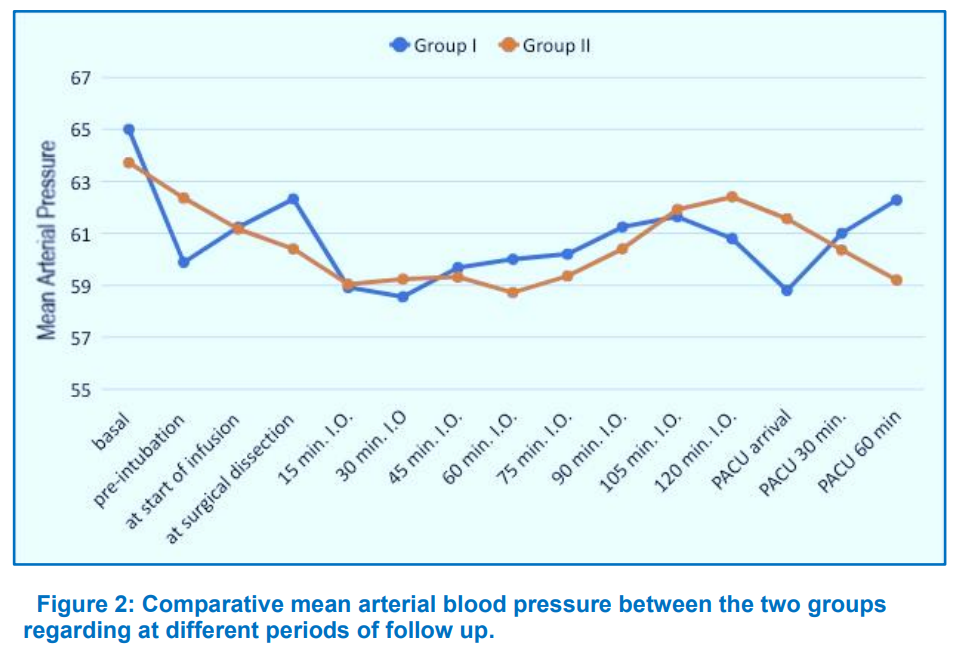
The study revealed no statistically significant difference in demographic and clinical characteristics of the study population; e.g., as age, weight, ASA status, and duration of surgery between the groups, as given in Table 1. Baseline HR and MAP measurements were comparable between the groups. MAP and HR showed insignificant difference between the groups at almost all of the time intervals throughout surgery till discharge from the PACU (Figures 1 and 2). As regards postoperative data, postoperative FLACC score showed insignificant difference between Group KD and Group OP at different periods of follow up (Table 2). Time from end of infusion till extubation was prolonged in Group KD compared to Group OP but this difference was insignificant (P > 0.05) (Table 3).
There was no need for opioid administration in PACU. Comparing both groups of the study, three patients in Group OP desaturated and needed oxygen by applying face mask. There was a significant statistical difference between the two groups as regards incidence of vomiting (Table 3).
The PACU emergence agitation score was significantly lower in Group KD compared to Group OP at 0, 5, 15 min postoperatively (P < 0.05). However, the difference became insignificant at 30 min in PACU (P > 0.05) (Table 4).
The intraoperative opioids have been used to provide analgesia and to stabilize the hemodynamics during GA. These have many benefits, but also some adverse effects such as nausea, vomiting, respiratory depression and tolerance.11 Opioid free anesthesia can be used preoperatively to block receptors in pain pathways.12 Opioid free anesthetic technique used non-opioid agents like sodium channel blockers, anti-inflammatory drugs, NMDA antagonist and alpha-2 agonists.13
We used ketodex as an opioid free anesthetic technique and compared it with standard opioid based anesthesia in cleft palate repair surgery. As this set of surgeries is associated with increased risk of postoperative airway compromise, we aimed to achieve adequate pain relief without respiratory depression. In the present study, there were insignificant differences regarding demographic data and clinical characteristics as well as the duration of surgery between the two groups. The current study showed that there was no statistically significant difference between the groups regarding HR and MAP at any intraoperative time till discharge from PACU. Surana et al. studied the effects of dexmedetomidine during cleft palate repair surgery.14 They found a significant difference as regards HR and MAP between the study groups (control and DEX groups) at all recorded times intraoperative. We didn't match those results of Surana et al. as regards the hemodynamics as we combined dexmedetomidine with ketamine to limit the tachycardia and hypertension of ketamine and the bradycardia and hypotension that may occur from dexmedetomidine.
As regards the time from end of infusion till extubation, it was prolonged in Group KD compared to Group OP without significant difference. This prolongation in Group KD can be attributed to sedative effect of dexmedetomidine. In a study done by Xu et al., and another one done by Jain et al., they found prolonged extubation time when studying the effect of dexmedetomidine on recovery from GA.15,16
Three patients in Group OP desaturated in PACU and they needed oxygen by applying a face mask and that did not occur in Group KD but this difference was statistically insignificant. A study done by Abdelrahman and Algharabawy.17 showed that there were statistically significant lower values in opioid based anesthesia group than opioid free anesthesia at 20 min, 30 min post extubation and at the first and second hours postoperatively. But this difference was insignificant immediately and after 10 min post extubation, third, fourth, and sixth hours postoperatively.
Our study showed that the incidence of vomiting was higher in the Group OP with significant difference than the Group KD. Hakim et al.18 also found that there was a significant difference in opioid based anesthesia group than opioid free anesthesia group as regards the incidence of nausea and vomiting.
As regards FLACC pain scores, patients in Group KD had a score from 0-2 compared to patients in Group OP, who had a score of 0-3, this difference was insignificant and there was no need for opioid administration in PACU. Dexmedetomidine has an effect on adrenoceptors that causes inhibition of norepinephrine release and so terminates the transmission of pain signals.19 Ketamine is NMDA receptor antagonist and its analgesic properties are well known.20 A study was done by Kayyal et al.21 on the treatment effect of dexmedetomidine and ketamine on postoperative analgesia in cleft palate repair. Their study was a retrospective analysis and they had control, Dexmedetomidine, and Ketamine groups. They found that patients in the later two groups had lower postoperative opioid requirements with no significant difference. Alghamdi et al.,22 in their pilot study, used opioid sparing technique for adenoidectomy operation and they found that FLACC pain scores were zero.
In the present study, emergence agitation score was significantly higher in the Group OP when compared with the Group KD at PACU arrival, at 5 min, and 15 min after that. That difference became insignificant at 30 min after surgery. A study done by Rao et al.23 as a meta-analysis, on the effect of dexmedetomidine on emergence agitation in children after anesthesia, found that dexmedetomidine significantly decreased the incidence of emergence agitation and delirium compared with placebo. Ibrahem Amin and Kamel.24 studied the effect of adding ketamine to dexmedetomidine on recovery from GA in adults undergoing endoscopic sinus surgery. They found that the Richmond Agitation-Sedation Scale (RASS) score was significantly higher in the control group than the Group KD. Many studies reported that ketamine decreased the incidence of emergence agitation when compared with placebo. Another study done by Demir and Yuzcat.25 studied the effect of ketamine on emergence agitation during rhinoplasty operation and reported a decreased incidence of emergence agitation significantly. Greg et al. reported that dexmedetomidine reduced the incidence of emergence agitation effectively in patients undergoing nasal surgery.26 A study done by Hadi et al. revealed that ketamine 0.15 mg /kg followed by dexmedetomidine 0.3 µg/kg I.V 10 min before end of surgery, significantly lowers the severity of emergence agitation compared to control group in children undergoing adenotonsillectomy.27 Yerramilli et al. reported that dexmedetomidine ketamine combination provided smooth recovery with one improving the adverse effects of the other, in day case surgeries in pediatrics.28
We conclude that using ketodex as opioid free anesthesia in cleft palate surgery in children under GA, provides adequate pain relief without respiratory depression.
6. Trial registration
NCT05402189
7. Data availability
The numerical data generated during this research is available with the authors.
8. Acknowledgement
We gratefully thank Faculty of Medicine, Ain Shams University, Heliopolis, Cairo, Egypt
9. Conflict of interest
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
MMM: Concept; design of the study; organization; data collection, statistical analysis; review and revision of the manuscript.
AFA, MAN: Data collection; organizing data; review the manuscript; Final approval
Author affiliations:
- Marwa Mamdouh Mohamed, Lecturer, Department of Anesthesia, Intensive Care & Pain Management, Faculty of Medicine, Ain Shams University, Heliopolis, Cairo, Egypt; E-mail: marwaemam600@gmail.com
- Ahmed Fayez Abdelraof, Lecturer, Department of Anesthesia, Intensive Care & Pain Management, Faculty of Medicine, Ain Shams University, Heliopolis, Cairo, Egypt; E-mail: awinnersid30@gmail.com
- Mohamed Abdallah Noser, Lecturer, Department of Anesthesia, Intensive Care & Pain Management, Faculty of Medicine, Ain Shams University, Heliopolis, Cairo, Egypt; E-mail: abyamoamed1981@gmail.com
ABSTRACT
Background & objective: Cleft palate is a congenital anomaly of the palate of the infants, and it may or may not be associated with cleft lip. The only remedy is surgical repair under general anesthesia. Postoperative pain relief in cleft palate repair surgery is very important to keep the infant or child calm and pain-free. It can only be achieved by use of analgesic drugs. We compared ketodex - a combination of ketamine and dexmedetomidine, versus opioid based anesthesia in cleft palate surgery as regards postoperative respiratory depression, need for oxygenation, and the emergence agitation.
Methodology: Fifty children, aged 6 months to 5 y, undergoing cleft palate surgery under general anesthesia, were randomly assigned to either ketodex group (Group KD) or opioids group (Group OP). Group KD received ketamine 1mg/kg loading dose followed by infusion @ 0.1 mg/kg/h, and dexmedetomidine loading dose 1 µg/kg followed by infusion @ 0.2 µg/kg/h], isoflurane anesthesia. Group OP received fentanyl in a loading dose of 2 µg/kg followed by infusion @ 0.5 µg/kg/h, followed by isoflurane anesthesia. Heart rate, mean blood pressure, emergence agitation, extubation time, FLACC score for pain, surgeons’ satisfaction, and the adverse effects were noted.
Results: Emergence agitation score was significantly lower in Group KD compared to Group OP. FLACC score showed insignificant difference between the two groups. There was no need for opioid administration in the PACU. Three patients in Group OP desaturated and needed oxygen supply. Incidence of vomiting was significantly higher in Group OP compared to Group KD (P = 0.022).
Conclusion: Ketodex used in opioid free anesthesia for cleft palate repair surgery in children provides adequate pain relief without respiratory depression, and is a good alternative of routine opioid based general anesthesia.
Key words: Ketamine; Dexmedetomidine; Opioids; Anesthesia; Recovery; Cleft Palate Surgery; Pediatrics
Citation: Mohamed MM, Abdelraof AF, Noser MA. Ketodex versus opioid based anesthesia in cleft palate. Anaesth. pain intensive care 2024;28(1):74-80; DOI: 10.35975/apic.v28i1.2239
Received: May 31, 2023; Reviewed: June 06, 2023; Accepted: January 02, 2024
1. INTRODUCTION
Cleft palate abnormalities are common in pediatric patients. all patients with cleft palate must be anesthetized and have surgery to correct this anomaly. The incidence of airway obstruction reaches up to18% in isolated cleft palate cases and this risk increases in symptomatic children.1The risk of postoperative airway compromise needs an ideal perioperative anesthetic technique that results in adequate pain relief without respiratory depression. It is also favorable if such anesthetic technique could provide good postoperative analgesia, so decreasing the need for opioids and other analgesics that may result in respiratory depression. In addition to that, postoperative pain can lead to vigorous crying, which in turn increases the risk of wound dehiscence and pulmonary complications that lead to delayed hospital discharge.2
Dexmedetomidine is a selective α2 receptor agonist with sympatholytic, sedative, analgesic, opioid, and anesthetic-sparing properties without inducing significant respiratory depression. It attenuates the stress response to surgery and ensures a stable hemodynamics.3-4
Ketamine is an N-methyl-D-aspartate (NMDA) receptor antagonist. It is an I.V anesthetic and it has analgesic and sedative effects.5 It causes stimulation of the cardiorespiratory systems. It leads to an increase in cerebral blood flow, cerebral metabolic rate and intracranial pressure.6
When combined with ketamine, dexmedetomidine may reduce the tachycardia, hypertension, salivation, and emergence events that occur when ketamine is used. Ketamine may mitigate the tachycardia and hypotension caused by dexmedetomidine. A combination of ketamine and dexmedetomidine [ Ketodex ] has low incidence of adverse effects, good analgesic, predictable recovery and no cardiorespiratory depression.7 So here in this study, our goal was to compare Ketodex versus opioid based anesthesia in cleft palate repair surgeries as regard postoperative respiratory depression and emergence agitation and need for oxygen therapy.
2. METHODOLOGY
Ethical approval for this study (No. FMASU R 10 2022) by the Research Ethics Committee of the
Faculty of Medicine, Ain Shams University, was obtained. The study was registered at ClinicalTrials.gov (No. NCT05402189). Written informed consent was obtained from all subjects.
Fifty patients of both genders, aged 6 months-5 y, ASA grade 1 and 2, scheduled for cleft palate repair under general anesthesia (GA) without other congenital anomalies were included. Patients who had an allergy to any of the study drugs, history of prematurity, delayed motor, mental or developmental milestones were excluded.
Patients were divided into 2 groups; Group KD and Group OP. Group KD patients received 1 mg/kg ketamine and 1 µg/kg dexmedetomidine diluted in 20 ml normal saline as a loading dose, followed by ketamine 0.1 mg/kg/h and dexmedetomidine 0.2 µg/kg/h as infusion.
Group OP patients received fentanyl 2 µg/kg diluted in 20 ml normal saline as a loading dose and then 0.5 µg/kg/h fentanyl as infusion.
2.1. Anesthesia and surgical procedure
In the operating room monitoring of pulse oximetry, non-invasive blood pressure, and an electrocardiogram was initiated.
An anesthesiologist, unaware of the patient's randomization status, administered study medications that had been prepared and sealed in an envelope by an anesthesiologist who had no further involvement in the research.
The study medication was given at induction and continued intraoperatively. Induction of anesthesia with sevoflurane inhalational anesthetic was done through a face mask and an IV line was secured. Suxamethonium 1.5 mg/kg IV was administered, an appropriate-sized ETT was inserted, and the throat was packed. A standard ventilator (Datex-Ohmeda, Helsinki, Finland) with a pediatric circle circuit was used for ventilation with appropriate settings. GA was maintained with isoflurane in 50% O2, with atracurium, titrated according to the hemodynamic parameters. Dexamethasone 0.2 mg/kg was given as an antiemetic and to reduce airway edema. Post-intubation, the loading doses of the study drugs were injected and infusions started. The urinary bladder of all patients was emptied with a sterile catheter before recovery.
At the end of surgery, patients were reversed with 0.05 mg/kg IV neostigmine and 0.02 mg/kg atropine, and extubated after meeting the standard extubation criteria.
2.2. Monitoring
Hemodynamic parameters, i.e., HR and MAP, were noted at following time points; pre-induction, pre-intubation, post-intubation and start of infusion, start of surgical dissection and every 15 min thereafter, end of surgery, extubation, and at 0.5, 1, and 2 h after shifting to PACU.
SpO2, EtCO2, end-tidal isoflurane concentration, and blood loss were monitored. Body temperature was stabilized using a warming mattress. The time taken from stopping the infusion to extubation was noted.
The PACU emergence agitation score was noted on the 0–4 point Watcha Emergence Agitation Scale.8
Pain was assessed with the FLACC (Faces, Legs, Activity, Cry and Consolability) score and Behavioral Pain Assessment Scale pain score.9
Time to requirement of first postoperative analgesic was noted. If the FLACC pain score was ≥ 4, then 1 µg/kg IV fentanyl was given as a rescue analgesic. The number of patients requiring rescue fentanyl in both groups was noted.
Surgeons' satisfaction score (0–10), based on bleeding from the surgical field, was noted at the end of surgery.
The children were shifted to the ward after 2 h. In the ward, children aged less than one year received a paracetamol suppository and children more than one year received a diclofenac suppository 3 times per day.
| Table 1: Comparative demographic and clinical data of the two groups. | ||||
| Parameter | Group KD
(n = 25) |
Group OP
(n = 25) |
Test applied | P value |
| Age (y) | 1.3 ± 0.6 | 1.7 ± 0.7 | T = 1.89 | 0.064 |
| Weight (Kg) | 10.4 ± 1.6 | 11.3 ± 1.4 | T = 1.78 | 0.081 |
| ASA
I II |
20 (80.0) 5 (20.0) |
20 (80.0) 5 (20.0) |
X2 = 0.611 |
0.500 |
| Duration of surgery (min) | 108.8 ± 8.5 | 106.4 ± 9.8 | T = 1.07 | 0.180 |
| Surgeons’ satisfaction | 2.6 ± 0.9 | 3.0 ± 0.6 | T = 1.77 | 0.082 |
| X2 = Chi square test; T = student t-test; P < 0.05 considered as significant | ||||
Hypertension and tachycardia were diagnosed intraoperatively if there was > 20% rise in MAP or HR above baseline on two or more readings in 2-3 min, which were managed by increasing isoflurane up to the maximum of 1% end-tidal concentration, followed by 1 µg/kg IV fentanyl if not controlled. Hypotension was defined as a reduction in SBP as per age-specific guidelines,10 and was corrected as required. Bradycardia was managed by stopping infusions and administering 0.01 mg/kg atropine. Postoperative desaturation, nausea, and vomiting were also noted.
| Table 2: Comparative FLACC scores in the two studied groups | |||||
| FLACC scores | Group KD
(n = 25) |
Group OP
(n = 25) |
X2
|
P value | |
| On admission | 0 | 8 (32.0) | 4 (16.0) | 2.600 |
0.457 |
| 1 | 7 (28.0) | 6 (24.0) | |||
| 2 | 8 (32.0) | 13 (52.0) | |||
| 3 | 2 (8.0) | 2 (8.0) | |||
| After 30 min | 0 | 7 (28.0) | 10 (40.0) | 2.255 |
0.521. |
| 1 | 9 (36.0) | 5 (20.0) | |||
| 2 | 7 (28.0) | 9 (36.0) | |||
| 3 | 2 (8.0) | 1 (4.0) | |||
| After 1 h | 0 | 4 (16.0) | 5 (20.0) | 2.771 |
0.438. |
| 1 | 12 (48.0) | 8 (32.0) | |||
| 2 | 8 (32.0) | 8 (32.0) | |||
| 3 | 1 (4.0) | 4 (16.0) | |||
| After 2 h | 0 | 7 (28.0) | 4 (16.0) | 3.324 |
0.344 |
| 1 | 5 (20.0) | 7 (28.0) | |||
| 2 | 13 (52.0) | 10 (40.0) | |||
| 3 | 0 (0.0) | 4 (16.0) | |||
| Postoperative
|
0 | 13 (52.0) | 8 (32.0) | 0.978 |
0.424 |
| 1 | 10 (40.0) | 10 (40.0) | |||
| 2 | 2 (8.0) | 5 (20.0) | |||
| 3 | 0 (0.0) | 2 (8.0) | |||
| X2 = Chi square test; P < 0.05 was significant; Data presented as n(%) | |||||
The primary outcome of this study was the incidence of postoperative respiratory depression, while the secondary outcomes were demographic characteristics like age, weight, height, gestation week, incidence of emergence agitation during recovery from anesthesia, the incidence of the requirement of postoperative oxygen therapy, and the need of postoperative opioids and paracetamol.
2.3. Statistical methods
Sample size was calculated by using G power program, setting power at 80% alpha error at 5% and assuming large effect size difference of 0.5. It was estimated that the sample of 25 patients per group can detect a statistically significant difference between 2 groups. Based on that band after considering 20% dropout rate a sample of at least 50 patients (25 in each group) would be sufficient to achieve study objectives.
The Data was collected and statistical analysis was done using Statistical Package for Social Sciences (SPSS version 24) software. Arithmetic means, standard deviations, numbers and percentages were calculated as appropriate. For categorized parameters chai square test was used while for numerical data Student’s t-test was used to compare two groups. The level of significance was 0.05.
3. RESULTS
The study revealed no statistically significant difference in demographic and clinical characteristics of the study population; e.g., as age, weight, ASA status, and duration of surgery between the groups, as given in Table 1. Baseline HR and MAP measurements were comparable between the groups. MAP and HR showed insignificant difference between the groups at almost all of the time intervals throughout surgery till discharge from the PACU (Figures 1 and 2). As regards postoperative data, postoperative FLACC score showed insignificant difference between Group KD and Group OP at different periods of follow up (Table 2). Time from end of infusion till extubation was prolonged in Group KD compared to Group OP but this difference was insignificant (P > 0.05) (Table 3).
| Table 3: Comparative postoperative data. | ||||
| Parameter | Group KD
(n = 25) |
Group OP
(n = 25) |
X2
|
P value |
| Vomiting | 0 (0.0) | 6 (24.0) | 3.11 | 0.022* |
| Desaturation | 0 (0.0) | 3 (12.0) | 0.987 | 0.234 |
| Need for oxygen | 0 (0.0) | 3 (12.0) | 0.987 | 0.234 |
| X2 = Chi square test; P < 0.05 was significant; * Significant difference | ||||
There was no need for opioid administration in PACU. Comparing both groups of the study, three patients in Group OP desaturated and needed oxygen by applying face mask. There was a significant statistical difference between the two groups as regards incidence of vomiting (Table 3).
The PACU emergence agitation score was significantly lower in Group KD compared to Group OP at 0, 5, 15 min postoperatively (P < 0.05). However, the difference became insignificant at 30 min in PACU (P > 0.05) (Table 4).
| Table 4: Comparison in the emergence agitation | ||||
| Observations | Group KD
(n = 25) |
Group OP
(n = 25) |
X2
|
P value |
| Emerge agitation T0 | ||||
| 1. The child makes eye contact with the caregiver | 9 (36.0) | 2 (8.0) | 2.97 |
0.021* |
| 2. The child’s action is purposeful | 16 (64.0) | 13 (52.0) | ||
| 3. The child is aware of his/her surroundings | 0 (0.0) | 9 (36.0) | ||
| 4. The child is restless | 0 (0.0) | 1 (4.0) | ||
| Emergence agitation T5 | ||||
| 1. The child makes eye contact with the caregiver | 15 (60.0) | 7 (28.0) | 5.55 |
0.036* |
| 2. The child’s action is purposeful | 10 (40.0) | 12 (48.0) | ||
| 3. The child is aware of his/her surroundings | 0 (0.0) | 6 (24.0) | ||
| 4. The child is restless | 0 (0.0) | 0 (0.0) | ||
| Emergence agitation T15 | ||||
| 1. The child makes eye contact with the caregiver | 22 (88.0) | 13 (52.0) | 4.58 |
0.039* |
| 2. The child’s action is purposeful | 3 (12.0) | 10 (40.0) | ||
| 3. The child is aware of his/her surroundings | 0 (0.0) | 2 (8.0) | ||
| 4. The child is restless | 0 (0.0) | 0 (0.0) | ||
| Emergence agitation T30 | ||||
| 1. The child makes eye contact with the caregiver | 25 (100.0) | 24 (96.0) | 0.214 |
0.861 |
| 2. The child’s action is purposeful | 0 (0.0) | 1 (4.0) | ||
| 3. The child is aware of his/her surroundings | 0 (0.0) | 0 (0.0) | ||
| 4. The child is restless | 0 (0.0) | 0 (0.0) | ||
| X2 = Chi square test; P < 0.05 was significant; * Significant difference | ||||
4. DISCUSSION
The intraoperative opioids have been used to provide analgesia and to stabilize the hemodynamics during GA. These have many benefits, but also some adverse effects such as nausea, vomiting, respiratory depression and tolerance.11 Opioid free anesthesia can be used preoperatively to block receptors in pain pathways.12 Opioid free anesthetic technique used non-opioid agents like sodium channel blockers, anti-inflammatory drugs, NMDA antagonist and alpha-2 agonists.13


We used ketodex as an opioid free anesthetic technique and compared it with standard opioid based anesthesia in cleft palate repair surgery. As this set of surgeries is associated with increased risk of postoperative airway compromise, we aimed to achieve adequate pain relief without respiratory depression. In the present study, there were insignificant differences regarding demographic data and clinical characteristics as well as the duration of surgery between the two groups. The current study showed that there was no statistically significant difference between the groups regarding HR and MAP at any intraoperative time till discharge from PACU. Surana et al. studied the effects of dexmedetomidine during cleft palate repair surgery.14 They found a significant difference as regards HR and MAP between the study groups (control and DEX groups) at all recorded times intraoperative. We didn't match those results of Surana et al. as regards the hemodynamics as we combined dexmedetomidine with ketamine to limit the tachycardia and hypertension of ketamine and the bradycardia and hypotension that may occur from dexmedetomidine.
As regards the time from end of infusion till extubation, it was prolonged in Group KD compared to Group OP without significant difference. This prolongation in Group KD can be attributed to sedative effect of dexmedetomidine. In a study done by Xu et al., and another one done by Jain et al., they found prolonged extubation time when studying the effect of dexmedetomidine on recovery from GA.15,16
Three patients in Group OP desaturated in PACU and they needed oxygen by applying a face mask and that did not occur in Group KD but this difference was statistically insignificant. A study done by Abdelrahman and Algharabawy.17 showed that there were statistically significant lower values in opioid based anesthesia group than opioid free anesthesia at 20 min, 30 min post extubation and at the first and second hours postoperatively. But this difference was insignificant immediately and after 10 min post extubation, third, fourth, and sixth hours postoperatively.
Our study showed that the incidence of vomiting was higher in the Group OP with significant difference than the Group KD. Hakim et al.18 also found that there was a significant difference in opioid based anesthesia group than opioid free anesthesia group as regards the incidence of nausea and vomiting.
As regards FLACC pain scores, patients in Group KD had a score from 0-2 compared to patients in Group OP, who had a score of 0-3, this difference was insignificant and there was no need for opioid administration in PACU. Dexmedetomidine has an effect on adrenoceptors that causes inhibition of norepinephrine release and so terminates the transmission of pain signals.19 Ketamine is NMDA receptor antagonist and its analgesic properties are well known.20 A study was done by Kayyal et al.21 on the treatment effect of dexmedetomidine and ketamine on postoperative analgesia in cleft palate repair. Their study was a retrospective analysis and they had control, Dexmedetomidine, and Ketamine groups. They found that patients in the later two groups had lower postoperative opioid requirements with no significant difference. Alghamdi et al.,22 in their pilot study, used opioid sparing technique for adenoidectomy operation and they found that FLACC pain scores were zero.
In the present study, emergence agitation score was significantly higher in the Group OP when compared with the Group KD at PACU arrival, at 5 min, and 15 min after that. That difference became insignificant at 30 min after surgery. A study done by Rao et al.23 as a meta-analysis, on the effect of dexmedetomidine on emergence agitation in children after anesthesia, found that dexmedetomidine significantly decreased the incidence of emergence agitation and delirium compared with placebo. Ibrahem Amin and Kamel.24 studied the effect of adding ketamine to dexmedetomidine on recovery from GA in adults undergoing endoscopic sinus surgery. They found that the Richmond Agitation-Sedation Scale (RASS) score was significantly higher in the control group than the Group KD. Many studies reported that ketamine decreased the incidence of emergence agitation when compared with placebo. Another study done by Demir and Yuzcat.25 studied the effect of ketamine on emergence agitation during rhinoplasty operation and reported a decreased incidence of emergence agitation significantly. Greg et al. reported that dexmedetomidine reduced the incidence of emergence agitation effectively in patients undergoing nasal surgery.26 A study done by Hadi et al. revealed that ketamine 0.15 mg /kg followed by dexmedetomidine 0.3 µg/kg I.V 10 min before end of surgery, significantly lowers the severity of emergence agitation compared to control group in children undergoing adenotonsillectomy.27 Yerramilli et al. reported that dexmedetomidine ketamine combination provided smooth recovery with one improving the adverse effects of the other, in day case surgeries in pediatrics.28
5. CONCLUSION
We conclude that using ketodex as opioid free anesthesia in cleft palate surgery in children under GA, provides adequate pain relief without respiratory depression.
6. Trial registration
NCT05402189
7. Data availability
The numerical data generated during this research is available with the authors.
8. Acknowledgement
We gratefully thank Faculty of Medicine, Ain Shams University, Heliopolis, Cairo, Egypt
9. Conflict of interest
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
MMM: Concept; design of the study; organization; data collection, statistical analysis; review and revision of the manuscript.
AFA, MAN: Data collection; organizing data; review the manuscript; Final approval
11. REFERENCES
- Liau JY, Sadove AM, van Aalst JA. An evidence-based approach to cleft palate repair. Plast Reconstr Surg. 2010;126(6):2216–2221. [PubMed] DOI: 1097/PRS.0b013e3181f830eb
- Obayah GM, Refaie A, Aboushanab O, Ibraheem N, Abdelazees M. Addition of dexmedetomidine to bupivacaine for greater palatine nerve block prolongs postoperative analgesia after cleft palate repair. Eur J Anaesthesiol. 2010;27(3):280–284. [PubMed] DOI: 1097/EJA.0b013e3283347c15
- Mason KP, Lerman J. Review article: dexmedetomidine in children: current knowledge and future applications. Anesth Analg. 2011;113:1129-42. [PubMed] DOI: 1213/ANE.0b013e31822b8629
- Mahmoud M, Mason KP. Dexmedetomidine: review, update, and future considerations of paediatric perioperative and periprocedural applications and limitations. Br J Anaesth. 2015;115:171-82. [PubMed] DOI: 1093/bja/aev226
- Elia N, Tramèr MR. Ketamine and postoperative pain—a quantitative systematic review of randomised trials. Pain. 2005;113(1–2):61–70. [PubMed] DOI: 1016/j.pain.2004.09.036
- Amornyotin S. Dexmedetomidine in gastrointestinal endoscopic procedures. World J Anesthesiol. 2016;5:1-14. DOI: 5313/wja.v5.i1.1
- Tobias JD. Dexmedetomidine and ketamine: an effective alternative for procedural sedation? Pediatr Crit Care Med. 2012;13:423-427. [PubMed] DOI: 1097/PCC.0b013e318238b81c
- Reduque LL, Verghese S. Paediatric emergence delirium. Contin Educ Anaesth Crit Care Pain. 2013;13:39-41. DOI: 1093/bjaceaccp/mks051
- Merkel SI, Voepel-Lewis T, Shayevitz JR, Malviya S. The FLACC: a behavioral scale for scoring postoperative pain in young children. Pediatr Nurs. 1997;23:293-7. [PubMed]
- de Caen AR, Berg MD, Chameides L, Gooden CK, Hickey RW, Scott HF, et al. Part 12: pediatric advanced life support: 2015 American Heart Association guidelines update for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation. 2015;132(18 Suppl2):S526-42. [PubMed] DOI: 1161/CIR.0000000000000266
- Aboalsoud R, Arida E, Sabry L, Elmolla AF, Ghoneim HEM. The effect of opioid free versus opioid based anaesthesia on breast cancer pain score and immune response. Egypt J Anaesth. 2021;37(1):472-482. DOI: 1080/11101849.2021.1983366
- Beloeil H. Opioid-free anesthesia. Best Pract Res Clin Anaesthesiol. 2019;33(3):353-360. [PubMed] DOI: 1016/j.bpa.2019.09.002
- Buvanendran A, Kroin JS. Multimodal analgesia for controlling acute postoperative pain. Curr Opin Anaesthesiol. 2009;22:588-93. [PubMed] DOI: 1097/ACO.0b013e328330373a
- Surana P, Parikh DA, Patkar GA, Tendolkar BA. A prospective randomized controlled double-blind trial to assess the effects of dexmedetomidine during cleft palate surgery. Korean J Anesthesiol. 2017 Dec;70(6):633-641. [PubMed] DOI: 4097/kjae.2017.70.6.633
- Xu K, Pan Y, Zhu M. Effect of dexmedetomidine on the recovery profiles from general anesthesia inpatients undergoing endoscopic sinus surgery. Int J Clin Exp Med. 2016;9(5):8405–8410. [FreeFullText]
- Jain V, Chaturvedi A, Pandia MP, Bithal PK. Effect of dexmedetomidine on recovery profile of patients undergoing anterior cervical discectomy and fusion. J Anaesthesiol Clin Pharmacol. 2019;35(1):92-98. [PubMed] DOI: 4103/joacp.JOACP_5_18
- Abdelrahman T, Algharabawy W. Opioid-free general anaesthesia for transthoracic oesophagectomy: does it improve postoperative analgesia and other recovery criteria? A prospective randomised study. Ain Shams J Anesthesiol. 2021;13(1):1-10. DOI: 1186/s42077-021-00170-6
- Hakim KYK, Wahba WZB. Opioid-Free Total Intravenous Anesthesia Improves Postoperative Quality of Recovery after Ambulatory Gynecologic Laparoscopy. Anesth Essays Res. 2019;13(2):199-203. [PubMed] DOI: 4103/aer.AER_74_19
- Gertler R, Brown HC, Mitchell DH, Silvius EN. Dexmedetomidine: a novel sedative-analgesic agent. Proc (Bayl Univ Med Cent). 2001;14(1):13–21 [PubMed] DOI: 1080/08998280.2001.11927725
- Elia N, Tramèr MR. Ketamine and postoperative pain—a quantitative systematic review of randomised trials. Pain. 2005;113(1–2):61–70. [PubMed] DOI: 1016/j.pain.2004.09.036
- Kayyal TA, Wolfswinkel EM, Weathers WM, Capehart SJ, Monson LA, Buchanan EP, et al. Treatment effects of dexmedetomidine and ketamine on postoperative analgesia after cleft palate repair. Craniomaxillofac Trauma Reconstr. 2014;7(2):131-8. [PubMed] DOI: 1055/s-0034-1371446
- Alghamdi F, Roth C, Jatana KR, Elmaraghy CA, Rice J, Tobias JD, et al. Opioid-Sparing Anesthetic Technique for Pediatric Patients Undergoing Adenoidectomy: A Pilot Study. J Pain Res. 2020;13:2997-3004. [PubMed] DOI: 2147/JPR.S281275
- Rao Y, Zeng R, Jiang X, Li J, Wang X. The Effect of Dexmedetomidine on Emergence Agitation or Delirium in Children After Anesthesia-A Systematic Review and Meta-Analysis of Clinical Studies. Front Pediatr. 202;8:329. [PubMed] DOI: 3389/fped.2020.00329
- Amin OAI, Kamel AAF. Effects of adding ketamine to dexmedetomidine on smooth recovery from isoflurane anesthesia in adults undergoing endoscopic sinus surgery. Egypt J Anaesth. 2020;36:1:50-55. DOI: 1080/11101849.2020.1761144
- Demir CY, Yuzkat N. Prevention of emergence agitation with ketamine in rhinoplasty. Aesthetic Plast Surg. 2018;42:847–853. [PubMed] DOI: 1007/s00266-018-1103-4
- Garg A, Kamal M, Mohammed S, Singariya G, Chouhan DS, Biyani G. Efficacy of dexmedetomidine for prevention of emergence agitation in patients posted for nasal surgery under desfluraneanaesthesia: A prospective double-blinded randomized controlled trial. Indian J Anaesth. 2008;62(7):524–530. [PubMed] DOI: 4103/ija.IJA_788_17
- Hadi SM, Saleh AJ, Tang YZ, Daoud A, Mei X, Ouyang W. The effect of KETODEX on the incidence and severity of emergence agitation in children undergoing adenotonsillectomy using sevoflurane based-anesthesia. Int J Pediatr Otorhinolaryngol. 2015;79(5):671-6. [PubMed] DOI: 1016/j.ijporl.2015.02.012
- Yerramilli RN, Aavula M. Ketamine and dexmedetomidine for day care anesthesia in pediatric surgery. J Evolution Med Dent Sci. 2016;5:5257–5260. DOI: 10.14260/jemds/2016/1191